df3a00e20d2ea5b9600fcb9024f6315b.ppt
- Количество слайдов: 107

Dyslipidemia & Evidence-Based Medicine : What have we behind statins monotherapy? Prof. A. Tenenbaum Director Cardioline Heart Institute Director of Research Cardiac Rehabilitation Institute, Sheba Medical Center 2010

Agenda Dyslipidemia & Atherogenic Particles ¨ Cholesterol Metabolism: Absorption, De Novo Synthesis and their Markers ¨ Clinical Significance of Atherogenic Dyslipidemia ¨ Beyond Statins: Clinical Evidences ¨ “Surrogate”-Endpoints Evidences ¨ Clinical “Hard”-Endpoints Evidences Summary

The basic structure of a lipoprotein. The core contains mostly lipids and the shell has a composition similar to cell membranes (protein + phospholipids + cholesterol). The shell protein molecules (apo-proteins) serve to activate surface receptors and enzymes essential for the uptake and metabolism of lipoproteins.
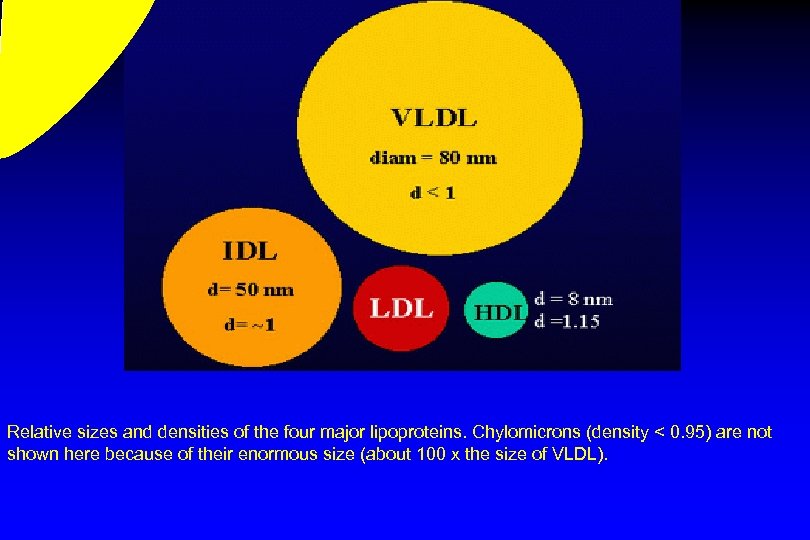
Relative sizes and densities of the four major lipoproteins. Chylomicrons (density < 0. 95) are not shown here because of their enormous size (about 100 x the size of VLDL).
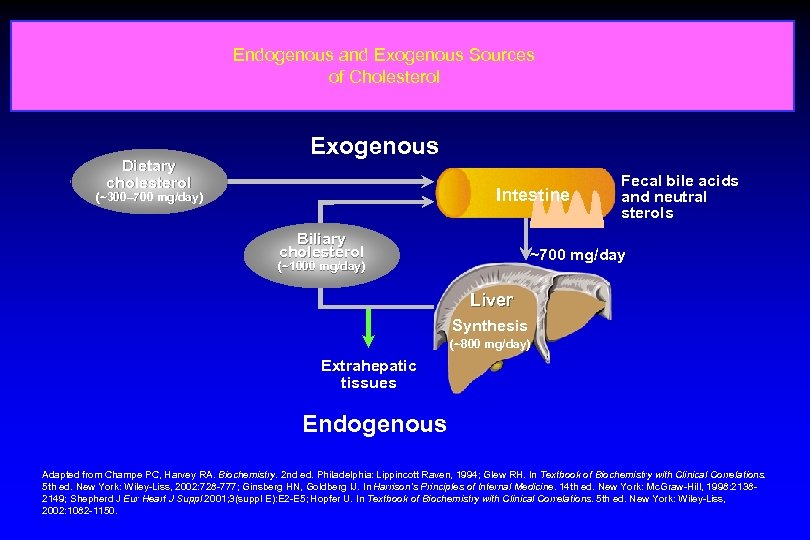
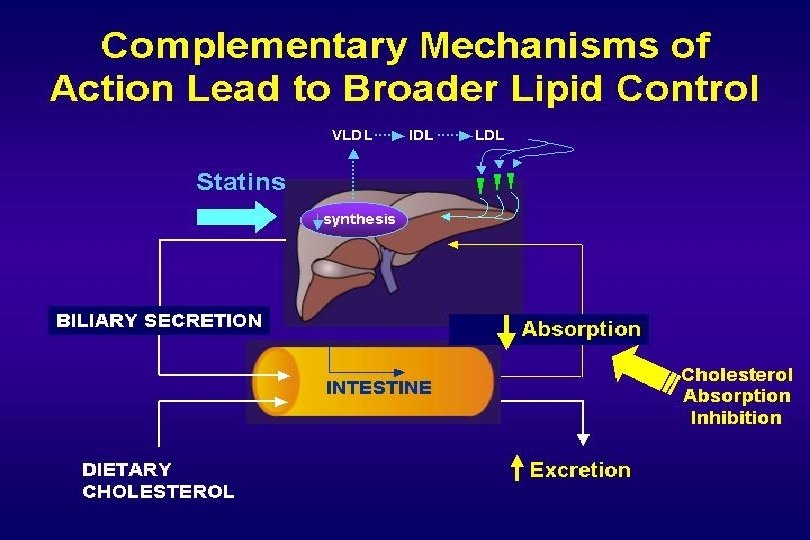
Endogenous and Exogenous Sources of Cholesterol Dietary cholesterol Exogenous Intestine (~300– 700 mg/day) Biliary cholesterol Fecal bile acids and neutral sterols ~700 mg/day (~1000 mg/day) Liver Synthesis (~800 mg/day) Extrahepatic tissues Endogenous Adapted from Champe PC, Harvey RA. Biochemistry. 2 nd ed. Philadelphia: Lippincott Raven, 1994; Glew RH. In Textbook of Biochemistry with Clinical Correlations. 5 th ed. New York: Wiley-Liss, 2002: 728 -777; Ginsberg HN, Goldberg IJ. In Harrison’s Principles of Internal Medicine. 14 th ed. New York: Mc. Graw-Hill, 1998: 21382149; Shepherd J Eur Heart J Suppl 2001; 3(suppl E): E 2 -E 5; Hopfer U. In Textbook of Biochemistry with Clinical Correlations. 5 th ed. New York: Wiley-Liss, 2002: 1082 -1150.
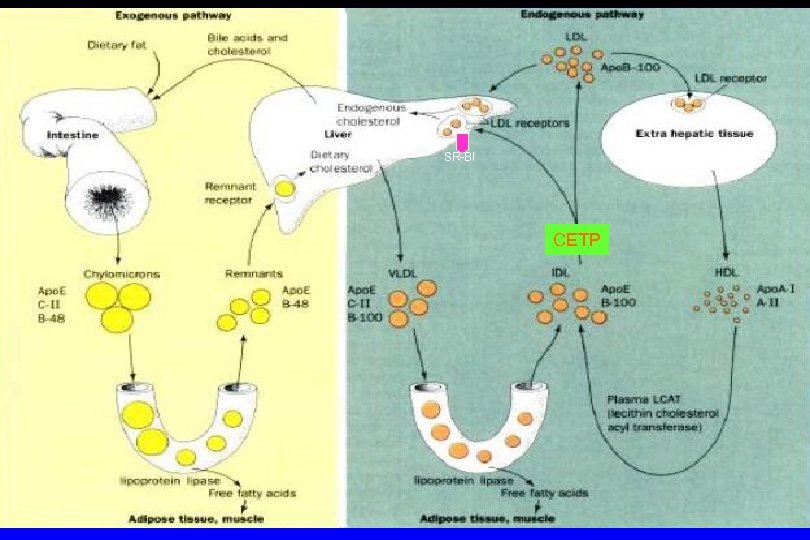
Cholesterol Metabolism: Uptake SR-BI CETP
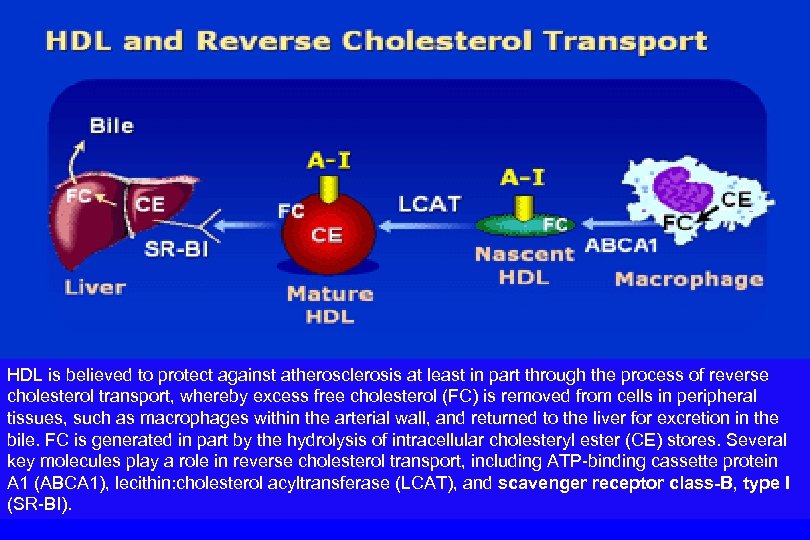
HDL is believed to protect against atherosclerosis at least in part through the process of reverse cholesterol transport, whereby excess free cholesterol (FC) is removed from cells in peripheral tissues, such as macrophages within the arterial wall, and returned to the liver for excretion in the bile. FC is generated in part by the hydrolysis of intracellular cholesteryl ester (CE) stores. Several key molecules play a role in reverse cholesterol transport, including ATP-binding cassette protein A 1 (ABCA 1), lecithin: cholesterol acyltransferase (LCAT), and scavenger receptor class-B, type I (SR-BI).
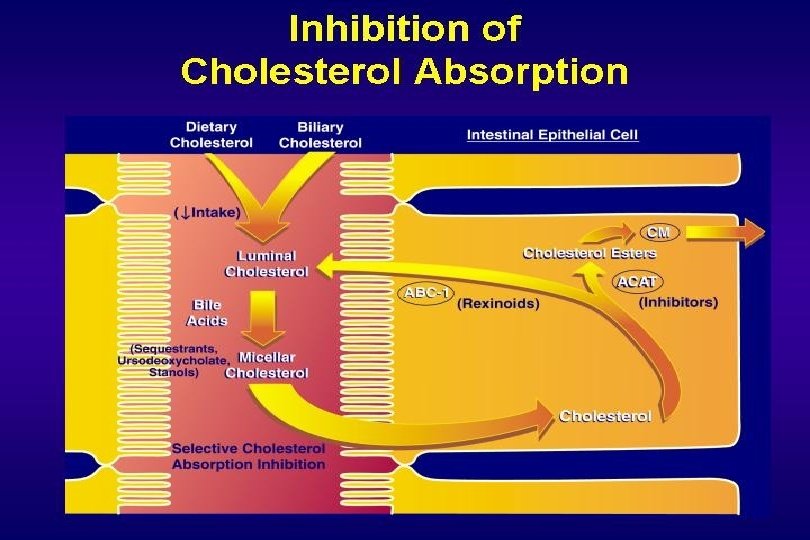
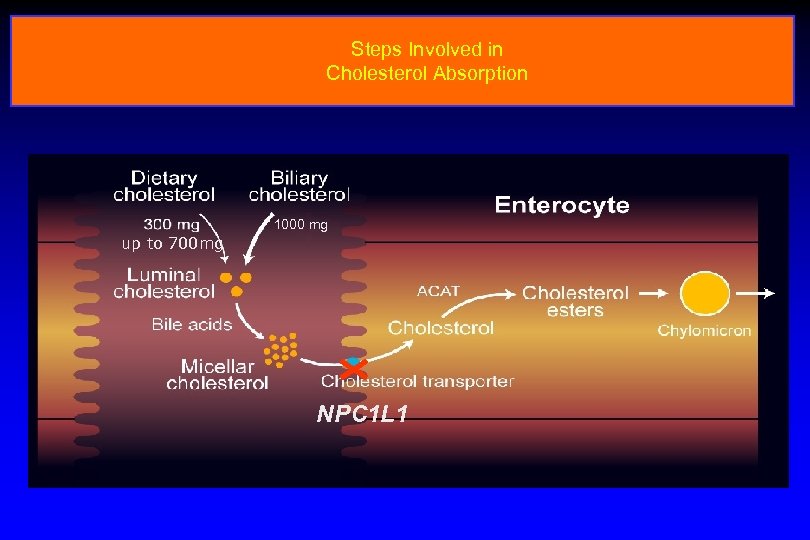
Steps Involved in Cholesterol Absorption 1000 mg up to 700 mg NPC 1 L 1
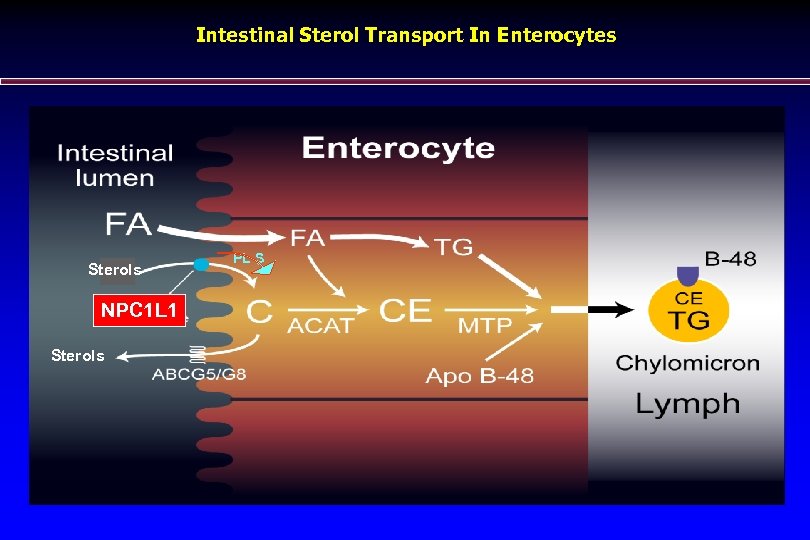
Intestinal Sterol Transport In Enterocytes Sterols NPC 1 L 1 Sterols PL S
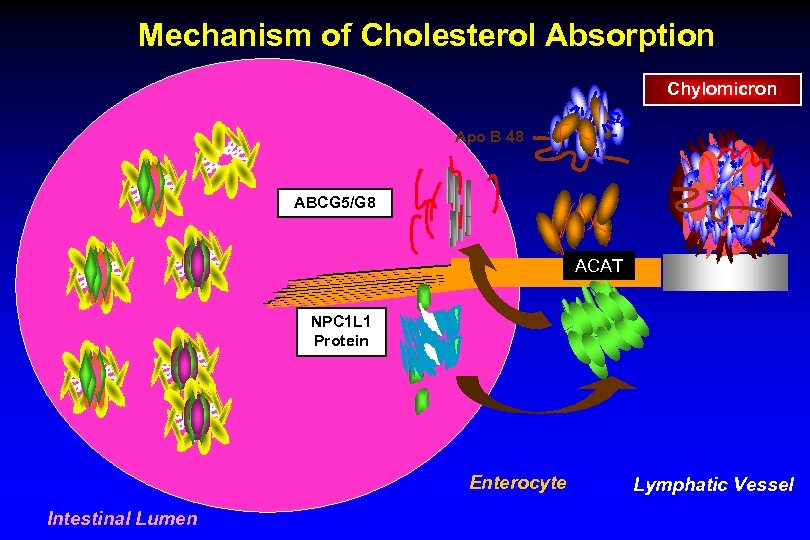
Mechanism of Cholesterol Absorption Chylomicron Apo B 48 ABCG 5/G 8 ACAT NPC 1 L 1 Protein Enterocyte Intestinal Lumen Lymphatic Vessel
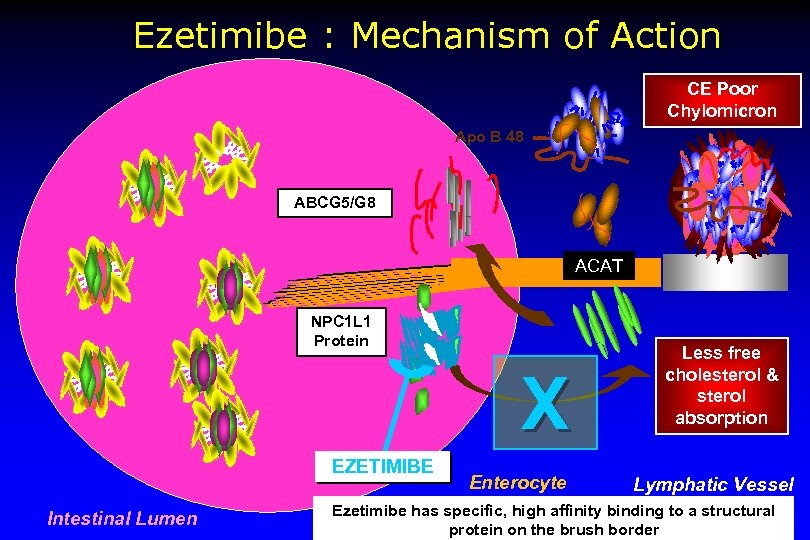
Ezetimibe : Mechanism of Action CE Poor Chylomicron Apo B 48 ABCG 5/G 8 ACAT NPC 1 L 1 Protein X EZETIMIBE Intestinal Lumen Enterocyte Less free cholesterol & sterol absorption Lymphatic Vessel Ezetimibe has specific, high affinity binding to a structural protein on the brush border
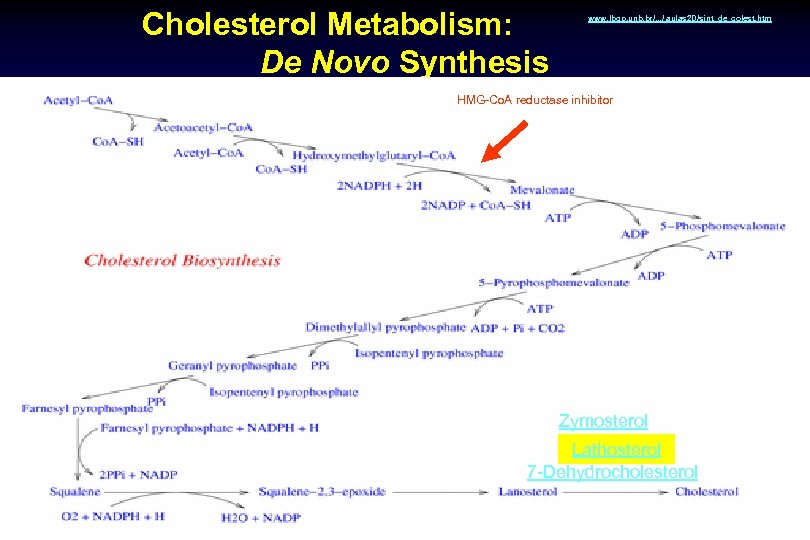
Cholesterol Metabolism: De Novo Synthesis www. lbqp. unb. br/. . . / aulas 2 D/sint_de_colest. htm HMG-Co. A reductase inhibitor Zymosterol Lathosterol 7 -Dehydrocholesterol

Absorption and Synthesis Markers • Lathosterol: is one of precursors of Cholesterol in the Mevalonate pathway thus is a marker of cholestrol synthesis. • Cholestanol and plant sterols: are positively correlated with the intestinal cholesterol (dietary plus endogenous) flux. * Lathosterol. C holestanol *Metabolism. 1989 Feb; 38(2): 136 -40 Synthesis Absorption

Absorption and Synthesis Markers Lathosterol. C holestanol Ratio Bigger Lathosterol. C holestanol Ratio Smaller More Synthesis More Absorption
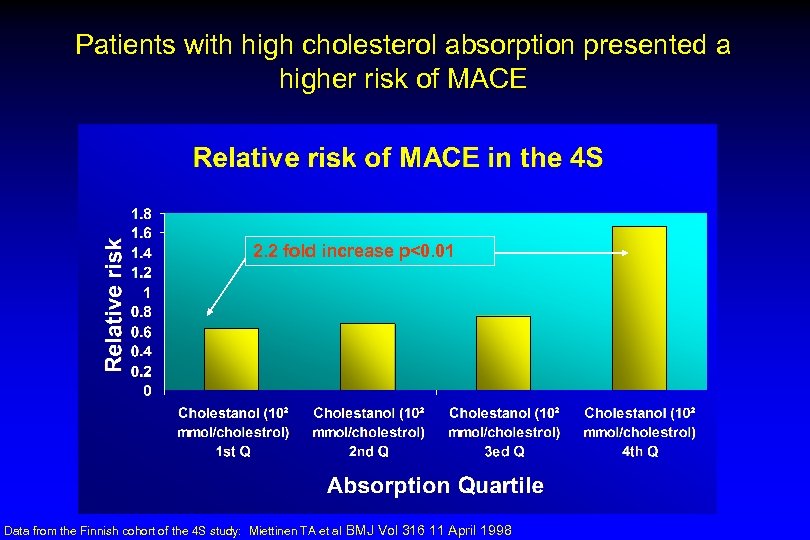
Patients with high cholesterol absorption presented a higher risk of MACE 2. 2 fold increase p<0. 01 Data from the Finnish cohort of the 4 S study: Miettinen TA et al BMJ Vol 316 11 April 1998
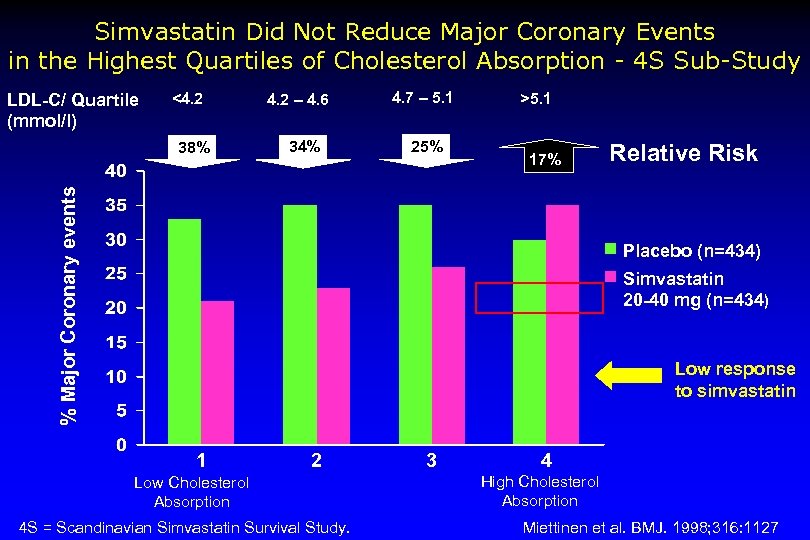
Simvastatin Did Not Reduce Major Coronary Events in the Highest Quartiles of Cholesterol Absorption - 4 S Sub-Study LDL-C/ Quartile (mmol/l) <4. 2 34% 4. 7 – 5. 1 25% >5. 1 17% % Major Coronary events 38% 4. 2 – 4. 6 Relative Risk Placebo (n=434) Simvastatin 20 -40 mg (n=434) Low response to simvastatin 1 2 Low Cholesterol Absorption 4 S = Scandinavian Simvastatin Survival Study. 3 4 High Cholesterol Absorption Miettinen et al. BMJ. 1998; 316: 1127

Dual Inhibition concept Assman G, Kannenberg F, Weng W et al. Effects of ezetimibe, simvastatin, and ezetimibe-simvastatin on non-cholesterols. Poster presented at the American Collegeof Cardiology meeting, New Orleans, Louisiana, USA, March 7– 10, 2004.
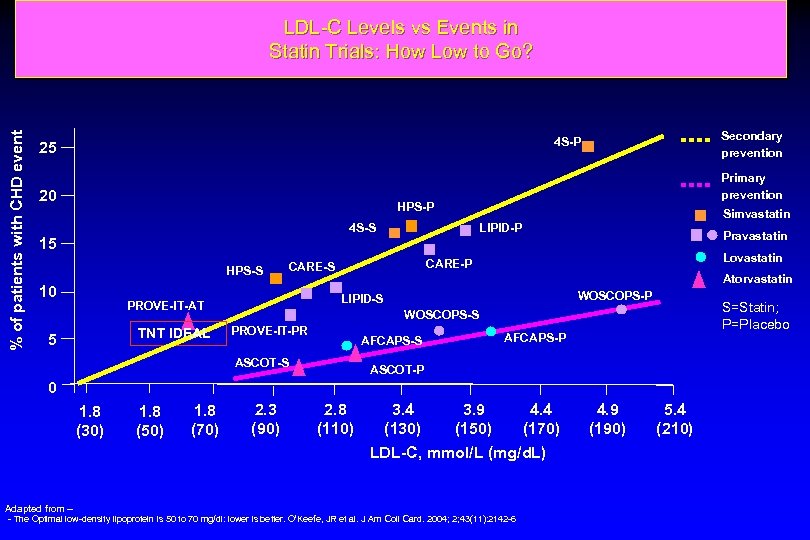
% of patients with CHD event LDL-C Levels vs Events in Statin Trials: How Low to Go? Secondary prevention 4 S-P 25 20 Primary prevention HPS-P 4 S-S LIPID-P 15 HPS-S 10 TNT IDEAL Pravastatin Lovastatin CARE-P CARE-S Atorvastatin WOSCOPS-P LIPID-S PROVE-IT-AT 5 Simvastatin S=Statin; P=Placebo WOSCOPS-S PROVE-IT-PR AFCAPS-S ASCOT-S AFCAPS-P ASCOT-P 0 1. 8 (30) 1. 8 (50) 1. 8 (70) 2. 3 (90) 2. 8 (110) 3. 4 3. 9 4. 4 (130) (150) (170) LDL-C, mmol/L (mg/d. L) Adapted from – - The Optimal low-density lipoprotein is 50 to 70 mg/dl: lower is better. O’Keefe, JR et al. J Am Coll Card. 2004; 2; 43(11): 2142 -6 4. 9 (190) 5. 4 (210)
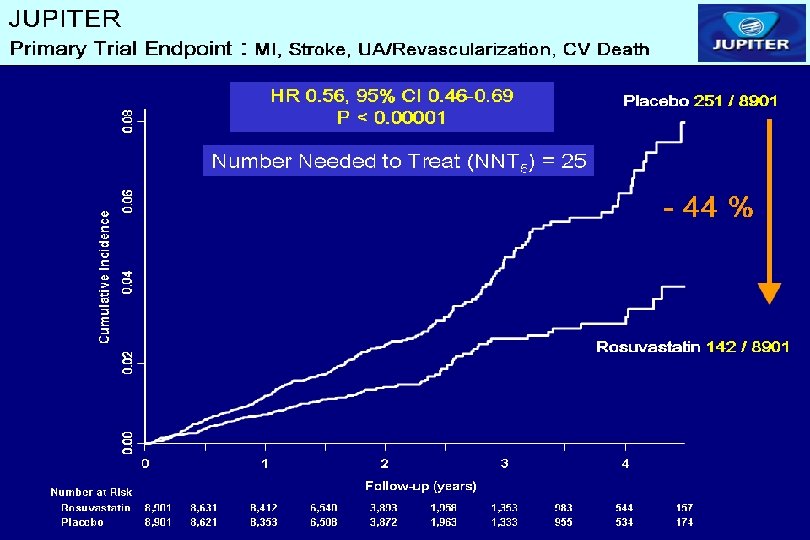
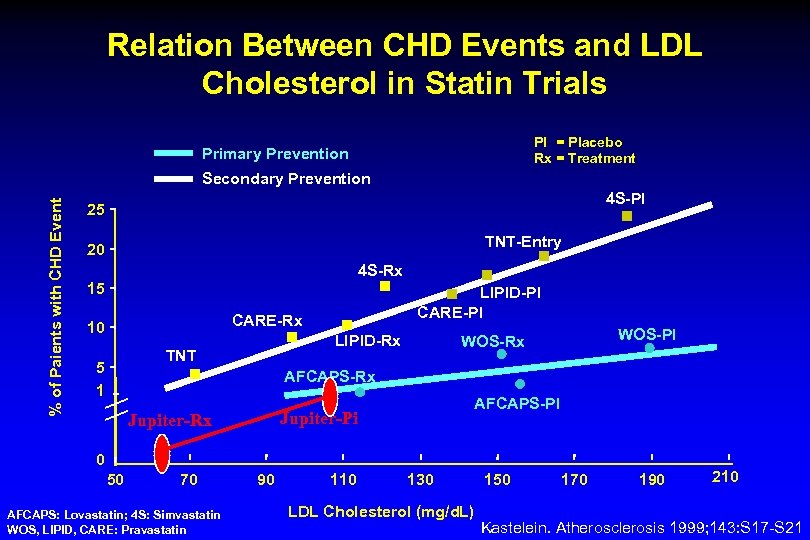
Relation Between CHD Events and LDL Cholesterol in Statin Trials PI = Placebo Rx = Treatment Primary Prevention % of Paients with CHD Event Secondary Prevention 4 S-PI 25 TNT-Entry 20 4 S-Rx 15 LIPID-PI CARE-Rx 10 LIPID-Rx TNT 5 1 WOS-PI WOS-Rx AFCAPS-PI Jupiter-Pi Jupiter-Rx 0 50 70 AFCAPS: Lovastatin; 4 S: Simvastatin WOS, LIPID, CARE: Pravastatin 90 110 130 LDL Cholesterol (mg/d. L) 150 170 190 210 Kastelein. Atherosclerosis 1999; 143: S 17 -S 21
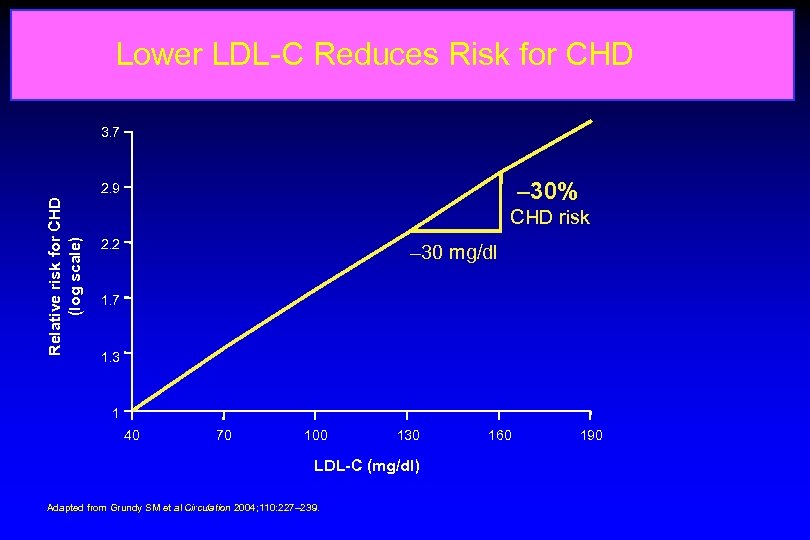
Lower LDL-C Reduces Risk for CHD 3. 7 – 30% Relative risk for CHD (log scale) 2. 9 CHD risk 2. 2 – 30 mg/dl 1. 7 1. 3 1 40 70 100 130 LDL-C (mg/dl) Adapted from Grundy SM et al Circulation 2004; 110: 227– 239. 160 190
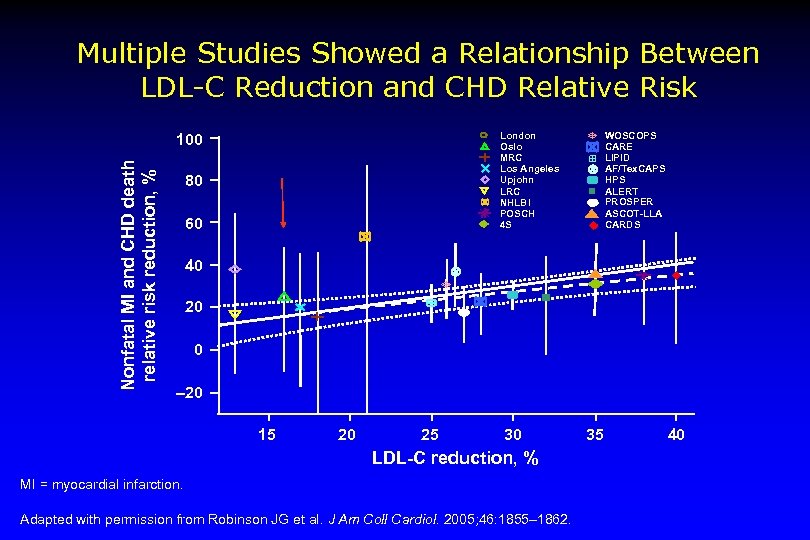
Multiple Studies Showed a Relationship Between LDL-C Reduction and CHD Relative Risk London Oslo MRC Los Angeles Upjohn LRC NHLBI POSCH 4 S Nonfatal MI and CHD death relative risk reduction, % 100 80 60 WOSCOPS CARE LIPID AF/Tex. CAPS HPS ALERT PROSPER ASCOT-LLA CARDS 40 20 0 – 20 15 20 25 30 LDL-C reduction, % MI = myocardial infarction. Adapted with permission from Robinson JG et al. J Am Coll Cardiol. 2005; 46: 1855– 1862. 35 40
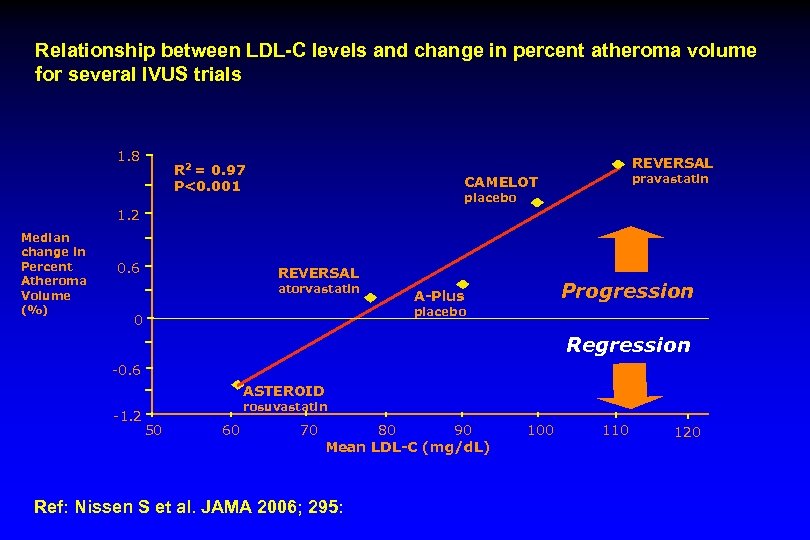
Relationship between LDL-C levels and change in percent atheroma volume for several IVUS trials 1. 8 REVERSAL R 2 = 0. 97 P<0. 001 pravastatin CAMELOT placebo 1. 2 Median change in Percent Atheroma Volume (%) 0. 6 REVERSAL atorvastatin Progression A-Plus placebo 0 Regression -0. 6 ASTEROID -1. 2 rosuvastatin 50 60 70 80 90 Mean LDL-C (mg/d. L) Ref: Nissen S et al. JAMA 2006; 295: 100 110 120
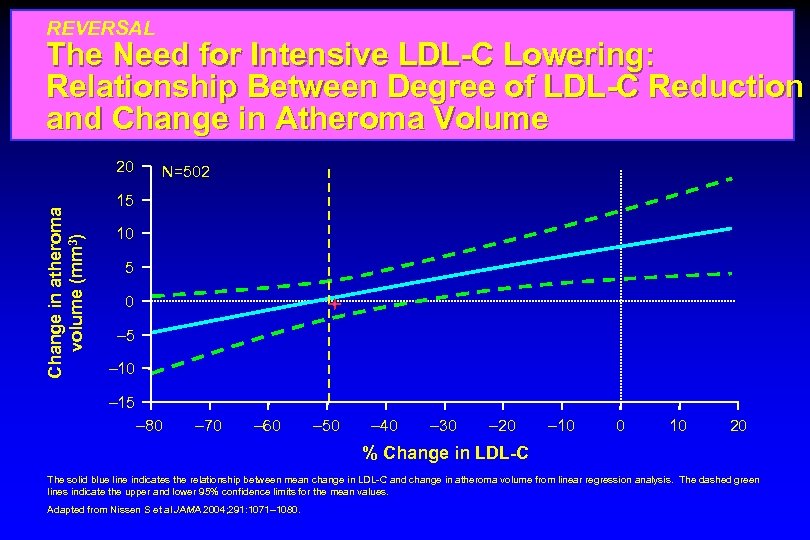
REVERSAL The Need for Intensive LDL-C Lowering: Relationship Between Degree of LDL-C Reduction and Change in Atheroma Volume Change in atheroma volume (mm 3) 20 N=502 15 10 5 0 – 5 – 10 – 15 – 80 – 70 – 60 – 50 – 40 – 30 – 20 – 10 0 10 20 % Change in LDL-C The solid blue line indicates the relationship between mean change in LDL-C and change in atheroma volume from linear regression analysis. The dashed green lines indicate the upper and lower 95% confidence limits for the mean values. Adapted from Nissen S et al JAMA 2004; 291: 1071– 1080.
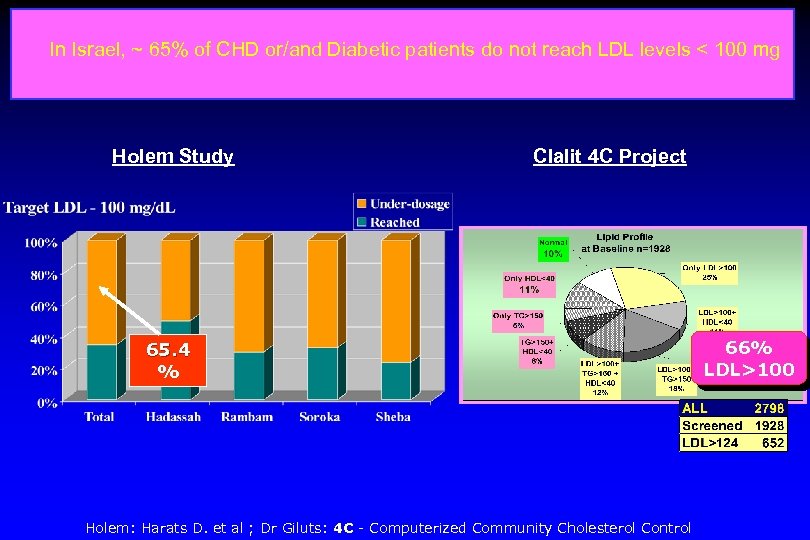
In Israel, ~ 65% of CHD or/and Diabetic patients do not reach LDL levels < 100 mg Holem Study Clalit 4 C Project 65. 4 % Holem: Harats D. et al ; Dr Giluts: 4 C - Computerized Community Cholesterol Control 66% LDL>100
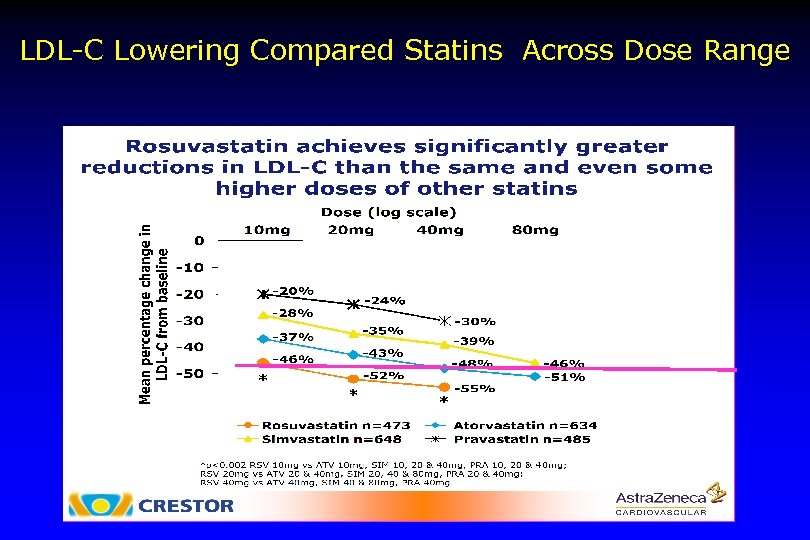
LDL-C Lowering Compared Statins Across Dose Range

Dual inhibition using Ezetrol and Statin provides greater Atherogenic burden relief ¨In add on trials ¨Vs Atrovaststin ¨Vs Rosuvastatin
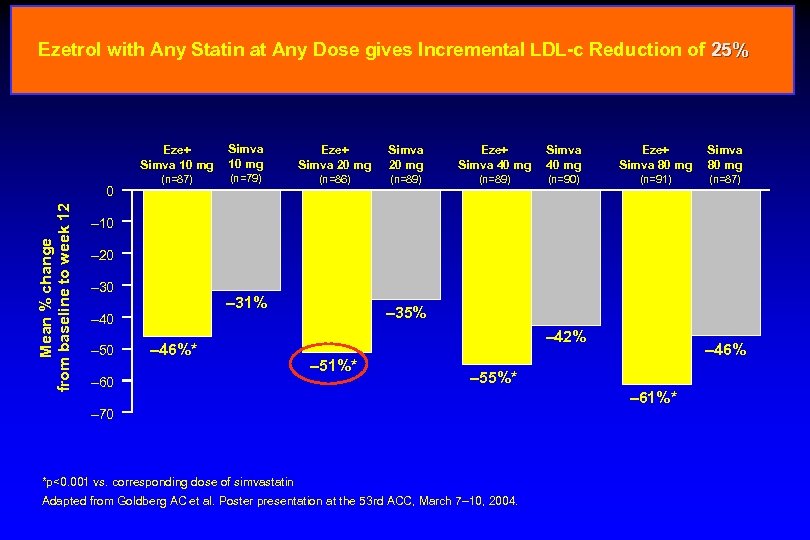
Ezetrol with Any Statin at Any Dose gives Incremental LDL-c Reduction of 25% Eze+ Simva 10 mg Mean % change from baseline to week 12 0 Simva 10 mg Eze+ Simva 20 mg Eze+ Simva 40 mg Eze+ Simva 80 mg (n=87) (n=79) (n=86) (n=89) (n=90) (n=91) (n=87) – 10 – 20 – 31% – 40 – 50 – 46%* – 60 – 35% – 42% – 51%* – 46% – 55%* – 70 *p<0. 001 vs. corresponding dose of simvastatin Adapted from Goldberg AC et al. Poster presentation at the 53 rd ACC, March 7– 10, 2004. – 61%*
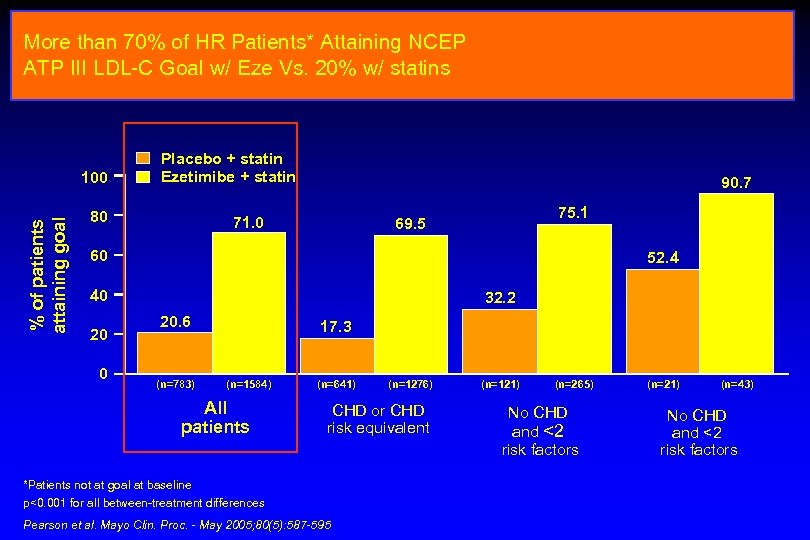
More than 70% of HR Patients* Attaining NCEP ATP III LDL-C Goal w/ Eze Vs. 20% w/ statins % of patients attaining goal 100 Placebo + statin Ezetimibe + statin 80 90. 7 71. 0 75. 1 69. 5 60 52. 4 40 20 0 32. 2 20. 6 (n=783) 17. 3 (n=1584) All patients (n=641) (n=1276) CHD or CHD risk equivalent *Patients not at goal at baseline p<0. 001 for all between-treatment differences Pearson et al. Mayo Clin. Proc. - May 2005; 80(5): 587 -595 (n=121) (n=265) No CHD and <2 risk factors (n=21) (n=43) No CHD and <2 risk factors
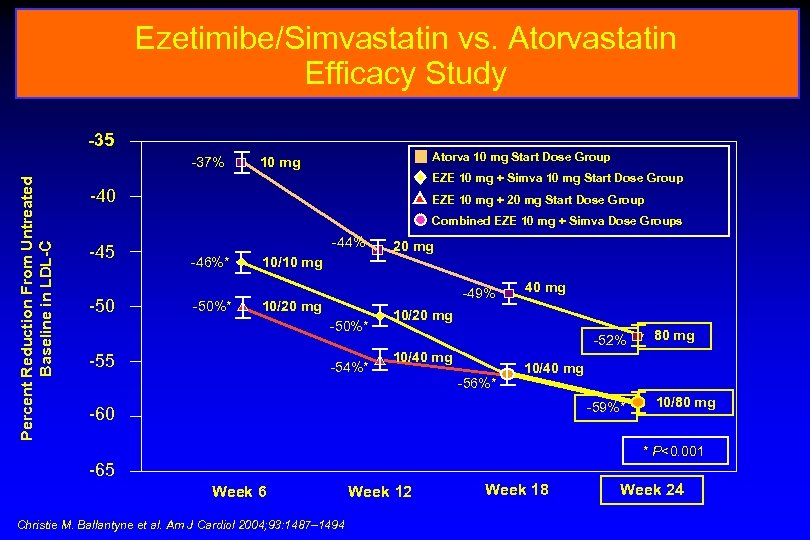
Ezetimibe/Simvastatin vs. Atorvastatin Efficacy Study -35 Percent Reduction From Untreated Baseline in LDL-C -37% Atorva 10 mg Start Dose Group 10 mg EZE 10 mg + Simva 10 mg Start Dose Group -40 EZE 10 mg + 20 mg Start Dose Group Combined EZE 10 mg + Simva Dose Groups -45 -50 -44% -46%* -50%* 20 mg 10/10 mg -49% 10/20 mg -50%* -55 -54%* 40 mg 10/20 mg -52% 10/40 mg -56%* 80 mg 10/40 mg -59%* -60 10/80 mg * P<0. 001 -65 Week 6 Christie M. Ballantyne et al. Am J Cardiol 2004; 93: 1487– 1494 Week 12 Week 18 Week 24
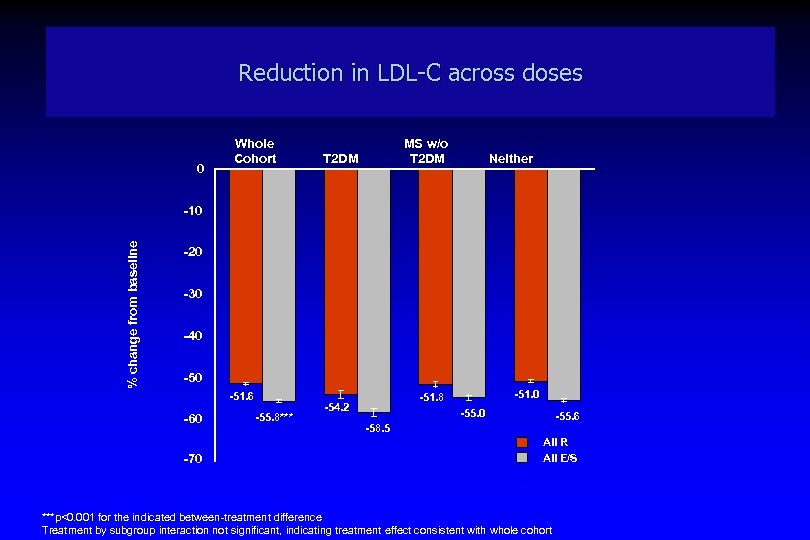
Reduction in LDL-C across doses 0 Whole MS w/o Cohort T 2 DM Neither % change from baseline -10 -20 -30 -40 -51. 6 -60 -70 -55. 8*** -51. 0 -51. 8 -54. 2 -55. 0 -55. 6 -58. 5 All R All E/S ***p<0. 001 for the indicated between-treatment difference Treatment by subgroup interaction not significant, indicating treatment effect consistent with whole cohort
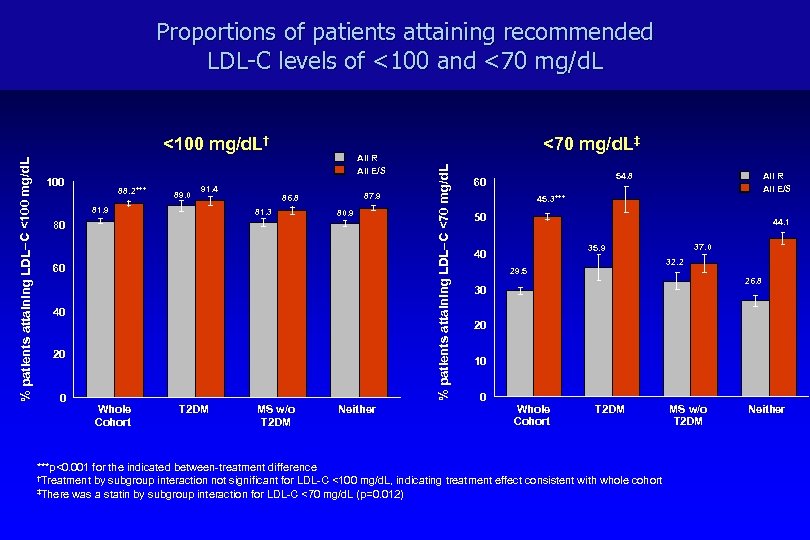
Proportions of patients attaining recommended LDL-C levels of <100 and <70 mg/d. L All R All E/S 100 88. 2*** 89. 0 91. 4 81. 9 87. 9 86. 8 81. 3 80. 9 80 60 40 20 0 Whole Cohort T 2 DM MS w/o T 2 DM Neither % patients attaining LDL–C <70 mg/d. L –C <100 mg/d. L % patients attaining LDL <100 mg/d. L† <70 mg/d. L‡ 54. 8 60 All R All E/S 45. 3*** 50 44. 1 37. 0 35. 9 40 32. 2 29. 5 26. 8 30 20 10 0 Whole Cohort T 2 DM ***p<0. 001 for the indicated between-treatment difference †Treatment by subgroup interaction not significant for LDL-C <100 mg/d. L, indicating treatment effect consistent with whole cohort ‡There was a statin by subgroup interaction for LDL-C <70 mg/d. L (p=0. 012) MS w/o T 2 DM Neither
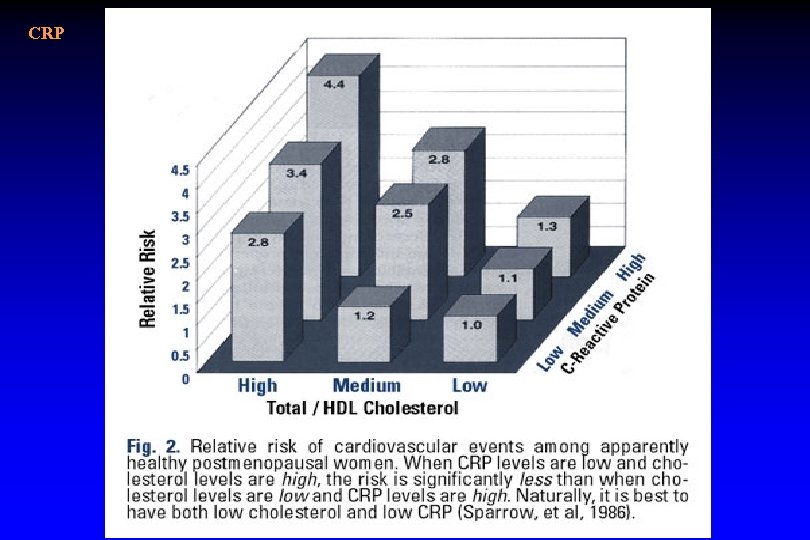
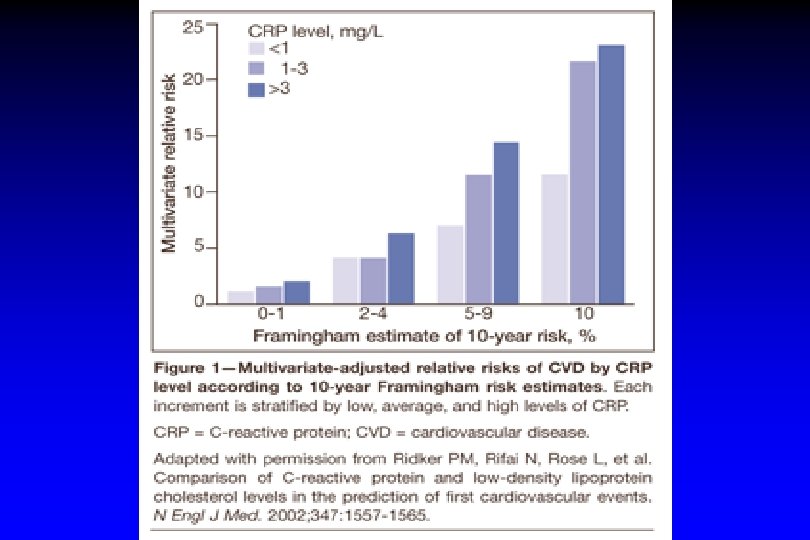
CRP
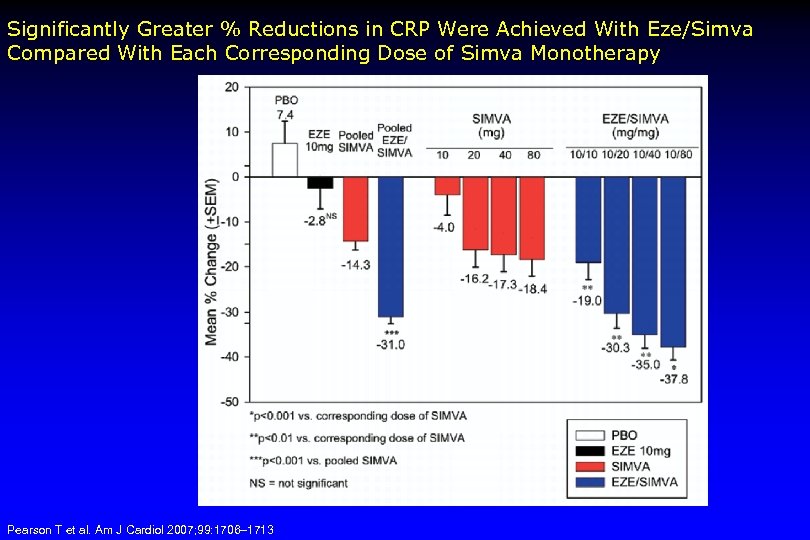
Significantly Greater % Reductions in CRP Were Achieved With Eze/Simva Compared With Each Corresponding Dose of Simva Monotherapy Pearson T et al. Am J Cardiol 2007; 99: 1706– 1713

METABOLIC SYNDROME

The evolution of mankind… 2. 5 mil. years 100 years
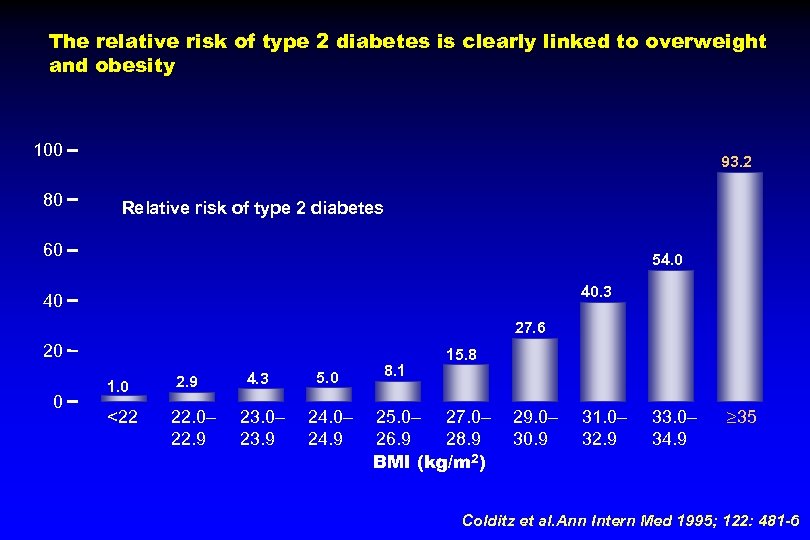
The relative risk of type 2 diabetes is clearly linked to overweight and obesity 100 80 93. 2 Relative risk of type 2 diabetes 60 54. 0 40. 3 40 27. 6 20 0 1. 0 2. 9 <22 22. 0– 22. 9 4. 3 5. 0 23. 0– 23. 9 24. 0– 24. 9 8. 1 15. 8 25. 0– 27. 0– 26. 9 28. 9 BMI (kg/m 2) 29. 0– 30. 9 31. 0– 32. 9 33. 0– 34. 9 35 Colditz et al. Ann Intern Med 1995; 122: 481 -6

What is an Atherogenic lipid profile? Atherogenic triad(? ): TG HDL, Small, dense LDL, Remnants Apo B 48 Apo B 100 Apo A
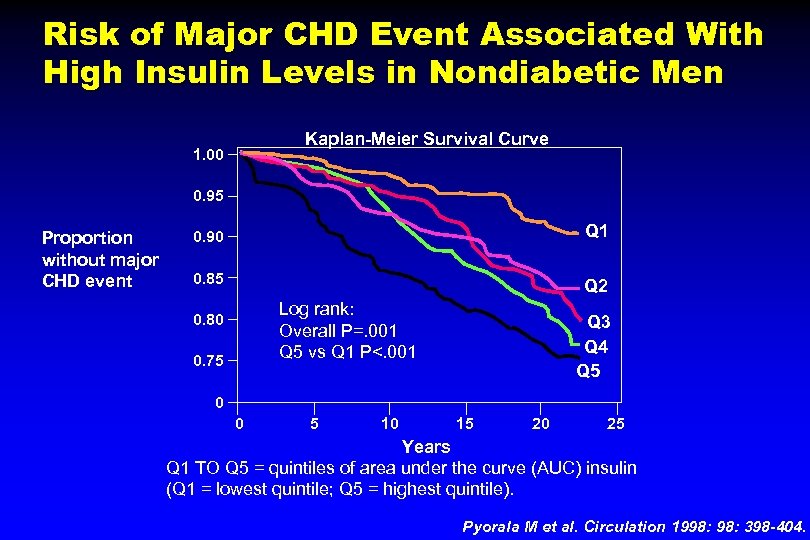
Risk of Major CHD Event Associated With High Insulin Levels in Nondiabetic Men 1. 00 Kaplan-Meier Survival Curve 0. 95 Proportion without major CHD event 0. 90 Q 1 0. 85 Q 2 0. 80 0. 75 Log rank: Overall P=. 001 Q 5 vs Q 1 P<. 001 Q 3 Q 4 Q 5 0 0 5 10 15 20 25 Years Q 1 TO Q 5 = quintiles of area under the curve (AUC) insulin (Q 1 = lowest quintile; Q 5 = highest quintile). Pyorala M et al. Circulation 1998: 398 -404.
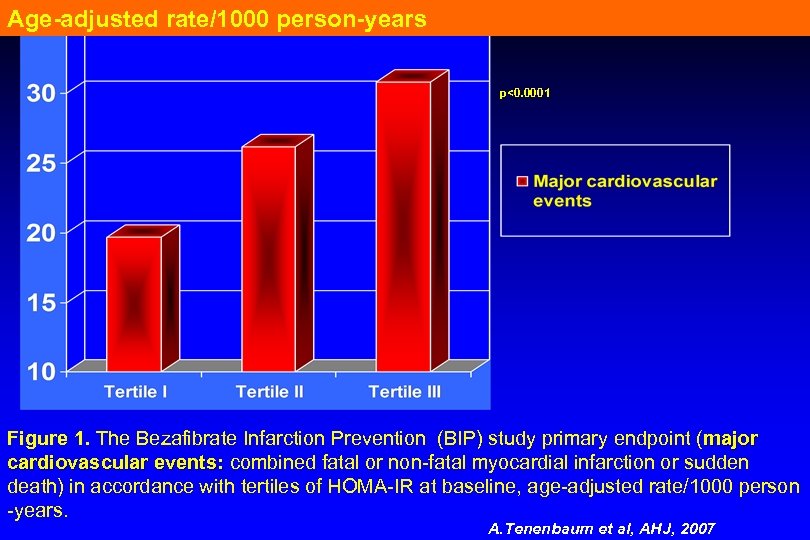
Age-adjusted rate/1000 person-years p<0. 0001 Figure 1. The Bezafibrate Infarction Prevention (BIP) study primary endpoint (major cardiovascular events: combined fatal or non-fatal myocardial infarction or sudden death) in accordance with tertiles of HOMA-IR at baseline, age-adjusted rate/1000 person -years. A. Tenenbaum et al, AHJ, 2007
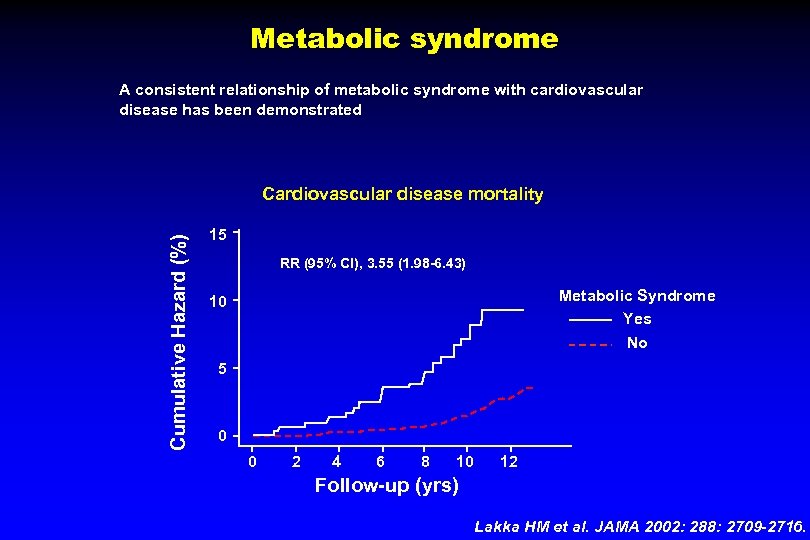
Metabolic syndrome A consistent relationship of metabolic syndrome with cardiovascular disease has been demonstrated Cumulative Hazard (%) Cardiovascular disease mortality 15 RR (95% CI), 3. 55 (1. 98 -6. 43) Metabolic Syndrome Yes No 10 5 0 0 2 4 6 8 10 12 Follow-up (yrs) Lakka HM et al. JAMA 2002: 288: 2709 -2716.

Current therapeutic use of statins as monotherapy is still leaving many patients with metabolic syndrome and mixed atherogenic dyslipidemia at high risk for coronary events.

Combined atherogenic dyslipidemia in metabolic syndrome and type 2 diabetes: Pharmacological options in addition to statin Nicotinic acid (or niacin) Fibrates Bile acid sequestrants CETP inhibition (Torcetrapib) Ezetimibe

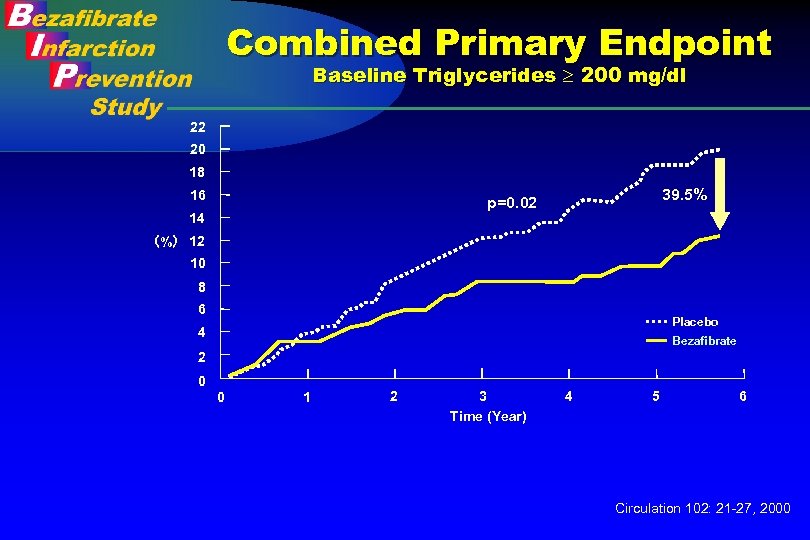
Combined Primary Endpoint Baseline Triglycerides 200 mg/dl 22 20 18 16 39. 5% p=0. 02 14 (%) 12 10 8 6 Placebo 4 Bezafibrate 2 0 0 1 2 3 Time (Year) 4 5 6 Circulation 102: 21 -27, 2000

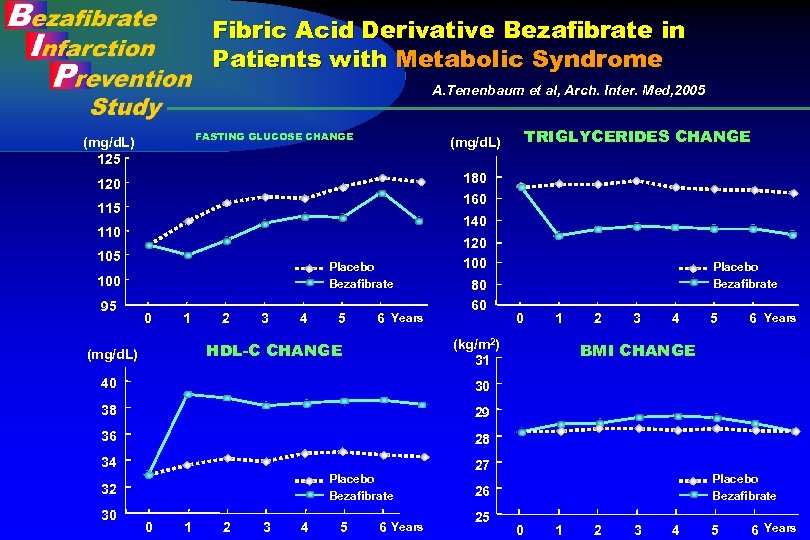
Fibric Acid Derivative Bezafibrate in Patients with Metabolic Syndrome A. Tenenbaum et al, Arch. Inter. Med, 2005 FASTING GLUCOSE CHANGE (mg/d. L) 125 180 120 160 115 140 110 105 Placebo Bezafibrate 100 95 TRIGLYCERIDES CHANGE (mg/d. L) 0 1 2 3 4 5 6 Years 80 60 Placebo Bezafibrate 0 1 (kg/m 2) 31 HDL-C CHANGE (mg/d. L) 120 100 40 4 5 6 Years BMI CHANGE 29 36 3 30 38 2 28 34 Placebo Bezafibrate 32 30 0 1 2 3 4 5 6 Years 27 Placebo Bezafibrate 26 25 0 1 2 3 4 5 6 Years
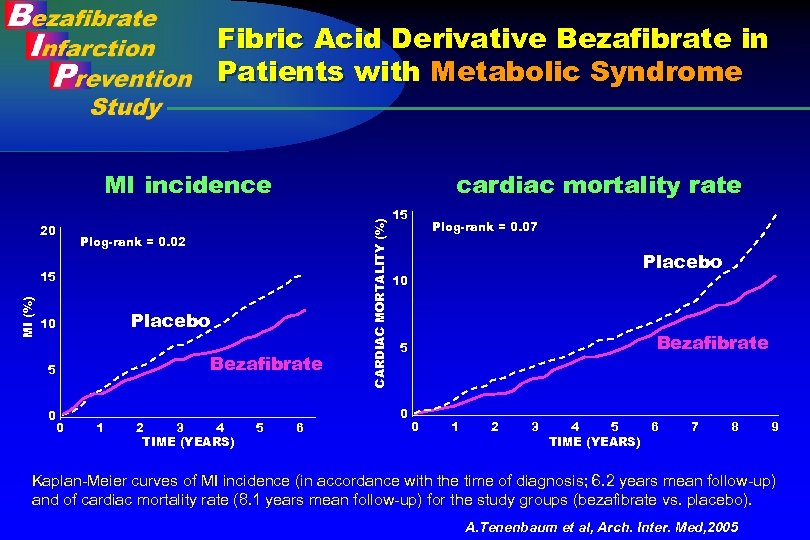
Fibric Acid Derivative Bezafibrate in Patients with Metabolic Syndrome 20 cardiac mortality rate Plog-rank = 0. 02 MI (%) 15 Placebo 10 Bezafibrate 5 0 0 1 2 3 4 TIME (YEARS) 5 6 CARDIAC MORTALITY (%) MI incidence 15 Plog-rank = 0. 07 Placebo 10 Bezafibrate 5 0 0 1 2 3 4 5 6 TIME (YEARS) 7 8 9 Kaplan-Meier curves of MI incidence (in accordance with the time of diagnosis; 6. 2 years mean follow-up) and of cardiac mortality rate (8. 1 years mean follow-up) for the study groups (bezafibrate vs. placebo). A. Tenenbaum et al, Arch. Inter. Med, 2005
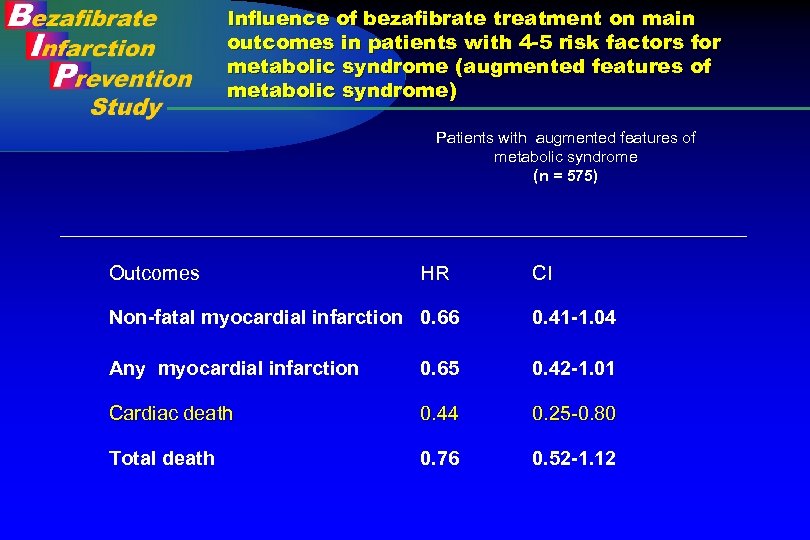
Influence of bezafibrate treatment on main outcomes in patients with 4 -5 risk factors for metabolic syndrome (augmented features of metabolic syndrome) Patients with augmented features of metabolic syndrome (n = 575) Outcomes HR CI Non-fatal myocardial infarction 0. 66 0. 41 -1. 04 Any myocardial infarction 0. 65 0. 42 -1. 01 Cardiac death 0. 44 0. 25 -0. 80 Total death 0. 76 0. 52 -1. 12
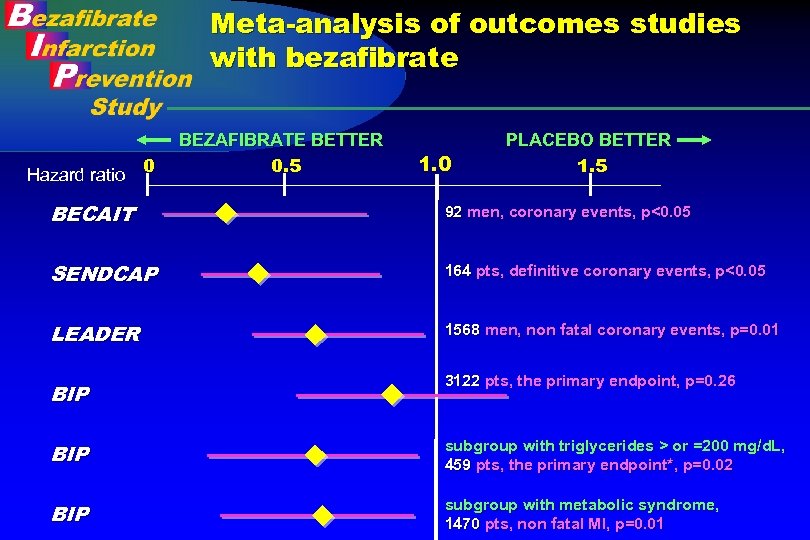
Meta-analysis of outcomes studies with bezafibrate Hazard ratio 0 BEZAFIBRATE BETTER 0. 5 1. 0 PLACEBO BETTER 1. 5 BECAIT 92 men, coronary events, p<0. 05 SENDCAP 164 pts, definitive coronary events, p<0. 05 LEADER 1568 men, non fatal coronary events, p=0. 01 BIP 3122 pts, the primary endpoint, p=0. 26 BIP subgroup with triglycerides > or =200 mg/d. L, 459 pts, the primary endpoint*, p=0. 02 BIP subgroup with metabolic syndrome, 1470 pts, non fatal MI, p=0. 01
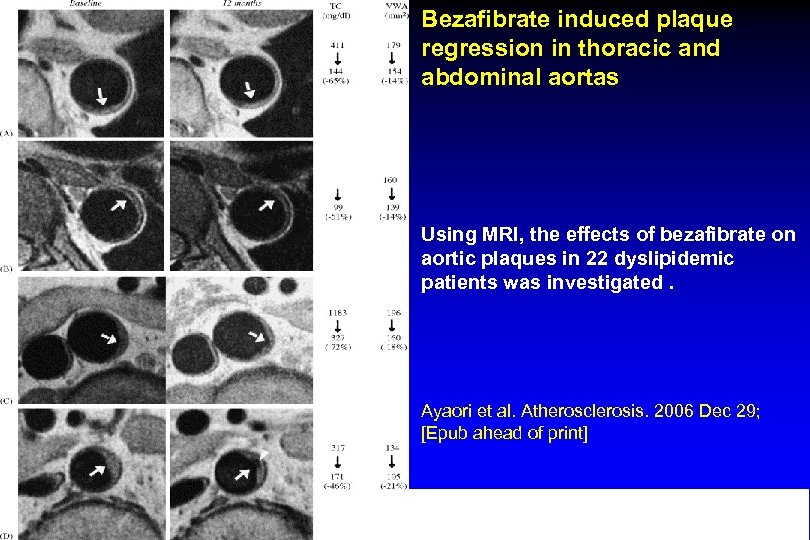
Bezafibrate induced plaque regression in thoracic and abdominal aortas Using MRI, the effects of bezafibrate on aortic plaques in 22 dyslipidemic patients was investigated. Ayaori et al. Atherosclerosis. 2006 Dec 29; [Epub ahead of print] MRI images at baseline and after 1 year of bezafibrate treatment.
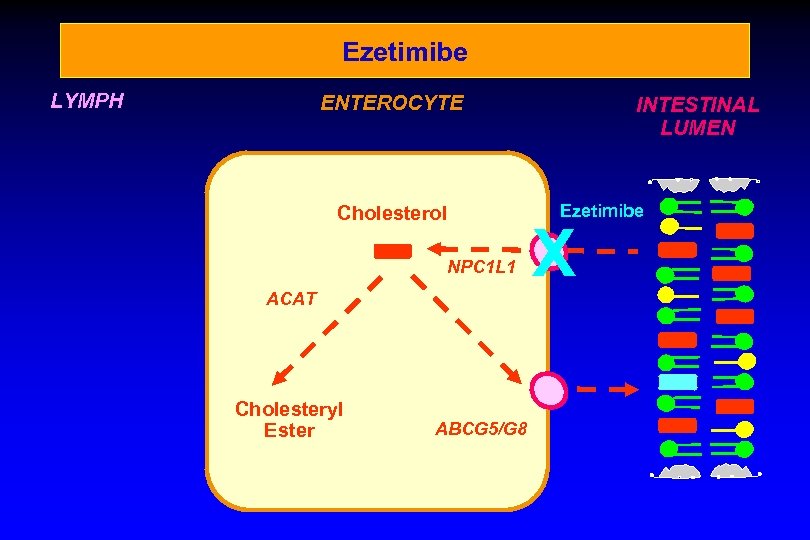
Ezetimibe LYMPH ENTEROCYTE Cholesterol NPC 1 L 1 ACAT Cholesteryl Ester ABCG 5/G 8 INTESTINAL LUMEN Ezetimibe X
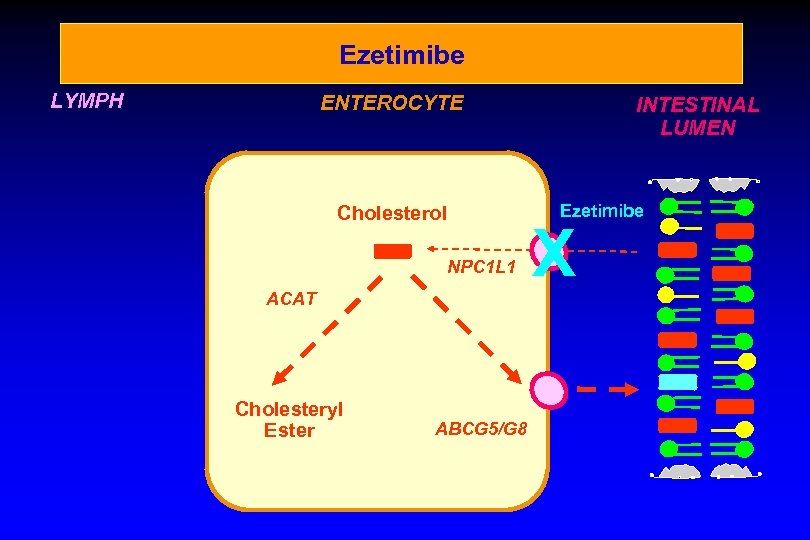
Ezetimibe LYMPH ENTEROCYTE Cholesterol NPC 1 L 1 ACAT Cholesteryl Ester ABCG 5/G 8 INTESTINAL LUMEN Ezetimibe X
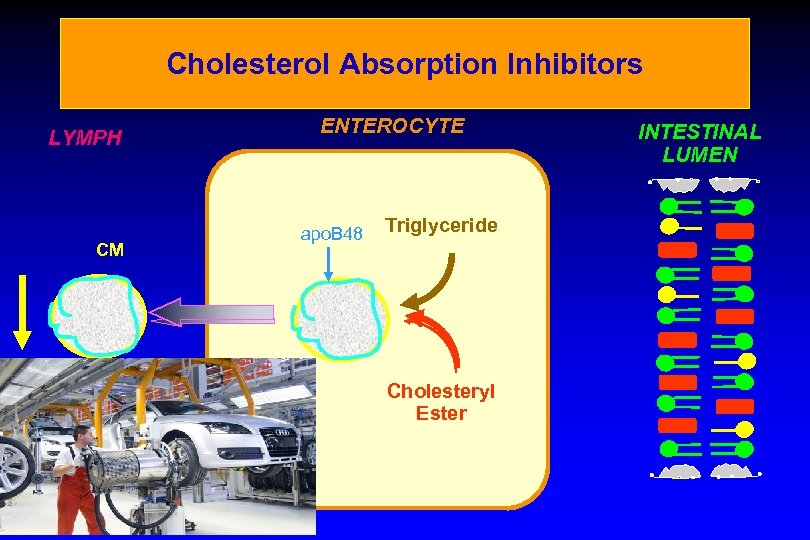
Cholesterol Absorption Inhibitors LYMPH CM ENTEROCYTE apo. B 48 Triglyceride Cholesteryl Ester INTESTINAL LUMEN
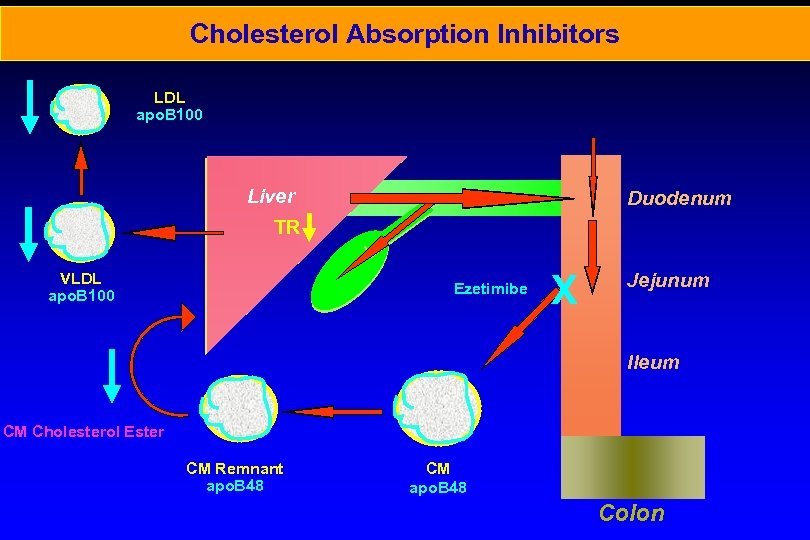
Cholesterol Absorption Inhibitors LDL apo. B 100 Liver Duodenum TR VLDL apo. B 100 Ezetimibe X Jejunum Ileum CM Cholesterol Ester CM Remnant apo. B 48 CM apo. B 48 Colon
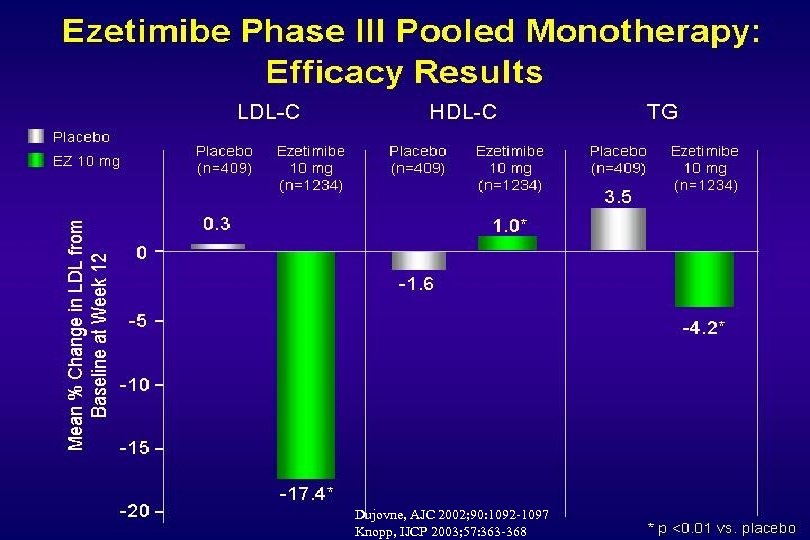
Dujovne, AJC 2002; 90: 1092 -1097 Knopp, IJCP 2003; 57: 363 -368
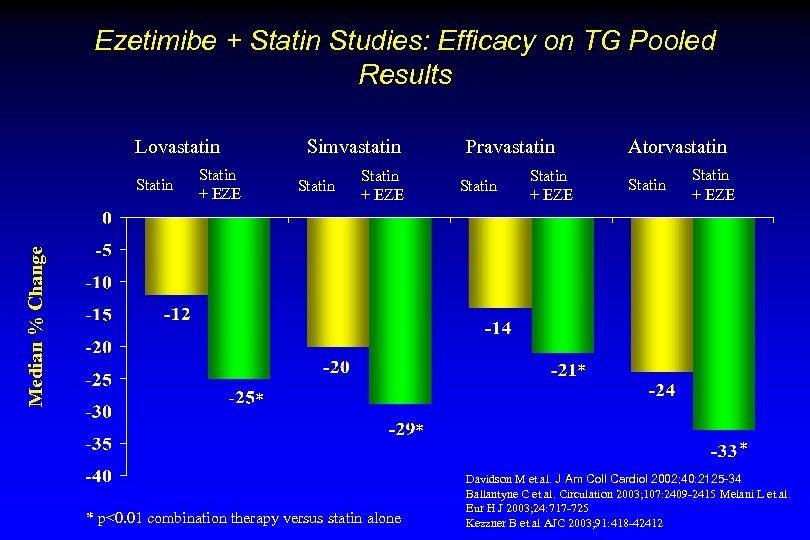
Ezetimibe + Statin Studies: Efficacy on TG Pooled Results Lovastatin Median % Change Statin Simvastatin Statin + EZE Statin Pravastatin Statin + EZE Statin Atorvastatin Statin + EZE * * p<0. 01 combination therapy versus statin alone * Davidson M et al. J Am Coll Cardiol 2002; 40: 2125 -34 Ballantyne C et al. Circulation 2003; 107: 2409 -2415 Melani L et al. Eur H J 2003; 24: 717 -725 Kezzner B et al AJC 2003; 91: 418 -42412
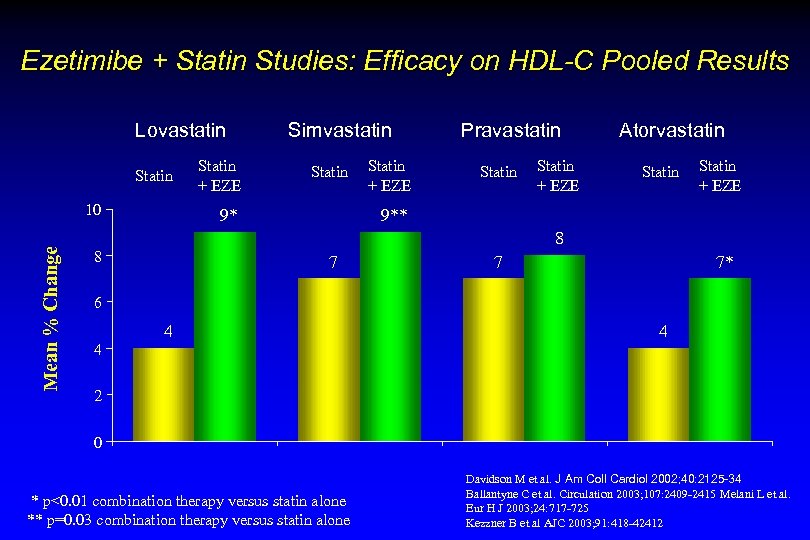
Ezetimibe + Statin Studies: Efficacy on HDL-C Pooled Results Lovastatin Statin Mean % Change 10 Statin + EZE Simvastatin Statin 9* Statin + EZE Pravastatin Statin + EZE Atorvastatin Statin + EZE 9** 8 8 7 7 7* 6 4 4 4 2 0 * p<0. 01 combination therapy versus statin alone ** p=0. 03 combination therapy versus statin alone Davidson M et al. J Am Coll Cardiol 2002; 40: 2125 -34 Ballantyne C et al. Circulation 2003; 107: 2409 -2415 Melani L et al. Eur H J 2003; 24: 717 -725 Kezzner B et al AJC 2003; 91: 418 -42412
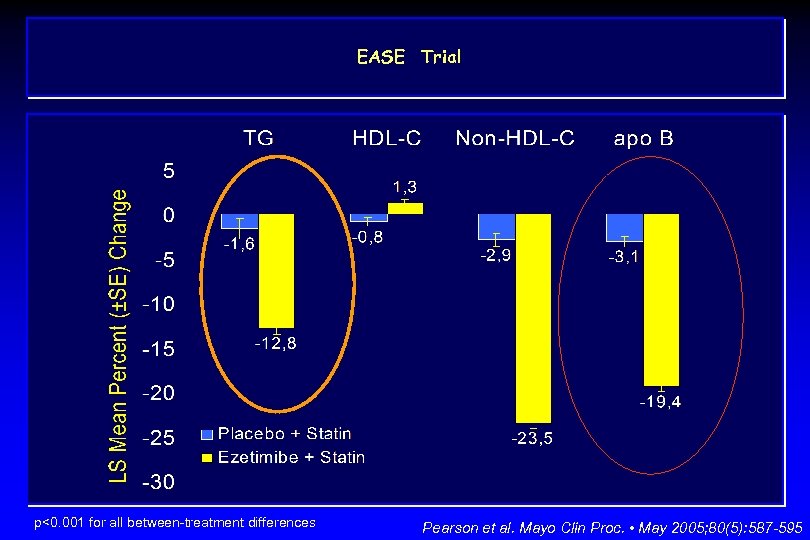
EASE Trial p<0. 001 for all between-treatment differences Pearson et al. Mayo Clin Proc. • May 2005; 80(5): 587 -595
Ezetimibe Clinical Trials Update

Surrogate cubs In a California zoo, a mother tiger gave birth to a rare set of triplet tiger cubs, but they died shortly after birth. The veterinarians felt that the loss of her litter had caused the tigress to fall into a depression. The veterinarians decided to try something that had never been tried in a zoo environment. The only "orphans" that could be found quickly, were a litter of wiener pigs.

“Surrogate”-endpoints Evidences Carotid IMT Trials Examining Clinical Efficacy of Statin & Ezetimibe Trial Patient population N Endpoint Treatment regimen ENHANCE Heterozygote Familial Hyperchol. 720 Carotid IMT 10/80 vs Simvastatin SANDS Diabetes 499 Carotid IMT Statins / statins + EZE
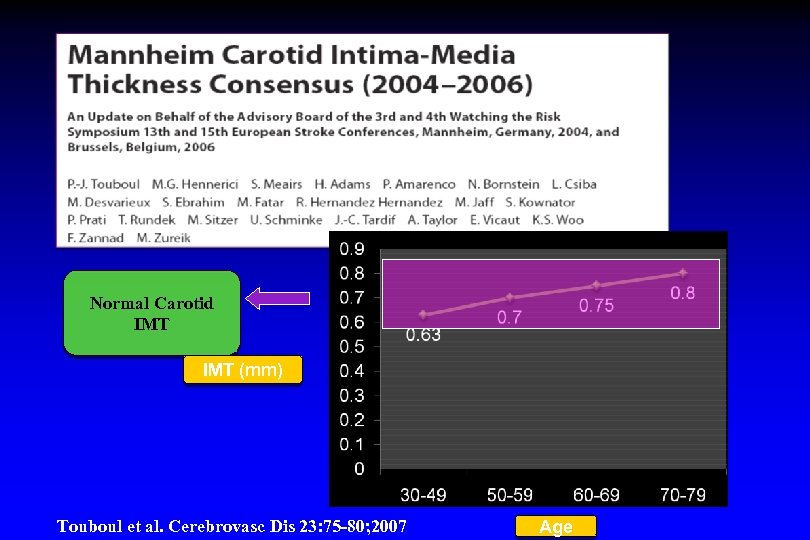
Normal Carotid IMT (mm) Touboul et al. Cerebrovasc Dis 23: 75 -80; 2007 Age
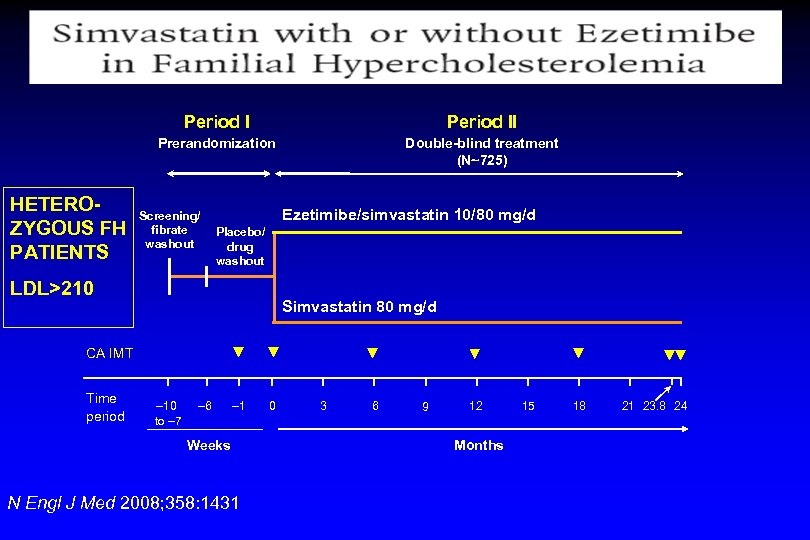
Period I Period II Prerandomization Double-blind treatment (N~725) HETEROZYGOUS FH PATIENTS Screening/ fibrate washout Ezetimibe/simvastatin 10/80 mg/d Placebo/ drug washout LDL>210 Simvastatin 80 mg/d CA IMT Time period – 10 to – 7 – 6 – 1 Weeks N Engl J Med 2008; 358: 1431 0 3 6 9 12 Months 15 18 21 23. 8 24
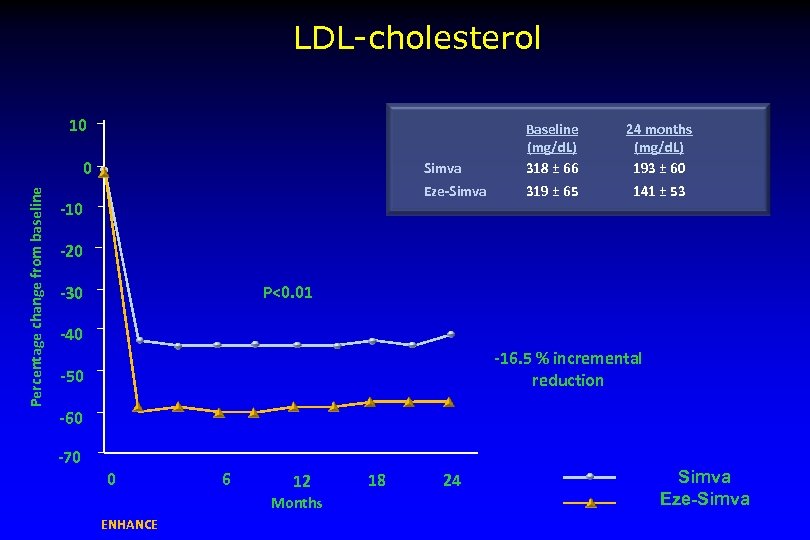
LDL-cholesterol 10 Simva Percentage change from baseline -10 24 months (mg/d. L) 193 ± 60 Eze-Simva 0 Baseline (mg/d. L) 318 ± 66 319 ± 65 141 ± 53 -20 P<0. 01 -30 -40 -16. 5 % incremental reduction -50 -60 -70 0 6 12 Months ENHANCE 18 24 Simva Eze-Simva
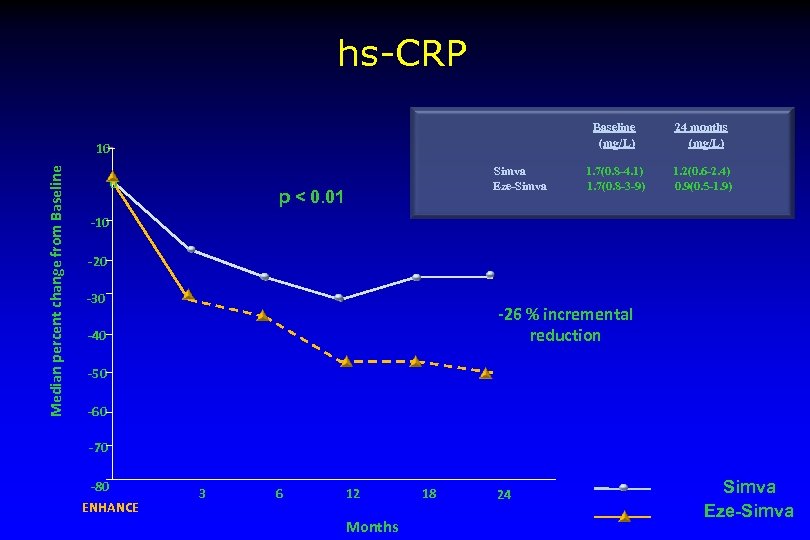
hs-CRP Baseline (mg/L) Median percent change from Baseline 10 0 Simva Eze-Simva p < 0. 01 24 months (mg/L) 1. 7(0. 8 -4. 1) 1. 7(0. 8 -3 -9) 1. 2(0. 6 -2. 4) 0. 9(0. 5 -1. 9) -10 -20 -30 -26 % incremental reduction -40 -50 -60 -70 -80 ENHANCE 3 6 12 Months 18 24 Simva Eze-Simva
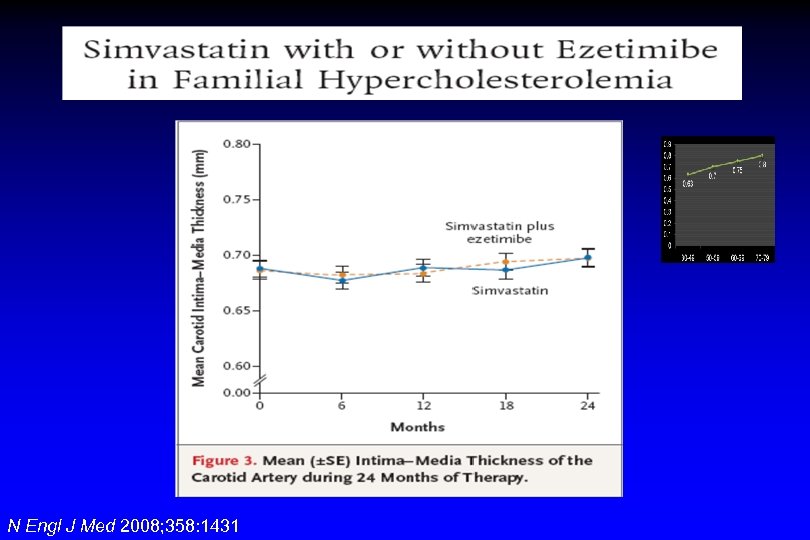
N Engl J Med 2008; 358: 1431

SANDS (Stop Atherosclerosis in Native Diabetics Study) (JAMA 2008; 299: 1678 -89) Effect of Statins Alone versus Statin plus Ezetimibe on Carotid Atherosclerosis in Type 2 Diabetes (J Am Coll Cardiol 2008
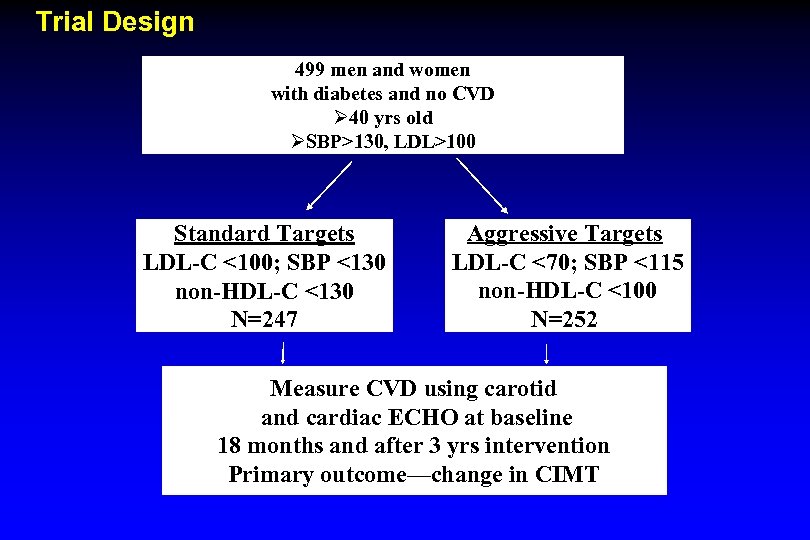
Trial Design 499 men and women with diabetes and no CVD Ø 40 yrs old ØSBP>130, LDL>100 Standard Targets LDL-C <100; SBP <130 non-HDL-C <130 N=247 Aggressive Targets LDL-C <70; SBP <115 non-HDL-C <100 N=252 Measure CVD using carotid and cardiac ECHO at baseline 18 months and after 3 yrs intervention Primary outcome—change in CIMT
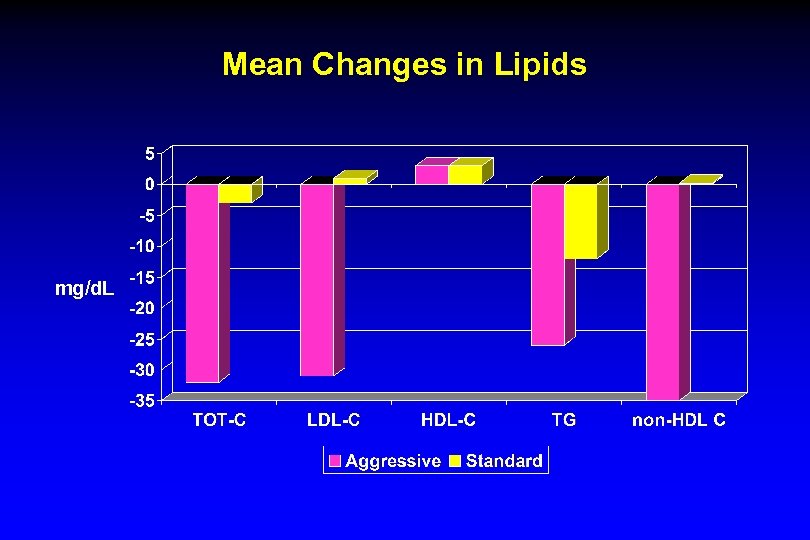
Mean Changes in Lipids mg/d. L

Change in IMT by Ezetimibe +/- 102 mg/d. L mm 108 to 78 mg/d. L -31% 102 mg/d. L 101 to 68 mg/d. L -32% Baseline c. IMT = 0. 81 * p<0. 001 for both E + and E- compared to Standard
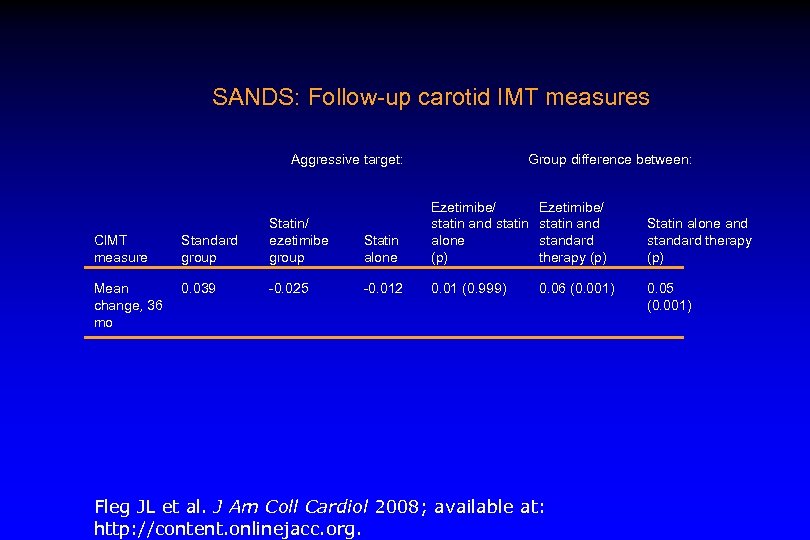
SANDS: Follow-up carotid IMT measures Aggressive target: CIMT measure Standard group Statin/ ezetimibe group Mean change, 36 mo 0. 039 -0. 025 Group difference between: Statin alone Ezetimibe/ statin and statin alone (p) -0. 012 0. 01 (0. 999) Ezetimibe/ statin and standard therapy (p) 0. 06 (0. 001) Fleg JL et al. J Am Coll Cardiol 2008; available at: http: //content. onlinejacc. org. Statin alone and standard therapy (p) 0. 05 (0. 001)

ARBITER 6
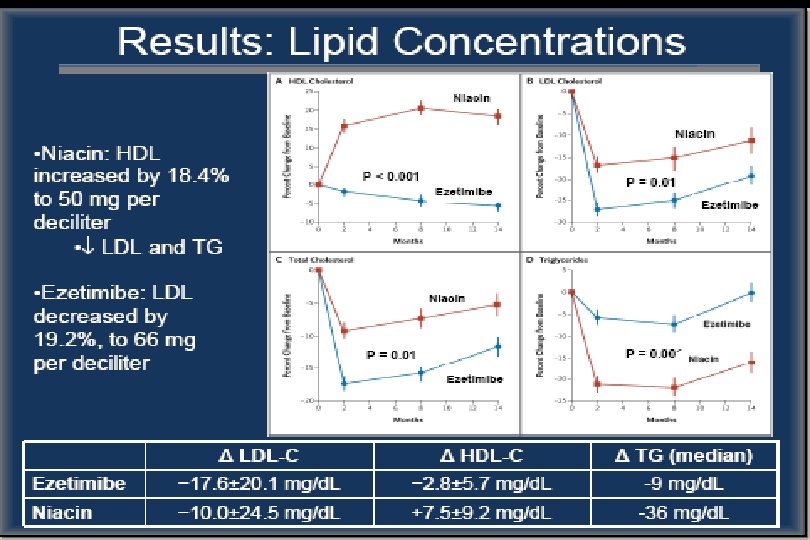
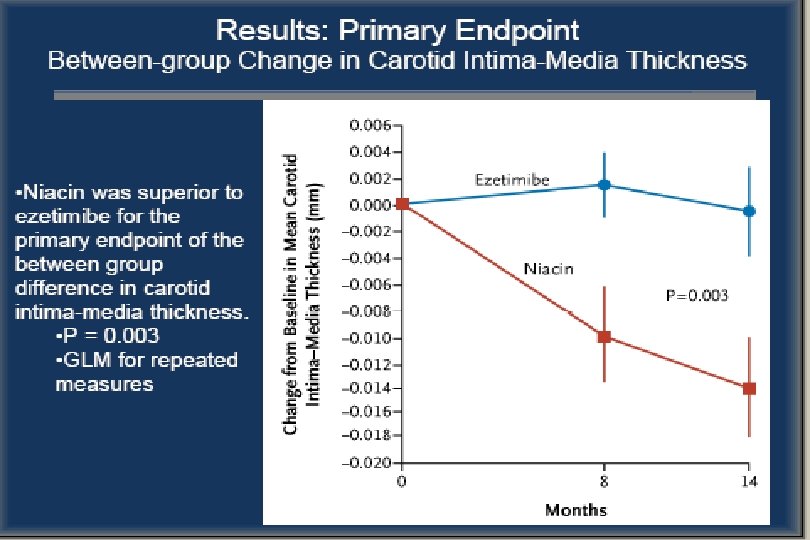
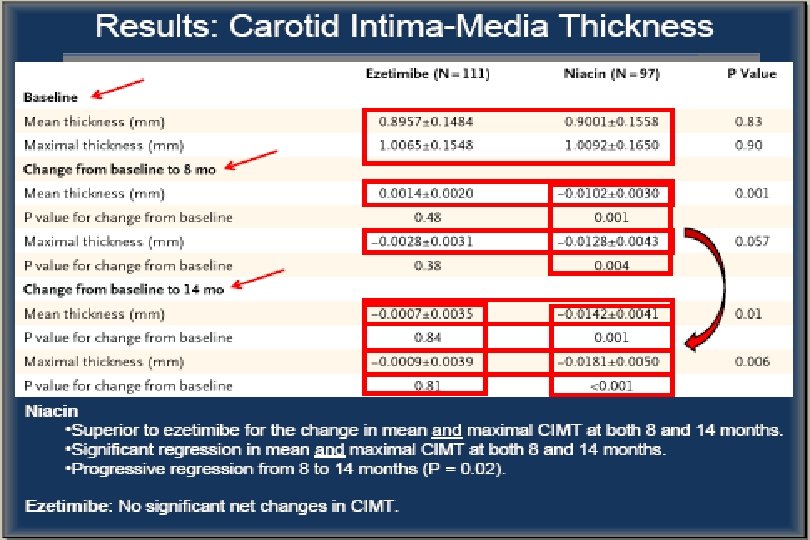
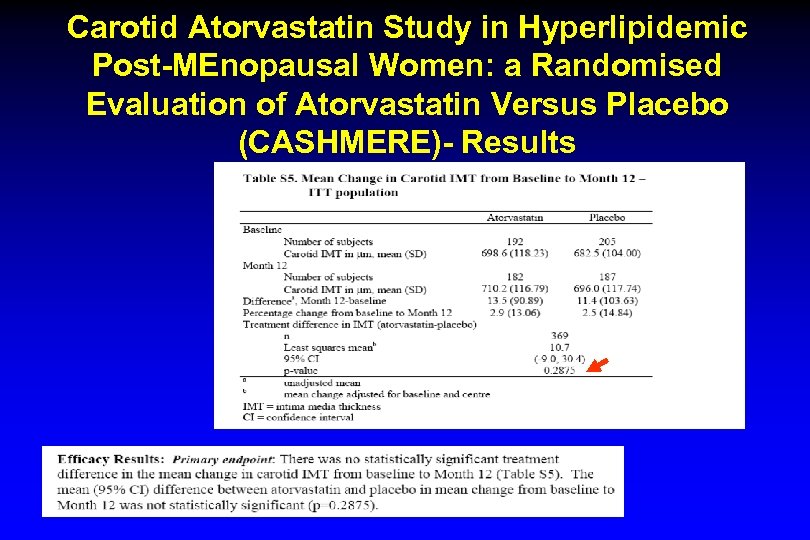
Carotid Atorvastatin Study in Hyperlipidemic Post-MEnopausal Women: a Randomised Evaluation of Atorvastatin Versus Placebo (CASHMERE)- Results

Evidence-Based Medicine: Cardiovascular Outcome Trials
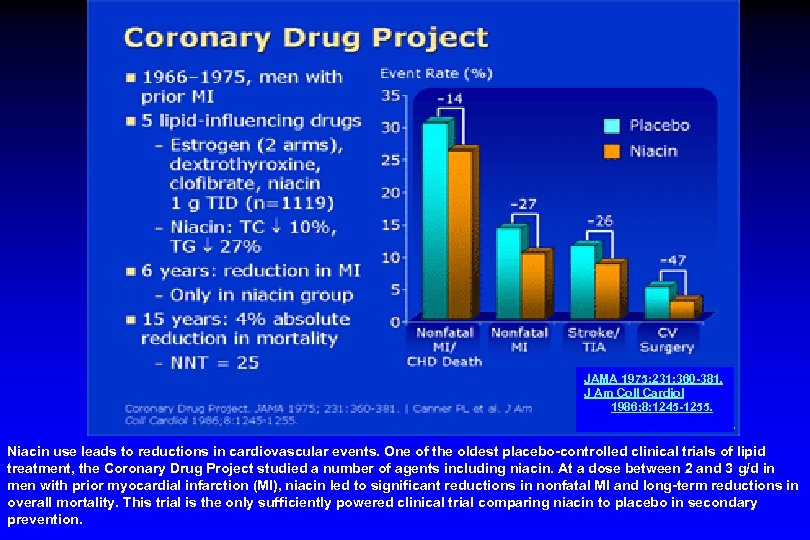
JAMA 1975; 231: 360 -381. J Am Coll Cardiol 1986; 8: 1245 -1255. Niacin use leads to reductions in cardiovascular events. One of the oldest placebo-controlled clinical trials of lipid treatment, the Coronary Drug Project studied a number of agents including niacin. At a dose between 2 and 3 g/d in men with prior myocardial infarction (MI), niacin led to significant reductions in nonfatal MI and long-term reductions in overall mortality. This trial is the only sufficiently powered clinical trial comparing niacin to placebo in secondary prevention.
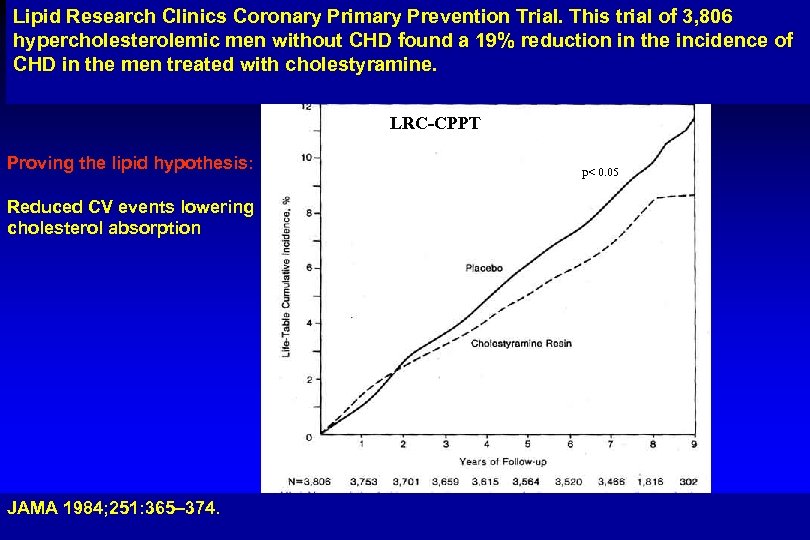
Lipid Research Clinics Coronary Primary Prevention Trial. This trial of 3, 806 hypercholesterolemic men without CHD found a 19% reduction in the incidence of CHD in the men treated with cholestyramine. LRC-CPPT Proving the lipid hypothesis: Reduced CV events lowering cholesterol absorption JAMA 1984; 251: 365– 374. p< 0. 05
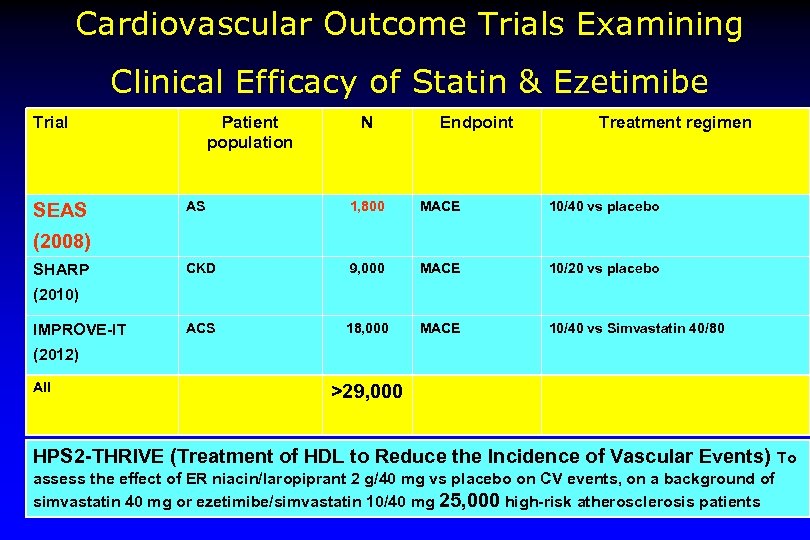
Cardiovascular Outcome Trials Examining Clinical Efficacy of Statin & Ezetimibe Trial SEAS Patient population N Endpoint Treatment regimen AS 1, 800 MACE 10/40 vs placebo CKD 9, 000 MACE 10/20 vs placebo ACS 18, 000 MACE 10/40 vs Simvastatin 40/80 (2008) SHARP (2010) IMPROVE-IT (2012) All >29, 000 HPS 2 -THRIVE (Treatment of HDL to Reduce the Incidence of Vascular Events) To assess the effect of ER niacin/laropiprant 2 g/40 mg vs placebo on CV events, on a background of simvastatin 40 mg or ezetimibe/simvastatin 10/40 mg 25, 000 high-risk atherosclerosis patients


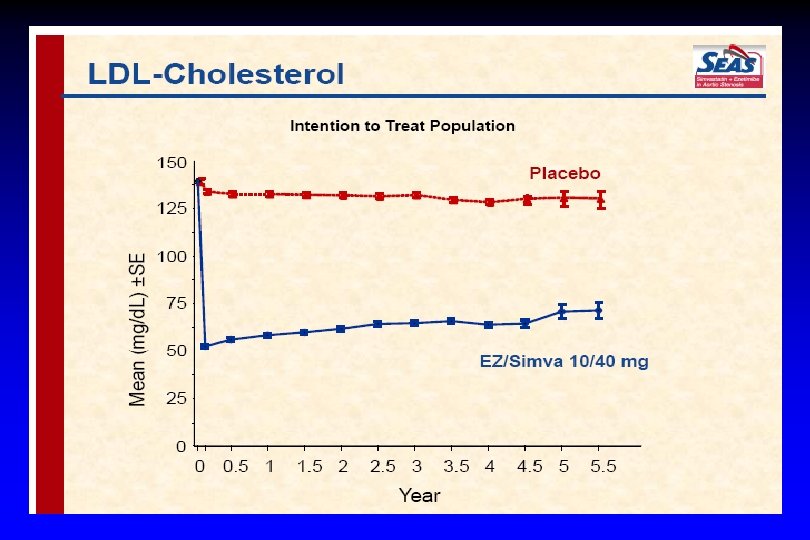
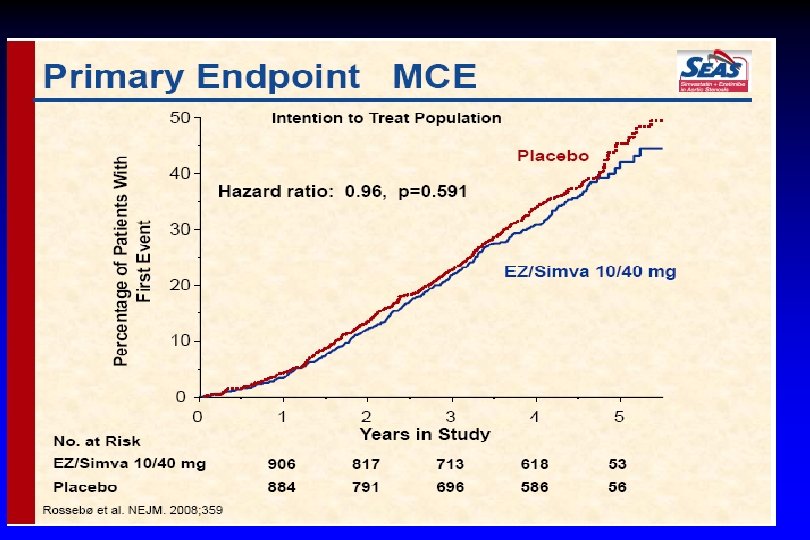
• Randomized, double-blind trial involving 1873 patients with mild-tomoderate, asymptomatic aortic stenosis. ¨ The patients received either 40 mg of simvastatin plus 10 mg of ezetimibe or placebo daily.

The primary outcome was a composite of major cardiovascular events, including death from cardiovascular causes, aortic-valve replacement, nonfatal myocardial infarction, hospitalization for unstable angina pectoris, heart failure, coronary-artery bypass grafting, percutaneous coronary intervention, and nonhemorrhagic stroke. Secondary outcomes were events related to aortic-valve stenosis and ischemic cardiovascular events.
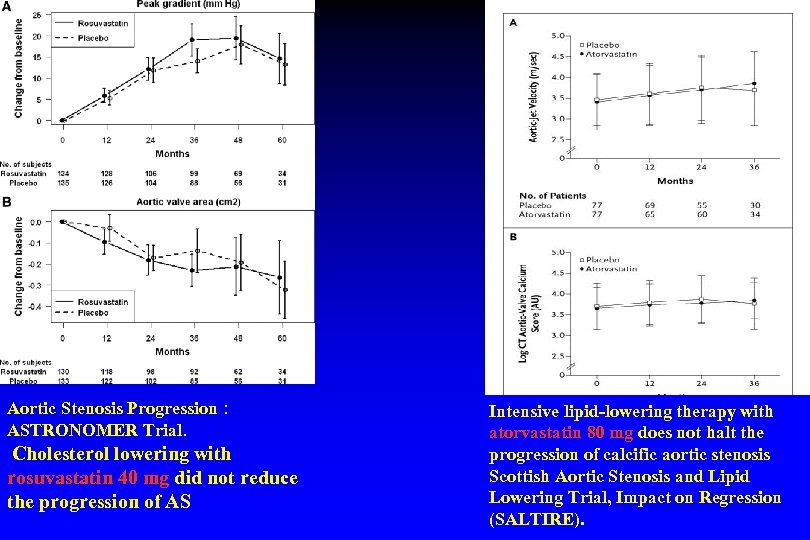
Aortic Stenosis Progression : ASTRONOMER Trial. Cholesterol lowering with rosuvastatin 40 mg did not reduce the progression of AS Intensive lipid-lowering therapy with atorvastatin 80 mg does not halt the progression of calcific aortic stenosis Scottish Aortic Stenosis and Lipid Lowering Trial, Impact on Regression (SALTIRE).
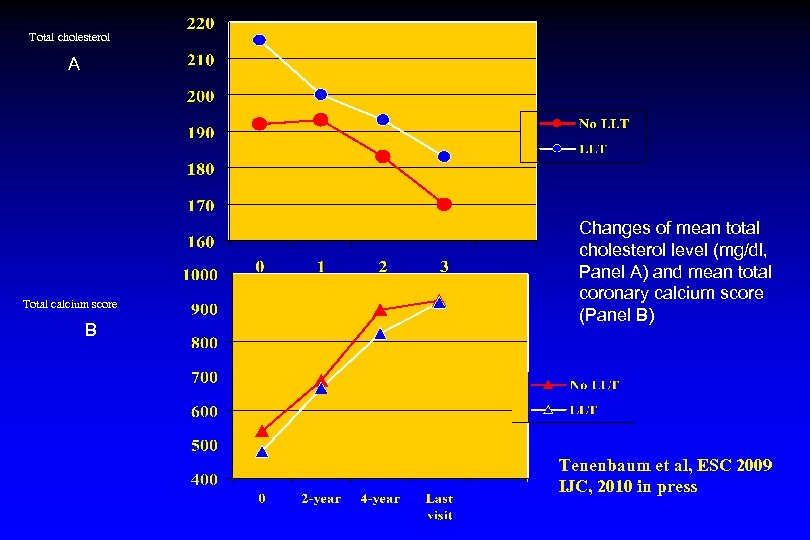
Total cholesterol A Total calcium score B Changes of mean total cholesterol level (mg/dl, Panel A) and mean total coronary calcium score (Panel B) Tenenbaum et al, ESC 2009 IJC, 2010 in press
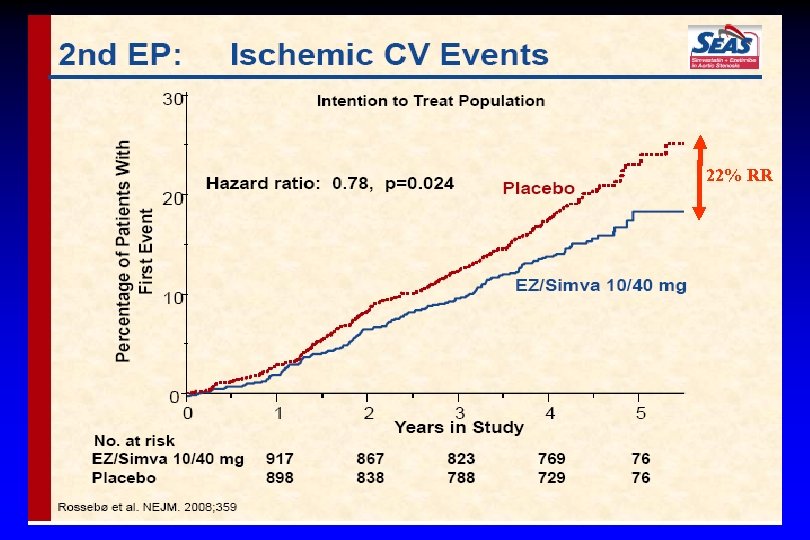
22% RR

Number Needed to Treat (NNT) Definition The NNT is the number of patients who need to be treated in order to prevent one additional bad outcome. It is the inverse of the Absolute Risk Reduction (ARR) How to Calculate NNTs NNT = 1/ARR = |CER - EER|
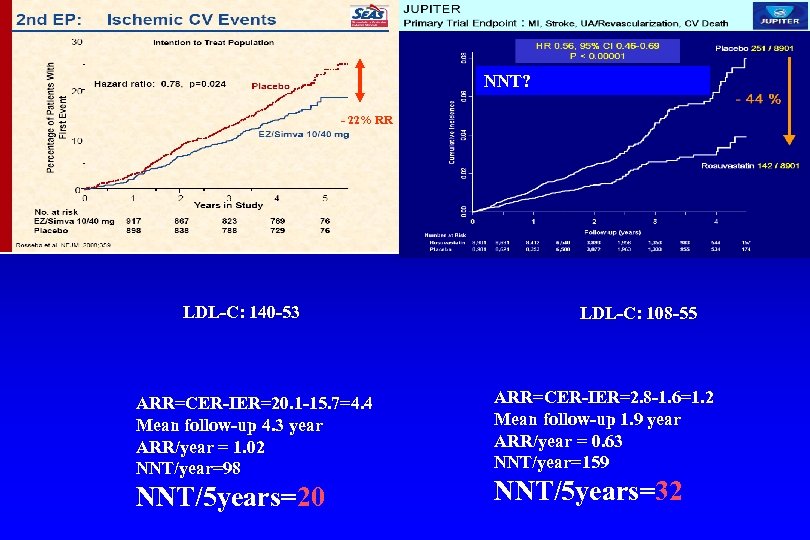
NNT? - 22% RR LDL-C: 140 -53 ARR=CER-IER=20. 1 -15. 7=4. 4 Mean follow-up 4. 3 year ARR/year = 1. 02 NNT/year=98 NNT/5 years=20 LDL-C: 108 -55 ARR=CER-IER=2. 8 -1. 6=1. 2 Mean follow-up 1. 9 year ARR/year = 0. 63 NNT/year=159 NNT/5 years=32



“Post-SEAS” Summary: 1. Can lipid-lowering therapy stop progression of aortic stenosis? No 2. Does ezetimibe associated with increase risk of cancer? No 3. Does ezetimibe/simvastatin combination significantly reduced the incidence of the ischemic cardiovascular events? Yes
Combined treatment more successfully target therapeutic goals in metabolic syndrome: a) low high-density lipoprotein (HDL)-cholesterol, b) high triglyceride levels) c) small dense LDL -cholesterol d) remnant-like particles cholesterol (RLP-C) d) Apo. B

A proper co-administration of statin and other lipid-lowering agent (Bezafibrate, Ezetimibe, Niacin) could be more effective in achieving a comprehensive lipid control as compared with monotherapy.

Conclusions (1) Cardiovascular disease is a major health problem world-wide. Epidemiologic and clinical evidences proved that elevated serum cholesterol, specifically low-density lipoprotein cholesterol (LDL-C), increases cardiovascular disease. Therefore, hypercholesterolaemia is a major target for primary and secondary prevention. Cholesterol homeostasis is regulated by both absorption and production. Each pathway may compensate for changes in the other. The LRC-CPPT have shown that lowering LDL-C levels follow diminishing cholesterol absorption by cholestyramine leads to reduce the incidence of CHD morbidity and mortality.

Conclusions (2) Inhibiting both cholesterol absorption and production, ezetimibe together with a statin provide superior LDL-C lowering vs statin monotherapy. Ezetimibe/simvastatin provides beneficial effect on those parameters beyond LDL-C, such as HDL-C, TG, CRP, endothelial function, oxidized and small dense LDL, platelet aggregation. Statin plus Ezetimibe can attenuate progression of carotid IMT in compare with natural history of carotid atherosclerosis. This combination was equal (ENHANCE) or better (SANDS) than statin alone in this Issue, but less effective than Niacin/Statin combination (ARBITER 6). In the completed clinical outcomes-based trial (SEAS), statin plus ezetimibe combination leads to significant 22% reduction of the ischemic cardiovascular events. This is first large randomised prospective controlled trial which proved the beneficial effect of ezetimibe/simvastatin on cardiovascular events risk reduction.

Conclusions (3) Even on optimal statin monotherapy, many patients fail to achieve all the desired lipid targets and remain at high residual risk of cardiovascular events. Targeting multiple lipid pathways can provide greater reductions in LDL cholesterol as well as improvements in other lipid parameters like low plasma levels of HDL cholesterol, as well as elevated triglycerides, both of which are associated with increased cardiovascular risk. Therefore, a proper co-administration of statins with other agents – fibrates, niacin or ezetimibe – selected on the basis of their safety and effectiveness, could be useful in patients with combined dyslipidemia. Tenenbaum, EVIDENCE-BASED MEDICINE , 2010, in press



df3a00e20d2ea5b9600fcb9024f6315b.ppt