026b0ac436b708a6ef37cc6773cd7f2e.ppt
- Количество слайдов: 25
 Effect of Levosimendan on the Short-Term Clinical Course of Patients With Acutely Decompensated Heart Failure Binu George , Heather Bury Critical care Journal Club May 2014
Effect of Levosimendan on the Short-Term Clinical Course of Patients With Acutely Decompensated Heart Failure Binu George , Heather Bury Critical care Journal Club May 2014
 Background l Over a million people hospitalised in the US for treatment of ADHF l Usually receive IV diuretics , peripheral vasdilators , positive inotropes l Unclear how haemodynamics translate to clinical benefits l Average hospital stay 5 days
Background l Over a million people hospitalised in the US for treatment of ADHF l Usually receive IV diuretics , peripheral vasdilators , positive inotropes l Unclear how haemodynamics translate to clinical benefits l Average hospital stay 5 days
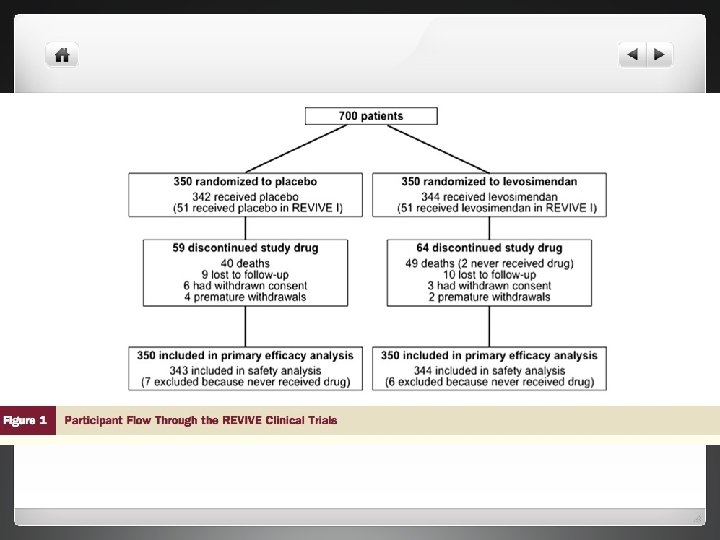
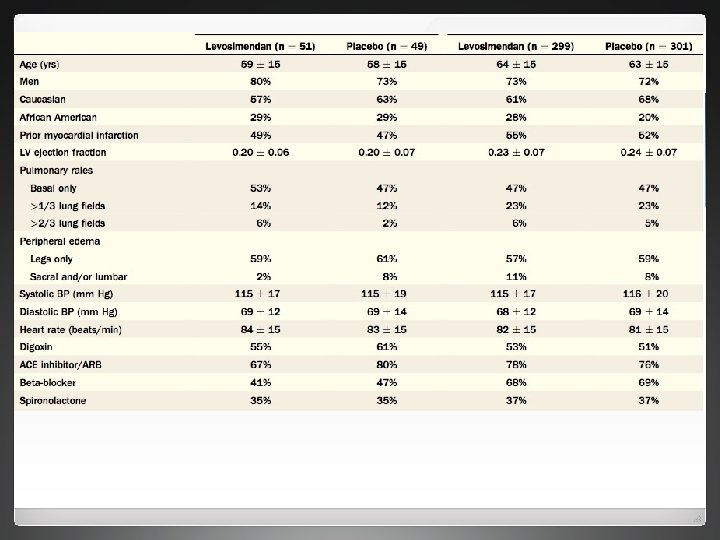
 Methods l REVIVE 1 and 2 carried out in 103 centres in the US, Australia and Israel between December 2001 and December 2004 l All patients with ADHF who remained dyspneic inspite of treatmet with intravenous diuretics
Methods l REVIVE 1 and 2 carried out in 103 centres in the US, Australia and Israel between December 2001 and December 2004 l All patients with ADHF who remained dyspneic inspite of treatmet with intravenous diuretics
 Study Plan l Randomly assigned (double blind) to treatments with placebo or levosimendan which was added to their existing treatment plan l Study endpoints- composite of clinically relevant measures
Study Plan l Randomly assigned (double blind) to treatments with placebo or levosimendan which was added to their existing treatment plan l Study endpoints- composite of clinically relevant measures
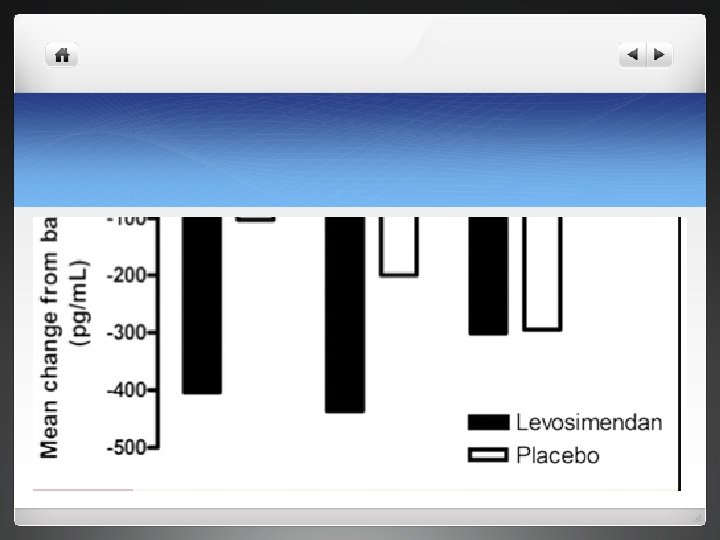
 Outcome measurement l Primary end point in both trials -clinical course during first 5 days characterized as improved , unchanged or worse. l Secondary endpoint- BNP at 24 hrs , changes in global assessment at 6 hrs , changes in perception of dyspnoea at 6 hrs , numer of days alive (1 -14 days after randomization), NHYA functional status at day 5, all cause mortality during first 90 days
Outcome measurement l Primary end point in both trials -clinical course during first 5 days characterized as improved , unchanged or worse. l Secondary endpoint- BNP at 24 hrs , changes in global assessment at 6 hrs , changes in perception of dyspnoea at 6 hrs , numer of days alive (1 -14 days after randomization), NHYA functional status at day 5, all cause mortality during first 90 days
 therapy in revive 1 and 2
therapy in revive 1 and 2
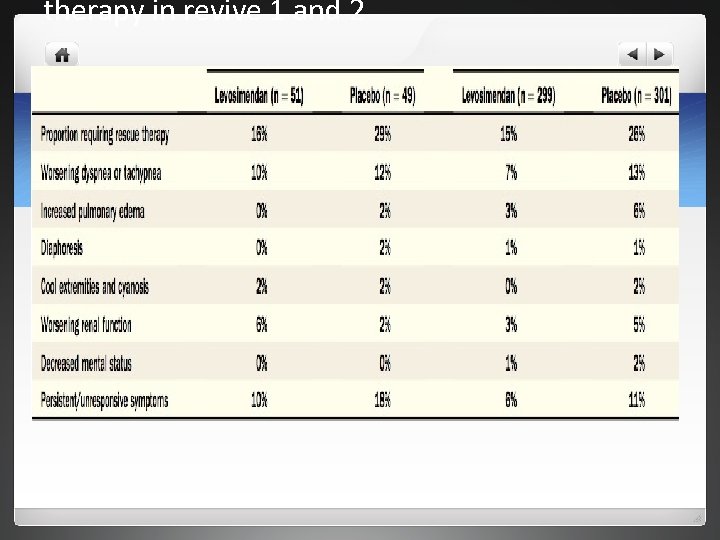
 Results l 12% of levosimendan group and 7% of placebo group discontinued before 24 hrs l Primary endpoints- patients improved on levosimendan compared to placebo l In both groups no differences in groupsin number of days alive over 14 days
Results l 12% of levosimendan group and 7% of placebo group discontinued before 24 hrs l Primary endpoints- patients improved on levosimendan compared to placebo l In both groups no differences in groupsin number of days alive over 14 days
 Results l Levosimendan arm briefer hospital stays-46%vs 37% l NYHA functional status was not significantly different between both groups l Safety- higher number of adverse effects with levosimendan
Results l Levosimendan arm briefer hospital stays-46%vs 37% l NYHA functional status was not significantly different between both groups l Safety- higher number of adverse effects with levosimendan
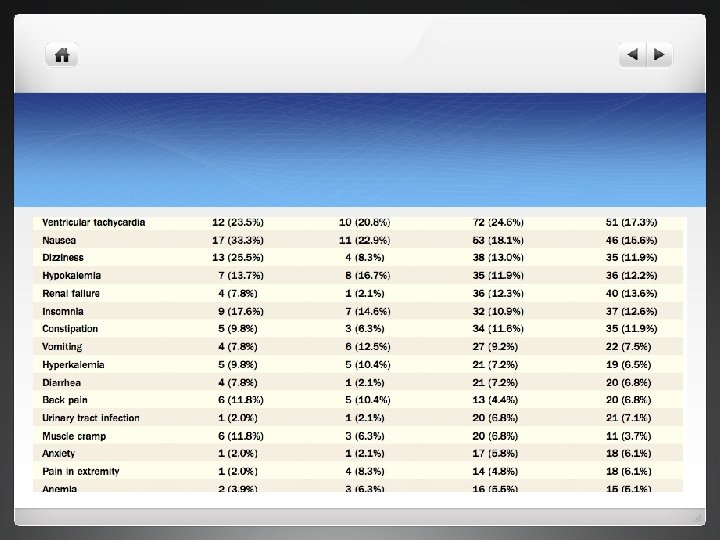
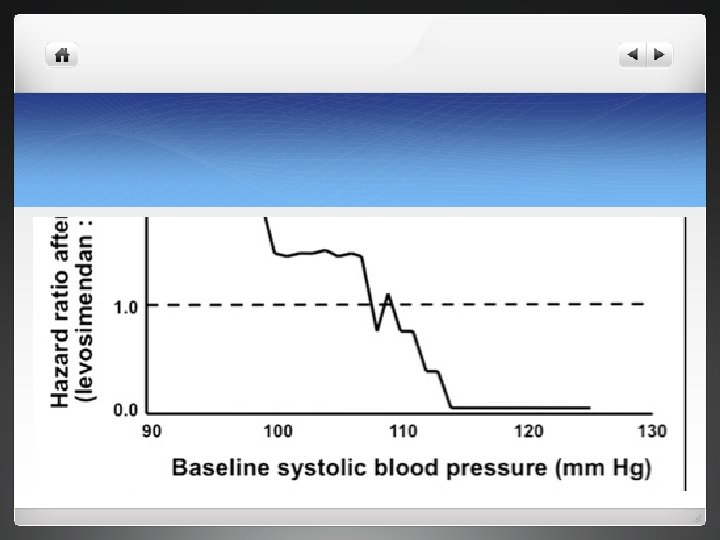
 discussion l Robust study , demonstrates favourable effect on short term clinical course of patients with ADHF l Likely reason for incresed mortality in levosimendan arm due to increased used of loading dose and approach which is no longer followed
discussion l Robust study , demonstrates favourable effect on short term clinical course of patients with ADHF l Likely reason for incresed mortality in levosimendan arm due to increased used of loading dose and approach which is no longer followed
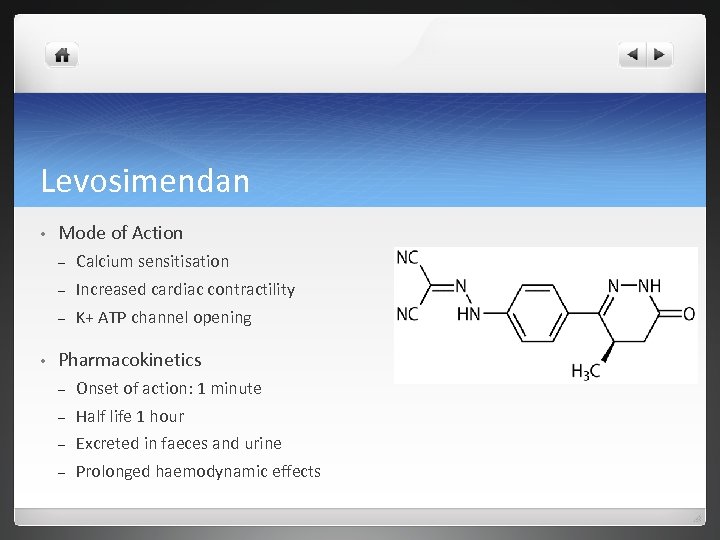 Levosimendan • Mode of Action – – Increased cardiac contractility – • Calcium sensitisation K+ ATP channel opening Pharmacokinetics – Onset of action: 1 minute – Half life 1 hour – Excreted in faeces and urine – Prolonged haemodynamic effects
Levosimendan • Mode of Action – – Increased cardiac contractility – • Calcium sensitisation K+ ATP channel opening Pharmacokinetics – Onset of action: 1 minute – Half life 1 hour – Excreted in faeces and urine – Prolonged haemodynamic effects
 Levosimendan effects…. l Cardioprotective effect l Anti inflammatory effect l Neurohormonal effect l Improves coronary circulation l Antistunning effect l Haemodynamic effects
Levosimendan effects…. l Cardioprotective effect l Anti inflammatory effect l Neurohormonal effect l Improves coronary circulation l Antistunning effect l Haemodynamic effects
 LIDO Study l Multicenter , double blind, double dummy. randomized study l Designed to compare clinical and haematological effects of levosimendan vs Dobutamine in low output heart failure l 203 patients- levosimendan improved haemodynamic performance and decreased mortality for upto 180 days
LIDO Study l Multicenter , double blind, double dummy. randomized study l Designed to compare clinical and haematological effects of levosimendan vs Dobutamine in low output heart failure l 203 patients- levosimendan improved haemodynamic performance and decreased mortality for upto 180 days
 RUSSLAN TRIAL l Double blind placebo controlled l Evaluating different doses of levosimendan vs placebo in patients with heart failure secondary to MI l 504 patients – higher dose , greater side effects l Overall mortality better than placebo group (at 14 days)
RUSSLAN TRIAL l Double blind placebo controlled l Evaluating different doses of levosimendan vs placebo in patients with heart failure secondary to MI l 504 patients – higher dose , greater side effects l Overall mortality better than placebo group (at 14 days)
 CASINO TRIAL l Randomised , double blinded, double dummy and parallel group study l 299 patients NYHA 4 (LVEF less than 35%) l Stopped prematurely – significant survival benefit with levosimendan compared with dobutamine
CASINO TRIAL l Randomised , double blinded, double dummy and parallel group study l 299 patients NYHA 4 (LVEF less than 35%) l Stopped prematurely – significant survival benefit with levosimendan compared with dobutamine
 SURVIVE TRIAL • Randomized , double blind, double dummy, prospective controlled study • 1327 patients (LVEF-less than 35%)- not responding to IV diureticsand vasodilator therapy • Primary endpoint- all cause mortality in 180 days – no statistical difference • 25% lower mortality compared to dobutamine 6 hrs, 24 hrs , 5, 14, 30 days
SURVIVE TRIAL • Randomized , double blind, double dummy, prospective controlled study • 1327 patients (LVEF-less than 35%)- not responding to IV diureticsand vasodilator therapy • Primary endpoint- all cause mortality in 180 days – no statistical difference • 25% lower mortality compared to dobutamine 6 hrs, 24 hrs , 5, 14, 30 days
 Levosimendan Facts !! • Well tolerated • Side effects – headache , hypotension, dizzyness, nausea • Can cause VT , AF at higher doses • Recommended dose 6 -24 μg/kg/min administerted in 10 -20 min and maintainance dose- 0. 05 -0. 2 μg/kg/min over 24 hrs
Levosimendan Facts !! • Well tolerated • Side effects – headache , hypotension, dizzyness, nausea • Can cause VT , AF at higher doses • Recommended dose 6 -24 μg/kg/min administerted in 10 -20 min and maintainance dose- 0. 05 -0. 2 μg/kg/min over 24 hrs
 Facts !! l Not licensed for use in the UK and USA however still used l Not recommended for use in patients with Bp less than 90 systolic l Can be used in conjunction with betablockers, noradrenaline l Can also be used in septic shock (some proven benefit)
Facts !! l Not licensed for use in the UK and USA however still used l Not recommended for use in patients with Bp less than 90 systolic l Can be used in conjunction with betablockers, noradrenaline l Can also be used in septic shock (some proven benefit)
 Discussion l New inodilator agent for therapy in end stage heart failure l Proven benefits compared to dobutamine l Further trials may help clarify its effects on mortality and use in clinical practice
Discussion l New inodilator agent for therapy in end stage heart failure l Proven benefits compared to dobutamine l Further trials may help clarify its effects on mortality and use in clinical practice
 Any Questions ? ? ?
Any Questions ? ? ?
 Thank You !!
Thank You !!


