a54028ce6f0cf40691646b6772afdb43.ppt
- Количество слайдов: 19

Downloaded from www. ezetrol. ae Ezetimibe Coadministered with Simvastatin in Patients with Hypercholesterolemia and Coronary Heart Disease Results of Two Randomized, Double-Blind, Placebo-Controlled Trials Slide 1

Downloaded from www. ezetrol. ae Study Objectives In CHD patients not at LDL-C goal (<2. 60 mmol/L) on simvastatin, to determine the effects of coadministration of ezetimibe on – LDL-C goal attainment – LDL-C reduction – TC, TG, HDL-C, and other lipid parameters – Safety profile (adverse experiences, laboratory values, and vital signs) CHD=coronary heart disease; LDL-C=low-density lipoprotein cholesterol; TC=total cholesterol; TG=triglycerides; HDL-C=high-density lipoprotein cholesterol Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 2
Downloaded from www. ezetrol. ae Participating Countries Belgium Croatia Czech Republic Egypt France Germany Greece Hungary Israel Italy Lebanon Netherlands Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. Norway Portugal Romania Russian Federation Saudi Arabia Slovenia Spain Sweden Switzerland Turkey United Arab Emirates 3
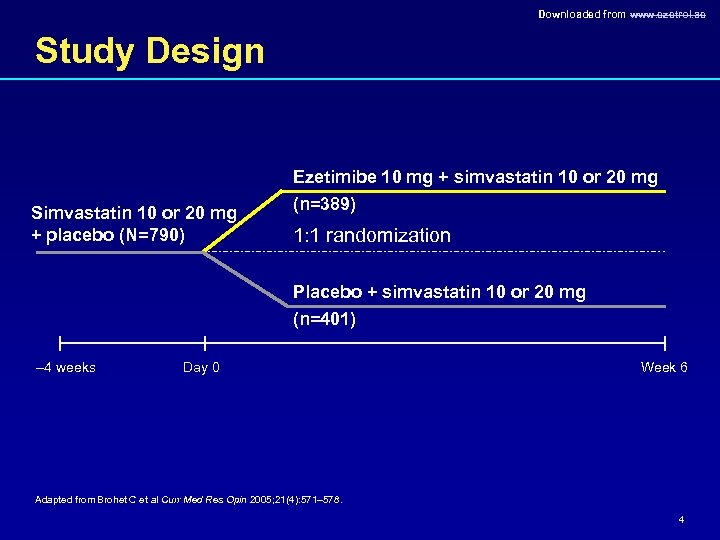
Downloaded from www. ezetrol. ae Study Design Ezetimibe 10 mg + simvastatin 10 or 20 mg Simvastatin 10 or 20 mg + placebo (N=790) (n=389) 1: 1 randomization Placebo + simvastatin 10 or 20 mg (n=401) – 4 weeks Day 0 Week 6 Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 4

Downloaded from www. ezetrol. ae Efficacy Endpoints • Primary – Percentage of patients achieving LDL-C goal of ≤ 2. 60 mmol/L (≤ 100 mg/dl) at study end (week 6) • Secondary – Percentage change from baseline in LDL-C, TG, HDL-C, and other lipid parameters Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 5
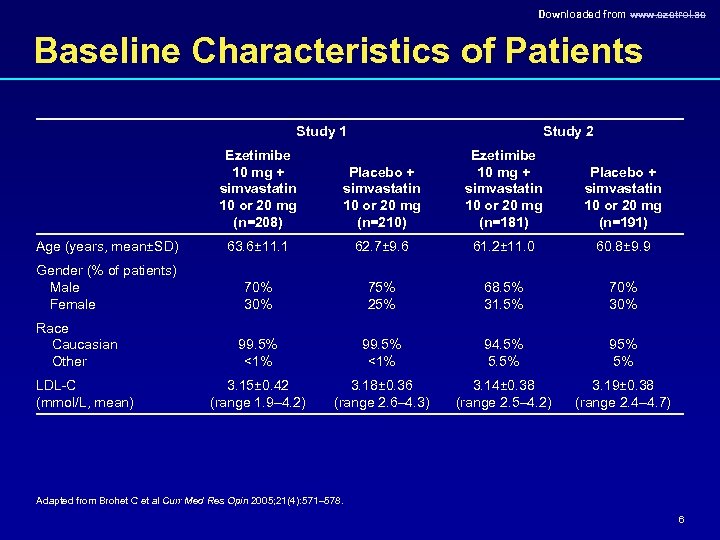
Downloaded from www. ezetrol. ae Baseline Characteristics of Patients Study 1 Ezetimibe 10 mg + simvastatin 10 or 20 mg (n=208) Age (years, mean±SD) Gender (% of patients) Male Female Race Caucasian Other LDL-C (mmol/L, mean) Study 2 Placebo + simvastatin 10 or 20 mg (n=210) Ezetimibe 10 mg + simvastatin 10 or 20 mg (n=181) Placebo + simvastatin 10 or 20 mg (n=191) 63. 6± 11. 1 62. 7± 9. 6 61. 2± 11. 0 60. 8± 9. 9 70% 30% 75% 25% 68. 5% 31. 5% 70% 30% 99. 5% <1% 94. 5% 5. 5% 95% 5% 3. 15± 0. 42 (range 1. 9– 4. 2) 3. 18± 0. 36 (range 2. 6– 4. 3) 3. 14± 0. 38 (range 2. 5– 4. 2) 3. 19± 0. 38 (range 2. 4– 4. 7) Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 6
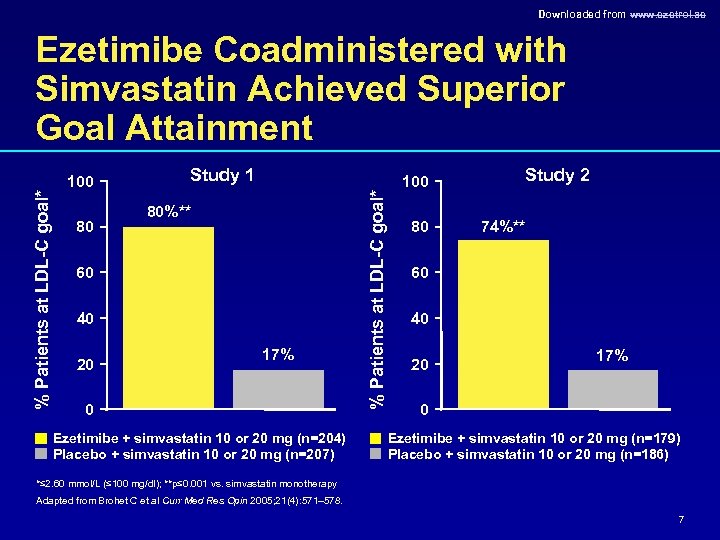
Downloaded from www. ezetrol. ae 100 80 Study 1 80%** 60 40 20 17% 0 Ezetimibe + simvastatin 10 or 20 mg (n=204) Placebo + simvastatin 10 or 20 mg (n=207) % Patients at LDL-C goal* Ezetimibe Coadministered with Simvastatin Achieved Superior Goal Attainment 100 80 Study 2 74%** 60 40 20 17% 0 Ezetimibe + simvastatin 10 or 20 mg (n=179) Placebo + simvastatin 10 or 20 mg (n=186) *≤ 2. 60 mmol/L (≤ 100 mg/dl); **p≤ 0. 001 vs. simvastatin monotherapy Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 7
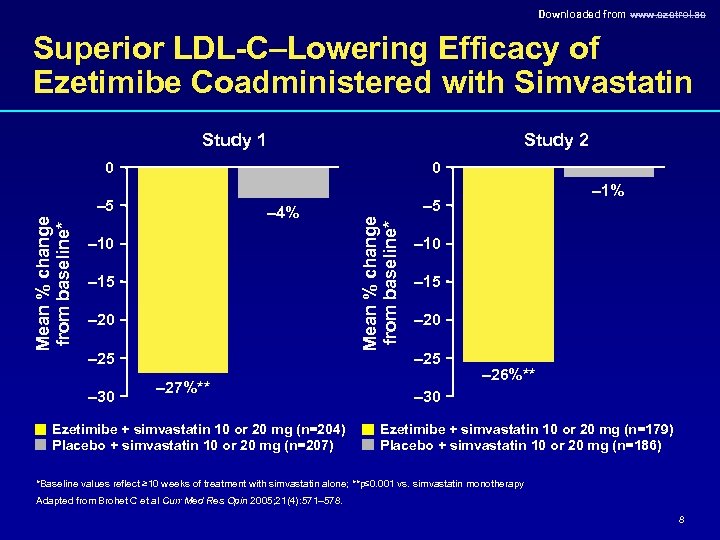
Downloaded from www. ezetrol. ae Superior LDL-C–Lowering Efficacy of Ezetimibe Coadministered with Simvastatin Study 1 Study 2 0 0 – 4% – 10 – 15 – 20 – 25 – 30 – 27%** Ezetimibe + simvastatin 10 or 20 mg (n=204) Placebo + simvastatin 10 or 20 mg (n=207) – 5 Mean % change from baseline* – 5 – 1% – 10 – 15 – 20 – 25 – 26%** – 30 Ezetimibe + simvastatin 10 or 20 mg (n=179) Placebo + simvastatin 10 or 20 mg (n=186) *Baseline values reflect ≥ 10 weeks of treatment with simvastatin alone; **p≤ 0. 001 vs. simvastatin monotherapy Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 8
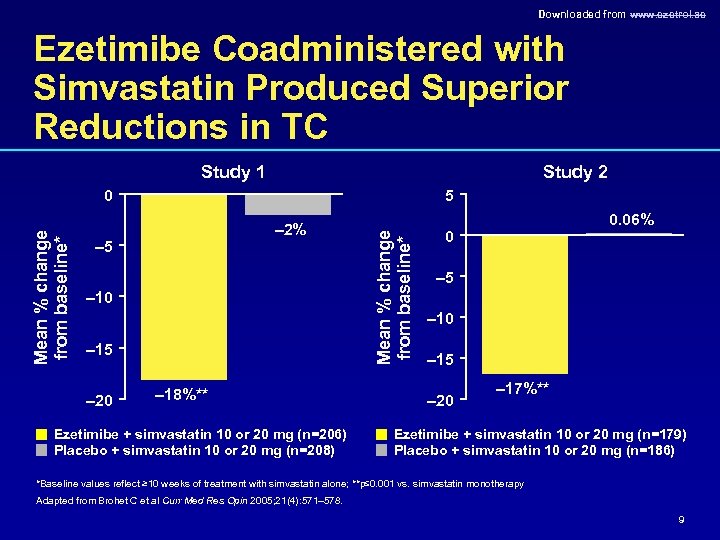
Downloaded from www. ezetrol. ae Ezetimibe Coadministered with Simvastatin Produced Superior Reductions in TC Study 1 Study 2 5 – 2% – 5 – 10 – 15 – 20 – 18%** Ezetimibe + simvastatin 10 or 20 mg (n=206) Placebo + simvastatin 10 or 20 mg (n=208) Mean % change from baseline* 0 0. 06% 0 – 5 – 10 – 15 – 20 – 17%** Ezetimibe + simvastatin 10 or 20 mg (n=179) Placebo + simvastatin 10 or 20 mg (n=186) *Baseline values reflect ≥ 10 weeks of treatment with simvastatin alone; **p≤ 0. 001 vs. simvastatin monotherapy Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 9
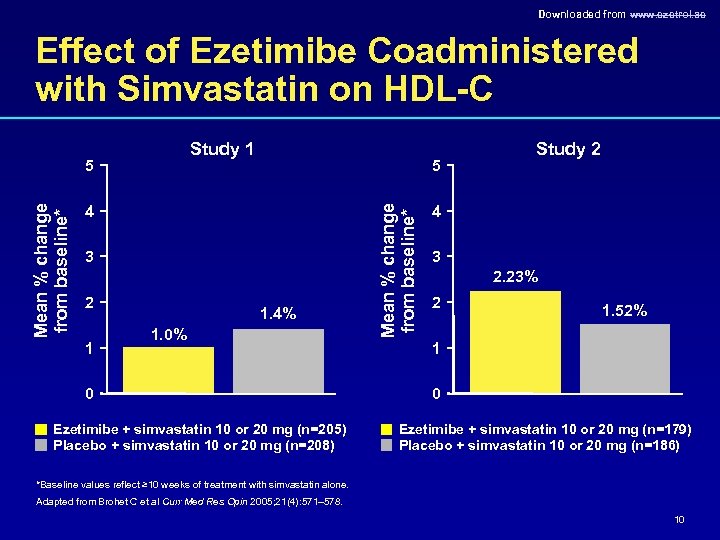
Downloaded from www. ezetrol. ae Effect of Ezetimibe Coadministered with Simvastatin on HDL-C Study 1 5 4 3 2 1 1. 4% 1. 0% 0 Ezetimibe + simvastatin 10 or 20 mg (n=205) Placebo + simvastatin 10 or 20 mg (n=208) Mean % change from baseline* 5 Study 2 4 3 2. 23% 2 1. 52% 1 0 Ezetimibe + simvastatin 10 or 20 mg (n=179) Placebo + simvastatin 10 or 20 mg (n=186) *Baseline values reflect ≥ 10 weeks of treatment with simvastatin alone. Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 10
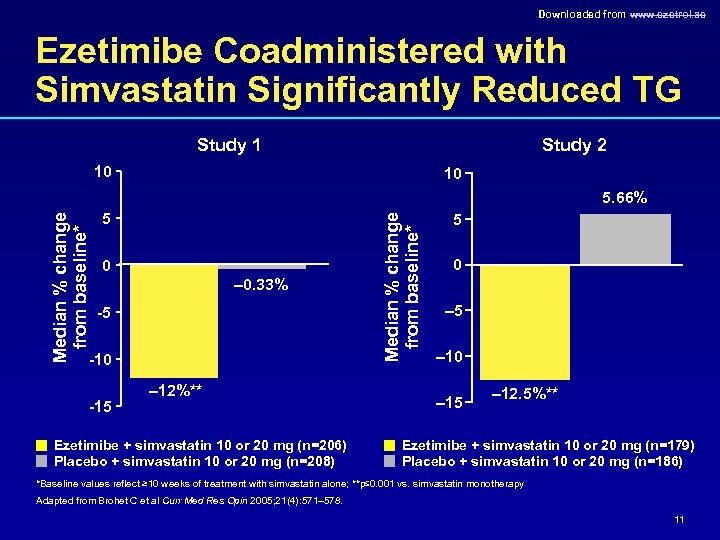
Downloaded from www. ezetrol. ae Ezetimibe Coadministered with Simvastatin Significantly Reduced TG Study 1 Study 2 10 10 5 0 – 0. 33% -5 -10 -15 – 12%** Ezetimibe + simvastatin 10 or 20 mg (n=206) Placebo + simvastatin 10 or 20 mg (n=208) Median % change from baseline* 5. 66% 5 0 – 5 – 10 – 15 – 12. 5%** Ezetimibe + simvastatin 10 or 20 mg (n=179) Placebo + simvastatin 10 or 20 mg (n=186) *Baseline values reflect ≥ 10 weeks of treatment with simvastatin alone; **p≤ 0. 001 vs. simvastatin monotherapy Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 11
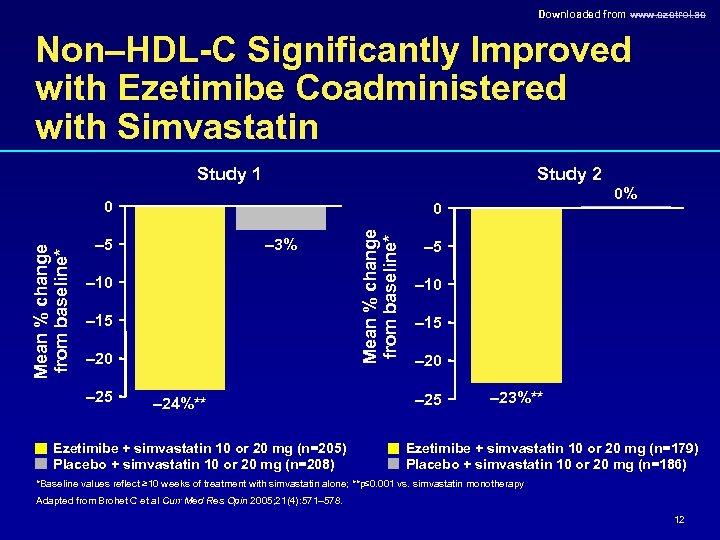
Downloaded from www. ezetrol. ae Non–HDL-C Significantly Improved with Ezetimibe Coadministered with Simvastatin Study 1 Study 2 0 – 3% – 5 – 10 – 15 – 20 – 25 – 24%** Ezetimibe + simvastatin 10 or 20 mg (n=205) Placebo + simvastatin 10 or 20 mg (n=208) Mean % change from baseline* 0 0% – 5 – 10 – 15 – 20 – 25 – 23%** Ezetimibe + simvastatin 10 or 20 mg (n=179) Placebo + simvastatin 10 or 20 mg (n=186) *Baseline values reflect ≥ 10 weeks of treatment with simvastatin alone; **p≤ 0. 001 vs. simvastatin monotherapy Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 12
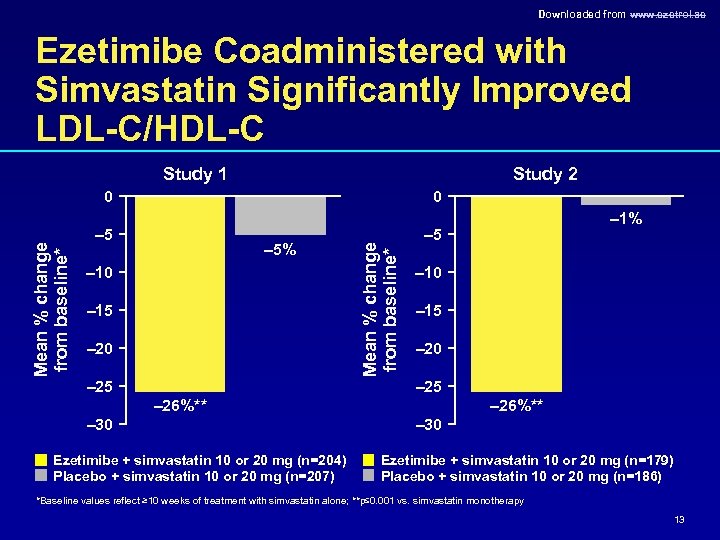
Downloaded from www. ezetrol. ae Ezetimibe Coadministered with Simvastatin Significantly Improved LDL-C/HDL-C Study 1 Study 2 0 – 5 – 5% – 10 – 15 – 20 – 25 Mean % change from baseline* 0 – 1% – 10 – 15 – 20 – 25 – 26%** – 30 Ezetimibe + simvastatin 10 or 20 mg (n=204) Placebo + simvastatin 10 or 20 mg (n=207) – 26%** – 30 Ezetimibe + simvastatin 10 or 20 mg (n=179) Placebo + simvastatin 10 or 20 mg (n=186) *Baseline values reflect ≥ 10 weeks of treatment with simvastatin alone; **p≤ 0. 001 vs. simvastatin monotherapy 13
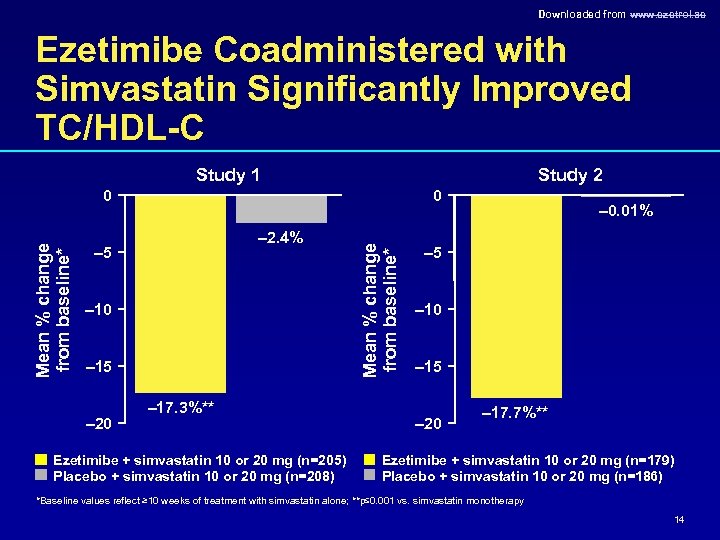
Downloaded from www. ezetrol. ae Ezetimibe Coadministered with Simvastatin Significantly Improved TC/HDL-C Study 1 Study 2 0 – 2. 4% – 5 – 10 – 15 – 20 – 17. 3%** Ezetimibe + simvastatin 10 or 20 mg (n=205) Placebo + simvastatin 10 or 20 mg (n=208) Mean % change from baseline* 0 – 0. 01% – 5 – 10 – 15 – 20 – 17. 7%** Ezetimibe + simvastatin 10 or 20 mg (n=179) Placebo + simvastatin 10 or 20 mg (n=186) *Baseline values reflect ≥ 10 weeks of treatment with simvastatin alone; **p≤ 0. 001 vs. simvastatin monotherapy 14
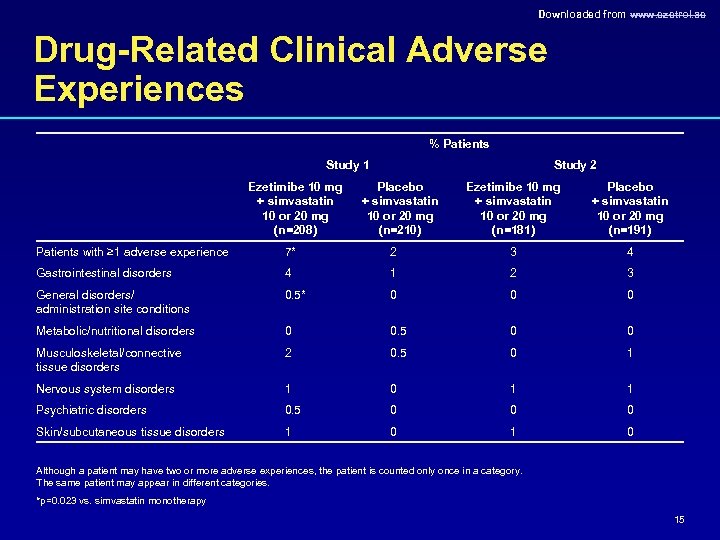
Downloaded from www. ezetrol. ae Drug-Related Clinical Adverse Experiences % Patients Study 1 Ezetimibe 10 mg + simvastatin 10 or 20 mg (n=208) Study 2 Placebo + simvastatin 10 or 20 mg (n=210) Ezetimibe 10 mg + simvastatin 10 or 20 mg (n=181) Placebo + simvastatin 10 or 20 mg (n=191) Patients with ≥ 1 adverse experience 7* 2 3 4 Gastrointestinal disorders 4 1 2 3 0. 5* 0 0 0 Metabolic/nutritional disorders 0 0. 5 0 0 Musculoskeletal/connective tissue disorders 2 0. 5 0 1 Nervous system disorders 1 0 1 1 Psychiatric disorders 0. 5 0 0 0 Skin/subcutaneous tissue disorders 1 0 General disorders/ administration site conditions Although a patient may have two or more adverse experiences, the patient is counted only once in a category. The same patient may appear in different categories. *p=0. 023 vs. simvastatin monotherapy 15
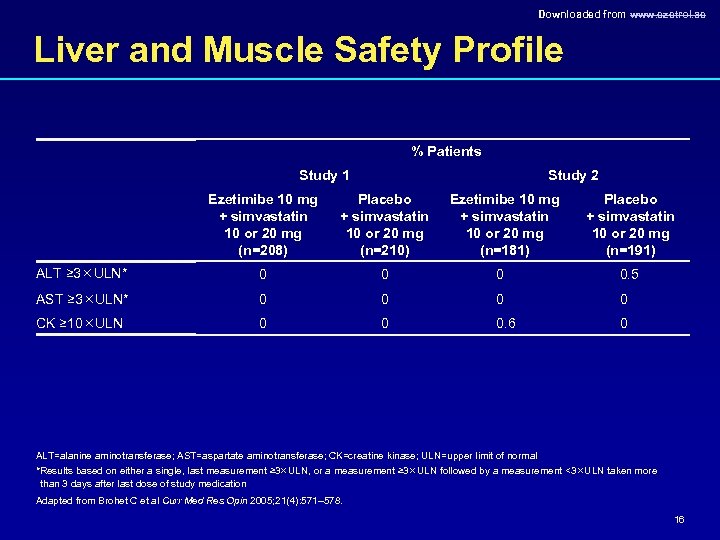
Downloaded from www. ezetrol. ae Liver and Muscle Safety Profile % Patients Study 1 Study 2 Ezetimibe 10 mg + simvastatin 10 or 20 mg (n=208) Placebo + simvastatin 10 or 20 mg (n=210) Ezetimibe 10 mg + simvastatin 10 or 20 mg (n=181) Placebo + simvastatin 10 or 20 mg (n=191) ALT ≥ 3 ULN* 0 0. 5 AST ≥ 3 ULN* 0 0 CK ≥ 10 ULN 0 0 0. 6 0 ALT=alanine aminotransferase; AST=aspartate aminotransferase; CK=creatine kinase; ULN=upper limit of normal *Results based on either a single, last measurement ≥ 3 ULN, or a measurement ≥ 3 ULN followed by a measurement <3 ULN taken more than 3 days after last dose of study medication Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 16

Downloaded from www. ezetrol. ae Summary and Conclusions of the Two Studies • By treating two sources of cholesterol, ezetimibe coadministered with a statin was superior to a statin alone in CHD patients with ü Significantly* greater reductions in LDL-C ü Significantly improved lipid profile—TC, TG, cholesterol ratios • Significantly* more patients achieved LDL-C goal (≤ 2. 60 mmol/L [≤ 100 mg/dl]) • Ezetimibe coadministration with a statin was well tolerated ü Similar to statin alone • Treating two sources of cholesterol (production and absorption) resulted in more patients achieving their LDL-C goals *p≤ 0. 001 Adapted from Brohet C et al Curr Med Res Opin 2005; 21(4): 571– 578. 17

Downloaded from www. ezetrol. ae References • Please see notes page. 18

Downloaded from www. ezetrol. ae Ezetimibe Coadministered with Simvastatin in Patients with Hypercholesterolemia and Coronary Heart Disease Before prescribing, please consult the manufacturers’ prescribing information. MSP does not recommend the use of any product in any different manner than as described in the prescribing information. Copyright © 2005 MSP Singapore Company, LLC. All rights reserved. 4 -08 EZT 2005 -W-166204 -SS Printed in USA 19
a54028ce6f0cf40691646b6772afdb43.ppt