Chronic Myeloid Leukemia.american.ppt
- Количество слайдов: 41
 Dr. Fineman Riva
Dr. Fineman Riva
 Myeloproliferative Neoplasms (MPNs): are a group of clonal myeloid neoplasms in which a genetic alteration occurs in a hematopoietic progenitor cell leading to its proliferation resulting in an increase in the peripheral blood white blood cells (WBCs), red blood cells (RBCs), platelets, or a combination of these cells.
Myeloproliferative Neoplasms (MPNs): are a group of clonal myeloid neoplasms in which a genetic alteration occurs in a hematopoietic progenitor cell leading to its proliferation resulting in an increase in the peripheral blood white blood cells (WBCs), red blood cells (RBCs), platelets, or a combination of these cells.
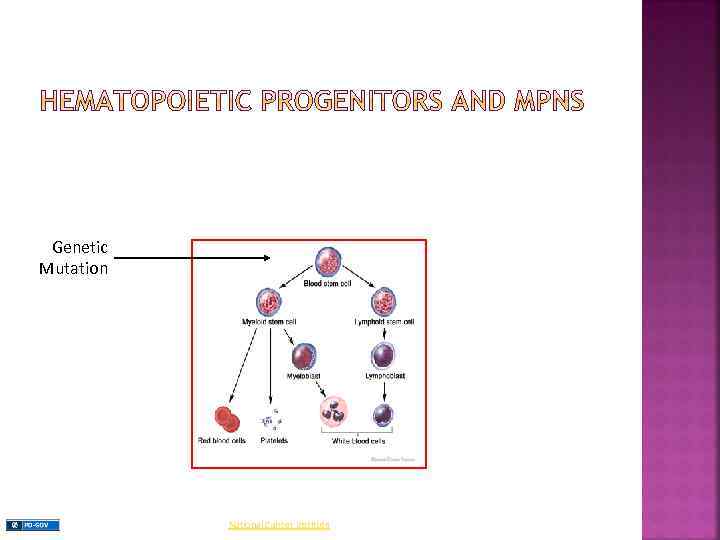 Genetic Mutation National Cancer Institute
Genetic Mutation National Cancer Institute
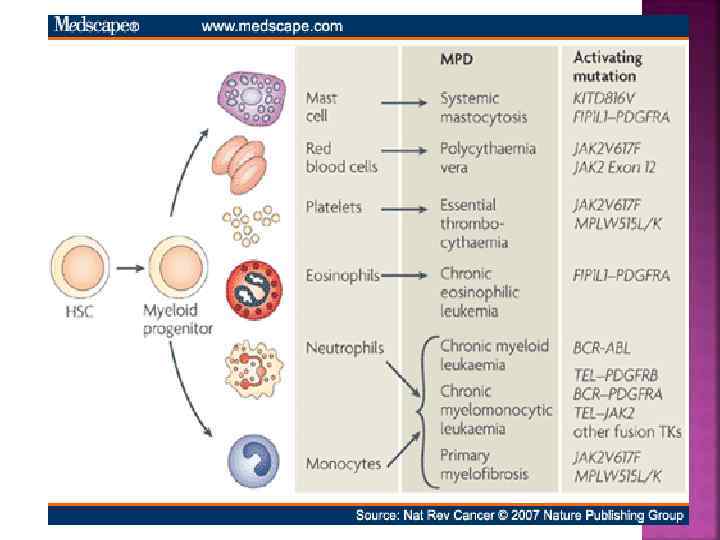
 The type of disorder is often based on the predominant cell line that is affected, but because blood counts are often abnormal in more than one cell line, diagnoses based upon blood counts and morphology alone may be inaccurate. Four Main MPNs: 1. Chronic Myelogenous Leukemia (CML) 2. Polycythemia Vera (PV) 3. Essential Thrombocytosis (ET) 4. Primary Myelofibrosis (PMF) Additional MPNs: 1. Systemic Mastocytosis 2. Hypereosinophilic Syndrome 3. Chronic Myelomonocytic Leukemia 4. Chronic Neutrophilic Leukemia 5. Chronic Eosinophilic Leukemia
The type of disorder is often based on the predominant cell line that is affected, but because blood counts are often abnormal in more than one cell line, diagnoses based upon blood counts and morphology alone may be inaccurate. Four Main MPNs: 1. Chronic Myelogenous Leukemia (CML) 2. Polycythemia Vera (PV) 3. Essential Thrombocytosis (ET) 4. Primary Myelofibrosis (PMF) Additional MPNs: 1. Systemic Mastocytosis 2. Hypereosinophilic Syndrome 3. Chronic Myelomonocytic Leukemia 4. Chronic Neutrophilic Leukemia 5. Chronic Eosinophilic Leukemia
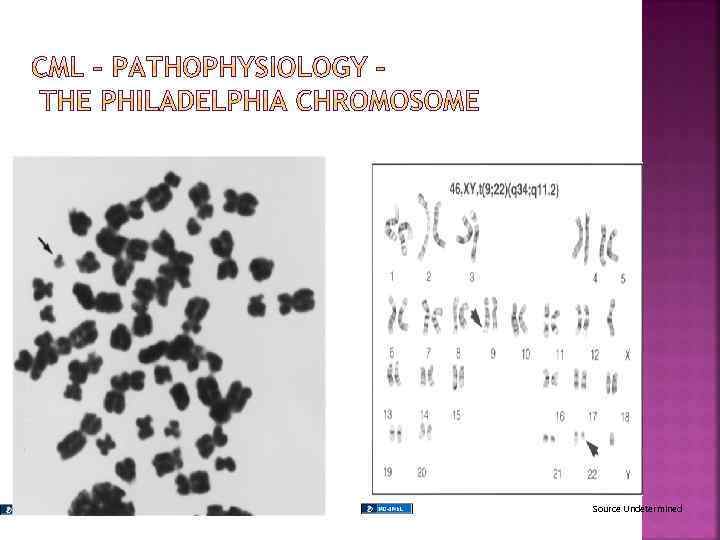
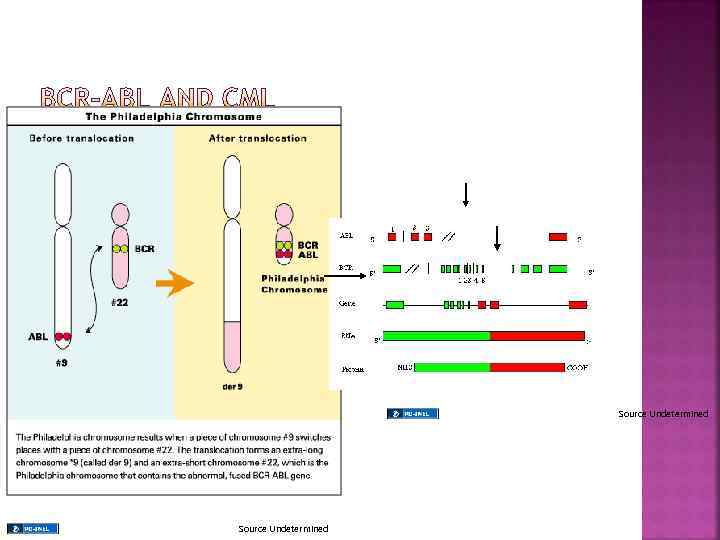
 A pluripotent stem cell disease characterized by anemia, extreme blood granulocytosis and granulocytic immaturity, basophilia, often thrombocytosis and splenomegaly The clonal hematopoietic cells contain a reciprocal translocations between chromosomes 9 and 22 in more than 95% of the patients, which leads to an overtly foreshortened long arm of chromosome 22 referred as the Philadelphia chromosome. Natural history - Phasic disease: chronic, accelerated and blast crisis
A pluripotent stem cell disease characterized by anemia, extreme blood granulocytosis and granulocytic immaturity, basophilia, often thrombocytosis and splenomegaly The clonal hematopoietic cells contain a reciprocal translocations between chromosomes 9 and 22 in more than 95% of the patients, which leads to an overtly foreshortened long arm of chromosome 22 referred as the Philadelphia chromosome. Natural history - Phasic disease: chronic, accelerated and blast crisis
 Approximately 5, 050 cases in the U. S. in 2009 (11% of all leukemias) with an incidence that increases significantly with age (median age ~ 55) Risk Factors include: ▪ prior high dose radiation exposure (WW II / Chernobyl / etc…) ▪ exposure to certain organic solvents (benzene) ▪ age ▪ gender (male > female)
Approximately 5, 050 cases in the U. S. in 2009 (11% of all leukemias) with an incidence that increases significantly with age (median age ~ 55) Risk Factors include: ▪ prior high dose radiation exposure (WW II / Chernobyl / etc…) ▪ exposure to certain organic solvents (benzene) ▪ age ▪ gender (male > female)
 ETIOLOGY OF CML The risk of getting CML does not seem to be affected by smoking, diet, or infections CML does not run in families since inherited mutations do not cause CML Instead, DNA changes related to CML occur during the patient’s life time
ETIOLOGY OF CML The risk of getting CML does not seem to be affected by smoking, diet, or infections CML does not run in families since inherited mutations do not cause CML Instead, DNA changes related to CML occur during the patient’s life time
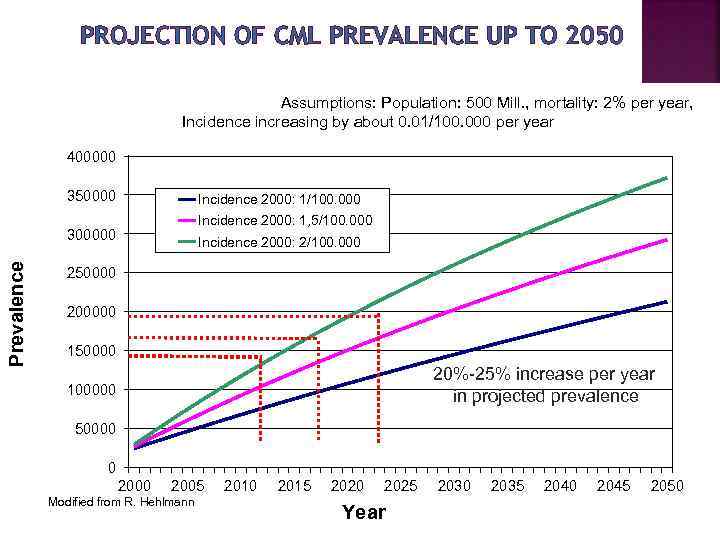 PROJECTION OF CML PREVALENCE UP TO 2050 Assumptions: Population: 500 Mill. , mortality: 2% per year, Incidence increasing by about 0. 01/100. 000 per year 400000 350000 Incidence 2000: 1/100. 000 Incidence 2000: 1, 5/100. 000 Prevalence 300000 Incidence 2000: 2/100. 000 250000 200000 150000 20%-25% increase per year in projected prevalence 100000 50000 0 2005 Modified from R. Hehlmann 2010 2015 2020 2025 Year 2030 2035 2040 2045 2050
PROJECTION OF CML PREVALENCE UP TO 2050 Assumptions: Population: 500 Mill. , mortality: 2% per year, Incidence increasing by about 0. 01/100. 000 per year 400000 350000 Incidence 2000: 1/100. 000 Incidence 2000: 1, 5/100. 000 Prevalence 300000 Incidence 2000: 2/100. 000 250000 200000 150000 20%-25% increase per year in projected prevalence 100000 50000 0 2005 Modified from R. Hehlmann 2010 2015 2020 2025 Year 2030 2035 2040 2045 2050
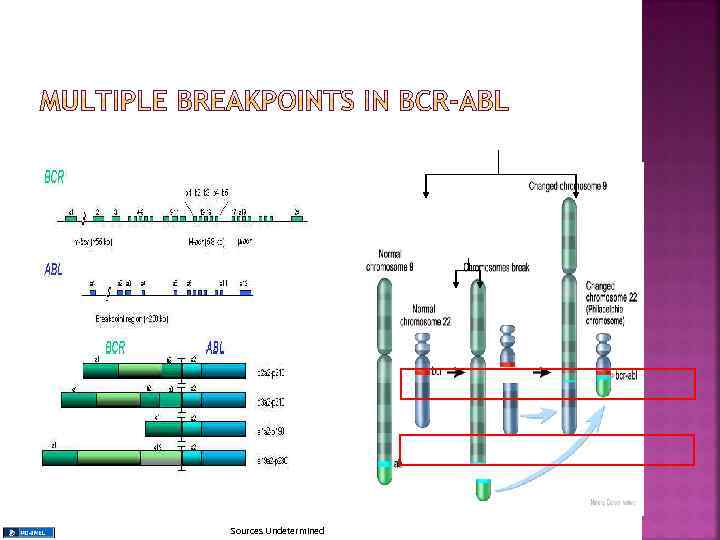
 The first malignancy with identified cytogenetic abnormality, molecular mechanism and specific therapy 1960 – Nowell and Hungerfold discover Ph chromosome 1973 – J. Rowley discovered that the translocation leads to fusion gene bcr/Abl 1983 – bcr/Abl encodes to unregulated tyrosine kinase 1996 – Tyrosine Kinase Inhibitor
The first malignancy with identified cytogenetic abnormality, molecular mechanism and specific therapy 1960 – Nowell and Hungerfold discover Ph chromosome 1973 – J. Rowley discovered that the translocation leads to fusion gene bcr/Abl 1983 – bcr/Abl encodes to unregulated tyrosine kinase 1996 – Tyrosine Kinase Inhibitor
 Source Undetermined
Source Undetermined
 The gene that breaks off from chromosome 9 is called ABL (after Abelson the scientist who first identified the gene), while the gene that splits from chromosome 22 is called BCR, short for breakpoint cluster region
The gene that breaks off from chromosome 9 is called ABL (after Abelson the scientist who first identified the gene), while the gene that splits from chromosome 22 is called BCR, short for breakpoint cluster region
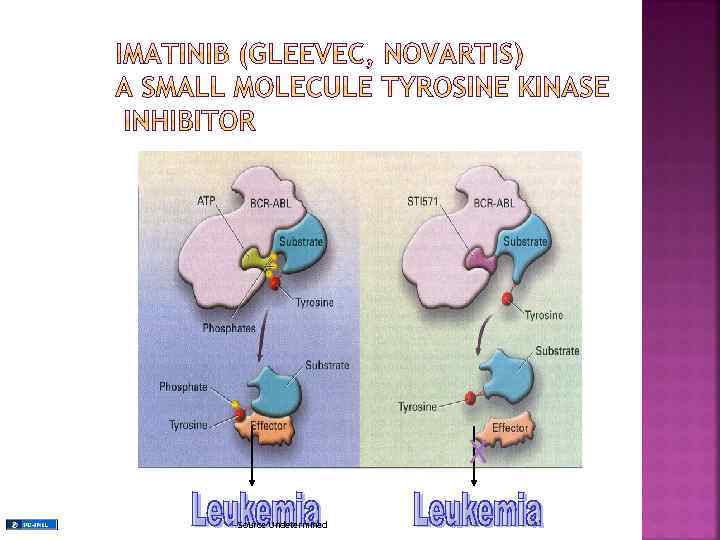
 WHAT IS PHILADELPHIA CHROMOSOME POSITIVE CML? The combination of BCR and ABL leads to the formation of an abnormal fusion gene responsible for the pathogenesis of CML In the words of Brian Druker the BCR-ABL gene in CML acts “like the gas pedal in a car stuck in the ‘on’ position fuelling the excess growth of white blood cells”
WHAT IS PHILADELPHIA CHROMOSOME POSITIVE CML? The combination of BCR and ABL leads to the formation of an abnormal fusion gene responsible for the pathogenesis of CML In the words of Brian Druker the BCR-ABL gene in CML acts “like the gas pedal in a car stuck in the ‘on’ position fuelling the excess growth of white blood cells”
 Source Undetermined
Source Undetermined
 Sources Undetermined
Sources Undetermined
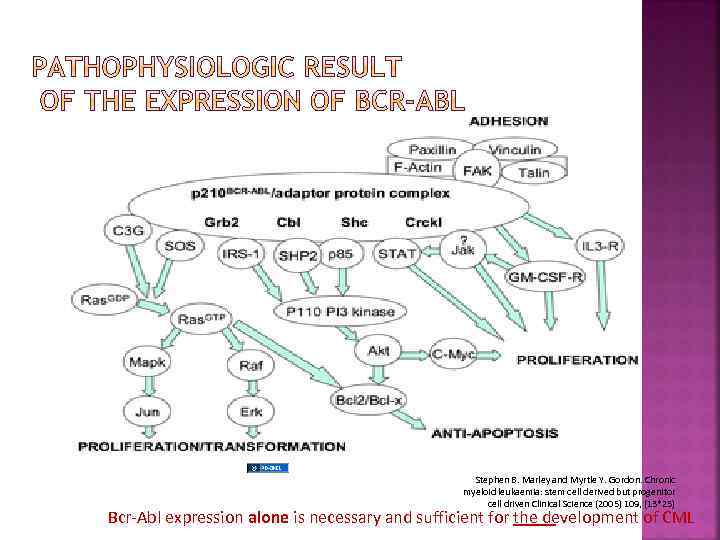 Stephen B. Marley and Myrtle Y. Gordon. Chronic myeloid leukaemia: stem cell derived but progenitor cell driven Clinical Science (2005) 109, (13*25) Bcr-Abl expression alone is necessary and sufficient for the development of CML
Stephen B. Marley and Myrtle Y. Gordon. Chronic myeloid leukaemia: stem cell derived but progenitor cell driven Clinical Science (2005) 109, (13*25) Bcr-Abl expression alone is necessary and sufficient for the development of CML
 ▪ Asymptomatic (~ 30%) ▪ Fatigue, weight loss, fever ▪ Abdominal fullness, pain and/or early satiety due to splenomegaly (~ 50 -90%) ▪ Easy bruising and purpura ▪ Leukostasis ▪ Pulmonary symptoms ▪ Neurologic symptoms
▪ Asymptomatic (~ 30%) ▪ Fatigue, weight loss, fever ▪ Abdominal fullness, pain and/or early satiety due to splenomegaly (~ 50 -90%) ▪ Easy bruising and purpura ▪ Leukostasis ▪ Pulmonary symptoms ▪ Neurologic symptoms
 Chronic phase 85% at diagnosis, asymptomatic or mild constitutional complaints, anemia or symptomatic splenomegaly, duration until progression 3 -5 years without treatment Accelerated phase Blast crisis – life expectancy <1 year, no effective treatment
Chronic phase 85% at diagnosis, asymptomatic or mild constitutional complaints, anemia or symptomatic splenomegaly, duration until progression 3 -5 years without treatment Accelerated phase Blast crisis – life expectancy <1 year, no effective treatment
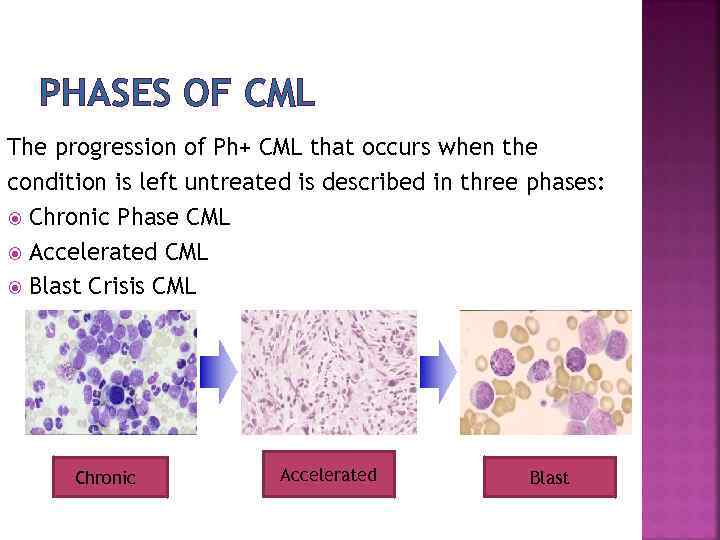 PHASES OF CML The progression of Ph+ CML that occurs when the condition is left untreated is described in three phases: Chronic Phase CML Accelerated CML Blast Crisis CML Chronic Accelerated Blast
PHASES OF CML The progression of Ph+ CML that occurs when the condition is left untreated is described in three phases: Chronic Phase CML Accelerated CML Blast Crisis CML Chronic Accelerated Blast
 D. Bixby
D. Bixby
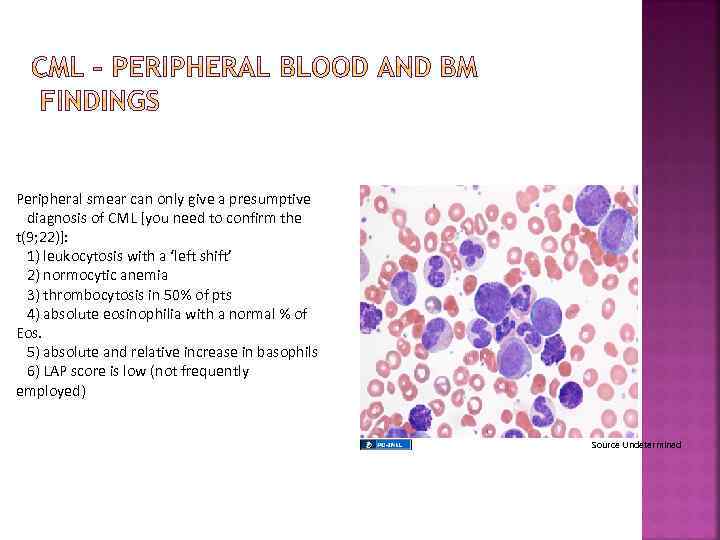 Peripheral smear can only give a presumptive diagnosis of CML [you need to confirm the t(9; 22)]: 1) leukocytosis with a ‘left shift’ 2) normocytic anemia 3) thrombocytosis in 50% of pts 4) absolute eosinophilia with a normal % of Eos. 5) absolute and relative increase in basophils 6) LAP score is low (not frequently employed) Source Undetermined
Peripheral smear can only give a presumptive diagnosis of CML [you need to confirm the t(9; 22)]: 1) leukocytosis with a ‘left shift’ 2) normocytic anemia 3) thrombocytosis in 50% of pts 4) absolute eosinophilia with a normal % of Eos. 5) absolute and relative increase in basophils 6) LAP score is low (not frequently employed) Source Undetermined
 Demonstrating the presence of the t(9; 22) or its gene product is absolutely essential in diagnosing a patient with CML Karyotyping in CML 1) Allows for the diagnosis of CML 2) Requires a bone marrow aspirate for optimal metaphases 3) Allows for evaluation of clonal evolution as well as additional chromosomal abnormalities Isochromosome 17; Double Philadelphia chromosome; Trisomia 8; Trisomia 19; Loss of Y chromosome 4) Occasional cryptic and complex karyotypes can result in the missed identification of the t(9; 22) Source Undetermined
Demonstrating the presence of the t(9; 22) or its gene product is absolutely essential in diagnosing a patient with CML Karyotyping in CML 1) Allows for the diagnosis of CML 2) Requires a bone marrow aspirate for optimal metaphases 3) Allows for evaluation of clonal evolution as well as additional chromosomal abnormalities Isochromosome 17; Double Philadelphia chromosome; Trisomia 8; Trisomia 19; Loss of Y chromosome 4) Occasional cryptic and complex karyotypes can result in the missed identification of the t(9; 22) Source Undetermined
 Fluorescence in-situ hybridization (FISH) in CML: 1) Allows for the diagnosis of CML 2) Does not require a bone marrow aspirate for optimal results 3) Allows for the identification of potential duplications of the Ph chromosome 4) Allows for the identification of the loss of the remainder (9) chromsome 5) Allows for the identification of cryptic translocations involving Bcr-Abl Bcr- Ch 22 Abl – Ch 9 Bcr-Abl Fusion Source Undetermined
Fluorescence in-situ hybridization (FISH) in CML: 1) Allows for the diagnosis of CML 2) Does not require a bone marrow aspirate for optimal results 3) Allows for the identification of potential duplications of the Ph chromosome 4) Allows for the identification of the loss of the remainder (9) chromsome 5) Allows for the identification of cryptic translocations involving Bcr-Abl Bcr- Ch 22 Abl – Ch 9 Bcr-Abl Fusion Source Undetermined
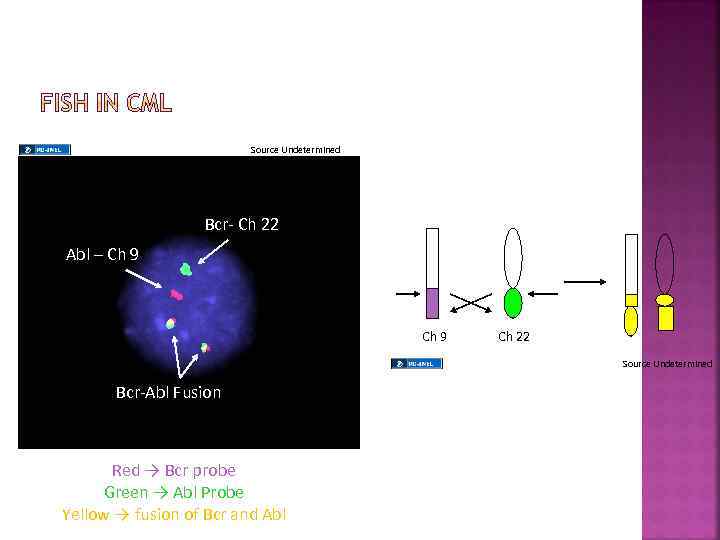 Source Undetermined Bcr- Ch 22 Abl – Ch 9 Ch 22 Source Undetermined Bcr-Abl Fusion Red → Bcr probe Green → Abl Probe Yellow → fusion of Bcr and Abl
Source Undetermined Bcr- Ch 22 Abl – Ch 9 Ch 22 Source Undetermined Bcr-Abl Fusion Red → Bcr probe Green → Abl Probe Yellow → fusion of Bcr and Abl
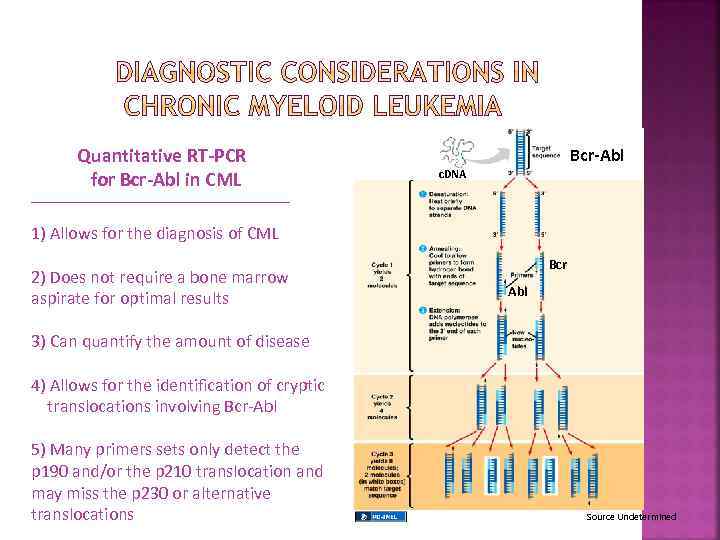 Quantitative RT-PCR for Bcr-Abl in CML Bcr-Abl c. DNA 1) Allows for the diagnosis of CML 2) Does not require a bone marrow aspirate for optimal results Bcr Abl 3) Can quantify the amount of disease 4) Allows for the identification of cryptic translocations involving Bcr-Abl 5) Many primers sets only detect the p 190 and/or the p 210 translocation and may miss the p 230 or alternative translocations Source Undetermined
Quantitative RT-PCR for Bcr-Abl in CML Bcr-Abl c. DNA 1) Allows for the diagnosis of CML 2) Does not require a bone marrow aspirate for optimal results Bcr Abl 3) Can quantify the amount of disease 4) Allows for the identification of cryptic translocations involving Bcr-Abl 5) Many primers sets only detect the p 190 and/or the p 210 translocation and may miss the p 230 or alternative translocations Source Undetermined
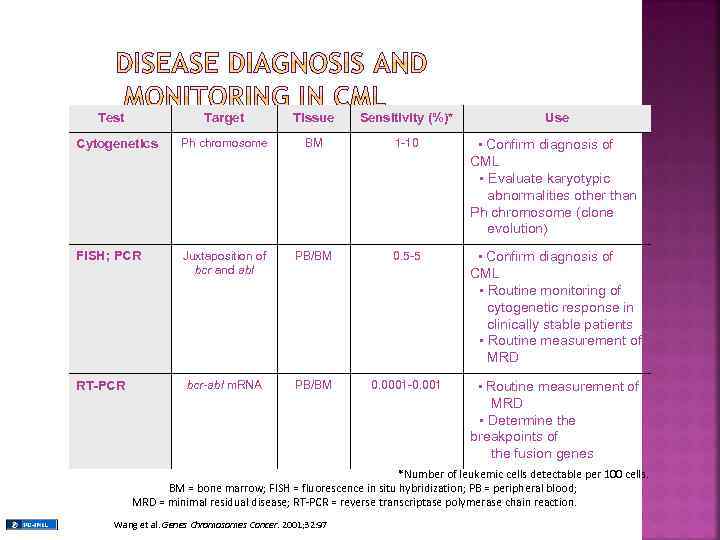 Test Target Tissue Sensitivity (%)* Cytogenetics Ph chromosome BM 1 -10 ¢▪ FISH; PCR Juxtaposition of bcr and abl PB/BM 0. 5 -5 ¢▪ bcr-abl m. RNA PB/BM 0. 0001 -0. 001 ¢▪ RT-PCR Use Confirm diagnosis of CML ¢▪ Evaluate karyotypic ¢ abnormalities other than Ph chromosome (clone ¢ evolution) Confirm diagnosis of CML ¢▪ Routine monitoring of ¢ cytogenetic response in ¢ clinically stable patients ¢▪ Routine measurement of ¢ MRD ¢▪ Determine the breakpoints of ¢ the fusion genes *Number of leukemic cells detectable per 100 cells. BM = bone marrow; FISH = fluorescence in situ hybridization; PB = peripheral blood; MRD = minimal residual disease; RT-PCR = reverse transcriptase polymerase chain reaction. Wang et al. Genes Chromosomes Cancer. 2001; 32: 97
Test Target Tissue Sensitivity (%)* Cytogenetics Ph chromosome BM 1 -10 ¢▪ FISH; PCR Juxtaposition of bcr and abl PB/BM 0. 5 -5 ¢▪ bcr-abl m. RNA PB/BM 0. 0001 -0. 001 ¢▪ RT-PCR Use Confirm diagnosis of CML ¢▪ Evaluate karyotypic ¢ abnormalities other than Ph chromosome (clone ¢ evolution) Confirm diagnosis of CML ¢▪ Routine monitoring of ¢ cytogenetic response in ¢ clinically stable patients ¢▪ Routine measurement of ¢ MRD ¢▪ Determine the breakpoints of ¢ the fusion genes *Number of leukemic cells detectable per 100 cells. BM = bone marrow; FISH = fluorescence in situ hybridization; PB = peripheral blood; MRD = minimal residual disease; RT-PCR = reverse transcriptase polymerase chain reaction. Wang et al. Genes Chromosomes Cancer. 2001; 32: 97
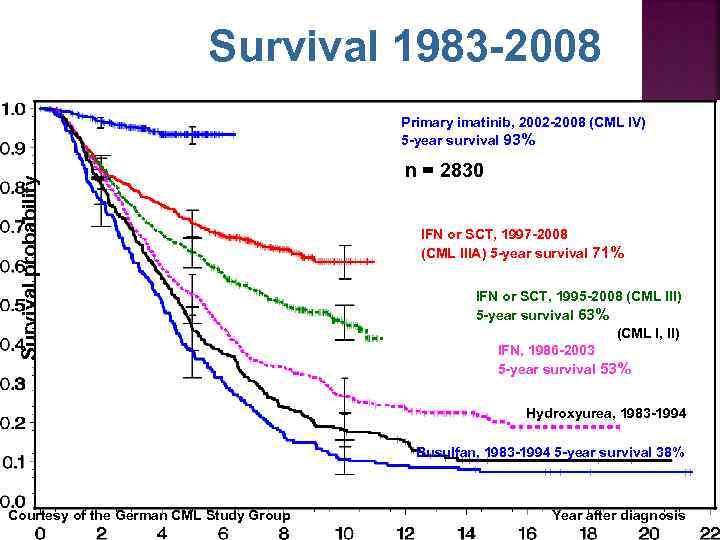 Survival 1983 -2008 Survival probability Primary imatinib, 2002 -2008 (CML IV) 5 -year survival 93% n = 2830 IFN or SCT, 1997 -2008 (CML IIIA) 5 -year survival 71% IFN or SCT, 1995 -2008 (CML III) 5 -year survival 63% (CML I, II) IFN, 1986 -2003 5 -year survival 53% Hydroxyurea, 1983 -1994 Busulfan, 1983 -1994 5 -year survival 38% Courtesy of the German CML Study Group Year after diagnosis
Survival 1983 -2008 Survival probability Primary imatinib, 2002 -2008 (CML IV) 5 -year survival 93% n = 2830 IFN or SCT, 1997 -2008 (CML IIIA) 5 -year survival 71% IFN or SCT, 1995 -2008 (CML III) 5 -year survival 63% (CML I, II) IFN, 1986 -2003 5 -year survival 53% Hydroxyurea, 1983 -1994 Busulfan, 1983 -1994 5 -year survival 38% Courtesy of the German CML Study Group Year after diagnosis
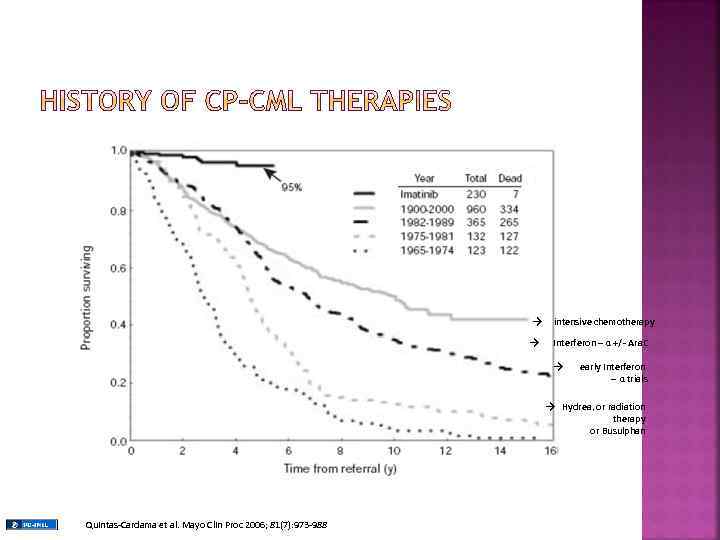 → intensive chemotherapy → Interferon – α +/- Ara. C → early Interferon – α trials → Hydrea, or radiation therapy or Busulphan Quintas-Cardama et al. Mayo Clin Proc 2006; 81(7): 973 -988
→ intensive chemotherapy → Interferon – α +/- Ara. C → early Interferon – α trials → Hydrea, or radiation therapy or Busulphan Quintas-Cardama et al. Mayo Clin Proc 2006; 81(7): 973 -988
 X Source Undetermined
X Source Undetermined
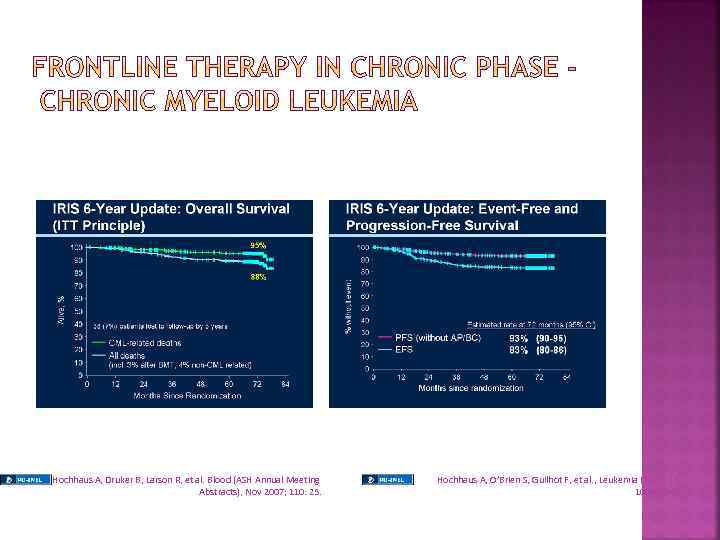 Hochhaus A, Druker B, Larson R, et al. Blood (ASH Annual Meeting Abstracts), Nov 2007; 110: 25. Hochhaus A, O’Brien S, Guilhot F, et al. , Leukemia (2009) 23, 1054– 1061.
Hochhaus A, Druker B, Larson R, et al. Blood (ASH Annual Meeting Abstracts), Nov 2007; 110: 25. Hochhaus A, O’Brien S, Guilhot F, et al. , Leukemia (2009) 23, 1054– 1061.
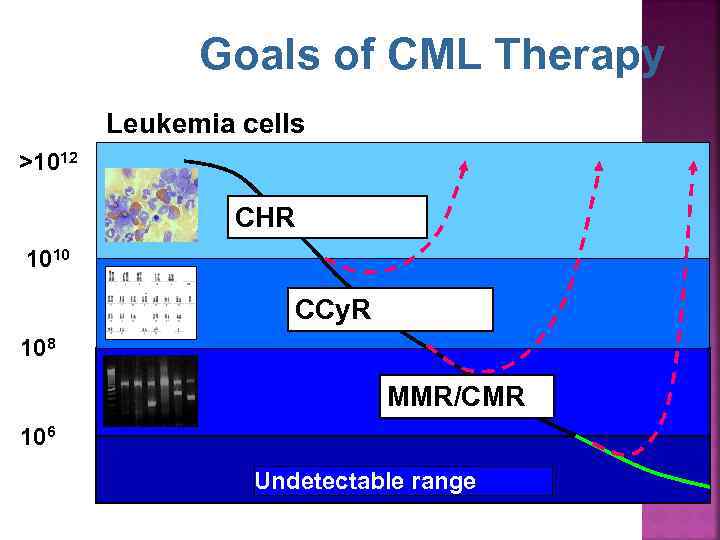 Goals of CML Therapy Leukemia cells >1012 CHR 1010 CCy. R 108 MMR/CMR 106 Undetectable range
Goals of CML Therapy Leukemia cells >1012 CHR 1010 CCy. R 108 MMR/CMR 106 Undetectable range
 Amount of Dz Definitions of Responses to Treatments 1 X 1012 1 X 1011 Hematologic Response Complete Hematologic response 1) Normal PB counts (WBC < 10 and plt < 450) 2) Normal WBC differential 3) No Dz symptoms 4) Normalization of the size of the liver and spleen Cytogenetic Responses: Ph+ Metaphases 1 X 1010 1) complete: 0% 2) partial: 1% - 35% 3) minor: 36% - 65% 4) minimal: 66% - 95% 5) none: 96% - 100% Molecular Responses: ratio of Bcr-Abl/Abl Major Molecular Response 3 -log 10 reduction from initial diagnosis sample (i. e. 25 → 0. 025) D. Bixby 1 X 10 8 -9
Amount of Dz Definitions of Responses to Treatments 1 X 1012 1 X 1011 Hematologic Response Complete Hematologic response 1) Normal PB counts (WBC < 10 and plt < 450) 2) Normal WBC differential 3) No Dz symptoms 4) Normalization of the size of the liver and spleen Cytogenetic Responses: Ph+ Metaphases 1 X 1010 1) complete: 0% 2) partial: 1% - 35% 3) minor: 36% - 65% 4) minimal: 66% - 95% 5) none: 96% - 100% Molecular Responses: ratio of Bcr-Abl/Abl Major Molecular Response 3 -log 10 reduction from initial diagnosis sample (i. e. 25 → 0. 025) D. Bixby 1 X 10 8 -9
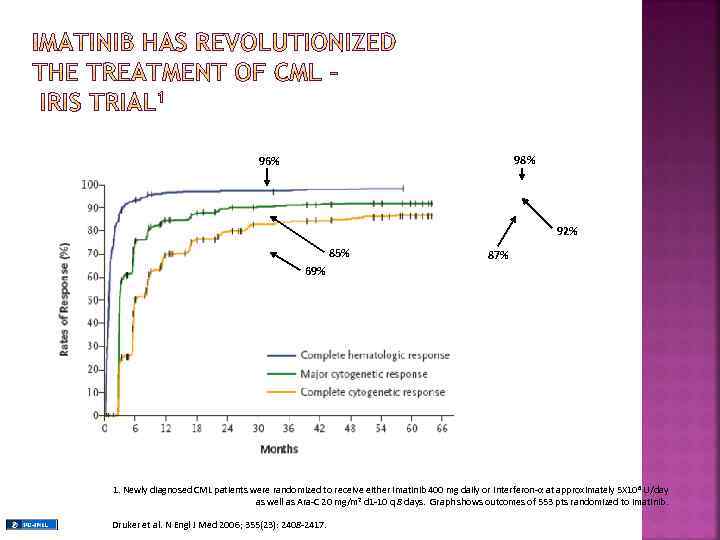 98% 96% 92% 85% 87% 69% 1. Newly diagnosed CML patients were randomized to receive either Imatinib 400 mg daily or Interferon-α at approximately 5 X 106 U/day as well as Ara-C 20 mg/m 2 d 1 -10 q 8 days. Graph shows outcomes of 553 pts randomized to Imatinib. Druker et al. N Engl J Med 2006; 355(23): 2408 -2417.
98% 96% 92% 85% 87% 69% 1. Newly diagnosed CML patients were randomized to receive either Imatinib 400 mg daily or Interferon-α at approximately 5 X 106 U/day as well as Ara-C 20 mg/m 2 d 1 -10 q 8 days. Graph shows outcomes of 553 pts randomized to Imatinib. Druker et al. N Engl J Med 2006; 355(23): 2408 -2417.
 Primary resistance ▪failure to achieve preset hematologic and/or cytogenetic milestones ▪IRIS data indicates a rate of ~ 15% by failing to a achieve a PCy. R at 12 months and 24% by failing to achieve a CCyr by 18 months of therapy. ▪rates higher in accelerated and blast phase disease Secondary resistance ▪loss of a previously achieved hematologic or cytogenetic milestone ▪rates may be 10 -15% on Imatinib, but become rarer as time on therapy progresses ▪rates higher in accelerated and blast phase disease Resistance Mechanisms 1) Bcr-Abl Kinase mutations ▪ > 50 known mutations within Abl sequence which inhibits Imatinib from binding ▪ mutations identified in 30 -80% of individuals with resistant disease 2) Bcr-Abl duplication of the Bcr-Abl sequence has been identified in cell lines with Im resistance 3) Pgp over-expression export pump of many chemotherapeuticsleading to lower intracellular Im concentration 4) h. Oct-1 under-expression import pump for Im which may lead to lower intracellular levels of IM 5) Src-Family kinase (SFK) expression activation may circumnavigate the Bcr-Abl ‘addiction’ of the transformed cell
Primary resistance ▪failure to achieve preset hematologic and/or cytogenetic milestones ▪IRIS data indicates a rate of ~ 15% by failing to a achieve a PCy. R at 12 months and 24% by failing to achieve a CCyr by 18 months of therapy. ▪rates higher in accelerated and blast phase disease Secondary resistance ▪loss of a previously achieved hematologic or cytogenetic milestone ▪rates may be 10 -15% on Imatinib, but become rarer as time on therapy progresses ▪rates higher in accelerated and blast phase disease Resistance Mechanisms 1) Bcr-Abl Kinase mutations ▪ > 50 known mutations within Abl sequence which inhibits Imatinib from binding ▪ mutations identified in 30 -80% of individuals with resistant disease 2) Bcr-Abl duplication of the Bcr-Abl sequence has been identified in cell lines with Im resistance 3) Pgp over-expression export pump of many chemotherapeuticsleading to lower intracellular Im concentration 4) h. Oct-1 under-expression import pump for Im which may lead to lower intracellular levels of IM 5) Src-Family kinase (SFK) expression activation may circumnavigate the Bcr-Abl ‘addiction’ of the transformed cell
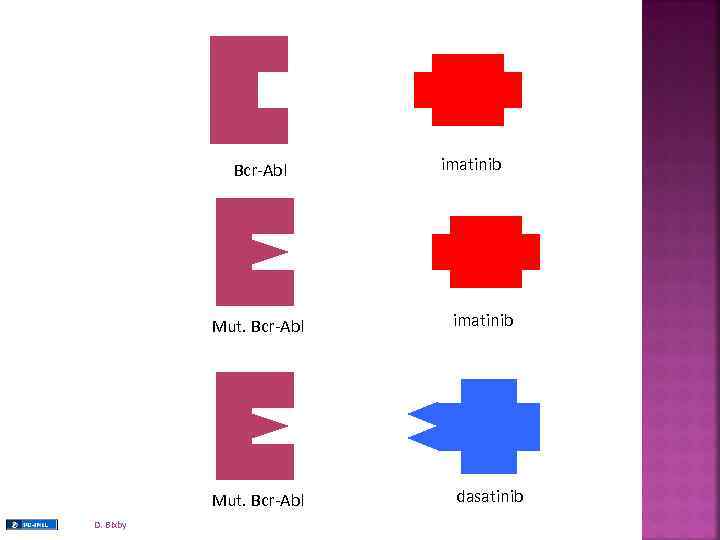 Bcr-Abl imatinib Mut. Bcr-Abl D. Bixby imatinib dasatinib
Bcr-Abl imatinib Mut. Bcr-Abl D. Bixby imatinib dasatinib
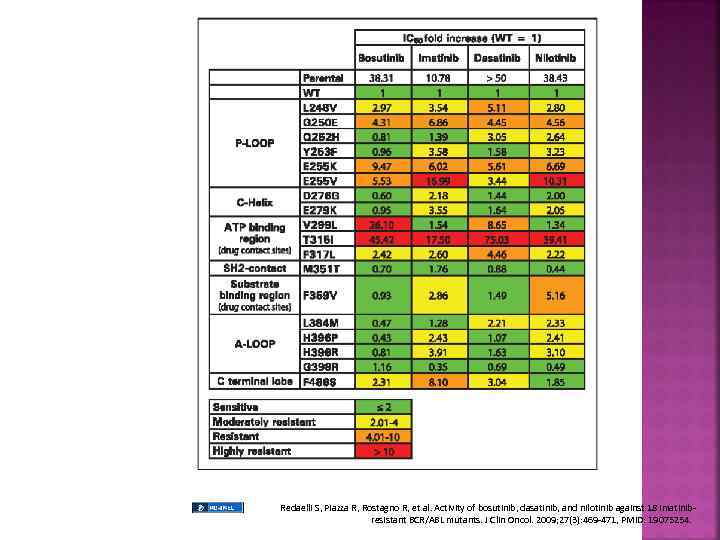 Redaelli S, Piazza R, Rostagno R, et al. Activity of bosutinib, dasatinib, and nilotinib against 18 imatinibresistant BCR/ABL mutants. J Clin Oncol. 2009; 27(3): 469 -471, PMID: 19075254.
Redaelli S, Piazza R, Rostagno R, et al. Activity of bosutinib, dasatinib, and nilotinib against 18 imatinibresistant BCR/ABL mutants. J Clin Oncol. 2009; 27(3): 469 -471, PMID: 19075254.
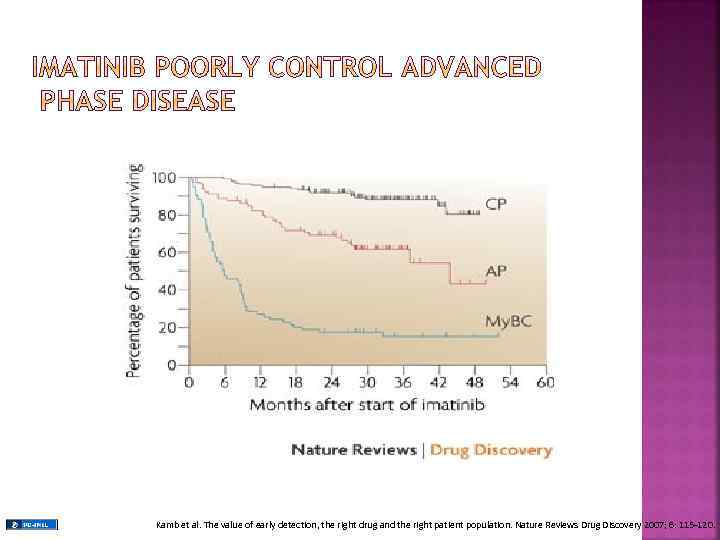 Kamb et al. The value of early detection, the right drug and the right patient population. Nature Reviews Drug Discovery 2007; 6: 115 -120.
Kamb et al. The value of early detection, the right drug and the right patient population. Nature Reviews Drug Discovery 2007; 6: 115 -120.
 1) Dose Escalation of imatinib 2) Second Generation TKIs 3) Bone Marrow Transplant 4) Clinical Trial Participation
1) Dose Escalation of imatinib 2) Second Generation TKIs 3) Bone Marrow Transplant 4) Clinical Trial Participation
 The FDA has approved 2 additional oral TKIs for the treatment of imatinib relapsed/refractory or imatinib intolerant CML dasatinib (Sprycel – BMS) nilotinib (Tasigna – Novartis) ▪ oral multi-kinase inhibitor ▪ ~ 325 times more potent than IM ▪ active against the ‘open’ and ‘closed confirmation of Bcr-Abl ▪ active against many of the identified kinase domain (KD) mutations ▪ active against the SFKs ▪ may not be a substrat for Pgp or h. Oct-1 ▪ oral multi-kinase inhibitor ▪ ~ 30 times more potent than IM ▪ active against only the closed confirmation of Bcr-Abl ▪ active against many of the KD mutations ▪ not active against the SKFs ▪ may not be a substrat for h. Oct-1
The FDA has approved 2 additional oral TKIs for the treatment of imatinib relapsed/refractory or imatinib intolerant CML dasatinib (Sprycel – BMS) nilotinib (Tasigna – Novartis) ▪ oral multi-kinase inhibitor ▪ ~ 325 times more potent than IM ▪ active against the ‘open’ and ‘closed confirmation of Bcr-Abl ▪ active against many of the identified kinase domain (KD) mutations ▪ active against the SFKs ▪ may not be a substrat for Pgp or h. Oct-1 ▪ oral multi-kinase inhibitor ▪ ~ 30 times more potent than IM ▪ active against only the closed confirmation of Bcr-Abl ▪ active against many of the KD mutations ▪ not active against the SKFs ▪ may not be a substrat for h. Oct-1
 § Allogeneic bone marrow transplantation remains the only known curative option in CML with Graft vs. Leukemia effect, in molecular relapse can achieve remission by Donor Lymphocyte Infusion § Associated with an increased morbidity and mortality (TRM -10%-30%) § Therefore, not typically applied for upfront therapy for CML ▪ considered only in cases of matched-related donor for extremely young pts (pediatrics) § However, often considered in those with relapsed/refractory disease to TKI based therapies ▪ efficacy of the transplant dependent upon the phase of the disease at the time of the transplant: CP>AP>BP
§ Allogeneic bone marrow transplantation remains the only known curative option in CML with Graft vs. Leukemia effect, in molecular relapse can achieve remission by Donor Lymphocyte Infusion § Associated with an increased morbidity and mortality (TRM -10%-30%) § Therefore, not typically applied for upfront therapy for CML ▪ considered only in cases of matched-related donor for extremely young pts (pediatrics) § However, often considered in those with relapsed/refractory disease to TKI based therapies ▪ efficacy of the transplant dependent upon the phase of the disease at the time of the transplant: CP>AP>BP
 thank you
thank you


