89213819cd5499cc5f3711a13d3259cd.ppt
- Количество слайдов: 34
 Dr Cheng Wing Ho Stephen ICU, Pamela Youde Nethersole Eastern Hospital Friday Meeting Jan 21, 2011 CARBAPENEM-RESISTANT ENTEROBACTERIACEAE (CRE)
Dr Cheng Wing Ho Stephen ICU, Pamela Youde Nethersole Eastern Hospital Friday Meeting Jan 21, 2011 CARBAPENEM-RESISTANT ENTEROBACTERIACEAE (CRE)
 What is CRE? Mechanism of resistance International cases 3 recent cases in Hong Kong Possible treatment options HA / CDC guidelines on infection control
What is CRE? Mechanism of resistance International cases 3 recent cases in Hong Kong Possible treatment options HA / CDC guidelines on infection control
 Enterobacteriaceae Gram -ve bacilli Facultative anaerobes Gut flora, or found in soil / water Common causative agents of UTI, pneumonia Examples: E. coli, Klebsiella, Salmonella, Shigella, Proteus
Enterobacteriaceae Gram -ve bacilli Facultative anaerobes Gut flora, or found in soil / water Common causative agents of UTI, pneumonia Examples: E. coli, Klebsiella, Salmonella, Shigella, Proteus
 Treatment options - Penicillin (e. g. ampicillin, amoxicillin, piperacillin) Cephalosporin (e. g. cefuroxime, ceftriaxone) (combined with β-lactamase inhibitor) Augmentin, Tazocin, Sulperazone - Carbapenem (e. g. imipenem, ertepenem, meropenem) Quinolone (eg. ciprofloxacin, levofloxacin) Extended-spectrum-β-lactamase strains - only sensitive to carbapenem group and limited penicillin/cephalosporin coupled with β-lactamase inhibitors
Treatment options - Penicillin (e. g. ampicillin, amoxicillin, piperacillin) Cephalosporin (e. g. cefuroxime, ceftriaxone) (combined with β-lactamase inhibitor) Augmentin, Tazocin, Sulperazone - Carbapenem (e. g. imipenem, ertepenem, meropenem) Quinolone (eg. ciprofloxacin, levofloxacin) Extended-spectrum-β-lactamase strains - only sensitive to carbapenem group and limited penicillin/cephalosporin coupled with β-lactamase inhibitors
 Enterobacteriaceae strains resistant to carbapenem have emerged in the past decade Wide-spread outbreaks have been reported in recent years No reliable treatment options Huge reservoir of carriers in healthy population Easy species-to-species spread Potential devastating health consequences
Enterobacteriaceae strains resistant to carbapenem have emerged in the past decade Wide-spread outbreaks have been reported in recent years No reliable treatment options Huge reservoir of carriers in healthy population Easy species-to-species spread Potential devastating health consequences
 Carbapenemase b-lactamase - hydrolyzing agent - 4 classes (A-D): serine vs zinc - produced by ‘Superbugs’ - Klebsiella Pneumoniae Carbapenemase (KPC) Verona integron-encoded metallo-β-lactamase (VIM) Oxallinase group β-lactamase (OXA) SME, IMI, NMC and Ccr. A (little clinical significance)
Carbapenemase b-lactamase - hydrolyzing agent - 4 classes (A-D): serine vs zinc - produced by ‘Superbugs’ - Klebsiella Pneumoniae Carbapenemase (KPC) Verona integron-encoded metallo-β-lactamase (VIM) Oxallinase group β-lactamase (OXA) SME, IMI, NMC and Ccr. A (little clinical significance)
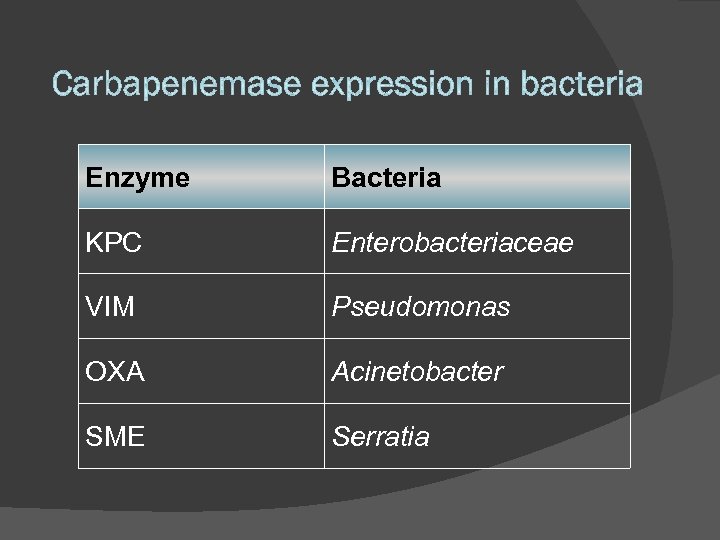 Enzyme Bacteria KPC Enterobacteriaceae VIM Pseudomonas OXA Acinetobacter SME Serratia
Enzyme Bacteria KPC Enterobacteriaceae VIM Pseudomonas OXA Acinetobacter SME Serratia
 Klebsiella Pneumoniae Carbapenemase (KPC) - first detected in 1996 most commonly expressed in K. pneumonia also reported in Citrobacter, Enterobacter , E. coli, Salmonella, Serratia - - class A b-lactamase causes resistance to all b-lactams including extendedspectrum cephalosporins and carbapenems
Klebsiella Pneumoniae Carbapenemase (KPC) - first detected in 1996 most commonly expressed in K. pneumonia also reported in Citrobacter, Enterobacter , E. coli, Salmonella, Serratia - - class A b-lactamase causes resistance to all b-lactams including extendedspectrum cephalosporins and carbapenems
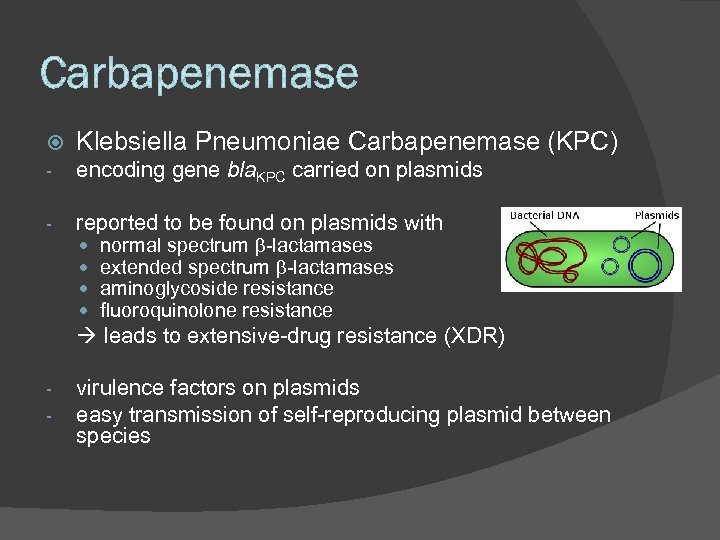 Klebsiella Pneumoniae Carbapenemase (KPC) - encoding gene bla. KPC carried on plasmids - reported to be found on plasmids with normal spectrum b-lactamases extended spectrum b-lactamases aminoglycoside resistance fluoroquinolone resistance leads to extensive-drug resistance (XDR) - virulence factors on plasmids easy transmission of self-reproducing plasmid between species
Klebsiella Pneumoniae Carbapenemase (KPC) - encoding gene bla. KPC carried on plasmids - reported to be found on plasmids with normal spectrum b-lactamases extended spectrum b-lactamases aminoglycoside resistance fluoroquinolone resistance leads to extensive-drug resistance (XDR) - virulence factors on plasmids easy transmission of self-reproducing plasmid between species
 European Antimicrobial Resistance Surveillance System (EARSS) - CRKP in 0. 3% of blood isolates prior to 2006 - following an outbreak in 2006 in Israel 11% in 2006, 22% in 2007 mortality associated with CRKP isolation up to 44% - standardized morality rate ~8 deaths per 100, 000 - compared to 6 deaths per 100, 000 of MRSA (2005) - Predictors of CRKP acquisition among hospitalized adults and effect of acquisition on mortality. Antimicrob Agents Chemother. 2008; 52(3): 1028 -1033
European Antimicrobial Resistance Surveillance System (EARSS) - CRKP in 0. 3% of blood isolates prior to 2006 - following an outbreak in 2006 in Israel 11% in 2006, 22% in 2007 mortality associated with CRKP isolation up to 44% - standardized morality rate ~8 deaths per 100, 000 - compared to 6 deaths per 100, 000 of MRSA (2005) - Predictors of CRKP acquisition among hospitalized adults and effect of acquisition on mortality. Antimicrob Agents Chemother. 2008; 52(3): 1028 -1033
 New Delhi metallo-b-lactamase (NDM-1) - class B b-lactamase plasmid carrying encoding gene bla. NDM - first detected in 2009 from an isolate of K. pneumoniae Swedish patient with antibiotic-resistant infection acquired and unsuccessfully treated in New Delhi, India novel gene identified in Sweden
New Delhi metallo-b-lactamase (NDM-1) - class B b-lactamase plasmid carrying encoding gene bla. NDM - first detected in 2009 from an isolate of K. pneumoniae Swedish patient with antibiotic-resistant infection acquired and unsuccessfully treated in New Delhi, India novel gene identified in Sweden
 New Delhi metallo-b-lactamase (NDM-1) - over a 3 -month period in Mumbai, India, 24 cases of carbapenam resistant isolates were identified 22 carry the NDM-1 gene 14 out of 24 patients K. pneumoniae (10) come from E. coli (9) ICU 14 samples from ICU Carbapenem-resistance 0% 8% in 3 years - - “New Delhi Metallo-β lactamase (NDM-1) in Enterobacteriaceae: Treatment options with Carbapenems Compromised”. Journal of Association of Physicians of India 58: 147– 150, March 2010
New Delhi metallo-b-lactamase (NDM-1) - over a 3 -month period in Mumbai, India, 24 cases of carbapenam resistant isolates were identified 22 carry the NDM-1 gene 14 out of 24 patients K. pneumoniae (10) come from E. coli (9) ICU 14 samples from ICU Carbapenem-resistance 0% 8% in 3 years - - “New Delhi Metallo-β lactamase (NDM-1) in Enterobacteriaceae: Treatment options with Carbapenems Compromised”. Journal of Association of Physicians of India 58: 147– 150, March 2010
 Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study Lancet Infect Dis. 2010 September ; 10(9): 597– 602 UK (37) - all had travelled to India/Pakistan Chennai (44), Haryana (26) other sites in India and Pakistan (73) K. pneumoniae (111) E. coli (36) Globally, estimated 250 identified isolates so far http: //www. ndm 1 bacteria. com
Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study Lancet Infect Dis. 2010 September ; 10(9): 597– 602 UK (37) - all had travelled to India/Pakistan Chennai (44), Haryana (26) other sites in India and Pakistan (73) K. pneumoniae (111) E. coli (36) Globally, estimated 250 identified isolates so far http: //www. ndm 1 bacteria. com
 - May 2010, first E. coli isolate in UK Indian man received dialysis in India 18 months ago June 2010, 3 Enterobacteriaciae strands in US all received medical procedures in Indian hospitals previously July 2010, 3 isolates of Acinetobacter baumannii in New Delhi, ICU August 2010, first identified case in Ontario, Canada September 2010, first case in Tokyo, Japan
- May 2010, first E. coli isolate in UK Indian man received dialysis in India 18 months ago June 2010, 3 Enterobacteriaciae strands in US all received medical procedures in Indian hospitals previously July 2010, 3 isolates of Acinetobacter baumannii in New Delhi, ICU August 2010, first identified case in Ontario, Canada September 2010, first case in Tokyo, Japan
 August 2010 - Belgian man - road-traffic-accident in Pakistan - hospitalized for major leg injury infected with an isolate of NDM-1 - succumbed despite use of Colistin -
August 2010 - Belgian man - road-traffic-accident in Pakistan - hospitalized for major leg injury infected with an isolate of NDM-1 - succumbed despite use of Colistin -
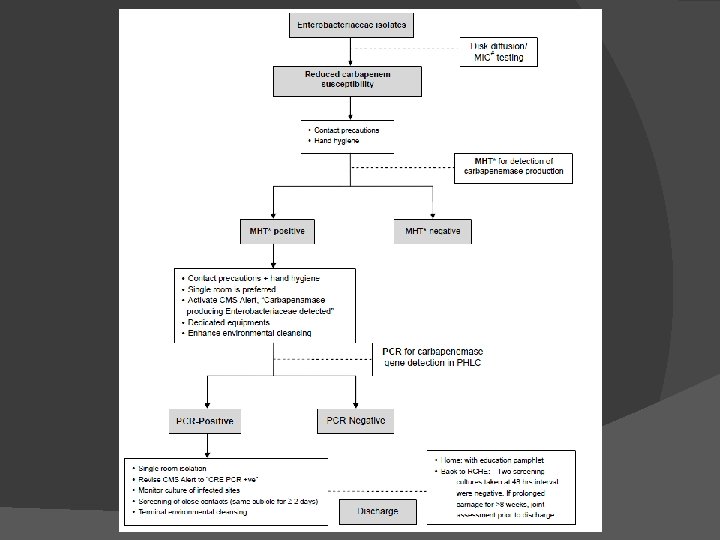
 Enterobacteriaceae resistant or moderately resistant to Carbapenem PCR test for carbapenemase gene Isolates that are susceptible but with increased minimal inhibitory concentrations (MIC) Modified Hodge Test test sample -ve control +ve control If positive Modified Hodge Test proceed to PCR test for gene identity manage as CRE Clinical and Laboratory Standards Institute. 2009 performance standards for antimicrobial susceptibility testing. 2009.
Enterobacteriaceae resistant or moderately resistant to Carbapenem PCR test for carbapenemase gene Isolates that are susceptible but with increased minimal inhibitory concentrations (MIC) Modified Hodge Test test sample -ve control +ve control If positive Modified Hodge Test proceed to PCR test for gene identity manage as CRE Clinical and Laboratory Standards Institute. 2009 performance standards for antimicrobial susceptibility testing. 2009.
 Incidence of CRE in 2009 is about 0. 05% 8 isolates carrying the metallo-b-lactamases (IMP type) E. coli, Klebsiella spp. and Citrobacter freundii Mostly colonizing flora No cases of KPC isolates detected so far 2 cases of recently identified New Delhi metallo-blactamase isolate An overview of surveillance of antimicrobialresistance by CHP in Hong Kong Communicable Diseases Watch 2010; Volume 7, Number 17. Aug 2010
Incidence of CRE in 2009 is about 0. 05% 8 isolates carrying the metallo-b-lactamases (IMP type) E. coli, Klebsiella spp. and Citrobacter freundii Mostly colonizing flora No cases of KPC isolates detected so far 2 cases of recently identified New Delhi metallo-blactamase isolate An overview of surveillance of antimicrobialresistance by CHP in Hong Kong Communicable Diseases Watch 2010; Volume 7, Number 17. Aug 2010
 Case 1 (October 2010) - 66 year old Indian man - out-patient - followed up for hypertension, diabetes treated for UTI that responded to ciprofloxacin - NDM-1 carrying E. coli detected in urine culture - history of spending 3 weeks in India in March 2010 - no history of hospitilization in India - - full recovery
Case 1 (October 2010) - 66 year old Indian man - out-patient - followed up for hypertension, diabetes treated for UTI that responded to ciprofloxacin - NDM-1 carrying E. coli detected in urine culture - history of spending 3 weeks in India in March 2010 - no history of hospitilization in India - - full recovery
 Case 2 (December 2010) - 54 year old Indian lady - treated for subdural hematoma in India from May to August 2010 attended and admitted to PMH on Dec 17 for respiratory distress - NDM-1 carrying E. coli detected in rectal swab - likely colonization due as no evidence of infection -
Case 2 (December 2010) - 54 year old Indian lady - treated for subdural hematoma in India from May to August 2010 attended and admitted to PMH on Dec 17 for respiratory distress - NDM-1 carrying E. coli detected in rectal swab - likely colonization due as no evidence of infection -
 ICU in-patient in D 10 (January 2010) - 83 year old Chinese man recent hospitilization in the Philippines biliary papillomatosis admitted for robotic assisted laparoscopic L hepatectomy, complicated with leakage - - CRE was cultured from bile only sensitive to Gentamicin (treated with Tazocin) PCR test positive for VIM gene (class B) patient discharged home case reported and contact tracing underway
ICU in-patient in D 10 (January 2010) - 83 year old Chinese man recent hospitilization in the Philippines biliary papillomatosis admitted for robotic assisted laparoscopic L hepatectomy, complicated with leakage - - CRE was cultured from bile only sensitive to Gentamicin (treated with Tazocin) PCR test positive for VIM gene (class B) patient discharged home case reported and contact tracing underway
 Antimicrobial Interpretation Amikacin I Chloramphenicol R Amox/clav R Ciprofloxacin R Ampicillin R Ertapenem R Aztreonam R Gentamicin R Cefazolin R Imipenem R Cefpodoxime R Meropenem R Cefotaxime R Pipercillin/Tazo R Cetotetan R Tobramycin R Cefoxitin R Trimeth/Sulfa R Ceftazidime R Polymyxin B MIC >4μg/ml Ceftriaxone R Colistin MIC >4μg/ml Cefepime R Tigecycline S
Antimicrobial Interpretation Amikacin I Chloramphenicol R Amox/clav R Ciprofloxacin R Ampicillin R Ertapenem R Aztreonam R Gentamicin R Cefazolin R Imipenem R Cefpodoxime R Meropenem R Cefotaxime R Pipercillin/Tazo R Cetotetan R Tobramycin R Cefoxitin R Trimeth/Sulfa R Ceftazidime R Polymyxin B MIC >4μg/ml Ceftriaxone R Colistin MIC >4μg/ml Cefepime R Tigecycline S
 Cyclic peptide with hydrophobic tail Produced by Bacillus polymyxa Selectively targets Gram-ve bacteria by disrupting both outer and inner membranes Polymyxin B Colistin (polymyxin E) Only effectively via parenteral route No guidelines for standard / optimum / max dose Nephrotoxic and neurotoxic Shown to be useful against MRAB, Pseudomonas
Cyclic peptide with hydrophobic tail Produced by Bacillus polymyxa Selectively targets Gram-ve bacteria by disrupting both outer and inner membranes Polymyxin B Colistin (polymyxin E) Only effectively via parenteral route No guidelines for standard / optimum / max dose Nephrotoxic and neurotoxic Shown to be useful against MRAB, Pseudomonas
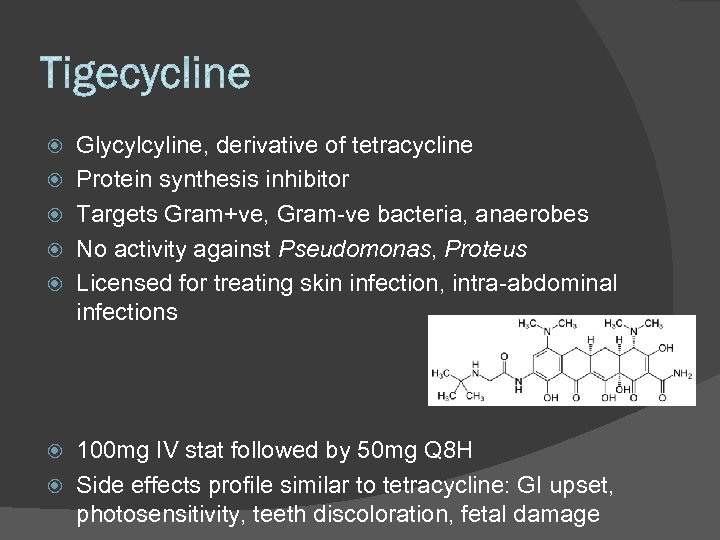 Glycylcyline, derivative of tetracycline Protein synthesis inhibitor Targets Gram+ve, Gram-ve bacteria, anaerobes No activity against Pseudomonas, Proteus Licensed for treating skin infection, intra-abdominal infections 100 mg IV stat followed by 50 mg Q 8 H Side effects profile similar to tetracycline: GI upset, photosensitivity, teeth discoloration, fetal damage
Glycylcyline, derivative of tetracycline Protein synthesis inhibitor Targets Gram+ve, Gram-ve bacteria, anaerobes No activity against Pseudomonas, Proteus Licensed for treating skin infection, intra-abdominal infections 100 mg IV stat followed by 50 mg Q 8 H Side effects profile similar to tetracycline: GI upset, photosensitivity, teeth discoloration, fetal damage
 NXL 104 - novel non-β-lactamase inhibitor - use in combination with Ceftazidime GSK 299423 (Glaxo. Smith. Kline 299423) - inhibits the enzyme topoisomerase - effective in clearing NDM-1 ACHN-490 and other Aminoglycosides - no activity against the NDM-1 isolates
NXL 104 - novel non-β-lactamase inhibitor - use in combination with Ceftazidime GSK 299423 (Glaxo. Smith. Kline 299423) - inhibits the enzyme topoisomerase - effective in clearing NDM-1 ACHN-490 and other Aminoglycosides - no activity against the NDM-1 isolates
 From data of other countries, KPC associated infections are mostly systemic infections NDM-1 associated infections are usually UTI, pneumonia, and blood stream infection Risk factors: - prolonged hospitalization ICU stay invasive devices immunosuppression prior use of multiple antibiotics - Infection Control Guideline on Carbapenem Resistant Enterobacteriaceae (CRE), Hospital Authority, 1 Dec 2010
From data of other countries, KPC associated infections are mostly systemic infections NDM-1 associated infections are usually UTI, pneumonia, and blood stream infection Risk factors: - prolonged hospitalization ICU stay invasive devices immunosuppression prior use of multiple antibiotics - Infection Control Guideline on Carbapenem Resistant Enterobacteriaceae (CRE), Hospital Authority, 1 Dec 2010

 1) Prudent antibiotic use Antibiotic Stewardship Program (ASP) should be in place to give advice on and monitor the appropriate use of antimicrobials in patient care 2) Early detection of CRE Active surveillance culture (ASC) - rectal swab/stool culture for patients hospitalized outside Hong Kong in the last 6 months Screen carriage for same cubicle in-patients (>=2 days) of PCR +ve CRE, inform Infection Control Branch if discharged to elderly home, institutions Infection Control Guideline on Carbapenem Resistant Enterobacteriaceae (CRE), Hospital Authority, 1 Dec 2010
1) Prudent antibiotic use Antibiotic Stewardship Program (ASP) should be in place to give advice on and monitor the appropriate use of antimicrobials in patient care 2) Early detection of CRE Active surveillance culture (ASC) - rectal swab/stool culture for patients hospitalized outside Hong Kong in the last 6 months Screen carriage for same cubicle in-patients (>=2 days) of PCR +ve CRE, inform Infection Control Branch if discharged to elderly home, institutions Infection Control Guideline on Carbapenem Resistant Enterobacteriaceae (CRE), Hospital Authority, 1 Dec 2010
 3) Review of microbiology record of previous 6 -12 months detect unrecognized cases if cases identified point prevalence survey (a single round of active surveillance cultures) in high risk units Example: - ICU - units where the identified case previously occupied - units exposed to broad-spectrum antimicrobials 4) Laboratory protocols for detecting CRE Guidance for Control of Infections with Carbapenem-Resistant or Carbapenemase-Producing Enterobacteriaceae in Acute Care Facilities, March 2009
3) Review of microbiology record of previous 6 -12 months detect unrecognized cases if cases identified point prevalence survey (a single round of active surveillance cultures) in high risk units Example: - ICU - units where the identified case previously occupied - units exposed to broad-spectrum antimicrobials 4) Laboratory protocols for detecting CRE Guidance for Control of Infections with Carbapenem-Resistant or Carbapenemase-Producing Enterobacteriaceae in Acute Care Facilities, March 2009
 1) Infection control measures: - Single room isolation discontinue when infected site cleared or eradication of carriage in gut (culture negative for at least 2 consecutive stool/ rectal swabs collected at 48 hours interval) Contact precautions Hand hygiene Dedicated equipments Enhance / terminal environmental cleansing Minimize patient transfer Advice to visitors Infection Control Guideline on Carbapenem Resistant Enterobacteriaceae (CRE), Hospital Authority, 1 Dec 2010
1) Infection control measures: - Single room isolation discontinue when infected site cleared or eradication of carriage in gut (culture negative for at least 2 consecutive stool/ rectal swabs collected at 48 hours interval) Contact precautions Hand hygiene Dedicated equipments Enhance / terminal environmental cleansing Minimize patient transfer Advice to visitors Infection Control Guideline on Carbapenem Resistant Enterobacteriaceae (CRE), Hospital Authority, 1 Dec 2010
 2) Alert and reporting: Inform the Chief Infection Control Officer’s Office (CICO Office) of any patient with PCR +ve CRE or when an outbreak of CRE is suspected MHT positive cases should label in the CMS alert ‘Carbapenemase producing Enterobacteriaceae detected’ Revise CMS alert to ‘CRE PCR +ve’ if PCR tested positive The alert should be removed when the patient has been eradicated of CRE When CRE PCR +ve patient is re-admitted, infection control precautions should taken and ICT informed Infection Control Guideline on Carbapenem Resistant Enterobacteriaceae (CRE), Hospital Authority, 1 Dec 2010
2) Alert and reporting: Inform the Chief Infection Control Officer’s Office (CICO Office) of any patient with PCR +ve CRE or when an outbreak of CRE is suspected MHT positive cases should label in the CMS alert ‘Carbapenemase producing Enterobacteriaceae detected’ Revise CMS alert to ‘CRE PCR +ve’ if PCR tested positive The alert should be removed when the patient has been eradicated of CRE When CRE PCR +ve patient is re-admitted, infection control precautions should taken and ICT informed Infection Control Guideline on Carbapenem Resistant Enterobacteriaceae (CRE), Hospital Authority, 1 Dec 2010
 3) Transfer / discharge issues ICT involved in all inter-hospital or intra-hospital transfer of CRE +ve patient Discharge to institutions / elderly home should have 2 consecutive negative screening culture at 48 hours interval. ICB must be informed asymptomatic prolonged CRE carriage (e. g. >=8 weeks) must be risk assessed jointly by the hospital ICT, CICO and ICB Patients discharged home should be given education pamphlet on infection control precautions Patients with CRE +ve confirmed after discharge should be contacted for information and education on infection control precautions Infection Control Guideline on Carbapenem Resistant Enterobacteriaceae (CRE), Hospital Authority, 1 Dec 2010
3) Transfer / discharge issues ICT involved in all inter-hospital or intra-hospital transfer of CRE +ve patient Discharge to institutions / elderly home should have 2 consecutive negative screening culture at 48 hours interval. ICB must be informed asymptomatic prolonged CRE carriage (e. g. >=8 weeks) must be risk assessed jointly by the hospital ICT, CICO and ICB Patients discharged home should be given education pamphlet on infection control precautions Patients with CRE +ve confirmed after discharge should be contacted for information and education on infection control precautions Infection Control Guideline on Carbapenem Resistant Enterobacteriaceae (CRE), Hospital Authority, 1 Dec 2010
 Carbapenem-Resistant Enterobacteriaceae is an emerging threat to hospitalized patients Global spread High mortality Treatment options are limited Active surveillance should be performed for high risk patients with overseas hospitalization in past 6 months Contact tracing / screening for confirmed cases Importance of infection control measures High vigilence in critically ill ICU patients
Carbapenem-Resistant Enterobacteriaceae is an emerging threat to hospitalized patients Global spread High mortality Treatment options are limited Active surveillance should be performed for high risk patients with overseas hospitalization in past 6 months Contact tracing / screening for confirmed cases Importance of infection control measures High vigilence in critically ill ICU patients
 Thank you! The End
Thank you! The End


