82d59fd09b9ee559aec764776a11e8a0.ppt
- Количество слайдов: 65

Dr Gerard Meachery

The objectives of the pre-anaesthetic assessment • Evaluate the patient’s medical condition from medical history, physical examination, investigations and, when appropriate, past medical records • Optimise the patient’s medical condition for anaesthesia and surgery • Determine and minimise risk factors for anaesthesia • Plan anaesthetic technique and peri-operative care • Develop a rapport with the patient to reduce anxiety and facilitate conduct of anaesthesia • Inform and educate the patient about anaesthesia, peri-operative care and pain management • Obtain consent for anaesthesia

Guidelines on the radical management of patients with lung cancer • Lim E, Baldwin D, Beckles M, et al. Thorax 2010, 65 Suppl III, iii 1 -iii 27 • A joint initiative by the British Thoracic Society and the Society for Cardiothoracic Surgery in Great Britain and Ireland undertaken to update the 2001 guidelines for the selection and assessment of patients with lung cancer who can potentially be managed by radical treatment

Guidelines on the radical management of patients with lung cancer • 2. 1. 3 Assessment of lung function • 43. Offer surgical resection to patients with low risk of postoperative dyspnoea. [C] • 44. Offer surgical resection to patients at moderate to high risk of postoperative dyspnoea if they are aware of and accept the risks of dyspnoea and associated complications. [D] • 47. Consider using shuttle walk testing as functional assessment in patients with moderate to high risk of postoperative dyspnoea using a distance walked of >400 m as a cut-off for good function. [C] • 48. Consider cardiopulmonary exercise testing to measure peak oxygen consumption as functional assessment in patients with moderate to high risk of postoperative dyspnoea using >15 ml/ kg/min as a cut-off for good function. [D]
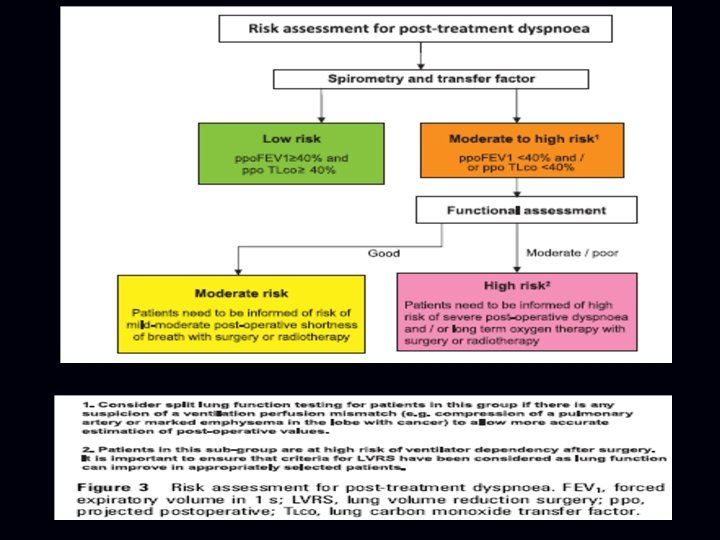


Guidelines on the radical management of patients with lung cancer • 49. RR Further studies with specific outcomes are required to define the role of exercise testing in the selection of patients for surgery • 51. Avoid taking pulmonary function and exercise tests as sole surrogates for quality of life evaluation. [C]

Pulmonary Function Testing

Objectives Categorise PFTs according to specific purposes Identify at least one indication for spirometry, lung volumes, and diffusing capacity Obstructive and restrictive ventilatory defects Relate respiratory history to indications for performing pulmonary function tests

Pulmonary Function Testing • Establish baseline lung function and evaluate the presence or absence of lung disease • Evaluate symptoms of dyspnoea • Evaluate if the lung disease is primarily an obstructive, restrictive or mixed ventilatory defect • Quantify the respiratory impairment and monitor the extent of known disease on lung function • Monitor effects of therapies used to treat respiratory disease

Pulmonary Function Testing • Evaluate operative risk • Perform surveillance for occupational-related lung disease • Evaluate disability or impairment • Assess for reversible components to optimise a patient’s clinical status

Spirometry • Forced expiratory volume in 1 second (FEV 1) – Volume exhaled in the first second of an FVC manoeuvre – (forced exhalation from maximal inspiration) • Vital capacity (VC) – Total volume exhaled by a exhalation from maximal inspiration – Can be a forced exhalation (FVC) or a relaxed exhalation (RVC) – best one taken as VC • FEV 1/VC – Ratio between FEV 1 and VC

Pulmonary Function Testing • In normal spirometry, FVC, FEV 1, and FEV 1 -to-FVC ratio are above the lower limit of normal • The lower limit of normal is defined as the result of the mean predicted value (based on the patient's sex, age, and height)
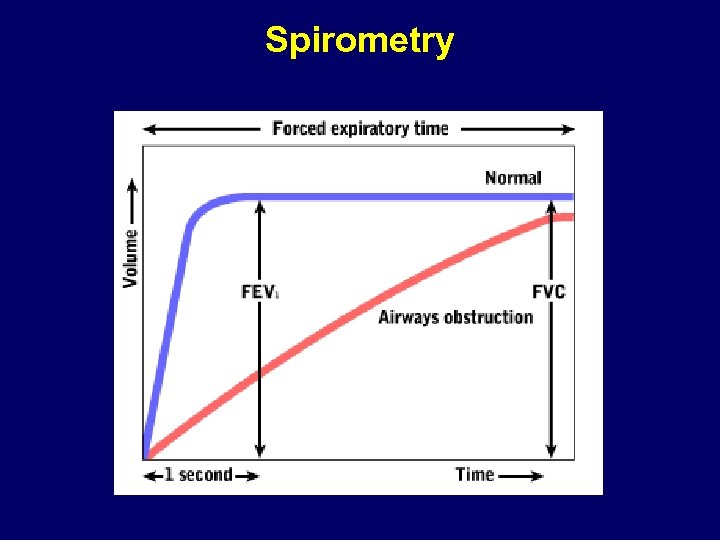
Spirometry

Reduction in FEV 1 • Airway obstruction is the most common cause of reduction in FEV 1 • Airflow obstruction may be secondary to Bronchospasm (Asthma/ COPD) Airway inflammation (Asthma/ COPD/ Bronchiectasis) Loss of lung elastic recoil (Emphysema) Increased secretions in the airway (Bronchitis/ Bronchiectasis/ Infection)

Assessing reversibility in airway obstruction • Response of FEV 1 to inhaled bronchodilators is used to assess the reversibility of airway obstruction (Post Bronchodilator challenge) • Methacholine Challenge – used to assess possible underlying asthma, (ie reversible airway obstruction). Baseline lung function may be normal when the patient is clinically stable.

Assessing reversibility in airway obstruction • Gibson Resp Med – 12% or 200 ml
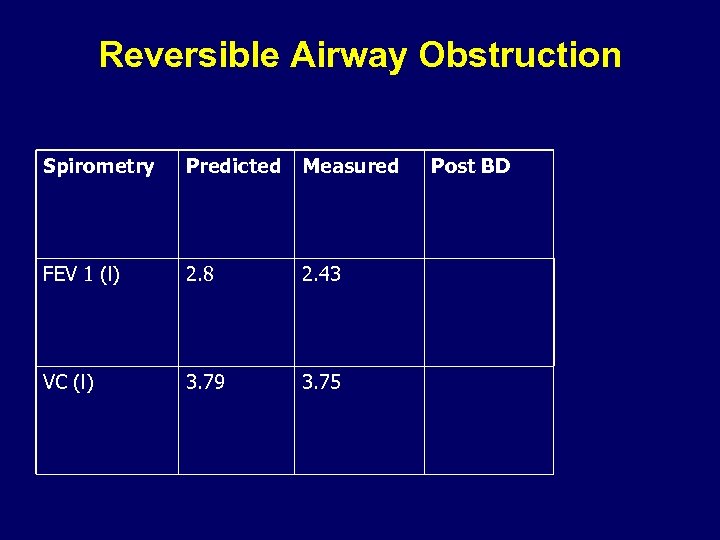
Reversible Airway Obstruction Spirometry Predicted Measured FEV 1 (l) 2. 8 2. 43 VC (l) 3. 79 3. 75 Post BD
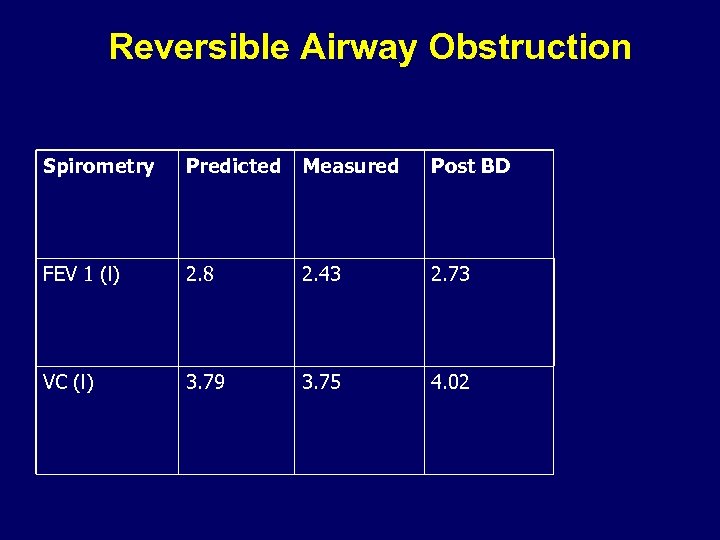
Reversible Airway Obstruction Spirometry Predicted Measured Post BD FEV 1 (l) 2. 8 2. 43 2. 73 VC (l) 3. 79 3. 75 4. 02

Indications for Lung Volume Tests • Diagnose or assess the severity of restrictive lung disease • Differentiate between obstructive and restrictive disease patterns • Assess the response to therapy • Make preoperative assessments of patients with compromised lung function

Static lung volumes • Total lung capacity (TLC) – Total volume of air in the lungs at the end of an maximal inspiration • Residual volume (RV) – Volume of air remaining in the lungs at the end of a maximal expiration • Functional residual volume (FRC) – Volume of air remaining in the lungs at the end of tidal expiration
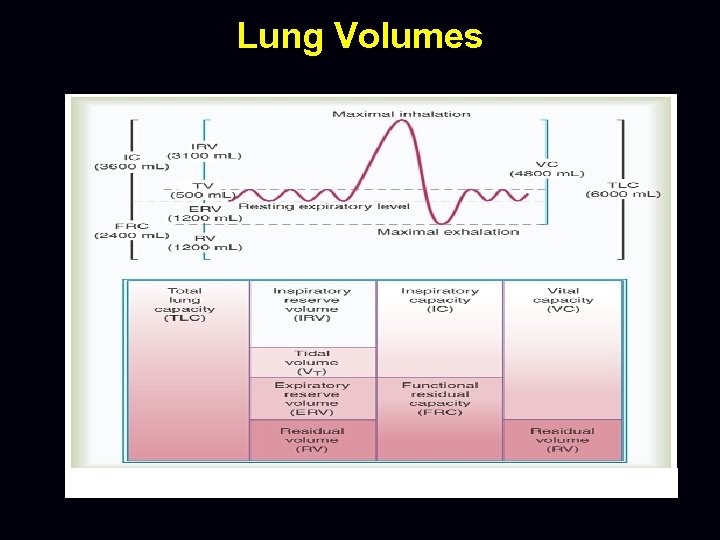
Lung Volumes

Reduction in FVC • A reduced FVC on spirometry in the absence of a reduced FEV 1 -to-FVC ratio suggests a restrictive ventilatory defect • An inappropriately shortened exhalation during spirometry can (and often does) result in a reduced FVC (i. e. Patient effort is important)

Causes of Abnormal Lung Volumes • Raised TLC – COPD esp. emphysema – Transiently raised during an asthma exacerbation or in the recovery phase of an asthma exacerbation • Increased RV – Airways disease (air-trapping), e. g. asthma or emphysema • Reduced TLC/ FVC/ RV – Restrictive defect (intrapulmonary or extrapulmonary)

Diffusion Capacity/ Transfer Factor • The diffusing capacity is a measure of the conductance of the CO molecule from the alveolar gas to Haemoglobin in the pulmonary capillary blood • CO (and oxygen) must pass through the alveolar epithelium, tissue interstitium, capillary endothelium, blood plasma, red cell membrane and cytoplasm before attaching to the Haemoglobin molecule
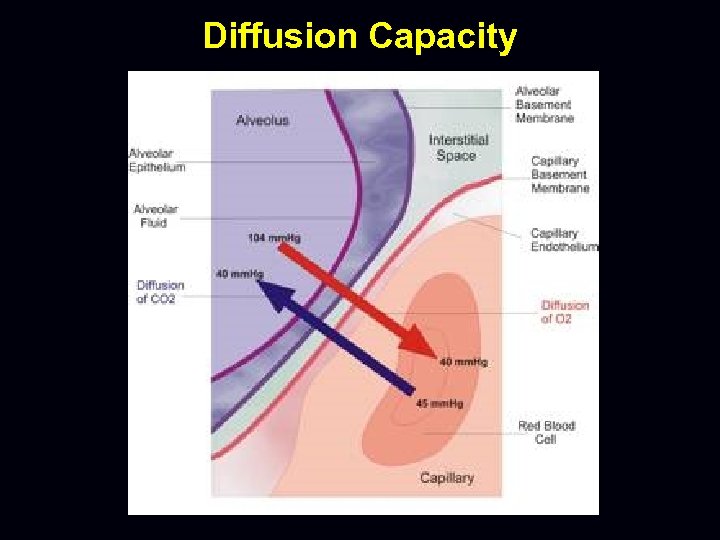
Diffusion Capacity

Indications for Diffusion Capacity • Evaluate or follow the progress of parenchymal/ interstitial lung disease • Evaluate pulmonary involvement in systemic disease • Evaluate obstructive lung disease • Quantify disability associated with interstitial lung disease • Evaluate pulmonary hemorrhage

Diffusion capacity • TLCO = transfer factor for the lung for carbon monoxide i. e. Total diffusing capacity for the lung – Same as DLCO • KCO = transfer coefficent i. e. Diffusing capacity of the lung per unit volume, standardised for alveolar volume (VA) • VA = Lung volume in which carbon monoxide diffuses into during a single breath-hold technique

Abnormal Diffusion Capacity • • Low TLCO Low/normal KCO = Intrapulmonary restrictive defect – Interstitial lung diseases – Pulmonary oedema • High TLC • Low TLCO • Low KCO – emphysema

Abnormal Diffusion Capacity Abnormal Diffusion C • Low TLCO • but high/N KCO • = extrapulmonary restrictive defect – Obesity – Neuromuscular disease (respiratory muscle weakness) – Pleural disease e. g. effusion, thickening, tumor – Skeletal deformity – Post pneumonectomy

Abnormal Diffusion Capacity Abnormal Diffusion C • Normal/raised TLCO • Raised KCO – Asthma – Pulmonary haemorrhage

Obstructive Lung Disease • Chronic Obstructive Pulmonary Disease (COPD) • Chronic Bronchitis “Excessive mucus production, with a productive cough on most days, for at least 3 months for 2 years or more. ” • Emphysema – Primarily caused by cigarette smoking. – Alpha -1 -antitrypsin deficiency – Environmental pollutants

Working Definition of COPD Chronic obstructive pulmonary disease (COPD) is characterised by airflow obstruction. The airflow obstruction is usually progressive, not fully reversible and does not change markedly over several months. The disease is predominantly caused by smoking. Airflow obstruction is defined as a reduced FEV 1 (forced expiratory volume in 1 second) and a reduced FEV 1/FVC ratio (where FVC is forced vital capacity), such that FEV 1 is less than 80% predicted and FEV 1/FVC is less than 0. 7. (www. nice. org. uk/CG 012 NICEguideline)

Chronic Obstructive Pulmonary Disease (COPD) Characterized by: – Dyspnoea at rest or with exertion – Productive cough – – – Barrel-chest (↑AP to Transverse diameter) Chest percussion: Hyper resonant Chest auscultation: Breath sounds distant or absent – Chest X-Ray • Flattened diaphragms • Hyperinflated lung fields/ bullae

Emphysema • Spirometry Reduction in FEV 1/ VC ratio • Lung Volumes Increased lung volumes (“air trapping”) • Diffusing Capacity Reduced

Obstructive Lung Disease • Asthma Airway obstruction is characterized by inflammation of Airway o the mucosal lining of the airways, bronchospasm and increased airway secretions Reversible airway obstruction

Obstructive Lung Disease • Asthma Triggers • Exercise/ Cold air • Allergic agents – Pollens, house dust mite, animal dander, moulds • Non-allergic agents – Viral infections, environmental pollutants, medication, food additives, emotional upset • Occupational exposure – Cotton/ wood dusts, grains, metal salts, insecticides

Obstructive Lung Disease • Asthma During Attacks – Peak Flow (PEF) is reduced/ Hypoxia – Response to bronchodilators • Spirometry Reduced FEV 1 • Lung Volumes Increased (Hyperinflation) • Diffusion Capacity Normal During stable state: Spirometry may be normal

Causes of Restrictive Spirometry • • • Pulmonary fibrosis Pleural effusion Pleural tumors Lung resection (lobectomy/ pneumonectomy) Diaphragm weakness or paralysis Neuromuscular disease Kyphoscoliosis Obesity Inadequate respiration secondary to pain Congestive heart failure Ascites Pregnancy

Restrictive Lung Disease • Idiopathic Pulmonary Fibrosis Or secondary to • Treatment with bleomycin, cyclophosphamide, • • • methotrexate or amiodarone Autoimmune diseases: Rheumatoid arthritis, systemic lupus erythematousus (SLE), scleroderma Sarcoidosis Pneumoconiosis – Silica dust – Asbestosis – Asbestos fibers

Restrictive Lung Disease • Idiopathic Pulmonary Fibrosis – – Increasing exertional dyspnoea Dry cough Finger clubbing Inspiratory crackles on auscultation – Chest X-Ray • Interstitial infiltrates are visible • Honeycombing pattern

Restrictive Lung Disease • Idiopathic Pulmonary Fibrosis • Spirometry Reduced VC • Lung volumes Reduced TLC/ RV • Diffusion capacity Reduced

Diseases of Chest Wall and Pleura Disorders involving the chest wall or pleura of the lungs result in restrictive ventilatory defects on pulmonary function testing. But, lung parenchyma is not affected.

Diseases of Chest Wall and Pleura • Spirometry Reduced FEV 1 and FVC • Lung Volumes Reduced TLC • Diffusion Capacity Reduced • KCo Normal

Obstructive v. Restrictive

Mixed Picture • Bronchiectasis Pathologic and irreversible dilatation of the bronchi, resulting from destruction of the bronchial wall by severe, repeated infections and inflammation

Bronchiectasis Post infective: Whooping cough/ TB Genetic: Cystic Fibrosis/ Primary Cliliary Dyskinesia (PCD) Immunodeficiency

Bronchiectasis – – – – Dyspnoea Significant productive cough Purulent, foul smelling sputum Haemoptysis Frequent pulmonary infections Chronically unwell Chest X-Ray / CT Scan • Airway Dilation
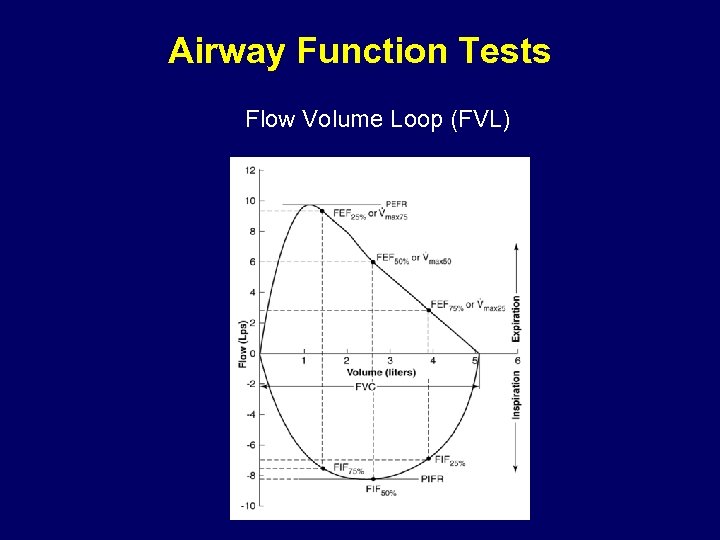
Airway Function Tests Flow Volume Loop (FVL)
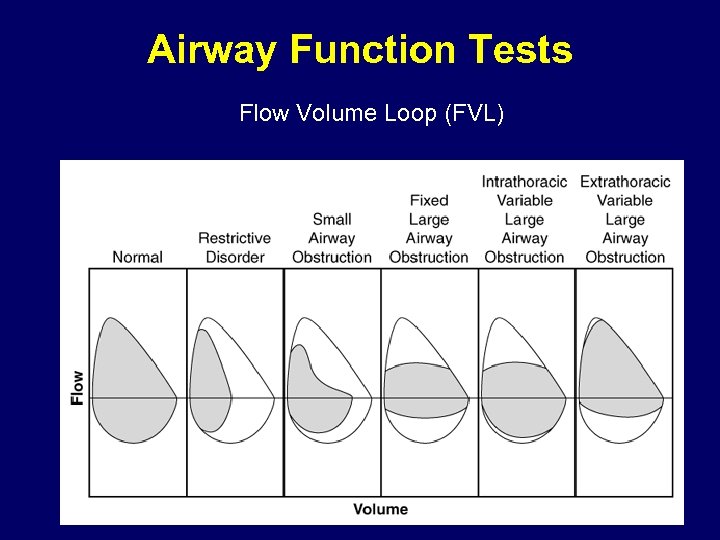
Airway Function Tests Flow Volume Loop (FVL)

Respiratory History • Dyspnoea: Do you get short of breath at the following times: • At rest? On exertion? At night? • Progression of dyspnoea • Cough: Do you ever cough? • In the morning? At night? • Dry or productive? • Blood? • Sputum/ Phlegm? (Color, volume, consistency) • Chest pain/ Orthopnoea/ Paroxysmal Nocturnal Dyspnoea • Family history of lung disease • Past History • TB/ Emphysema/ Chronic Bronchitis/ Asthma • Recurrent lung infection/ Pneumonia or pleurisy • Allergies or hay fever • Previous chest injury or chest surgery

Respiratory History • Current Medications • Inhalers/ Steroids/ Nebulised bronchodilators or antibiotics/ • • Oxygen/ Mucolytics Cardiac medications Oncology drugs or immunosuppressives • Smoking Habits • Cigarettes/ Cigars/ Pipe/ Illicit drugs • How many years? • Current or ex smoker? • Occupation • Asbestos (Direct/ Bystander exposure) • Mining, quarry, foundry

Invisible Lives Report - BLF 1. The disease is far from invisible statistically: it is the UK’s fifth biggest killer disease, claiming more lives than breast, bowel or prostate cancer (estimated 30 000 lives/ year) 2. The second most common cause of emergency admission to hospital and one of the most costly inpatient conditions treated by the NHS 3. It is estimated that the direct cost of providing care in the NHS for people with COPD is almost £ 500 million a year – more than half of which relates to hospital care

Invisible Lives Report - BLF 1. The epidemiological evidence published in 2006 suggesting that out of an estimated 3. 7 million people in the UK with COPD, only 900, 000 were currently diagnosed and receiving treatment and care 2. The remaining 2. 8 million people were still unaware they had a disease which, if left untreated, could severely restrict their lives and would eventually kill them

Diagnosis • Clinical suspicion in patients (usually smokers or exsmokers, age >35 yrs) with: – exertional breathlessness – chronic cough – regular sputum production – frequent “winter bronchitis” – wheeze with a risk factor (usually smoking) • Airflow obstruction should be confirmed with spirometry

Spirometry for COPD Diagnosis: NICE 2010 – FEV 1 <80% predicted – Post Bronchodilator FEV 1: FVC ratio <0. 7 – Stage 1 Mild: FEV 1 80% (+ Symptoms) – Stage 2 Moderate: FEV 1 50 -79% – Stage 3 Severe: FEV 1 30 -49% – Stage 4 Very Severe: FEV 1 <30% or – Stage 4 Very Severe: FEV 1 <50% (+Respiratory Failure)

General principles of management of stable COPD – NICE guidelines • Lifestyle modification – Smoking cessation (Behavioural support/ Nicotene replacement/ Bupropion/ Varenicline) – Pulmonary rehabilitation • Optimisation of pharmacological therapies – Inhalers • Short-acting bronchodilators • Long-acting bronchodilators regularly, often combined with… • Corticosteroids (FEV 1 </= 50% with 2+ exacerbations requiring antibiotics or oral steroids in 1 year)
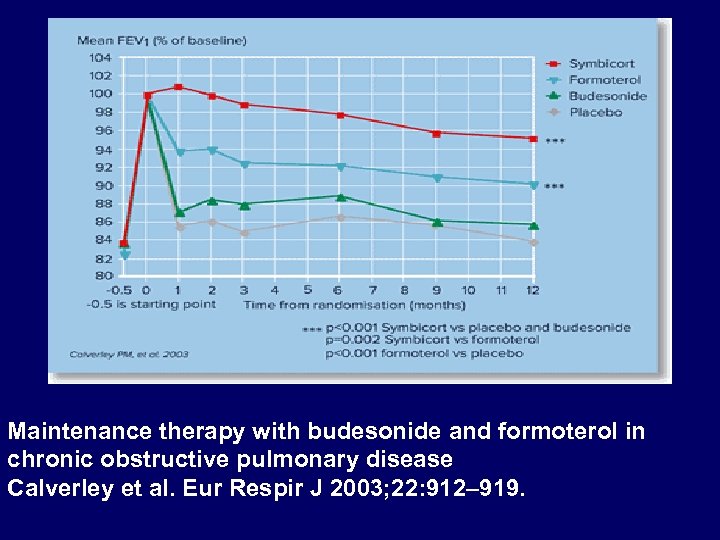
Maintenance therapy with budesonide and formoterol in chronic obstructive pulmonary disease Calverley et al. Eur Respir J 2003; 22: 912– 919.
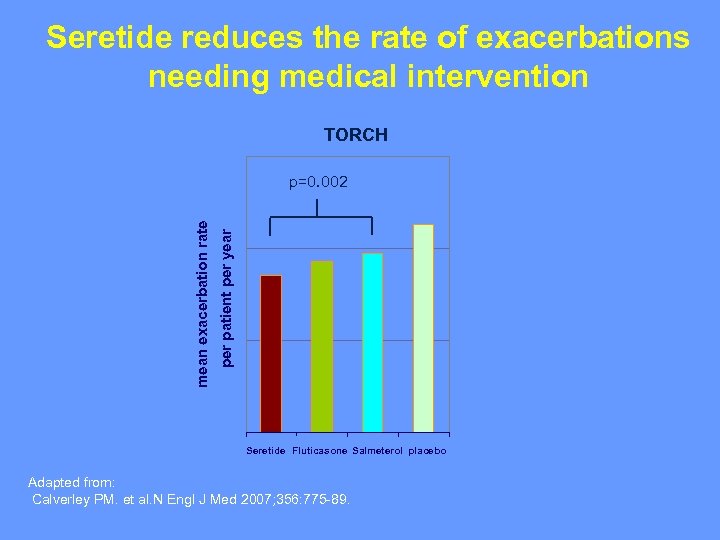
Seretide reduces the rate of exacerbations needing medical intervention TORCH per patient per year mean exacerbation rate p=0. 002 Seretide Fluticasone Salmeterol placebo Adapted from: Calverley PM. et al. N Engl J Med 2007; 356: 775 -89.

General Principles of Guidelines • Theophylline, oral steroids, diuretics, mucolytics • Prophylaxis – Immunisations (influenza, pneumococcus, H 1 N 1) • Long Term Oxygen Therapy (LTOT) • Surgery – Bullectomy, lung volume reduction, transplant • Management of anxiety and depression • Palliation and end of life support

General Principles - in addition • Early detection + Patient education + Smoking cessation • Treatment of acute exacerbations • Pulmonary Rehabilitation: Increases threshold for perception of dyspnoea Improves quality of life Substantially reduces health care costs Under resourced • NIPPV: Reduces need for invasive ventilation Reduces admissions Bridging measure prior to surgery Under resourced

Referral for Specialist advice - NICE • • Diagnostic uncertainty Suspected severe COPD Onset of cor pulmonale Assessment for oxygen therapy, long-term nebuliser therapy or oral corticosteroid therapy Bullous lung disease Rapid decline in FEV 1 Assessment for pulmonary rehabilitation • Assessment for lung • • volume reduction surgery or transplantation Patient aged under 40 years or a family history of alpha-1 antitrypsin deficiency Symptoms disproportionate to lung function deficit Frequent infections Haemoptysis

Complications of COPD • Respiratory failure • Cor pulmonale • Bullae • Pneumothorax • Pneumonia • Increased risk of malignancy (shared risk factor)

Preoperative measures • Cessation of cigarette smoking for at least 8 weeks • • before surgery Treat airflow obstruction Treat respiratory infection if present Educate for lung-expansion manouvres Mucolytics/ Physiotherapy and chest clearance • Postoperative measures • Epidural analgesia or intercostals nerve bloc for pain • • • control Early mobilization Chest physical therapy (including deep breathing and incentive spirometry) Continuous positive airway pressure in selected patients

Pre Operative Assessment • • • Be wary of a “presumed diagnosis” of lung disease Beware of “no previous diagnosis of known lung disease” Evaluate lung function systematically Careful history and examination If in doubt…. . Find a friendly respiratory physician
82d59fd09b9ee559aec764776a11e8a0.ppt