6baaa6a96210c0522aca58bd5e36c11c.ppt
- Количество слайдов: 17
 EFFECT OF INTRAVENOUS CORTICOSTEROIDS ON DEATH WITHIN 14 DAYS IN 10, 008 ADULTS WITH CLINICALLY SIGNIFICANT HEAD INJURY (MRC CRASH TRIAL): RANDOMISED PLACEBOCONTROLLED TRIAL Dr. Hussein Ssenyonjo Resident, Neurosurgery Mulago Hospital
EFFECT OF INTRAVENOUS CORTICOSTEROIDS ON DEATH WITHIN 14 DAYS IN 10, 008 ADULTS WITH CLINICALLY SIGNIFICANT HEAD INJURY (MRC CRASH TRIAL): RANDOMISED PLACEBOCONTROLLED TRIAL Dr. Hussein Ssenyonjo Resident, Neurosurgery Mulago Hospital
 Summary n Cortical steroids have been used to treat head injuries for more than 30 years. In 1997, findings after systematic review suggested that these drugs reduce the risk of death by 1 -2%. The CRASH trial- a muliticentre international collaboration aimed to confirm or refute such an effect by recruiting 20, 000 patients. In May, 2004, the data monitoring committee disclosed the unmasked results to the steering committee, which stopped recruitment.
Summary n Cortical steroids have been used to treat head injuries for more than 30 years. In 1997, findings after systematic review suggested that these drugs reduce the risk of death by 1 -2%. The CRASH trial- a muliticentre international collaboration aimed to confirm or refute such an effect by recruiting 20, 000 patients. In May, 2004, the data monitoring committee disclosed the unmasked results to the steering committee, which stopped recruitment.
 Patients and methods n Adults 16 yrs and above who presented within 8 hrs of injury and were noted in hospital to be having GCS of 14 and less. These were recruited if the treating doctor was uncertain whether or not to use steroids-(UNCERTANITY PRINCIPLE)
Patients and methods n Adults 16 yrs and above who presented within 8 hrs of injury and were noted in hospital to be having GCS of 14 and less. These were recruited if the treating doctor was uncertain whether or not to use steroids-(UNCERTANITY PRINCIPLE)
 Procedures n Patients randomised in one of two ways. 1. reliable telephone access 2. Local pack system n We randomly allocated patients to 48 h infusion of either methylprednisolone or placebo. Loading dose 2 g over 1 h in 100 mls of infusion. Maintenance dose 0. 4 g/h for 48 h at rate of 20 ml/h. n Primary outcome measures were death from any cause within 2 weeks of injury and death or disability at 6 months.
Procedures n Patients randomised in one of two ways. 1. reliable telephone access 2. Local pack system n We randomly allocated patients to 48 h infusion of either methylprednisolone or placebo. Loading dose 2 g over 1 h in 100 mls of infusion. Maintenance dose 0. 4 g/h for 48 h at rate of 20 ml/h. n Primary outcome measures were death from any cause within 2 weeks of injury and death or disability at 6 months.
 Statistical analysis n n n Estimated risk of death allocated to placebo – 15%. 2% survival difference would be clinically important so the trial size had to be large enough to detect a difference of this size. A trial of 20, 000 patients would have a good chance of showing a 2% survival difference at convincing levels of significance ie >90% power to achieve p<0. 01 Precision-95%CI for overall risk and 99%CI for subgroup results Homogenicity in treatment effects assessed within subgroups by X 2 test at 5% significance level.
Statistical analysis n n n Estimated risk of death allocated to placebo – 15%. 2% survival difference would be clinically important so the trial size had to be large enough to detect a difference of this size. A trial of 20, 000 patients would have a good chance of showing a 2% survival difference at convincing levels of significance ie >90% power to achieve p<0. 01 Precision-95%CI for overall risk and 99%CI for subgroup results Homogenicity in treatment effects assessed within subgroups by X 2 test at 5% significance level.
 Results n n n 239 hospitals from 49 countries. 10, 008 patients randomised to steroid or placebo infusion 62 <16 years, 21 enrolled > 8 h after injury, 3 stopped at request of a relative.
Results n n n 239 hospitals from 49 countries. 10, 008 patients randomised to steroid or placebo infusion 62 <16 years, 21 enrolled > 8 h after injury, 3 stopped at request of a relative.
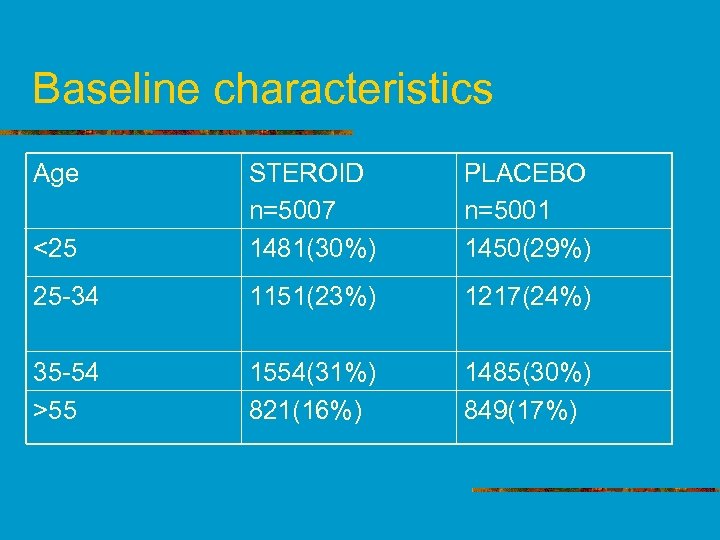 Baseline characteristics Age <25 STEROID n=5007 1481(30%) PLACEBO n=5001 1450(29%) 25 -34 1151(23%) 1217(24%) 35 -54 >55 1554(31%) 821(16%) 1485(30%) 849(17%)
Baseline characteristics Age <25 STEROID n=5007 1481(30%) PLACEBO n=5001 1450(29%) 25 -34 1151(23%) 1217(24%) 35 -54 >55 1554(31%) 821(16%) 1485(30%) 849(17%)
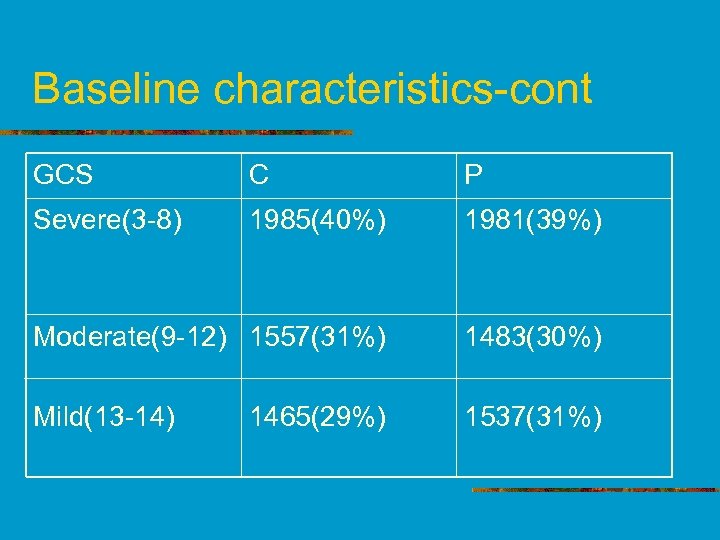 Baseline characteristics-cont GCS C P Severe(3 -8) 1985(40%) 1981(39%) Moderate(9 -12) 1557(31%) 1483(30%) Mild(13 -14) 1537(31%) 1465(29%)
Baseline characteristics-cont GCS C P Severe(3 -8) 1985(40%) 1981(39%) Moderate(9 -12) 1557(31%) 1483(30%) Mild(13 -14) 1537(31%) 1465(29%)
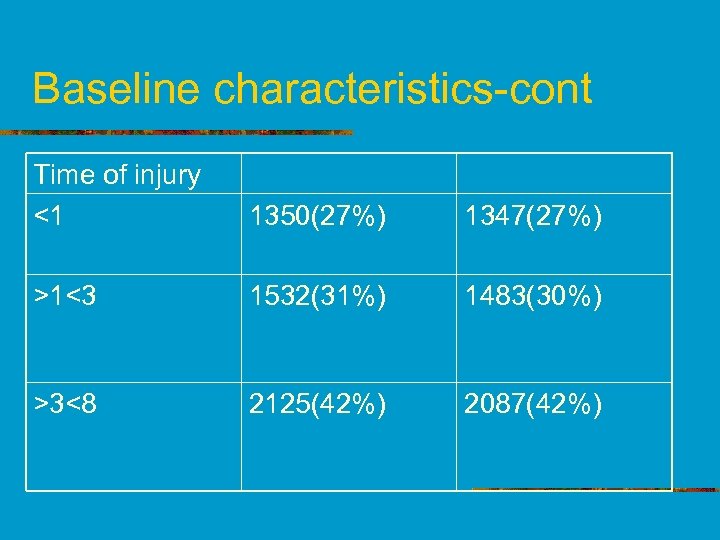 Baseline characteristics-cont Time of injury <1 1350(27%) 1347(27%) >1<3 1532(31%) 1483(30%) >3<8 2125(42%) 2087(42%)
Baseline characteristics-cont Time of injury <1 1350(27%) 1347(27%) >1<3 1532(31%) 1483(30%) >3<8 2125(42%) 2087(42%)
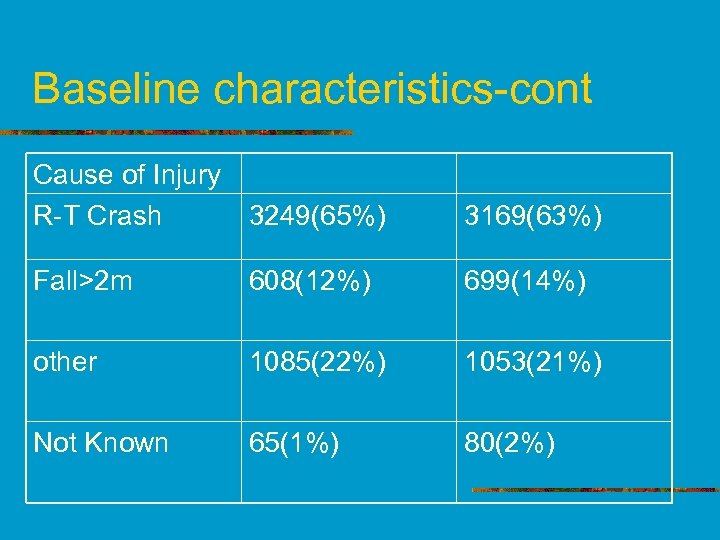 Baseline characteristics-cont Cause of Injury R-T Crash 3249(65%) 3169(63%) Fall>2 m 608(12%) 699(14%) other 1085(22%) 1053(21%) Not Known 65(1%) 80(2%)
Baseline characteristics-cont Cause of Injury R-T Crash 3249(65%) 3169(63%) Fall>2 m 608(12%) 699(14%) other 1085(22%) 1053(21%) Not Known 65(1%) 80(2%)
 Baseline characteristics-cont Brain CT Scan Done Yes 3, 916(78%) 3, 896(78%) No/Not Known 1, 091(22%) 1, 105(22%)
Baseline characteristics-cont Brain CT Scan Done Yes 3, 916(78%) 3, 896(78%) No/Not Known 1, 091(22%) 1, 105(22%)
 Baseline characteristics-cont CT Scan Results Normal 1 petechial Hrrge C 23% 29% P 22% 28% 3 rd V and basal cistern obliteration 23% 24% SAH 31% 32% Shift>5 14% 15% Non evacuated haematoma 27% Evacuated H 12% 13% Cortical contusion > 1 cm 22% 23%
Baseline characteristics-cont CT Scan Results Normal 1 petechial Hrrge C 23% 29% P 22% 28% 3 rd V and basal cistern obliteration 23% 24% SAH 31% 32% Shift>5 14% 15% Non evacuated haematoma 27% Evacuated H 12% 13% Cortical contusion > 1 cm 22% 23%
 Baseline characteristics-cont n n n Adherence to treatment-9, 848(98%) 9, 748(99%)received full loading dose. Mortality data obtained for 9, 964 patients, 4, 985 CS M-1. 052(21%) in 2 wks, 4, 979 -P M 893(18%) RR of death from all causes in 2 wks in CS group with placebo 1. 18 RR of death at 2 wks didn’t differ by injury severity (p=0. 22) or time since injury(p=0. 05)
Baseline characteristics-cont n n n Adherence to treatment-9, 848(98%) 9, 748(99%)received full loading dose. Mortality data obtained for 9, 964 patients, 4, 985 CS M-1. 052(21%) in 2 wks, 4, 979 -P M 893(18%) RR of death from all causes in 2 wks in CS group with placebo 1. 18 RR of death at 2 wks didn’t differ by injury severity (p=0. 22) or time since injury(p=0. 05)
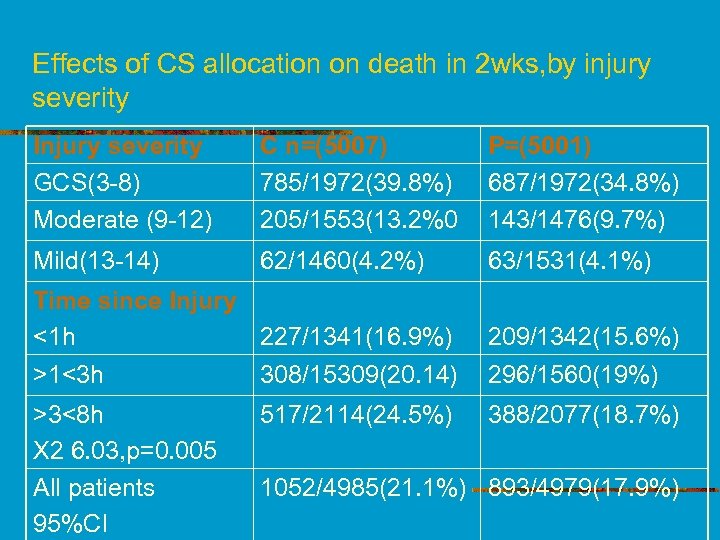 Effects of CS allocation on death in 2 wks, by injury severity Injury severity GCS(3 -8) Moderate (9 -12) C n=(5007) 785/1972(39. 8%) 205/1553(13. 2%0 P=(5001) 687/1972(34. 8%) 143/1476(9. 7%) Mild(13 -14) 62/1460(4. 2%) 63/1531(4. 1%) Time since Injury <1 h 227/1341(16. 9%) >1<3 h 308/15309(20. 14) >3<8 h X 2 6. 03, p=0. 005 All patients 95%CI 517/2114(24. 5%) 209/1342(15. 6%) 296/1560(19%) 388/2077(18. 7%) 1052/4985(21. 1%) 893/4979(17. 9%)
Effects of CS allocation on death in 2 wks, by injury severity Injury severity GCS(3 -8) Moderate (9 -12) C n=(5007) 785/1972(39. 8%) 205/1553(13. 2%0 P=(5001) 687/1972(34. 8%) 143/1476(9. 7%) Mild(13 -14) 62/1460(4. 2%) 63/1531(4. 1%) Time since Injury <1 h 227/1341(16. 9%) >1<3 h 308/15309(20. 14) >3<8 h X 2 6. 03, p=0. 005 All patients 95%CI 517/2114(24. 5%) 209/1342(15. 6%) 296/1560(19%) 388/2077(18. 7%) 1052/4985(21. 1%) 893/4979(17. 9%)
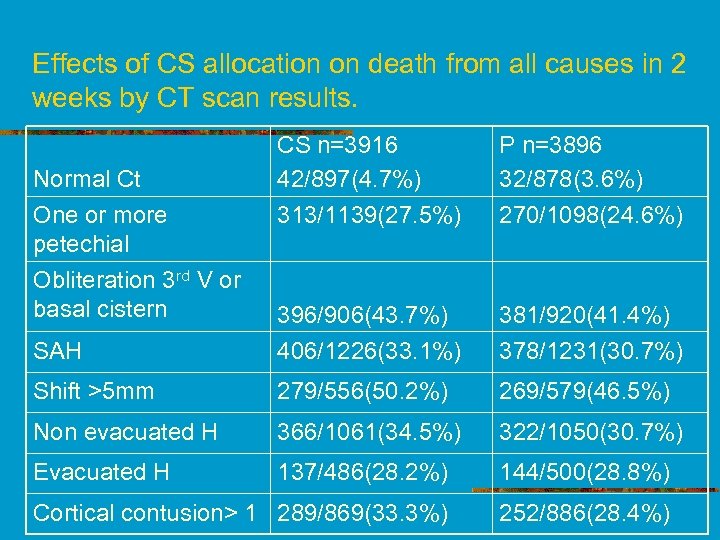 Effects of CS allocation on death from all causes in 2 weeks by CT scan results. CS n=3916 42/897(4. 7%) 313/1139(27. 5%) P n=3896 32/878(3. 6%) 270/1098(24. 6%) SAH 396/906(43. 7%) 406/1226(33. 1%) 381/920(41. 4%) 378/1231(30. 7%) Shift >5 mm 279/556(50. 2%) 269/579(46. 5%) Non evacuated H 366/1061(34. 5%) 322/1050(30. 7%) Evacuated H 137/486(28. 2%) 144/500(28. 8%) Cortical contusion> 1 289/869(33. 3%) 252/886(28. 4%) Normal Ct One or more petechial Obliteration 3 rd V or basal cistern
Effects of CS allocation on death from all causes in 2 weeks by CT scan results. CS n=3916 42/897(4. 7%) 313/1139(27. 5%) P n=3896 32/878(3. 6%) 270/1098(24. 6%) SAH 396/906(43. 7%) 406/1226(33. 1%) 381/920(41. 4%) 378/1231(30. 7%) Shift >5 mm 279/556(50. 2%) 269/579(46. 5%) Non evacuated H 366/1061(34. 5%) 322/1050(30. 7%) Evacuated H 137/486(28. 2%) 144/500(28. 8%) Cortical contusion> 1 289/869(33. 3%) 252/886(28. 4%) Normal Ct One or more petechial Obliteration 3 rd V or basal cistern
 Comment on results n Relative Risk of death at 2 wks wasn’t different in any of the 8 Ct diagnosis subgroups examined
Comment on results n Relative Risk of death at 2 wks wasn’t different in any of the 8 Ct diagnosis subgroups examined
 Conclusion Corticosteroids should not be used routinely to use had injury whatever the severity.
Conclusion Corticosteroids should not be used routinely to use had injury whatever the severity.


