136e577bbb3fda408032335696791a80.ppt
- Количество слайдов: 17

Drug Re-Formulation - Creating New Business Opportunities Dr. Alex Nivorozhkin, Chief Operating Officer Amorsa Therapeutics Inc. , USA Pharmaceutics & Novel Drug Delivery Systems, March 16 -18, 2015, Dubai www. amorsatx. com 1

The Need in Pain Management A Potent Non-Opioid Without the Adverse Effects Morphine has been the gold standard for treating moderate-to-severe pain for nearly 200 years! It’s time for a change: • 16, 000 deaths per year due to opioid overdoses • High potential for addiction • Disabling side effects • Tolerance development

Why Reformulate Existing Drugs? • Extending life cycle of the product (IP issues) – salt formation, route of administration, proprietary delivery • Improve patient compliance, frequency, ease of administration -depot formulations (CNS) • Serve newly found therapeutics indications -adjusting dose, route of administration, delivery • Improving poor intrinsic physico-chemical properties – Poor bioavailability (due to poor solubility and/or permeability) – TCI reports (2014) identified 30 recently launched drugs with poor bioavailability – BCS Class IV oral drugs sales >$145 B
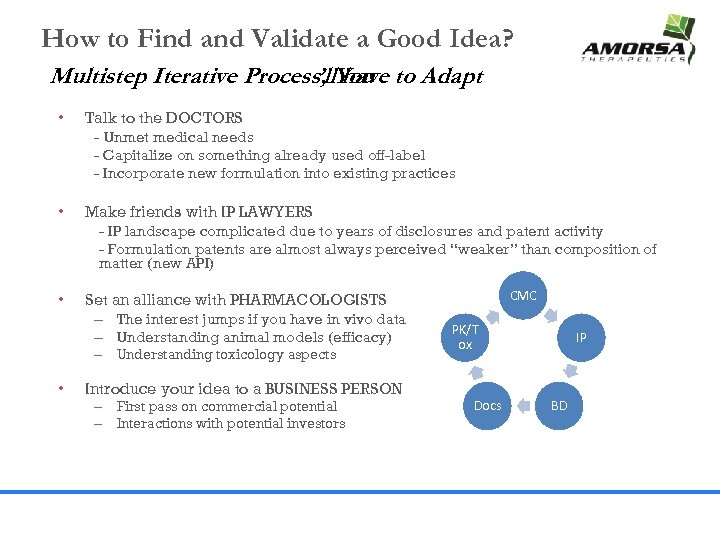
How to Find and Validate a Good Idea? Multistep Iterative Process, You to Adapt ’ll have • Talk to the DOCTORS - Unmet medical needs - Capitalize on something already used off-label - Incorporate new formulation into existing practices • Make friends with IP LAWYERS - IP landscape complicated due to years of disclosures and patent activity - Formulation patents are almost always perceived “weaker” than composition of matter (new API) • Set an alliance with PHARMACOLOGISTS – The interest jumps if you have in vivo data – Understanding animal models (efficacy) – Understanding toxicology aspects • Introduce your idea to a BUSINESS PERSON – First pass on commercial potential – Interactions with potential investors CMC PK/T ox Docs IP BD

Unmet Medical Need for Pain Management U. S. Market for Opioid Analgesics - $8. 3 Billion (2013) Post-Trauma Pain • Opioids are associated with serious side effects including respiratory depression • Military priority for non-opioid treatments Post-Operative Pain • 70 million surgeries each year in the U. S. • >50% report moderate to severe pain Refractory Cancer Pain • Up to 25% of advanced cancer patients have refractory pain – nothing works • No FDA approved treatment for patients 5 5

Unlocking Ketamine’s Potential “The medical community is missing out on one of the best pain drugs there is. ” Director, Defense Center for Integrative Pain Management • Ketamine was approved as an anesthetic in 1970 • Recent studies have shown ketamine’s analgesic and antidepressant effects but with undesirable side effects • Majority of therapeutic benefits are due to the conversion of ketamine to norketamine • Amorsa is developing proprietary formulations of novel ketamine analogs designed to: – Deliver the potency of ketamine with fewer adverse effects – Be administered as convenient oral formulations – Offer patients an effective alternative to opioids
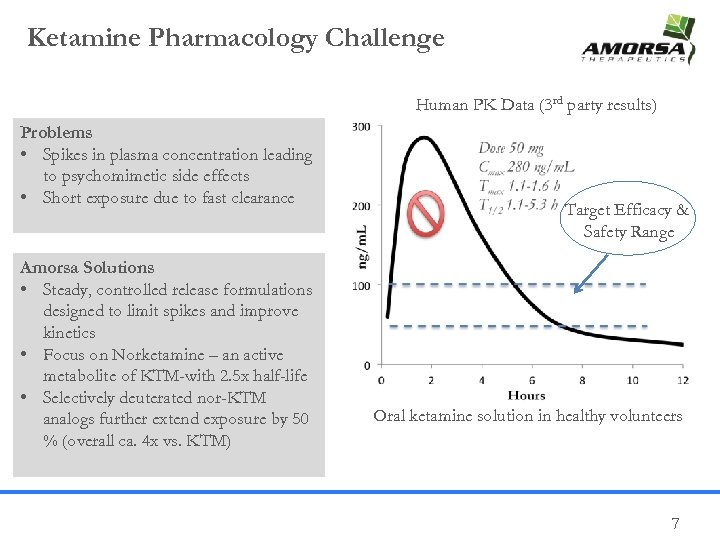
Ketamine Pharmacology Challenge Human PK Data (3 rd party results) Problems • Spikes in plasma concentration leading to psychomimetic side effects • Short exposure due to fast clearance Amorsa Solutions • Steady, controlled release formulations designed to limit spikes and improve kinetics • Focus on Norketamine – an active metabolite of KTM-with 2. 5 x half-life • Selectively deuterated nor-KTM analogs further extend exposure by 50 % (overall ca. 4 x vs. KTM) Target Efficacy & Safety Range Oral ketamine solution in healthy volunteers 7
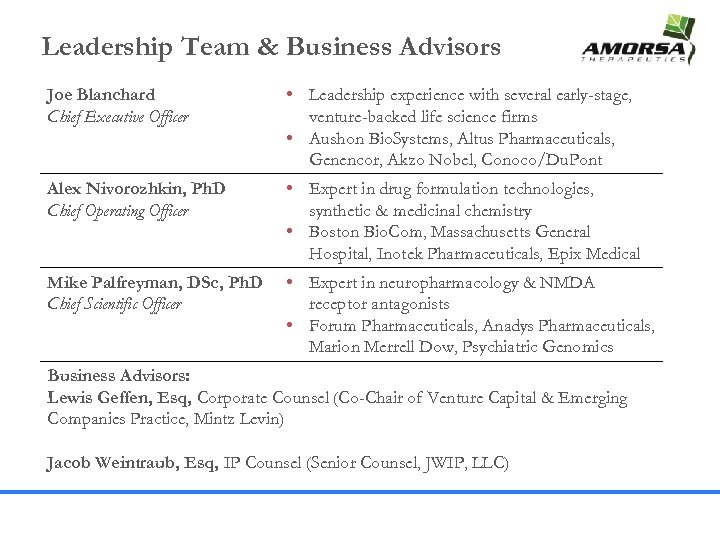
Leadership Team & Business Advisors Joe Blanchard Chief Executive Officer • Leadership experience with several early-stage, venture-backed life science firms • Aushon Bio. Systems, Altus Pharmaceuticals, Genencor, Akzo Nobel, Conoco/Du. Pont Alex Nivorozhkin, Ph. D Chief Operating Officer • Expert in drug formulation technologies, synthetic & medicinal chemistry • Boston Bio. Com, Massachusetts General Hospital, Inotek Pharmaceuticals, Epix Medical Mike Palfreyman, DSc, Ph. D Chief Scientific Officer • Expert in neuropharmacology & NMDA receptor antagonists • Forum Pharmaceuticals, Anadys Pharmaceuticals, Marion Merrell Dow, Psychiatric Genomics Business Advisors: Lewis Geffen, Esq, Corporate Counsel (Co-Chair of Venture Capital & Emerging Companies Practice, Mintz Levin) Jacob Weintraub, Esq, IP Counsel (Senior Counsel, JWIP, LLC)
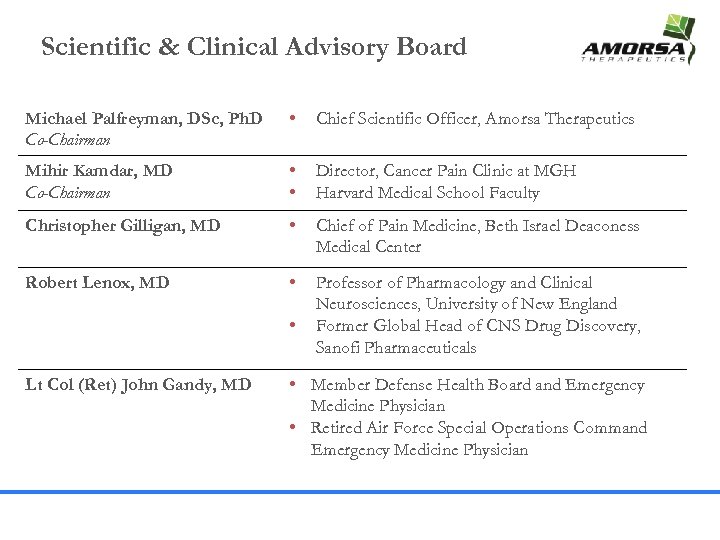
Scientific & Clinical Advisory Board Michael Palfreyman, DSc, Ph. D Co-Chairman • Chief Scientific Officer, Amorsa Therapeutics Mihir Kamdar, MD Co-Chairman • • Director, Cancer Pain Clinic at MGH Harvard Medical School Faculty Christopher Gilligan, MD • Chief of Pain Medicine, Beth Israel Deaconess Medical Center Robert Lenox, MD • Professor of Pharmacology and Clinical Neurosciences, University of New England Former Global Head of CNS Drug Discovery, Sanofi Pharmaceuticals • Lt Col (Ret) John Gandy, MD • Member Defense Health Board and Emergency Medicine Physician • Retired Air Force Special Operations Command Emergency Medicine Physician
Amorsa’s Drug Development Platform Novel Deuterated Ketamine Analogs Proprietary Formulations Safe & Effective NMDA Receptor Antagonists • Built on known pharmacology • Expedited clinical path possible • Novel analogs expected to deliver improved efficacy and/or safety • Convenient oral tablets • Neuro-attenuating features designed to limit side effects • All FDA approved ingredients • Expected efficacy without the adverse effects • Pipeline of high value applications
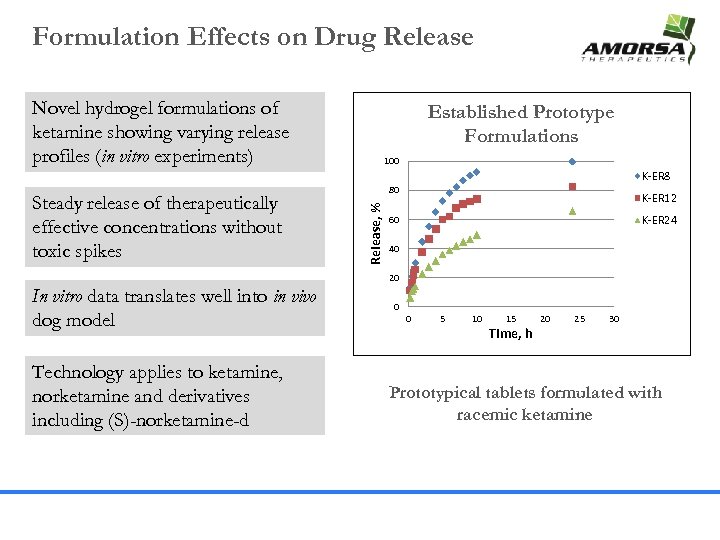
Formulation Effects on Drug Release Novel hydrogel formulations of ketamine showing varying release profiles (in vitro experiments) Established Prototype Formulations 100 K-ER 8 Release, % Steady release of therapeutically effective concentrations without toxic spikes 80 K-ER 12 K-ER 24 60 40 20 In vitro data translates well into in vivo dog model Technology applies to ketamine, norketamine and derivatives including (S)-norketamine-d 0 0 5 10 15 Time, h 20 25 30 Prototypical tablets formulated with racemic ketamine
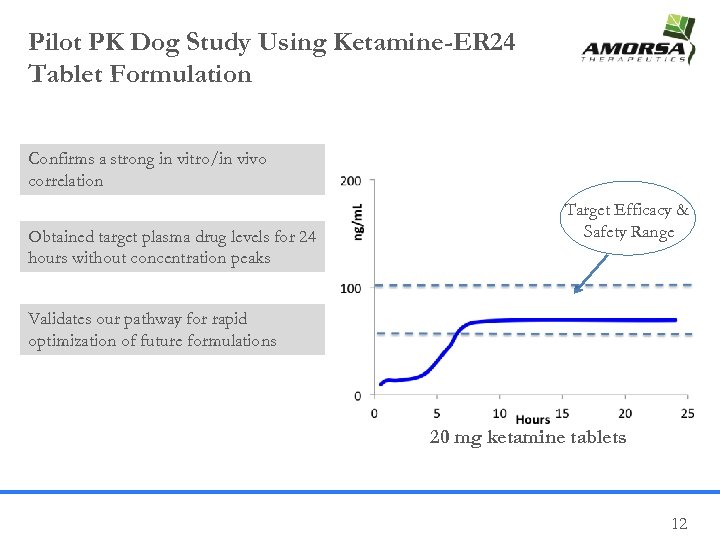
Pilot PK Dog Study Using Ketamine-ER 24 Tablet Formulation Confirms a strong in vitro/in vivo correlation Obtained target plasma drug levels for 24 hours without concentration peaks Target Efficacy & Safety Range Validates our pathway for rapid optimization of future formulations 20 mg ketamine tablets 12
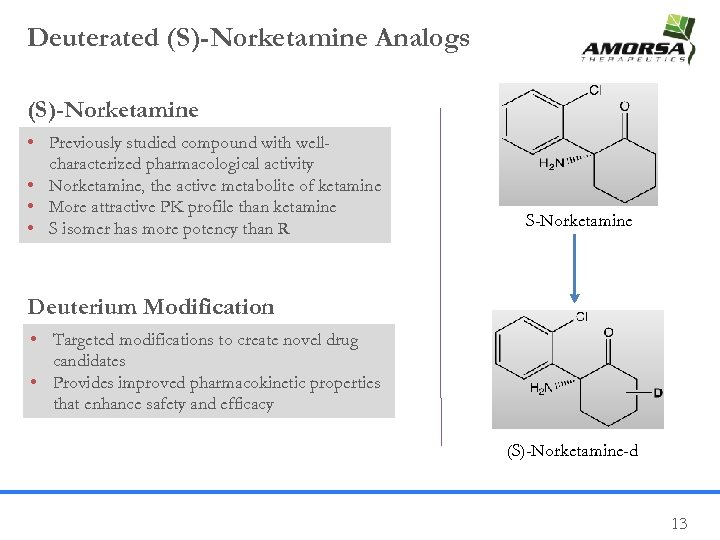
Deuterated (S)-Norketamine Analogs (S)-Norketamine • Previously studied compound with wellcharacterized pharmacological activity • Norketamine, the active metabolite of ketamine • More attractive PK profile than ketamine • S isomer has more potency than R S-Norketamine Deuterium Modification • Targeted modifications to create novel drug candidates • Provides improved pharmacokinetic properties that enhance safety and efficacy (S)-Norketamine-d 13
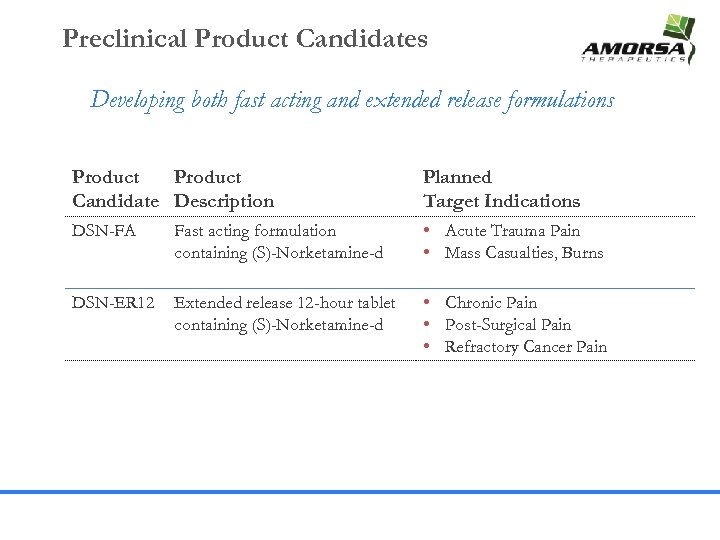
Preclinical Product Candidates Developing both fast acting and extended release formulations Product Candidate Description Planned Target Indications DSN-FA Fast acting formulation containing (S)-Norketamine-d • Acute Trauma Pain • Mass Casualties, Burns DSN-ER 12 Extended release 12 -hour tablet containing (S)-Norketamine-d • Chronic Pain • Post-Surgical Pain • Refractory Cancer Pain
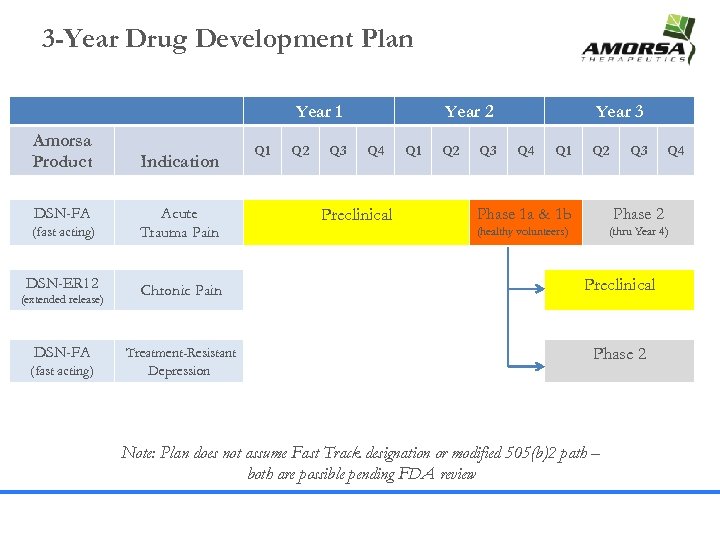
3 -Year Drug Development Plan Year 1 Amorsa Product DSN-FA (fast acting) DSN-ER 12 (extended release) DSN-FA (fast acting) Indication Acute Trauma Pain Chronic Pain Treatment-Resistant Depression Q 1 Q 2 Q 3 Year 2 Q 4 Preclinical Q 1 Q 2 Q 3 Year 3 Q 4 Q 1 Q 2 Q 3 Q 4 Phase 1 a & 1 b Phase 2 (healthy volunteers) (thru Year 4) Preclinical Phase 2 Note: Plan does not assume Fast Track designation or modified 505(b)2 path – both are possible pending FDA review

Who Dares, Wins

Contacts: Alex Nivorozhkin, Ph. D. Chief Operating Officer (617) 921 -0114 aniv@amorsatx. com www. amorsatx. com
136e577bbb3fda408032335696791a80.ppt