f3c767d8bdcc61902889d81637c5d2d5.ppt
- Количество слайдов: 84
 ECMO FOR ARDS: Does high Frequency Ventilation Make a Difference? Jeff Heltborg RRT Respiratory Clinical Specialist
ECMO FOR ARDS: Does high Frequency Ventilation Make a Difference? Jeff Heltborg RRT Respiratory Clinical Specialist
 Objectives 1. Review the current definition for ARDS. 2. To describe ARDS patients that may qualify for ECMO 3. To identify different High Frequency ventilation devices 4. Explore current ECMO literature and techniques.
Objectives 1. Review the current definition for ARDS. 2. To describe ARDS patients that may qualify for ECMO 3. To identify different High Frequency ventilation devices 4. Explore current ECMO literature and techniques.
 ARDSnet findings: 2000 Ø Completely changed current day treatment for ARDS/ALI by: Ø Instituting Vt of 4 -6 ml/kg based on IBW Ø PEEP level’s based on fio 2 requirement Ø Inclusion data included: Ø P/F ratio <300 Ø Bilateral infiltrates on CXR Ø No L sided heart failure as explanation for pulmonary edema
ARDSnet findings: 2000 Ø Completely changed current day treatment for ARDS/ALI by: Ø Instituting Vt of 4 -6 ml/kg based on IBW Ø PEEP level’s based on fio 2 requirement Ø Inclusion data included: Ø P/F ratio <300 Ø Bilateral infiltrates on CXR Ø No L sided heart failure as explanation for pulmonary edema
 The Berlin definition says:
The Berlin definition says:
 P/F ratios Ø Who are the patients at risk for severe hypoxemic respiratory failure? Ø Ø Ø Mild ARDS Pa. O 2/FIO 2 ≤ 300 Moderate RDS Pa. O 2/FIO 2 ≤ 200 Severe hypoxemic respiratory failure Pa. O 2/FIO 2 ≤ 100
P/F ratios Ø Who are the patients at risk for severe hypoxemic respiratory failure? Ø Ø Ø Mild ARDS Pa. O 2/FIO 2 ≤ 300 Moderate RDS Pa. O 2/FIO 2 ≤ 200 Severe hypoxemic respiratory failure Pa. O 2/FIO 2 ≤ 100

 Direct ARDS Ø Pneumonia Ø Pulmonary contusion Ø Inhalation
Direct ARDS Ø Pneumonia Ø Pulmonary contusion Ø Inhalation
 Indirect ARDS : The inflammatory cascade Ø SIRS/Sepsis Ø Trauma/shock Ø This inflammatory process incites alveolar injury Ø Capillary permeability Ø Surfactant deactivation Ø Alveolar flooding
Indirect ARDS : The inflammatory cascade Ø SIRS/Sepsis Ø Trauma/shock Ø This inflammatory process incites alveolar injury Ø Capillary permeability Ø Surfactant deactivation Ø Alveolar flooding
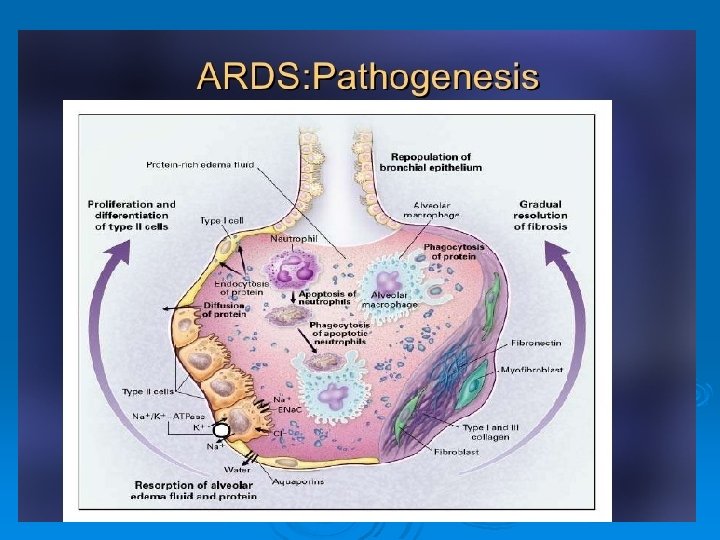
 What is severe hypoxemic respiratory failure? Acute onset of severe respiratory distress resulting in the need for: High oxygenation – FIO 2 ≥ 70% or Oxygenation Index>30 ventilator requirements – PEEP > 15 cm. H 2 O or Plateau pressure >30 cm. H 2 O with a tidal volume < 6 m. L/kg IBW Synonymous with severe ARDS Pa. O 2/FIO 2 ≤ 100 mm. Hg
What is severe hypoxemic respiratory failure? Acute onset of severe respiratory distress resulting in the need for: High oxygenation – FIO 2 ≥ 70% or Oxygenation Index>30 ventilator requirements – PEEP > 15 cm. H 2 O or Plateau pressure >30 cm. H 2 O with a tidal volume < 6 m. L/kg IBW Synonymous with severe ARDS Pa. O 2/FIO 2 ≤ 100 mm. Hg
 Approximately 16% of deaths in patients with ARDS results from severe hypoxemic respiratory failure. 1 l l l 1. 2. 3. 4. Degree of respiratory failure may be a direct predictor of poor prognosis. 2 ARDS patients on lung-protective mechanical ventilation who improve in oxygenation and disease severity in 24 h 3, 4 • 13 – 23% mortality ARDS patients who don’t improve in Pa. O 2/FIO 2 ratio in the first 24 h of lung-protective ventilation 3, 4 • 53 – 68% mortality Montgomery AB, et al. Am Rev Respir Dis 1985; 132: 485 -489 Luhr OR, et al. Am J Respir Crit Care Med 1999; 159: 1849 -186 Villar J, et al. Intensive Care Med. 1999; 25 (9): 930 -935 Ferguson ND, et al. Intensive Care Med. 2004; 30 (6): 1111 -1116
Approximately 16% of deaths in patients with ARDS results from severe hypoxemic respiratory failure. 1 l l l 1. 2. 3. 4. Degree of respiratory failure may be a direct predictor of poor prognosis. 2 ARDS patients on lung-protective mechanical ventilation who improve in oxygenation and disease severity in 24 h 3, 4 • 13 – 23% mortality ARDS patients who don’t improve in Pa. O 2/FIO 2 ratio in the first 24 h of lung-protective ventilation 3, 4 • 53 – 68% mortality Montgomery AB, et al. Am Rev Respir Dis 1985; 132: 485 -489 Luhr OR, et al. Am J Respir Crit Care Med 1999; 159: 1849 -186 Villar J, et al. Intensive Care Med. 1999; 25 (9): 930 -935 Ferguson ND, et al. Intensive Care Med. 2004; 30 (6): 1111 -1116
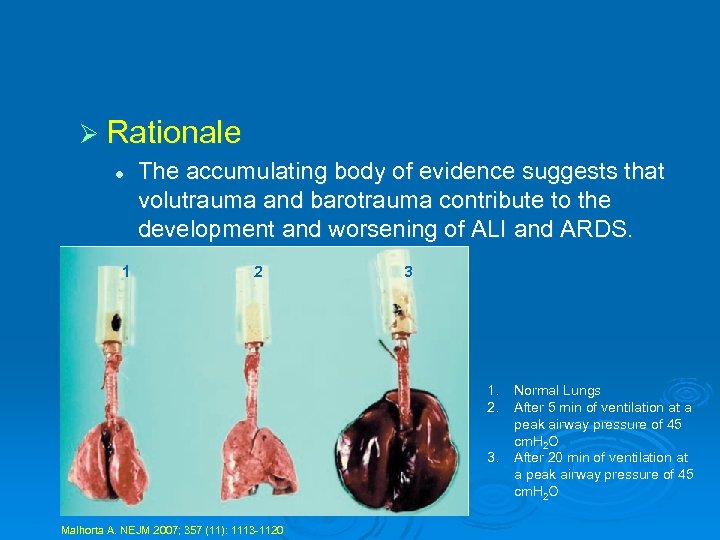 Ø Rationale l 1 The accumulating body of evidence suggests that volutrauma and barotrauma contribute to the development and worsening of ALI and ARDS. 2 3 1. 2. 3. Malhorta A. NEJM 2007; 357 (11): 1113 -1120 Normal Lungs After 5 min of ventilation at a peak airway pressure of 45 cm. H 2 O After 20 min of ventilation at a peak airway pressure of 45 cm. H 2 O
Ø Rationale l 1 The accumulating body of evidence suggests that volutrauma and barotrauma contribute to the development and worsening of ALI and ARDS. 2 3 1. 2. 3. Malhorta A. NEJM 2007; 357 (11): 1113 -1120 Normal Lungs After 5 min of ventilation at a peak airway pressure of 45 cm. H 2 O After 20 min of ventilation at a peak airway pressure of 45 cm. H 2 O
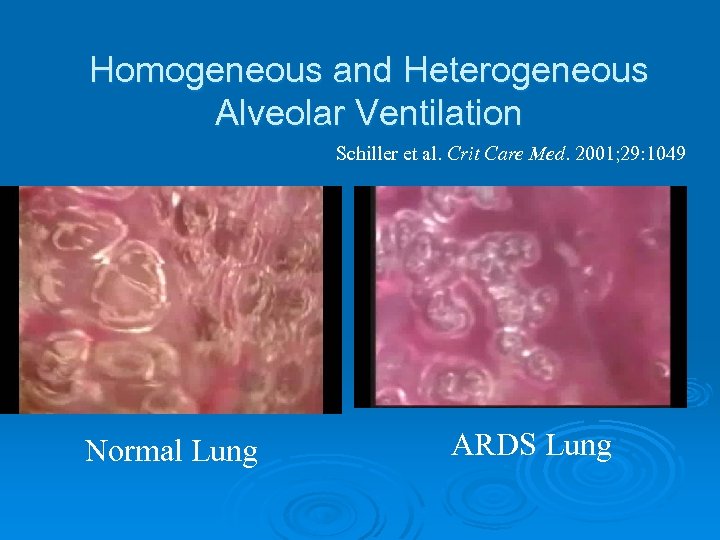 Homogeneous and Heterogeneous Alveolar Ventilation Schiller et al. Crit Care Med. 2001; 29: 1049 Normal Lung ARDS Lung
Homogeneous and Heterogeneous Alveolar Ventilation Schiller et al. Crit Care Med. 2001; 29: 1049 Normal Lung ARDS Lung
 MECHANICAL VENTILATION AND ALI/ARDS ◊ BECAUSE LUNG INJURY IS NON-HOMOGENOUS, VENTILATION IS DISTRIBUTED UNEVENLY. ◊ VENTILATION IS DISTRIBUTED TO THE REGIONS OF GREATEST COMPLIANCE AND THE LEAST RESISTANCE OR THE PREFERENTIAL AIRWAY. WHEN THIS MAL-DISTRIBUTION OF VENTILATION DOES FURTHER LUNG DAMAGE THIS IS KNOWN AS (VILI).
MECHANICAL VENTILATION AND ALI/ARDS ◊ BECAUSE LUNG INJURY IS NON-HOMOGENOUS, VENTILATION IS DISTRIBUTED UNEVENLY. ◊ VENTILATION IS DISTRIBUTED TO THE REGIONS OF GREATEST COMPLIANCE AND THE LEAST RESISTANCE OR THE PREFERENTIAL AIRWAY. WHEN THIS MAL-DISTRIBUTION OF VENTILATION DOES FURTHER LUNG DAMAGE THIS IS KNOWN AS (VILI).
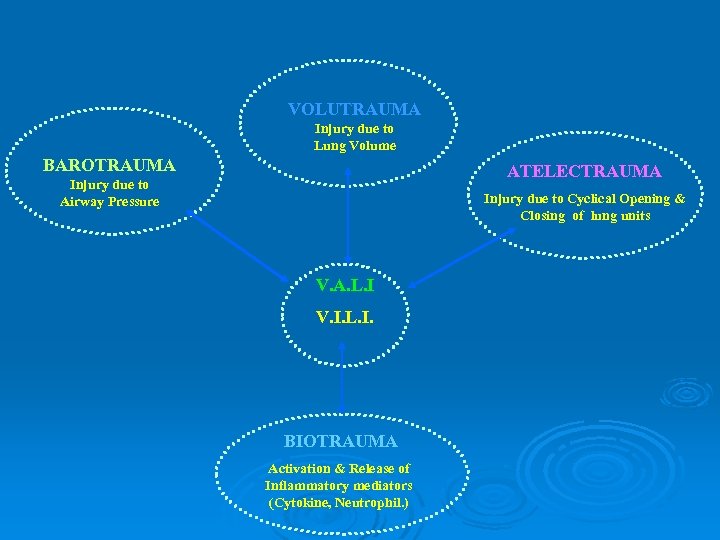 VOLUTRAUMA Injury due to Lung Volume BAROTRAUMA ATELECTRAUMA Injury due to Airway Pressure Injury due to Cyclical Opening & Closing of lung units V. A. L. I V. I. L. I. BIOTRAUMA Activation & Release of Inflammatory mediators (Cytokine, Neutrophil. )
VOLUTRAUMA Injury due to Lung Volume BAROTRAUMA ATELECTRAUMA Injury due to Airway Pressure Injury due to Cyclical Opening & Closing of lung units V. A. L. I V. I. L. I. BIOTRAUMA Activation & Release of Inflammatory mediators (Cytokine, Neutrophil. )
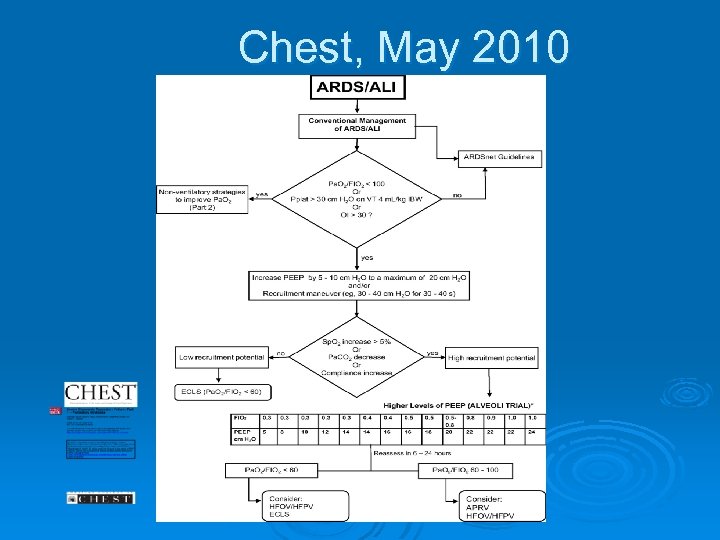 Chest, May 2010
Chest, May 2010
 Ventilator strategies in severe hypoxemic respiratory failure A. Esan, D. Hess, C. Sessler, L. George, C. Oribabor, F. Khusid and S. Raoof Summary Mechanical ventilation of patients with acute lung injury/acute respiratory distress syndrome (ARDS) should commence with low tidal volume (VT), low stretch and adequate positive endexpiratory pressure (PEEP), as proposed by the first ARDSnet trial. The majority of patients with ARDS will achieve their goals of oxygenation and plateau pressure, utilizing the lung protective strategy. In the remaining minority of patients, these end-points may not be achieved. Such patients have a significantly high mortality and should be considered for rescue strategies relatively early on. If the patients respond positively to lung recruitment trials, using rescue strategies may open atelectactic alveoli and allow oxygenation or plateau pressure targets to be achieved. None of these rescue strategies have been shown to reduce mortality, although shortterm objectives of improvement in oxygenation or reduction in plateau pressures may be achieved. Therefore, the selection of these strategies should be based on availability and level of comfort of the operators. European Respiratory Society Monographs 2012; 55 (New Developments in Mechanical Ventilation): 19 -39
Ventilator strategies in severe hypoxemic respiratory failure A. Esan, D. Hess, C. Sessler, L. George, C. Oribabor, F. Khusid and S. Raoof Summary Mechanical ventilation of patients with acute lung injury/acute respiratory distress syndrome (ARDS) should commence with low tidal volume (VT), low stretch and adequate positive endexpiratory pressure (PEEP), as proposed by the first ARDSnet trial. The majority of patients with ARDS will achieve their goals of oxygenation and plateau pressure, utilizing the lung protective strategy. In the remaining minority of patients, these end-points may not be achieved. Such patients have a significantly high mortality and should be considered for rescue strategies relatively early on. If the patients respond positively to lung recruitment trials, using rescue strategies may open atelectactic alveoli and allow oxygenation or plateau pressure targets to be achieved. None of these rescue strategies have been shown to reduce mortality, although shortterm objectives of improvement in oxygenation or reduction in plateau pressures may be achieved. Therefore, the selection of these strategies should be based on availability and level of comfort of the operators. European Respiratory Society Monographs 2012; 55 (New Developments in Mechanical Ventilation): 19 -39
 No definitive conclusions can be made about the role of HFPV/HFOV in the ventilatory management of ALI / ARDS patients. Oxygenation and ventilation improve at lower airway pressures in comparison to conventional ventilation; however, no mortality benefit has been demonstrated to date. Like the other alternative / rescue strategies that have been described, large RCTs are needed to accurately elucidate their role in the ventilatory management of ALI / ARDS patients. European Respiratory Society Monographs 2012; 55 (New Developments in Mechanical Ventilation): 19 -39
No definitive conclusions can be made about the role of HFPV/HFOV in the ventilatory management of ALI / ARDS patients. Oxygenation and ventilation improve at lower airway pressures in comparison to conventional ventilation; however, no mortality benefit has been demonstrated to date. Like the other alternative / rescue strategies that have been described, large RCTs are needed to accurately elucidate their role in the ventilatory management of ALI / ARDS patients. European Respiratory Society Monographs 2012; 55 (New Developments in Mechanical Ventilation): 19 -39
 What about treatment strategies before ECMO Ø 1. ARDS network Ø 2. Proning Ø 3. APRV / Bi-level Ø 4. Delivery of nitric oxide or an inhaled prostacyclin (flolan, epoprostenol). Ø 5. high frequency ventilation Ø HFOV Ø HFPV
What about treatment strategies before ECMO Ø 1. ARDS network Ø 2. Proning Ø 3. APRV / Bi-level Ø 4. Delivery of nitric oxide or an inhaled prostacyclin (flolan, epoprostenol). Ø 5. high frequency ventilation Ø HFOV Ø HFPV
 We have discussed the ARDS Network
We have discussed the ARDS Network
 Lets start with proning
Lets start with proning
 Positional Therapy Strategies for Skin Protection
Positional Therapy Strategies for Skin Protection
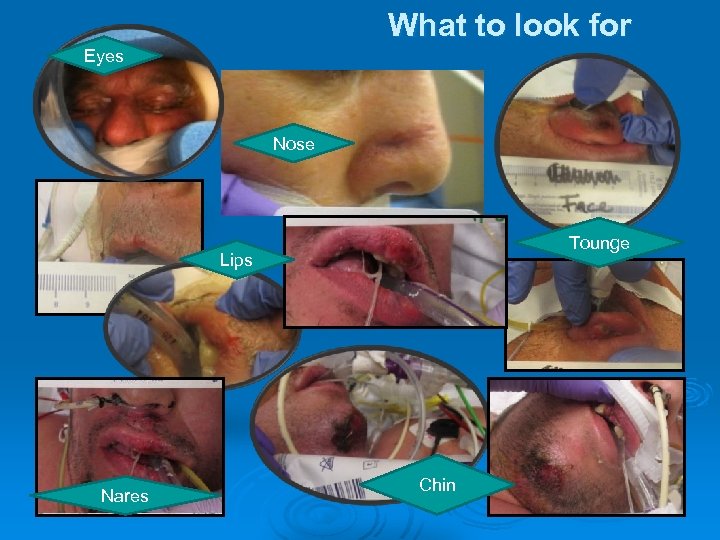 What to look for Eyes Nose Tounge Lips Nares Chin
What to look for Eyes Nose Tounge Lips Nares Chin
 APRV Defined APRV: Airway Pressure Release Ventilation A variant of CPAP where the patient spontaneously breathes at an elevated baseline pressure, and ventilation is augmented by briefly allowing pressure to fall to a lower level (0) and rapidly return to baseline.
APRV Defined APRV: Airway Pressure Release Ventilation A variant of CPAP where the patient spontaneously breathes at an elevated baseline pressure, and ventilation is augmented by briefly allowing pressure to fall to a lower level (0) and rapidly return to baseline.
 APRV When properly applied, APRV decreases intratidal alveolar collapse Recruitment Zone Derecruitment Zone Syring RS, Otto CM, Spivack RE, Markstaller K, Baumgardner JE: Maintenance of End-expiratory Recruitment with Increased Respiratory Rate after Saline-lavage Lung Injury. J Appl Physiol 102: 331– 339, 2007
APRV When properly applied, APRV decreases intratidal alveolar collapse Recruitment Zone Derecruitment Zone Syring RS, Otto CM, Spivack RE, Markstaller K, Baumgardner JE: Maintenance of End-expiratory Recruitment with Increased Respiratory Rate after Saline-lavage Lung Injury. J Appl Physiol 102: 331– 339, 2007
 APRV Goals Ø To fully recruit the lung and to keep it open. l l l Decreasing injury from intratidal alveolar recruitment and de-recruitment (low lung volume injury) Prevent overdistension and stretch from high inspiratory pressures (high lung volume injury) Utilize the patients spontaneous breathing efforts to assist in recruitment
APRV Goals Ø To fully recruit the lung and to keep it open. l l l Decreasing injury from intratidal alveolar recruitment and de-recruitment (low lung volume injury) Prevent overdistension and stretch from high inspiratory pressures (high lung volume injury) Utilize the patients spontaneous breathing efforts to assist in recruitment
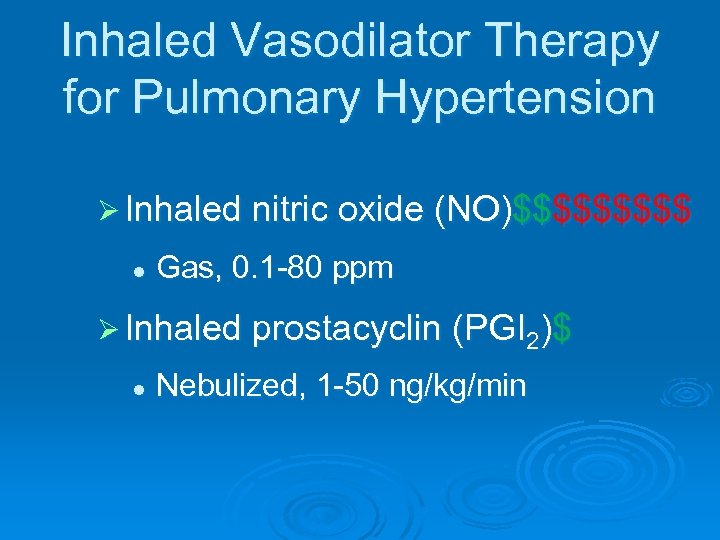 Inhaled Vasodilator Therapy for Pulmonary Hypertension Ø Inhaled nitric oxide (NO)$$$$$ l Gas, 0. 1 -80 ppm Ø Inhaled prostacyclin (PGI 2)$ l Nebulized, 1 -50 ng/kg/min
Inhaled Vasodilator Therapy for Pulmonary Hypertension Ø Inhaled nitric oxide (NO)$$$$$ l Gas, 0. 1 -80 ppm Ø Inhaled prostacyclin (PGI 2)$ l Nebulized, 1 -50 ng/kg/min
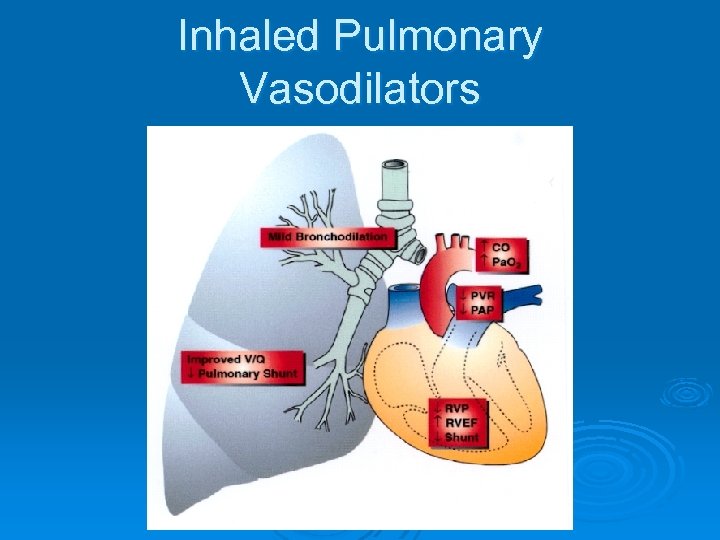 Inhaled Pulmonary Vasodilators
Inhaled Pulmonary Vasodilators
 Inhaled Pulmonary Vasodilators Ø Selective pulmonary vasodilation- PVR, while maintaining MAP and coronary perfusion pressure Ø May improve oxygenation in certain patients- Vasodilation is limited to ventilated areas
Inhaled Pulmonary Vasodilators Ø Selective pulmonary vasodilation- PVR, while maintaining MAP and coronary perfusion pressure Ø May improve oxygenation in certain patients- Vasodilation is limited to ventilated areas
 High Frequency Ventilation 3100 B VDR-4
High Frequency Ventilation 3100 B VDR-4
 What is Considered HF Ø Following the 1976 Medical Device Act FDA called anything over 150 BPM “High Frequency” Ø No definition about HOW the “High Frequency” is created or delivered.
What is Considered HF Ø Following the 1976 Medical Device Act FDA called anything over 150 BPM “High Frequency” Ø No definition about HOW the “High Frequency” is created or delivered.
 HFV Goal Ø The PRIMARY goal is to manage patients who are at a risk of Ventilator Induced Lung Injury (VILI) when conventional methods of ventilation require higher PIP’s, Fi. O 2, PEEP and higher respiratory rates.
HFV Goal Ø The PRIMARY goal is to manage patients who are at a risk of Ventilator Induced Lung Injury (VILI) when conventional methods of ventilation require higher PIP’s, Fi. O 2, PEEP and higher respiratory rates.
 That Means That HFV Are: Ø Able to ventilate at low “pressure-costs” and “volume-costs” while still managing to provide adequate gas exchange which is to say: l Adequate ventilation and oxygenation using low airway pressures and tidal volumes that are smaller than anatomic dead space!
That Means That HFV Are: Ø Able to ventilate at low “pressure-costs” and “volume-costs” while still managing to provide adequate gas exchange which is to say: l Adequate ventilation and oxygenation using low airway pressures and tidal volumes that are smaller than anatomic dead space!
 HF Classifications Ø VDR 4 is unclassified (no restrictions) l l Can be used on any patient age Can be used with any patient weight Ø Oscillator is classified (with restrictions) l l 3100 A on patients < 35 kg and in respiratory Failure( only in NICU) 3100 B on patients > 35 kg
HF Classifications Ø VDR 4 is unclassified (no restrictions) l l Can be used on any patient age Can be used with any patient weight Ø Oscillator is classified (with restrictions) l l 3100 A on patients < 35 kg and in respiratory Failure( only in NICU) 3100 B on patients > 35 kg
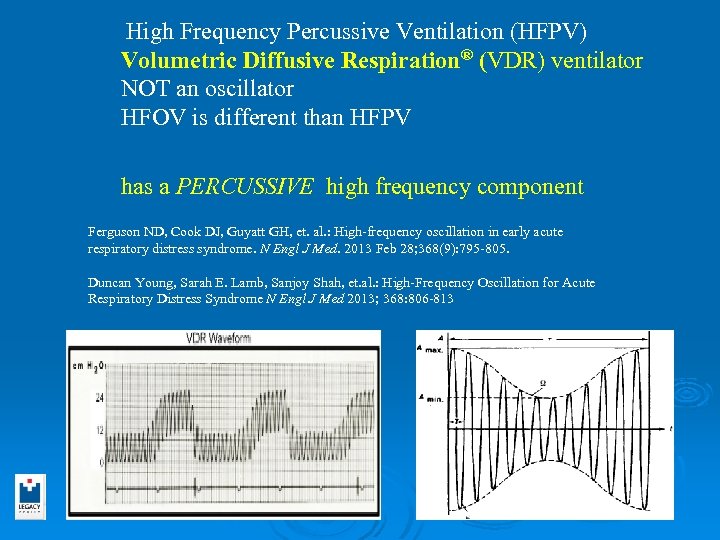 High Frequency Percussive Ventilation (HFPV) Volumetric Diffusive Respiration® (VDR) ventilator NOT an oscillator HFOV is different than HFPV has a PERCUSSIVE high frequency component Ferguson ND, Cook DJ, Guyatt GH, et. al. : High-frequency oscillation in early acute respiratory distress syndrome. N Engl J Med. 2013 Feb 28; 368(9): 795 -805. Duncan Young, Sarah E. Lamb, Sanjoy Shah, et. al. : High-Frequency Oscillation for Acute Respiratory Distress Syndrome N Engl J Med 2013; 368: 806 -813
High Frequency Percussive Ventilation (HFPV) Volumetric Diffusive Respiration® (VDR) ventilator NOT an oscillator HFOV is different than HFPV has a PERCUSSIVE high frequency component Ferguson ND, Cook DJ, Guyatt GH, et. al. : High-frequency oscillation in early acute respiratory distress syndrome. N Engl J Med. 2013 Feb 28; 368(9): 795 -805. Duncan Young, Sarah E. Lamb, Sanjoy Shah, et. al. : High-Frequency Oscillation for Acute Respiratory Distress Syndrome N Engl J Med 2013; 368: 806 -813
 Volume Measurement with HFV Ø Oscillator l Ø Estimated max volume per “burst” is approx 30 -40 cc’s VDR 4 l Estimated volume per “burst” ranges from approx 2050 cc’s
Volume Measurement with HFV Ø Oscillator l Ø Estimated max volume per “burst” is approx 30 -40 cc’s VDR 4 l Estimated volume per “burst” ranges from approx 2050 cc’s
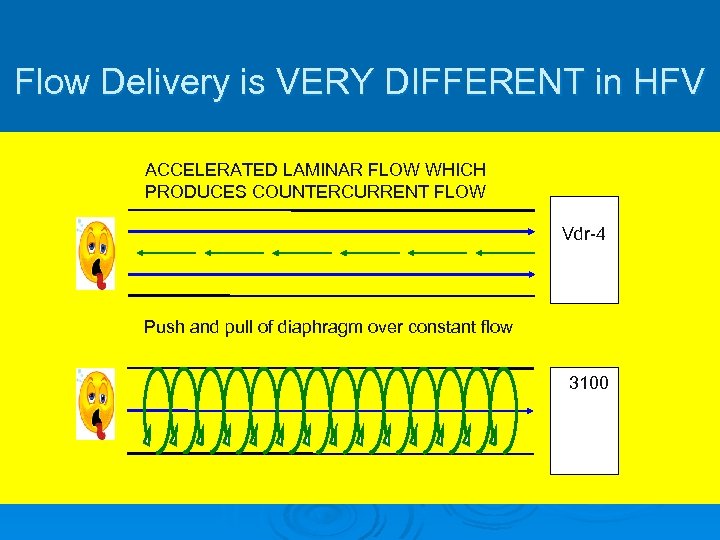 Flow Delivery is VERY DIFFERENT in HFV ACCELERATED LAMINAR FLOW WHICH PRODUCES COUNTERCURRENT FLOW Vdr-4 Push and pull of diaphragm over constant flow 3100
Flow Delivery is VERY DIFFERENT in HFV ACCELERATED LAMINAR FLOW WHICH PRODUCES COUNTERCURRENT FLOW Vdr-4 Push and pull of diaphragm over constant flow 3100
 COUNTER CURRENT FLOW IN ACTION
COUNTER CURRENT FLOW IN ACTION
 When to use which high frequency device
When to use which high frequency device
 HFOV 3100 A/B Ø Difficult to Oxygenate patients with mild hemodynamic compromise without major ventilation issues Ø Homogeneous Lung Disease
HFOV 3100 A/B Ø Difficult to Oxygenate patients with mild hemodynamic compromise without major ventilation issues Ø Homogeneous Lung Disease
 HFPV Ø VDR 4 l l l Able to improve oxygenation AND Able to improve ventilation Able to mobilize and remove secretions Able to ventilate neonate through adult Able to ventilate 600 gm through 256 kg
HFPV Ø VDR 4 l l l Able to improve oxygenation AND Able to improve ventilation Able to mobilize and remove secretions Able to ventilate neonate through adult Able to ventilate 600 gm through 256 kg
 High-Frequency Oscillation in Early Acute Respiratory Distress Syndrome Niall D. Ferguson, M. D. , for the OSCILLATE Trial Investigators and the Canadian Critical Care Trials Grou January 22, 2013, NEJM. org. Conclusions In adults with moderate-to-severe ARDS, early application of HFOV, as compared with a ventilation strategy of low tidal volum and high positive end-expiratory pressure, does not reduce, and may increase, in-hospital mortality. High-Frequency Oscillation for Acute Respiratory Distress Syndrome Duncan Young, D. M. , for the OSCAR Study Group* January 22, 2013, NEJM. org. Conclusions The use of HFOVhad no significant effect 30 -day on mortality in patients undergo- ing mechanical ventilation for ARDS.
High-Frequency Oscillation in Early Acute Respiratory Distress Syndrome Niall D. Ferguson, M. D. , for the OSCILLATE Trial Investigators and the Canadian Critical Care Trials Grou January 22, 2013, NEJM. org. Conclusions In adults with moderate-to-severe ARDS, early application of HFOV, as compared with a ventilation strategy of low tidal volum and high positive end-expiratory pressure, does not reduce, and may increase, in-hospital mortality. High-Frequency Oscillation for Acute Respiratory Distress Syndrome Duncan Young, D. M. , for the OSCAR Study Group* January 22, 2013, NEJM. org. Conclusions The use of HFOVhad no significant effect 30 -day on mortality in patients undergo- ing mechanical ventilation for ARDS.
 Oscillate Trial-2013 Ø 548 new-onset, moderate-to-severe ARDS patients Ø 548 of a planned 1200 patients 47% vs. 35% in-hospital mortality Patients were given more midazolam, more NMBs, more vasopressors Trial stopped early due to potential harm from HFOV Ø Ø Ø
Oscillate Trial-2013 Ø 548 new-onset, moderate-to-severe ARDS patients Ø 548 of a planned 1200 patients 47% vs. 35% in-hospital mortality Patients were given more midazolam, more NMBs, more vasopressors Trial stopped early due to potential harm from HFOV Ø Ø Ø
 All right What about ECMO?
All right What about ECMO?
 2009 was a big year for ECMO Quadrox D oxygenator Avalon DLVV cannula CESAR trial H 1 N 1 pandemic
2009 was a big year for ECMO Quadrox D oxygenator Avalon DLVV cannula CESAR trial H 1 N 1 pandemic
 Italian 9 days (7 -15) The Italian ECMO network experience during the 2009 influenza A(H 1 N 1) pandemic: preparation for severe respiratory emergency outbreaks. ntensive I Care Med. 2011 Sep; 37(9): 1447 -57. Japanese 9 days (6. 5– 12. 5) Extracorporeal membrane oxygenation for 2009 influenza A(H 1 N 1) severe respiratory failure in Japan. Anesthesia 2012 J Australian/New Zealand 10 days (7 -15) Extracorporeal Membrane Oxygenation for 2009 Influenza A(H 1 N 1) Acute Respiratory Distress Syndrome. AMA. 2009 Nov 4; 302(17): 1888 J Canadian 15 days (14 -15) Extracorporeal lung support for patients who had severe respiratory failure secondary to influenza A (H 1 N 1) 2009 infection in Canada. an J Anesthesia C 2010 Mar; 57(3): 240 -7 Swedish 16 days (9. 5 -30. 5) Extracorporeal membrane oxygenation for pandemic H 1 N 1 2009 respiratory failure. Minerva Anestethio. 2010 Dec; 76(12): 1043 -51. Chinese 18 days (2. 8– 90) Extracorporeal Membrane Oxygenation for Critically Ill Patients With 2009 Influenza A (H 1 N 1)-Related Acute Respiratory Distress Syndrome: Preliminary Experience From a Single Center. Artif Organs. 2012 Sep; 36(9): 780 -6 French -47) 23 days (3 Extracorporeal membrane oxygenation for 2009 influenza A (H 1 N 1) acute respiratory distress syndrome: single-centre experience with 1 -year follow-up. Eur J Cardiothorac Surg. 2012 Mar; 41(3): 691 -5
Italian 9 days (7 -15) The Italian ECMO network experience during the 2009 influenza A(H 1 N 1) pandemic: preparation for severe respiratory emergency outbreaks. ntensive I Care Med. 2011 Sep; 37(9): 1447 -57. Japanese 9 days (6. 5– 12. 5) Extracorporeal membrane oxygenation for 2009 influenza A(H 1 N 1) severe respiratory failure in Japan. Anesthesia 2012 J Australian/New Zealand 10 days (7 -15) Extracorporeal Membrane Oxygenation for 2009 Influenza A(H 1 N 1) Acute Respiratory Distress Syndrome. AMA. 2009 Nov 4; 302(17): 1888 J Canadian 15 days (14 -15) Extracorporeal lung support for patients who had severe respiratory failure secondary to influenza A (H 1 N 1) 2009 infection in Canada. an J Anesthesia C 2010 Mar; 57(3): 240 -7 Swedish 16 days (9. 5 -30. 5) Extracorporeal membrane oxygenation for pandemic H 1 N 1 2009 respiratory failure. Minerva Anestethio. 2010 Dec; 76(12): 1043 -51. Chinese 18 days (2. 8– 90) Extracorporeal Membrane Oxygenation for Critically Ill Patients With 2009 Influenza A (H 1 N 1)-Related Acute Respiratory Distress Syndrome: Preliminary Experience From a Single Center. Artif Organs. 2012 Sep; 36(9): 780 -6 French -47) 23 days (3 Extracorporeal membrane oxygenation for 2009 influenza A (H 1 N 1) acute respiratory distress syndrome: single-centre experience with 1 -year follow-up. Eur J Cardiothorac Surg. 2012 Mar; 41(3): 691 -5
 Italian 9 days (7 -15) The Italian ECMO network experience during the 2009 influenza A(H 1 N 1) pandemic: preparation for severe respiratory emergency outbreaks. ntensive Care I Med. 2011 Sep; 37(9): 1447 -57. Japanese 9 days (6. 5– 12. 5) Extracorporeal membrane oxygenation for 2009 influenza A(H 1 N 1) severe respiratory failure in Japan. Anesthesia 2012 J Australian/New Zealand 10 days (7 -15) Extracorporeal Membrane Oxygenation for 2009 Influenza A(H 1 N 1) Acute Respiratory Distress Syndrome. AMA. 2009 Nov 4; 302(17): 1888 J Canadian 15 days (14 -15) Extracorporeal lung support for patients who had severe respiratory failure secondary to influenza A (H 1 N 1) 2009 infection in Canada. an J Anesthesia 2010 C Mar; 57(3): 240 -7 Swedish 16 days (9. 5 -30. 5) Extracorporeal membrane oxygenation for pandemic H 1 N 1 2009 respiratory failure. Minerva Anestethio. 2010 Dec; 76(12): 1043 -51. Chinese 18 days (2. 8– 90) Extracorporeal Membrane Oxygenation for Critically Ill Patients With 2009 Influenza A (H 1 N 1)-Related Acute Respiratory Distress Syndrome: Preliminary Experience From a Single Center. Artif Organs. 2012 Sep; 36(9): 780 -6 French 23 days (3 -47) Extracorporeal membrane oxygenation for 2009 influenza A (H 1 N 1) acute respiratory distress syndrome: single-centre experience with 1 -year follow-up. ur J E Cardiothorac Surg. 2012 Mar; 41(3): 691 -5 Legacy/Emanuel (193 hours) 8 days (3 -15)
Italian 9 days (7 -15) The Italian ECMO network experience during the 2009 influenza A(H 1 N 1) pandemic: preparation for severe respiratory emergency outbreaks. ntensive Care I Med. 2011 Sep; 37(9): 1447 -57. Japanese 9 days (6. 5– 12. 5) Extracorporeal membrane oxygenation for 2009 influenza A(H 1 N 1) severe respiratory failure in Japan. Anesthesia 2012 J Australian/New Zealand 10 days (7 -15) Extracorporeal Membrane Oxygenation for 2009 Influenza A(H 1 N 1) Acute Respiratory Distress Syndrome. AMA. 2009 Nov 4; 302(17): 1888 J Canadian 15 days (14 -15) Extracorporeal lung support for patients who had severe respiratory failure secondary to influenza A (H 1 N 1) 2009 infection in Canada. an J Anesthesia 2010 C Mar; 57(3): 240 -7 Swedish 16 days (9. 5 -30. 5) Extracorporeal membrane oxygenation for pandemic H 1 N 1 2009 respiratory failure. Minerva Anestethio. 2010 Dec; 76(12): 1043 -51. Chinese 18 days (2. 8– 90) Extracorporeal Membrane Oxygenation for Critically Ill Patients With 2009 Influenza A (H 1 N 1)-Related Acute Respiratory Distress Syndrome: Preliminary Experience From a Single Center. Artif Organs. 2012 Sep; 36(9): 780 -6 French 23 days (3 -47) Extracorporeal membrane oxygenation for 2009 influenza A (H 1 N 1) acute respiratory distress syndrome: single-centre experience with 1 -year follow-up. ur J E Cardiothorac Surg. 2012 Mar; 41(3): 691 -5 Legacy/Emanuel (193 hours) 8 days (3 -15)
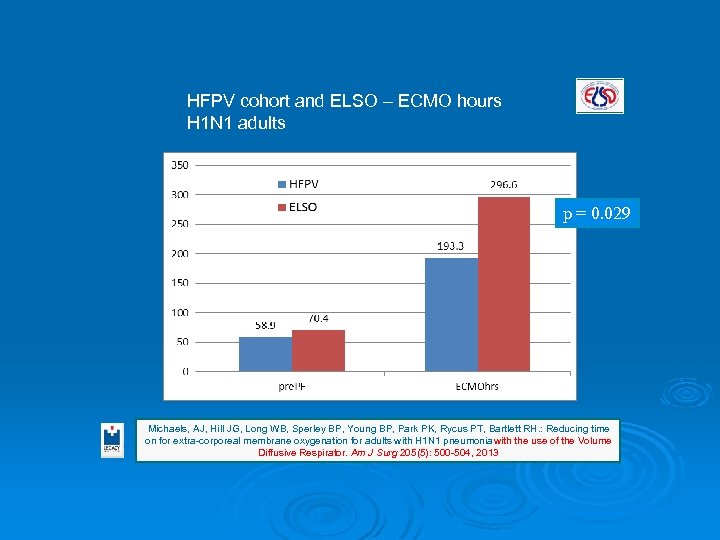 HFPV cohort and ELSO – ECMO hours H 1 N 1 adults p = 0. 029 Michaels, AJ, Hill JG, Long WB, Sperley BP, Young BP, Park PK, Rycus PT, Bartlett RH. : Reducing time on for extra-corporeal membrane oxygenation for adults with H 1 N 1 pneumonia with the use of the Volume Diffusive Respirator. Am J Surg 205(5): 500 -504, 2013
HFPV cohort and ELSO – ECMO hours H 1 N 1 adults p = 0. 029 Michaels, AJ, Hill JG, Long WB, Sperley BP, Young BP, Park PK, Rycus PT, Bartlett RH. : Reducing time on for extra-corporeal membrane oxygenation for adults with H 1 N 1 pneumonia with the use of the Volume Diffusive Respirator. Am J Surg 205(5): 500 -504, 2013
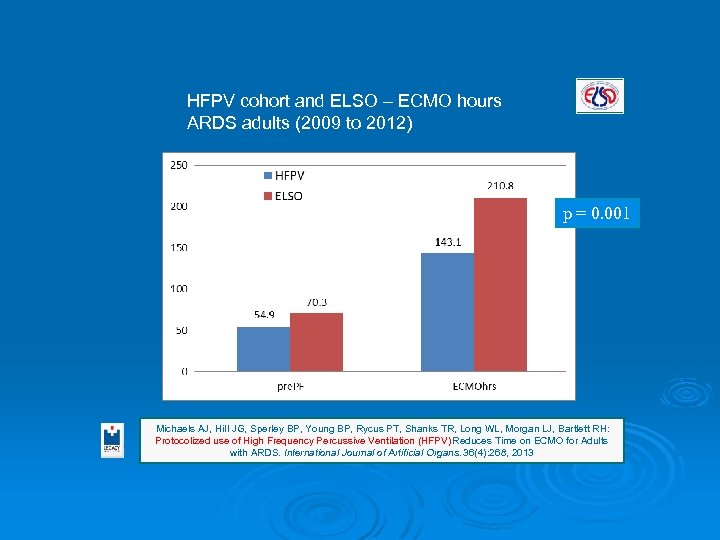 HFPV cohort and ELSO – ECMO hours ARDS adults (2009 to 2012) p = 0. 001 Michaels AJ, Hill JG, Sperley BP, Young BP, Rycus PT, Shanks TR, Long WL, Morgan LJ, Bartlett RH: Protocolized use of High Frequency Percussive Ventilation (HFPV) Reduces Time on ECMO for Adults with ARDS. International Journal of Artificial Organs. 36(4): 268, 2013
HFPV cohort and ELSO – ECMO hours ARDS adults (2009 to 2012) p = 0. 001 Michaels AJ, Hill JG, Sperley BP, Young BP, Rycus PT, Shanks TR, Long WL, Morgan LJ, Bartlett RH: Protocolized use of High Frequency Percussive Ventilation (HFPV) Reduces Time on ECMO for Adults with ARDS. International Journal of Artificial Organs. 36(4): 268, 2013
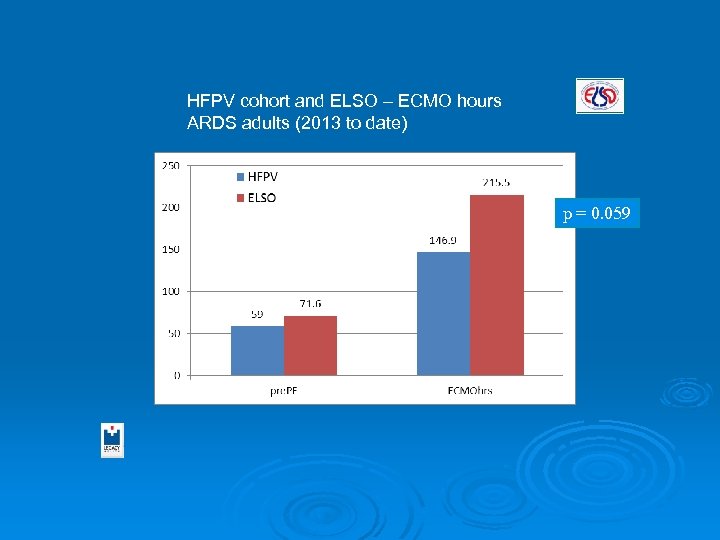 HFPV cohort and ELSO – ECMO hours ARDS adults (2013 to date) p = 0. 059
HFPV cohort and ELSO – ECMO hours ARDS adults (2013 to date) p = 0. 059
 5 basic phases to an ECMO case for ARDS
5 basic phases to an ECMO case for ARDS
 5 basic phases to an adult ECMO case for ARDS Gain support “rest the lungs” eliminate VILI…
5 basic phases to an adult ECMO case for ARDS Gain support “rest the lungs” eliminate VILI…
 5 basic phases to an ECMO case for ARDS Gain support “rest the lungs” eliminate VILI… Treat the source stop the inflammation…
5 basic phases to an ECMO case for ARDS Gain support “rest the lungs” eliminate VILI… Treat the source stop the inflammation…
 5 basic phases to an ECMO case for ARDS Gain support “rest the lungs” eliminate VILI… Treat the source stop the inflammation… Recovery restore “normal” physiology
5 basic phases to an ECMO case for ARDS Gain support “rest the lungs” eliminate VILI… Treat the source stop the inflammation… Recovery restore “normal” physiology
 5 basic phases to an ECMO case for ARDS Gain support “rest the lungs” eliminate VILI… Treat the source stop the inflammation… Recovery restore “normal” physiology Recruitment regain FRC and capillary: alveolar function…
5 basic phases to an ECMO case for ARDS Gain support “rest the lungs” eliminate VILI… Treat the source stop the inflammation… Recovery restore “normal” physiology Recruitment regain FRC and capillary: alveolar function…
 5 basic phases to an adult ECMO case for ARDS Gain support “rest the lungs” eliminate VILI… Treat the source stop the inflammation… Recovery restore “normal” physiology Recruitment regain FRC and capillary: alveolar function… Get off ECMO before something bad happens…
5 basic phases to an adult ECMO case for ARDS Gain support “rest the lungs” eliminate VILI… Treat the source stop the inflammation… Recovery restore “normal” physiology Recruitment regain FRC and capillary: alveolar function… Get off ECMO before something bad happens…
 Recruitment Tracheobronchial BAL Parenchymal positional RX diuresis / CRRT Thoracic domain drain effusions reduce edema abdominal pressures Alveolar HFPV “rest” followed by increasing “recruitment”
Recruitment Tracheobronchial BAL Parenchymal positional RX diuresis / CRRT Thoracic domain drain effusions reduce edema abdominal pressures Alveolar HFPV “rest” followed by increasing “recruitment”
 Whats it look like with a patient
Whats it look like with a patient
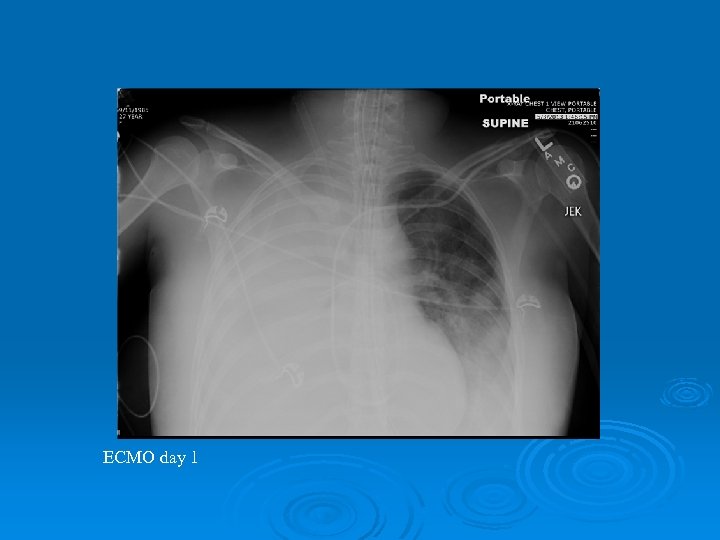 ECMO day 1
ECMO day 1
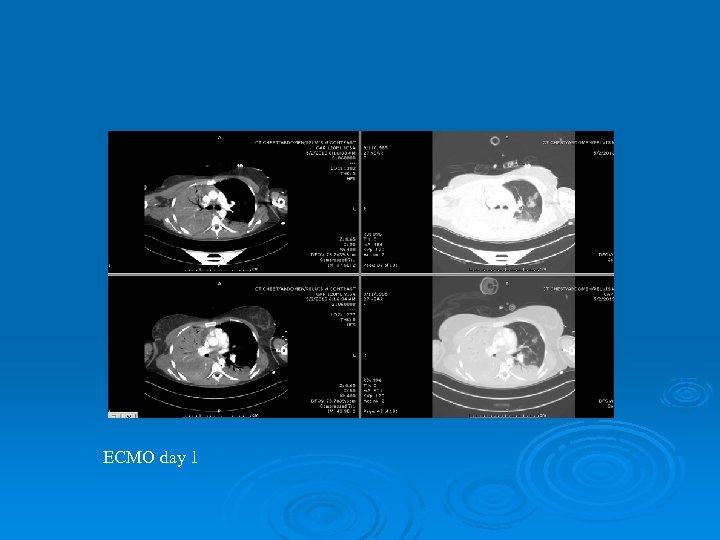 ECMO day 1
ECMO day 1
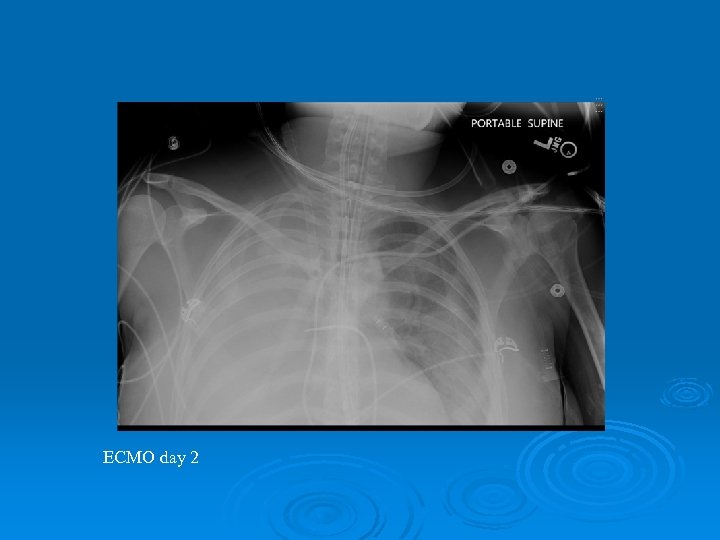 ECMO day 2
ECMO day 2
 Prone Positioning on ECMO
Prone Positioning on ECMO
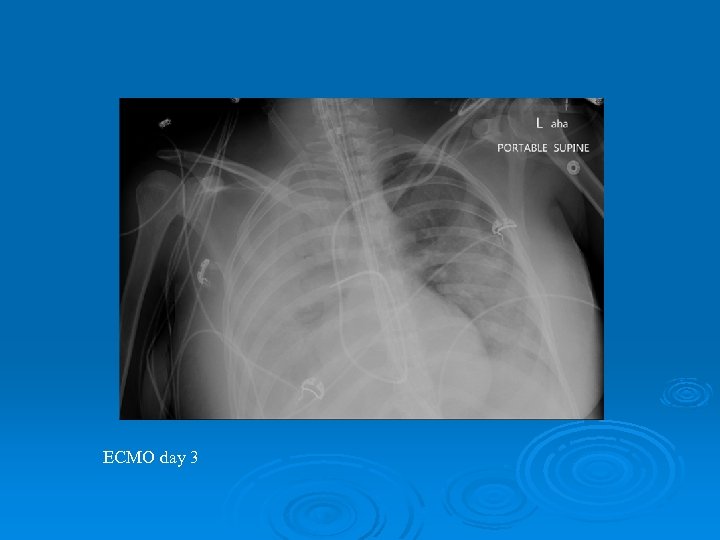 ECMO day 3
ECMO day 3
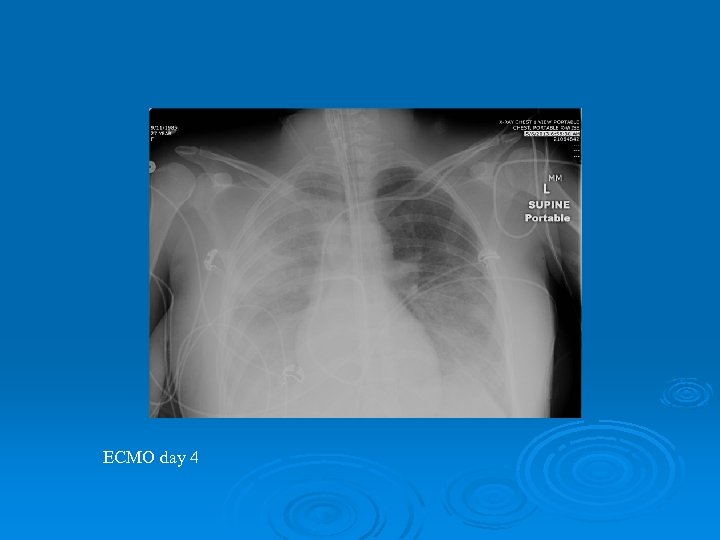 ECMO day 4
ECMO day 4
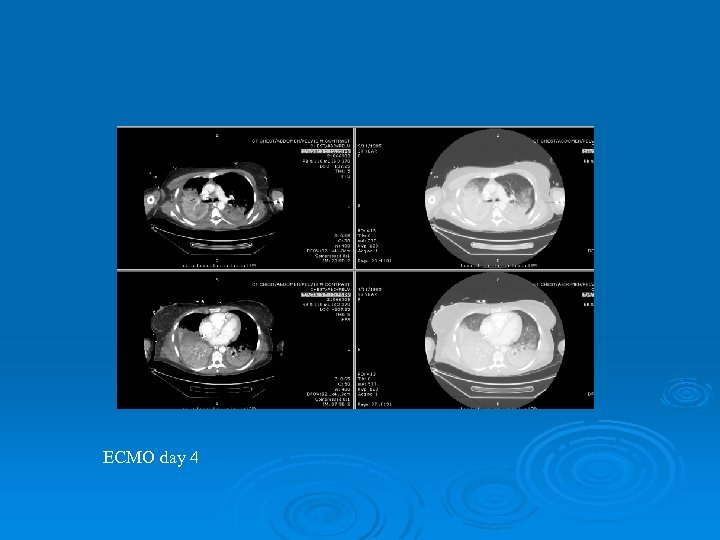 ECMO day 4
ECMO day 4
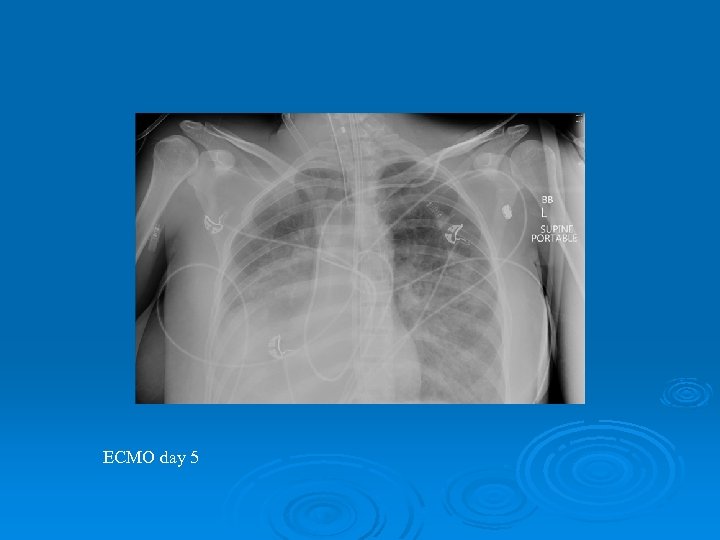 ECMO day 5
ECMO day 5
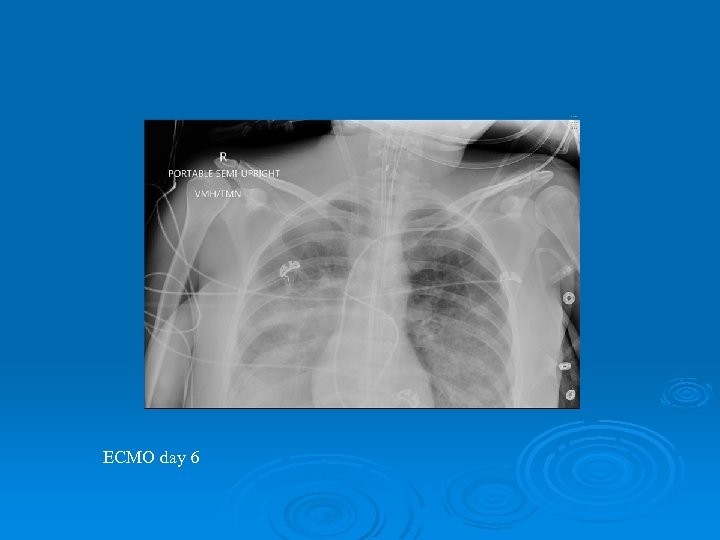 ECMO day 6
ECMO day 6
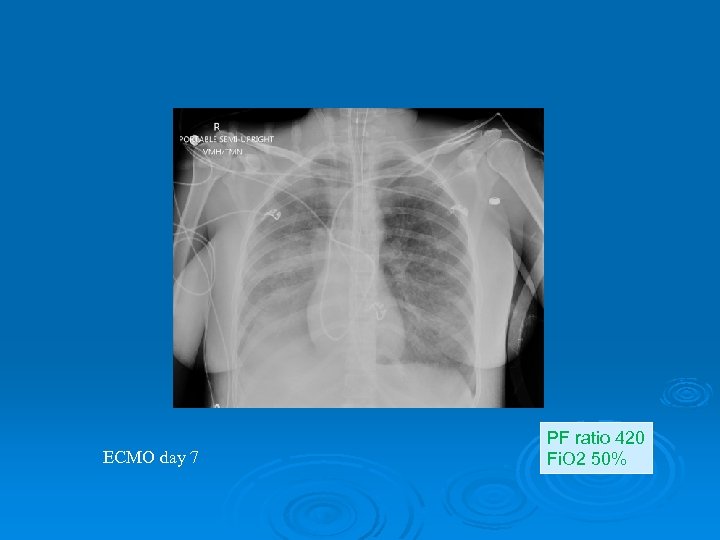 ECMO day 7 PF ratio 420 Fi. O 2 50%
ECMO day 7 PF ratio 420 Fi. O 2 50%
 How about walking an Ecmo patient to the Garden?
How about walking an Ecmo patient to the Garden?
 Anybody else using the VDR?
Anybody else using the VDR?
 CHILDREN’S HOSPITAL OF PHILADELPHIA EXPERIENCE HIGH FREQUENCY PERCUSSIVE VENTILATION
CHILDREN’S HOSPITAL OF PHILADELPHIA EXPERIENCE HIGH FREQUENCY PERCUSSIVE VENTILATION
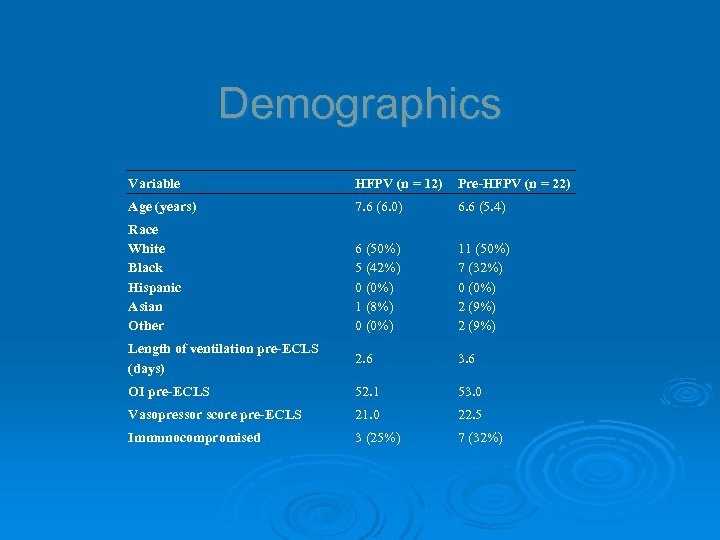 Demographics Variable HFPV (n = 12) Pre-HFPV (n = 22) Age (years) 7. 6 (6. 0) 6. 6 (5. 4) Race White Black Hispanic Asian Other 6 (50%) 5 (42%) 0 (0%) 1 (8%) 0 (0%) 11 (50%) 7 (32%) 0 (0%) 2 (9%) Length of ventilation pre-ECLS (days) 2. 6 3. 6 OI pre-ECLS 52. 1 53. 0 Vasopressor score pre-ECLS 21. 0 22. 5 Immunocompromised 3 (25%) 7 (32%)
Demographics Variable HFPV (n = 12) Pre-HFPV (n = 22) Age (years) 7. 6 (6. 0) 6. 6 (5. 4) Race White Black Hispanic Asian Other 6 (50%) 5 (42%) 0 (0%) 1 (8%) 0 (0%) 11 (50%) 7 (32%) 0 (0%) 2 (9%) Length of ventilation pre-ECLS (days) 2. 6 3. 6 OI pre-ECLS 52. 1 53. 0 Vasopressor score pre-ECLS 21. 0 22. 5 Immunocompromised 3 (25%) 7 (32%)
 Comparison Ø Prospectively collected database of consecutive children placed on ECLS in CHOP PICU (starting June, 2008) Ø After May, 2011, all children received HFPV and therapeutic bronchoscopies while on ECLS as an adjunct to pulmonary toilet (HFPV cohort) Ø Compared the HFPV cohort to the cohort immediately preceding protocolization of HFPV and bronchoscopies (pre-HFPV cohort)
Comparison Ø Prospectively collected database of consecutive children placed on ECLS in CHOP PICU (starting June, 2008) Ø After May, 2011, all children received HFPV and therapeutic bronchoscopies while on ECLS as an adjunct to pulmonary toilet (HFPV cohort) Ø Compared the HFPV cohort to the cohort immediately preceding protocolization of HFPV and bronchoscopies (pre-HFPV cohort)
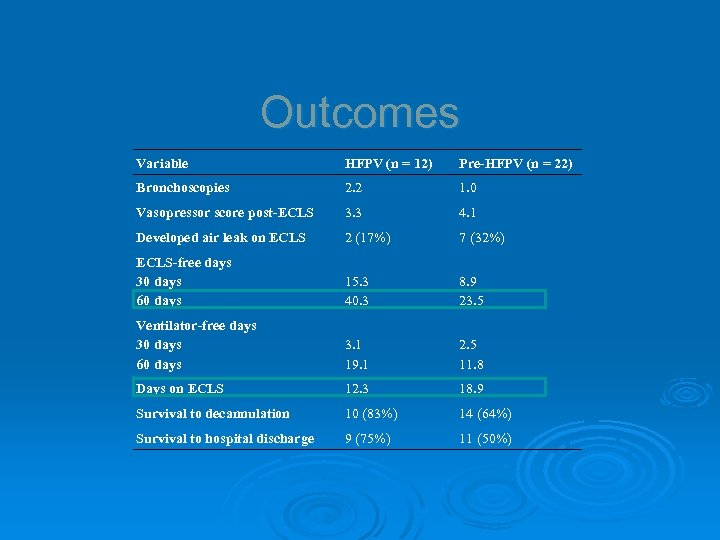 Outcomes Variable HFPV (n = 12) Pre-HFPV (n = 22) Bronchoscopies 2. 2 1. 0 Vasopressor score post-ECLS 3. 3 4. 1 Developed air leak on ECLS 2 (17%) 7 (32%) ECLS-free days 30 days 60 days 15. 3 40. 3 8. 9 23. 5 Ventilator-free days 30 days 60 days 3. 1 19. 1 2. 5 11. 8 Days on ECLS 12. 3 18. 9 Survival to decannulation 10 (83%) 14 (64%) Survival to hospital discharge 9 (75%) 11 (50%)
Outcomes Variable HFPV (n = 12) Pre-HFPV (n = 22) Bronchoscopies 2. 2 1. 0 Vasopressor score post-ECLS 3. 3 4. 1 Developed air leak on ECLS 2 (17%) 7 (32%) ECLS-free days 30 days 60 days 15. 3 40. 3 8. 9 23. 5 Ventilator-free days 30 days 60 days 3. 1 19. 1 2. 5 11. 8 Days on ECLS 12. 3 18. 9 Survival to decannulation 10 (83%) 14 (64%) Survival to hospital discharge 9 (75%) 11 (50%)
 Conclusions Ø Use of HFPV during ECLS is associated with an increased number of days alive and off of ECLS Ø HFPV can be safely applied with minimum complications while on ECLS
Conclusions Ø Use of HFPV during ECLS is associated with an increased number of days alive and off of ECLS Ø HFPV can be safely applied with minimum complications while on ECLS
 What if in the process of going and getting pre ecmo patients you could place some of them on a ventilator and bring them home and not use ECMO?
What if in the process of going and getting pre ecmo patients you could place some of them on a ventilator and bring them home and not use ECMO?
 3/18/2018 LEGACY HEALTH 80
3/18/2018 LEGACY HEALTH 80
 HFPV STUDY Ø 1. To determine if early use of High Frequency Percussive Ventilation (HFPV) reduces need for Veno-Venous ECMO (V-V ECMO) in patients who have failed conventional modes of ventilation and were accepted for ECMO EVALUATION to our facility Ø 2. Patients referred to our facility for V-V ECMO have severe Refractory Hypoxemic Respiratory Failure. They have failed traditional treatment and conventional modes of ventilation including ARDSnet, APRV, PCV and HFOV, prone positioning and inhaled Nitric Oxide and / or Epoprostenol. Ø 3. Patients are considered for ECMO when the P/F ratio is <100 and have a treatable and reversible cause. Patients found to be unstable or in extremis upon arrival to the referring hospital were not considered candidates for HFPV alone. These candidates were immediately placed on ECMO Ø
HFPV STUDY Ø 1. To determine if early use of High Frequency Percussive Ventilation (HFPV) reduces need for Veno-Venous ECMO (V-V ECMO) in patients who have failed conventional modes of ventilation and were accepted for ECMO EVALUATION to our facility Ø 2. Patients referred to our facility for V-V ECMO have severe Refractory Hypoxemic Respiratory Failure. They have failed traditional treatment and conventional modes of ventilation including ARDSnet, APRV, PCV and HFOV, prone positioning and inhaled Nitric Oxide and / or Epoprostenol. Ø 3. Patients are considered for ECMO when the P/F ratio is <100 and have a treatable and reversible cause. Patients found to be unstable or in extremis upon arrival to the referring hospital were not considered candidates for HFPV alone. These candidates were immediately placed on ECMO Ø
 Ø We reviewed calendar years 2014 and 2015 to compare patients accepted for ECMO, but were placed on HFPV first, before determining further need for extracorporeal support. We found that ~ 20% of patients in 2014 and ~40% of patients in 2015 were treated successfully WITH HFPV alone and were not placed on ECMO. Ø
Ø We reviewed calendar years 2014 and 2015 to compare patients accepted for ECMO, but were placed on HFPV first, before determining further need for extracorporeal support. We found that ~ 20% of patients in 2014 and ~40% of patients in 2015 were treated successfully WITH HFPV alone and were not placed on ECMO. Ø
 Ø Early implementation of HFPV in patients with Severe Hypoxemic and/or Hypercapnic Respiratory Failure who have qualified for extracorporeal support, can be managed with HFPV without ECMO in some cases. By doing this, we avoid the inherent mortality risks associated with extracorporeal support, decrease overall hospital length of stay and ventilator days. We also contribute to a significant reduction in ECMO associated costs. A combined savings for calendar years 2014 and 2015 of $3. 98 million. Ø
Ø Early implementation of HFPV in patients with Severe Hypoxemic and/or Hypercapnic Respiratory Failure who have qualified for extracorporeal support, can be managed with HFPV without ECMO in some cases. By doing this, we avoid the inherent mortality risks associated with extracorporeal support, decrease overall hospital length of stay and ventilator days. We also contribute to a significant reduction in ECMO associated costs. A combined savings for calendar years 2014 and 2015 of $3. 98 million. Ø
 Conclusions Ø ARDS continues to affect many patients and as respiratory therapists we can greatly affect the outcome of these patients by early detection and identification of disease. Then we must use the treatment strategies quickly and efficiently to include: ARDS network, proning, APRV, inhaled prostacyclins and if they are not working consider aggressive strategies such as HFPV and ECMO! Ø WE CAN BE THE DIFFERENCE MAKER IN THIS DISEASE AS PART OF THE TEAM! Ø
Conclusions Ø ARDS continues to affect many patients and as respiratory therapists we can greatly affect the outcome of these patients by early detection and identification of disease. Then we must use the treatment strategies quickly and efficiently to include: ARDS network, proning, APRV, inhaled prostacyclins and if they are not working consider aggressive strategies such as HFPV and ECMO! Ø WE CAN BE THE DIFFERENCE MAKER IN THIS DISEASE AS PART OF THE TEAM! Ø
 Ø THANK YOU VERY MUCH
Ø THANK YOU VERY MUCH


