4720890e6cac78d60587f7aaa891ddd7.ppt
- Количество слайдов: 48
 down staging of cervical cancer In India “Your cooperation is needed” Dr. Sharda Jain Director: Global Institute of Gynaecoloy at Pushpanjali Crosslay Hospital Secretary general: Delhi Gynaecologist Forum
down staging of cervical cancer In India “Your cooperation is needed” Dr. Sharda Jain Director: Global Institute of Gynaecoloy at Pushpanjali Crosslay Hospital Secretary general: Delhi Gynaecologist Forum
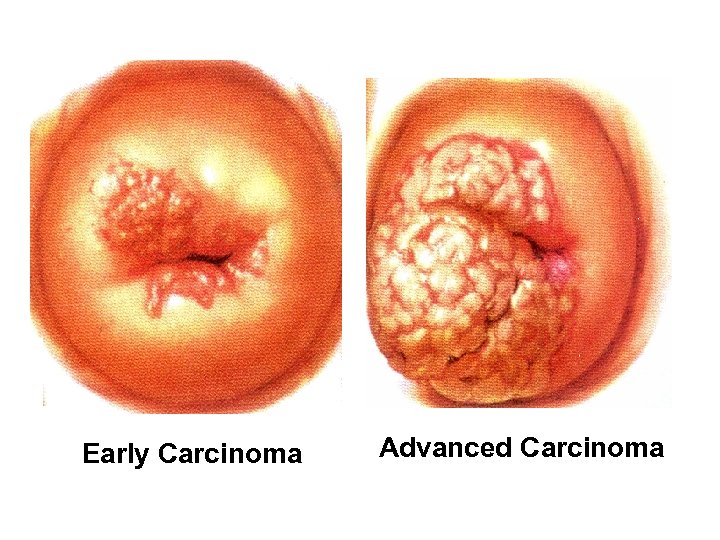 Early Carcinoma Advanced Carcinoma
Early Carcinoma Advanced Carcinoma
 Global Burden of Cervical Cancer • Worldwide, • 500, 000 women diagnosed per year 1 • 270, 000 deaths per year 1 • >1 million new cases of cervical cancer each year, 20502 One Death every 2 minutes
Global Burden of Cervical Cancer • Worldwide, • 500, 000 women diagnosed per year 1 • 270, 000 deaths per year 1 • >1 million new cases of cervical cancer each year, 20502 One Death every 2 minutes
 cancer cervix World New Deaths - No. - 2 - 5 Lack 2. 75 India New Deaths Number One – - 1. 32 lack o. 74 lack One death every 7 th minutes in India
cancer cervix World New Deaths - No. - 2 - 5 Lack 2. 75 India New Deaths Number One – - 1. 32 lack o. 74 lack One death every 7 th minutes in India
 cancer cervix life time risk India = 20 – 35 / lack (35 – 64 yrs) Developed countries = 1 -8/ lack It is expected by 2050 = double If no action is taken
cancer cervix life time risk India = 20 – 35 / lack (35 – 64 yrs) Developed countries = 1 -8/ lack It is expected by 2050 = double If no action is taken
 path (prog. of appro. tech in health) Most effective program of down staging of cervical Cancer is paps smear screening
path (prog. of appro. tech in health) Most effective program of down staging of cervical Cancer is paps smear screening
 mass paps smear screening {IARC - Int. agency of research on cancer } 35 – 64 yrs = 93% reduction if Screening 1 -3 years = 84 % Reduction = 64% reduction India 5 years 10 years No Govt. Effort for public Screening (non availability of Tech/ doctors to read paps smear )
mass paps smear screening {IARC - Int. agency of research on cancer } 35 – 64 yrs = 93% reduction if Screening 1 -3 years = 84 % Reduction = 64% reduction India 5 years 10 years No Govt. Effort for public Screening (non availability of Tech/ doctors to read paps smear )
 alternative methods for down staging of the cervical cancer 1. VISUAL INSPECTION OF CERVIX WITH ACETIC ACID. (VIA) 2. Use of MAGNASCOPE instead of colposcope 3. SINGLE VISIT APPROACH i. e. Treatment with cryosurgery for VIA +ve women 4. SELF COLLECTED SAMPLE for cytology or HPV – DNA testing
alternative methods for down staging of the cervical cancer 1. VISUAL INSPECTION OF CERVIX WITH ACETIC ACID. (VIA) 2. Use of MAGNASCOPE instead of colposcope 3. SINGLE VISIT APPROACH i. e. Treatment with cryosurgery for VIA +ve women 4. SELF COLLECTED SAMPLE for cytology or HPV – DNA testing
 alternative methods for down staging of the cervical cancer 5. Education and counseling 6. Increase coverage by camp approach 7. Low cost HPV test 8. HPV Vaccines.
alternative methods for down staging of the cervical cancer 5. Education and counseling 6. Increase coverage by camp approach 7. Low cost HPV test 8. HPV Vaccines.
 via AFTER APPLYING 3 % ACETIC ACID TO CERVIX “WHITE PATCHES” APPEARS DUE TO COAGULATION OF CELLULAR PROTEINS AND INDICATE THE ABNORMAL EPITHELIUM WHICH IS THICK AND DOES NOT ALLOW THE LIGHT REACTION TO PASS THROUGH. CERVICAL BIOPSIES - LSIL/HSIL
via AFTER APPLYING 3 % ACETIC ACID TO CERVIX “WHITE PATCHES” APPEARS DUE TO COAGULATION OF CELLULAR PROTEINS AND INDICATE THE ABNORMAL EPITHELIUM WHICH IS THICK AND DOES NOT ALLOW THE LIGHT REACTION TO PASS THROUGH. CERVICAL BIOPSIES - LSIL/HSIL
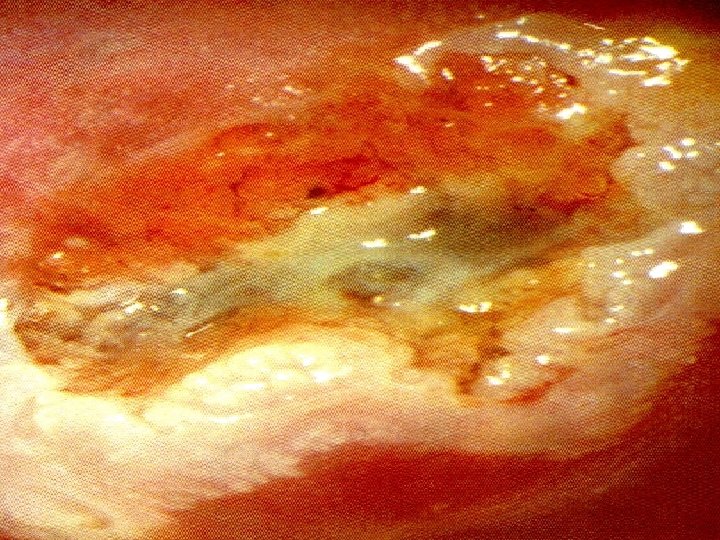
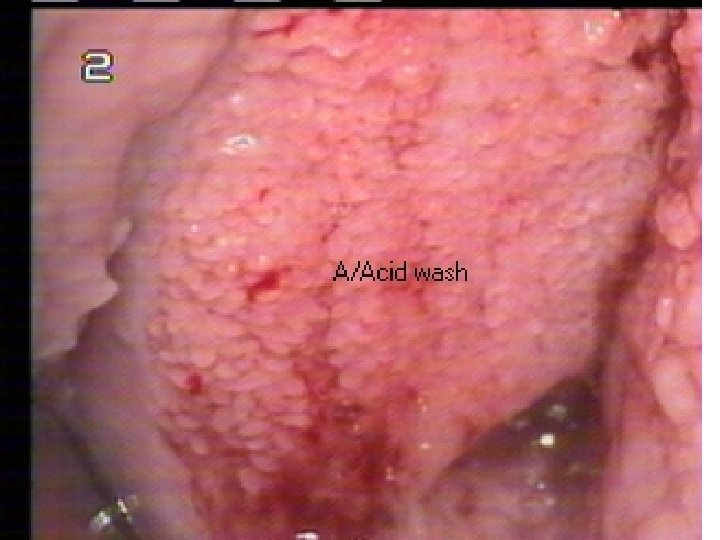
 Women were examined visually by simple speculum and colposcopically after application of 3 % acetic acid to cervix. Equal detection rates of cervical abnormalities by both techniques. Ottaviano and la torre 1982,
Women were examined visually by simple speculum and colposcopically after application of 3 % acetic acid to cervix. Equal detection rates of cervical abnormalities by both techniques. Ottaviano and la torre 1982,
 vili WHEN LUGOL’S IODINE IS APPLIED TO THE CERVIX, THE NORMAL CELLS CONTAINING GLYCOGEN STAIN DARK BROWN. THE ABNORMAL CELLS ARE RAPIDLY DIVIDING AND ARE DEFICIENT IN GLYCOGEN HENCE, REMAIN UNSTAINED WHICH ARE FURTHER EVALUATED BY COLPOSCOPY & BIOPSY.
vili WHEN LUGOL’S IODINE IS APPLIED TO THE CERVIX, THE NORMAL CELLS CONTAINING GLYCOGEN STAIN DARK BROWN. THE ABNORMAL CELLS ARE RAPIDLY DIVIDING AND ARE DEFICIENT IN GLYCOGEN HENCE, REMAIN UNSTAINED WHICH ARE FURTHER EVALUATED BY COLPOSCOPY & BIOPSY.
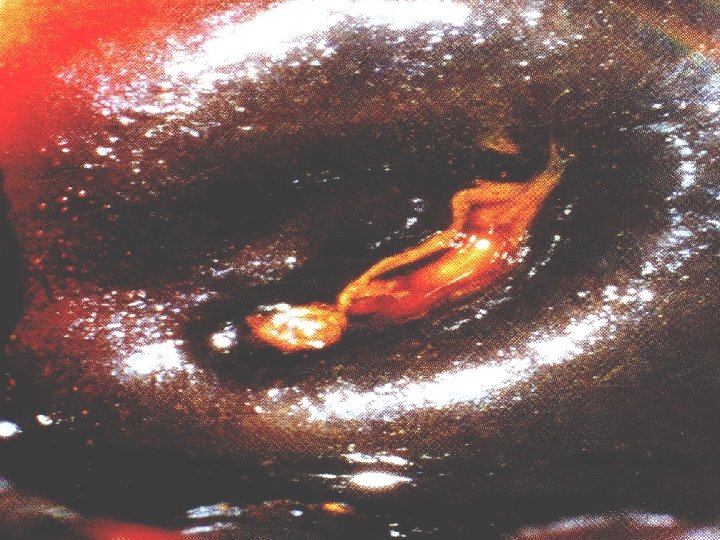
 iarc studies in india and africa • proved that VIA performed by trained paramedics has sensitivity of 64 to 90% and specificity of 73 to 91% which is comparable to conventional cytology. • specificity of VIA was increased by adding adjunctive test like VILI. • Advantages of visual technique are immediate results, making cost effective and has more than 99% negative predictive value. muwonge R (2007)
iarc studies in india and africa • proved that VIA performed by trained paramedics has sensitivity of 64 to 90% and specificity of 73 to 91% which is comparable to conventional cytology. • specificity of VIA was increased by adding adjunctive test like VILI. • Advantages of visual technique are immediate results, making cost effective and has more than 99% negative predictive value. muwonge R (2007)
 Sensitivity and specificity are often used to summarise the performance of a diagnostic test. Sensitivity is the probability of testing positive if the disease is truly present. Specificity is the probability of testing negative if the disease is truly absent.
Sensitivity and specificity are often used to summarise the performance of a diagnostic test. Sensitivity is the probability of testing positive if the disease is truly present. Specificity is the probability of testing negative if the disease is truly absent.
 who needs colposcopy ± biopsy Ten to fifteen percent VIA+ve women require referral for colposcopy & colposcopic guided biopsies.
who needs colposcopy ± biopsy Ten to fifteen percent VIA+ve women require referral for colposcopy & colposcopic guided biopsies.
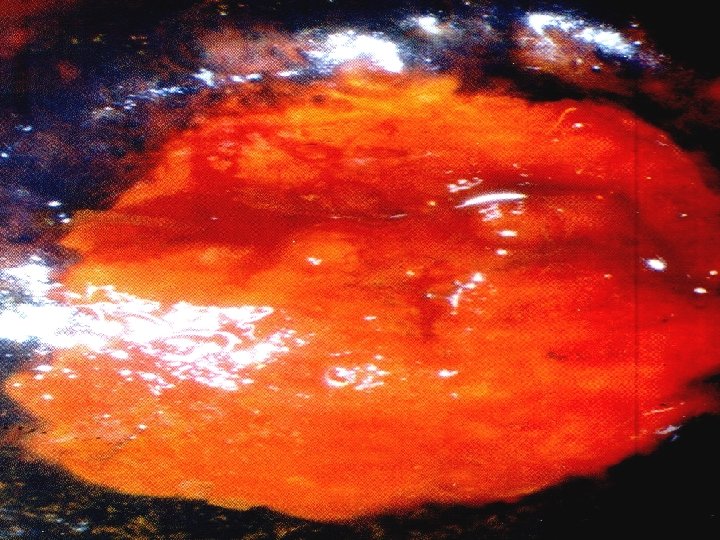
 lsil / lgsil Conservative treatment and follow up subsequently 6 monthly.
lsil / lgsil Conservative treatment and follow up subsequently 6 monthly.
 hsil / cancer Specialized Treatment
hsil / cancer Specialized Treatment
 what is single visit approach? In single visit approach cryo therapy is offered to all those women who are VIA +ve and cannot visit more than once for treatment.
what is single visit approach? In single visit approach cryo therapy is offered to all those women who are VIA +ve and cannot visit more than once for treatment.
 tamil nadu study Single Visit approach - follow up after 7 years showed • 25% reduction in cervical cancer incidence • 35% reduction in cervical cancer mortality • 27. 5% reduction in the incidence of stage II or advanced cancer compared to control group.
tamil nadu study Single Visit approach - follow up after 7 years showed • 25% reduction in cervical cancer incidence • 35% reduction in cervical cancer mortality • 27. 5% reduction in the incidence of stage II or advanced cancer compared to control group.
 what should be indian govt. approach Cervical cancer Should be taken seriously for ↓ number & reduction in mortality. • Camp approach • Single visit approach Both can help to down stage the disease in resource poor settings like India. Self sampling in rural areas/ slums Pap Smear/ HPV
what should be indian govt. approach Cervical cancer Should be taken seriously for ↓ number & reduction in mortality. • Camp approach • Single visit approach Both can help to down stage the disease in resource poor settings like India. Self sampling in rural areas/ slums Pap Smear/ HPV
 HPV Infection
HPV Infection
 100% of cervical cancers are caused by HPV
100% of cervical cancers are caused by HPV
 93. 5% of all cancer caused by HPV is Cervical Cancer
93. 5% of all cancer caused by HPV is Cervical Cancer
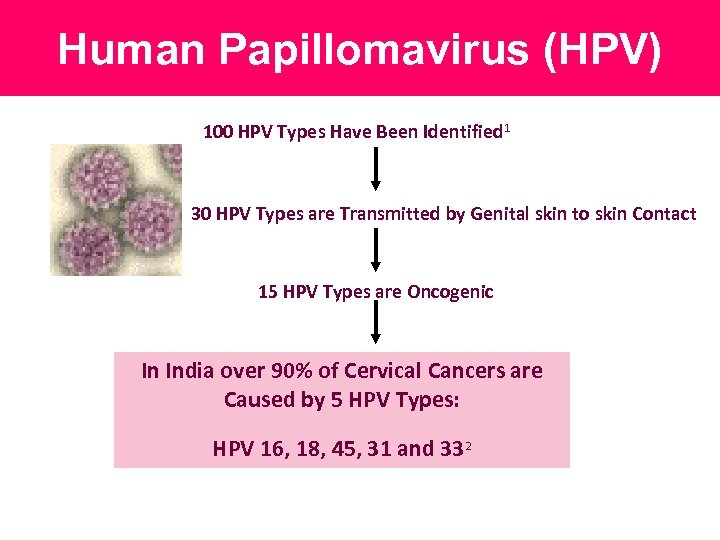 Human Papillomavirus (HPV) 100 HPV Types Have Been Identified 1 30 HPV Types are Transmitted by Genital skin to skin Contact 15 HPV Types are Oncogenic In India over 90% of Cervical Cancers are Caused by 5 HPV Types: HPV 16, 18, 45, 31 and 332
Human Papillomavirus (HPV) 100 HPV Types Have Been Identified 1 30 HPV Types are Transmitted by Genital skin to skin Contact 15 HPV Types are Oncogenic In India over 90% of Cervical Cancers are Caused by 5 HPV Types: HPV 16, 18, 45, 31 and 332
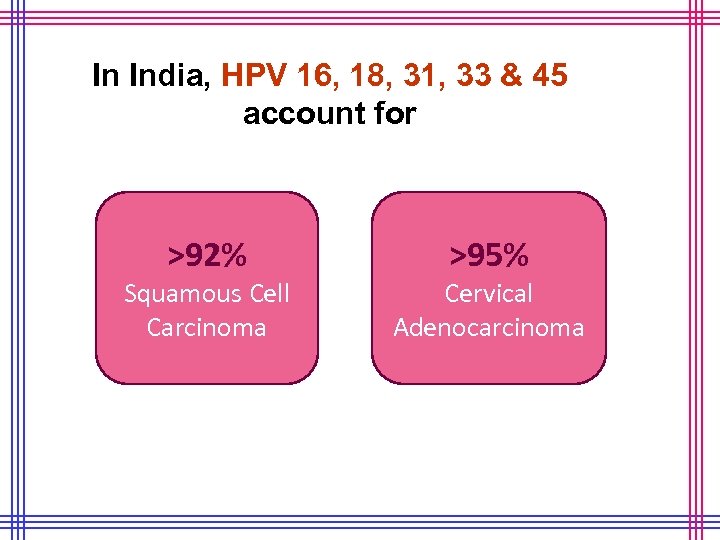 In India, HPV 16, 18, 31, 33 & 45 account for >92% Squamous Cell Carcinoma >95% Cervical Adenocarcinoma
In India, HPV 16, 18, 31, 33 & 45 account for >92% Squamous Cell Carcinoma >95% Cervical Adenocarcinoma
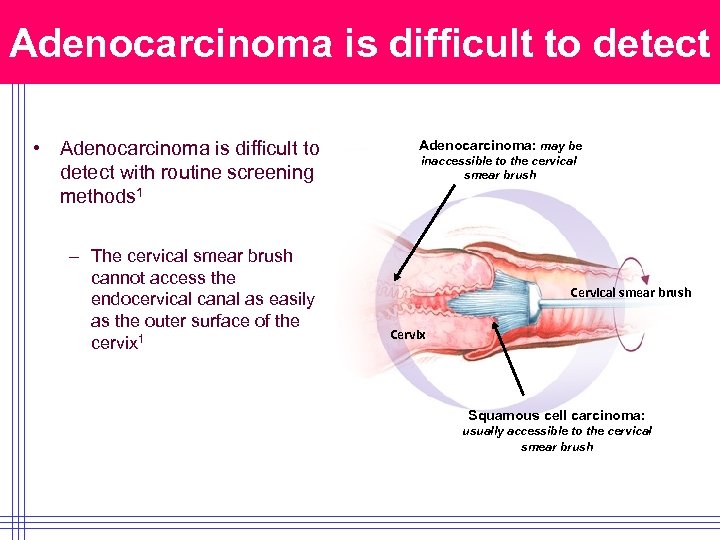 Adenocarcinoma is difficult to detect • Adenocarcinoma is difficult to detect with routine screening methods 1 – The cervical smear brush cannot access the endocervical canal as easily as the outer surface of the cervix 1 Adenocarcinoma: may be inaccessible to the cervical smear brush Cervix Squamous cell carcinoma: usually accessible to the cervical smear brush
Adenocarcinoma is difficult to detect • Adenocarcinoma is difficult to detect with routine screening methods 1 – The cervical smear brush cannot access the endocervical canal as easily as the outer surface of the cervix 1 Adenocarcinoma: may be inaccessible to the cervical smear brush Cervix Squamous cell carcinoma: usually accessible to the cervical smear brush
 Adenocarcinoma of the cervix. An Emerging concern • Incidence increasing (20– 25% of all cervical cancers), not prevented with traditional pap screening • More aggressive and occurs in younger women • > 90% of adenocarcinomas result from HPV 16, 18, 45, 33 and 311 • HPV 18 confers the highest risk
Adenocarcinoma of the cervix. An Emerging concern • Incidence increasing (20– 25% of all cervical cancers), not prevented with traditional pap screening • More aggressive and occurs in younger women • > 90% of adenocarcinomas result from HPV 16, 18, 45, 33 and 311 • HPV 18 confers the highest risk
 every woman is at risk of cervical cancer • HPV infections are very common • The risk starts from sexual debut 1 and continues throughout life 2
every woman is at risk of cervical cancer • HPV infections are very common • The risk starts from sexual debut 1 and continues throughout life 2
 • Up to 80% of women will acquire an HPV infection in their lifetime 5– 7 • HPV infections continue to occur in women over 25 years of age
• Up to 80% of women will acquire an HPV infection in their lifetime 5– 7 • HPV infections continue to occur in women over 25 years of age
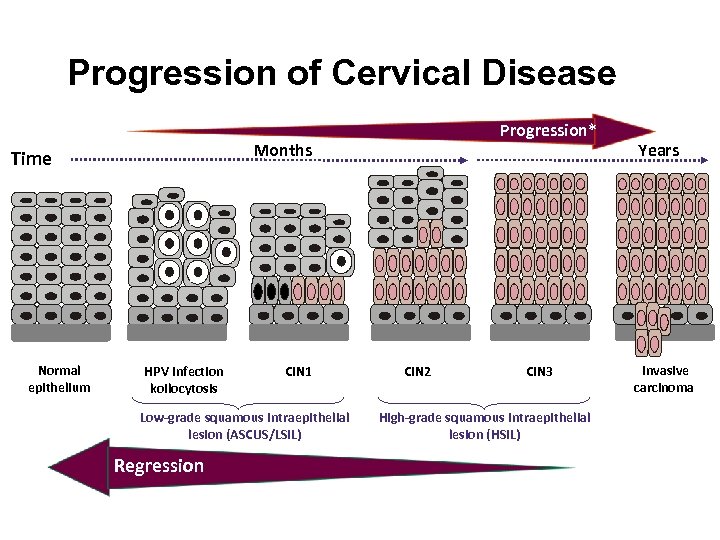 Progression of Cervical Disease Months Time Normal epithelium Progression* HPV infection koilocytosis CIN 1 Low-grade squamous intraepithelial lesion (ASCUS/LSIL) Regression CIN 2 CIN 3 High-grade squamous intraepithelial lesion (HSIL) Years Invasive carcinoma
Progression of Cervical Disease Months Time Normal epithelium Progression* HPV infection koilocytosis CIN 1 Low-grade squamous intraepithelial lesion (ASCUS/LSIL) Regression CIN 2 CIN 3 High-grade squamous intraepithelial lesion (HSIL) Years Invasive carcinoma
 The need for Vaccination against Cervical Cancer
The need for Vaccination against Cervical Cancer
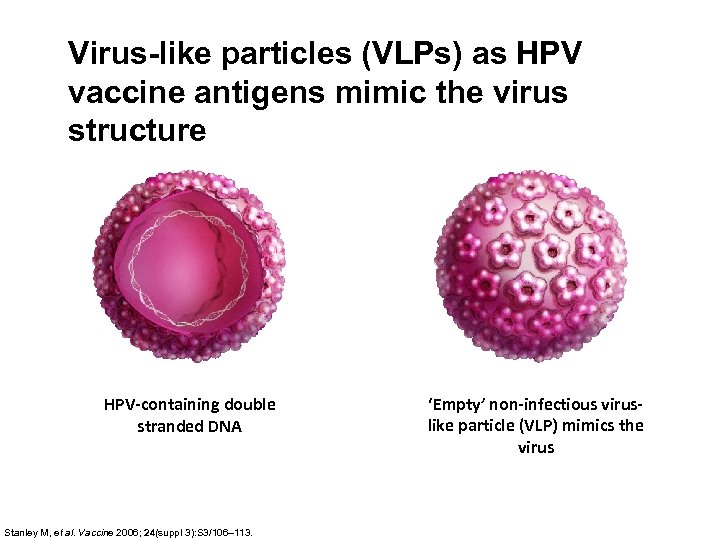 Virus-like particles (VLPs) as HPV vaccine antigens mimic the virus structure HPV-containing double stranded DNA Stanley M, et al. Vaccine 2006; 24(suppl 3): S 3/106– 113. ‘Empty’ non-infectious viruslike particle (VLP) mimics the virus
Virus-like particles (VLPs) as HPV vaccine antigens mimic the virus structure HPV-containing double stranded DNA Stanley M, et al. Vaccine 2006; 24(suppl 3): S 3/106– 113. ‘Empty’ non-infectious viruslike particle (VLP) mimics the virus
 why vaccination is needed ? ? • No protection through natural infection as HPV evades the immune system • Vaccines are highly immunogenic • Higher the serum antibodies, more is local neutralising antibody & longer the protection
why vaccination is needed ? ? • No protection through natural infection as HPV evades the immune system • Vaccines are highly immunogenic • Higher the serum antibodies, more is local neutralising antibody & longer the protection
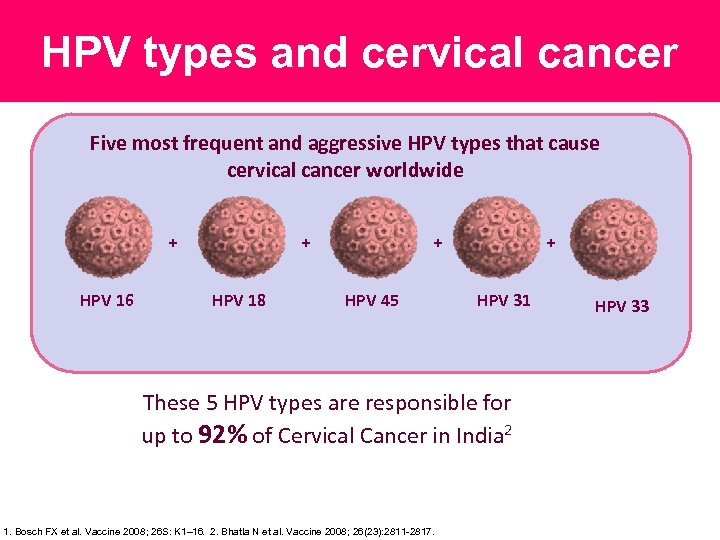 HPV types and cervical cancer Five most frequent and aggressive HPV types that cause cervical cancer worldwide + HPV 16 + HPV 18 + + HPV 45 HPV 31 These 5 HPV types are responsible for up to 92% of Cervical Cancer in India 2 1. Bosch FX et al. Vaccine 2008; 26 S: K 1– 16. 2. Bhatla N et al. Vaccine 2008; 26(23): 2811 -2817. HPV 33
HPV types and cervical cancer Five most frequent and aggressive HPV types that cause cervical cancer worldwide + HPV 16 + HPV 18 + + HPV 45 HPV 31 These 5 HPV types are responsible for up to 92% of Cervical Cancer in India 2 1. Bosch FX et al. Vaccine 2008; 26 S: K 1– 16. 2. Bhatla N et al. Vaccine 2008; 26(23): 2811 -2817. HPV 33
 2 vaccines • Gardasil • Cerverix
2 vaccines • Gardasil • Cerverix
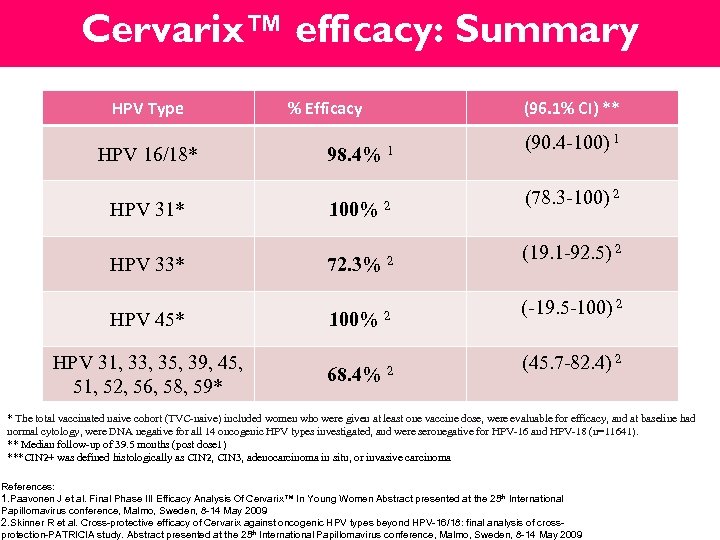 Cervarix™ efficacy: Summary HPV Type % Efficacy (96. 1% CI) ** (90. 4 -100) 1 HPV 16/18* 98. 4% 1 HPV 31* 100% 2 (78. 3 -100) 2 HPV 33* 72. 3% 2 (19. 1 -92. 5) 2 HPV 45* 100% 2 (-19. 5 -100) 2 HPV 31, 33, 35, 39, 45, 51, 52, 56, 58, 59* 68. 4% 2 (45. 7 -82. 4) 2 * The total vaccinated naive cohort (TVC-naive) included women who were given at least one vaccine dose, were evaluable for efficacy, and at baseline had normal cytology, were DNA negative for all 14 oncogenic HPV types investigated, and were seronegative for HPV-16 and HPV-18 (n=11641). ** Median follow-up of 39. 5 months (post dose 1) ***CIN 2+ was defined histologically as CIN 2, CIN 3, adenocarcinoma in situ, or invasive carcinoma References: 1. Paavonen J et al. Final Phase III Efficacy Analysis Of Cervarix™ In Young Women Abstract presented at the 25 th International Papillomavirus conference, Malmo, Sweden, 8 -14 May 2009 2. Skinner R et al. Cross-protective efficacy of Cervarix against oncogenic HPV types beyond HPV-16/18: final analysis of crossprotection-PATRICIA study. Abstract presented at the 25 th International Papillomavirus conference, Malmo, Sweden, 8 -14 May 2009
Cervarix™ efficacy: Summary HPV Type % Efficacy (96. 1% CI) ** (90. 4 -100) 1 HPV 16/18* 98. 4% 1 HPV 31* 100% 2 (78. 3 -100) 2 HPV 33* 72. 3% 2 (19. 1 -92. 5) 2 HPV 45* 100% 2 (-19. 5 -100) 2 HPV 31, 33, 35, 39, 45, 51, 52, 56, 58, 59* 68. 4% 2 (45. 7 -82. 4) 2 * The total vaccinated naive cohort (TVC-naive) included women who were given at least one vaccine dose, were evaluable for efficacy, and at baseline had normal cytology, were DNA negative for all 14 oncogenic HPV types investigated, and were seronegative for HPV-16 and HPV-18 (n=11641). ** Median follow-up of 39. 5 months (post dose 1) ***CIN 2+ was defined histologically as CIN 2, CIN 3, adenocarcinoma in situ, or invasive carcinoma References: 1. Paavonen J et al. Final Phase III Efficacy Analysis Of Cervarix™ In Young Women Abstract presented at the 25 th International Papillomavirus conference, Malmo, Sweden, 8 -14 May 2009 2. Skinner R et al. Cross-protective efficacy of Cervarix against oncogenic HPV types beyond HPV-16/18: final analysis of crossprotection-PATRICIA study. Abstract presented at the 25 th International Papillomavirus conference, Malmo, Sweden, 8 -14 May 2009
 hpv vaccines: safety and approval • WHO’s Global Advisory Committee on Vaccine Safety (GACVS) concluded that the HPV vaccines had good safety profiles 1 • U. S. Food and Drug Administration (FDA) approved both vaccines.
hpv vaccines: safety and approval • WHO’s Global Advisory Committee on Vaccine Safety (GACVS) concluded that the HPV vaccines had good safety profiles 1 • U. S. Food and Drug Administration (FDA) approved both vaccines.
 potential impact of hpv vaccination • HPV vaccination is the primary prevention strategy against cervical cancer • HPV vaccination is predicted to have a major impact on the burden of cervical cancer, especially in settings without optimal screening programs
potential impact of hpv vaccination • HPV vaccination is the primary prevention strategy against cervical cancer • HPV vaccination is predicted to have a major impact on the burden of cervical cancer, especially in settings without optimal screening programs
 fogsi recommendations • Cervical cancer causes significant morbidity/ mortality • HPV vaccine to be offered to all appropriate females who can afford the vaccine • Vaccine should be given prior to sexual debut www. fogsi. org/hpv vaccine
fogsi recommendations • Cervical cancer causes significant morbidity/ mortality • HPV vaccine to be offered to all appropriate females who can afford the vaccine • Vaccine should be given prior to sexual debut www. fogsi. org/hpv vaccine
 • When is the best time to vaccinate? • Upto what age can the Vaccine be given
• When is the best time to vaccinate? • Upto what age can the Vaccine be given
 fogsi recommendations – vaccine schedule • Age for initiation of vaccination is 10 - 12 years. – Catch up vaccination is permitted up to 45 yrs • Three doses at 0, 2 and 6 months with quadrivalent vaccine (Gardasil) • Three doses 0, 1 and 6 months with bivalent vaccine (Cerverix) Intra muscular – Deltoid reason
fogsi recommendations – vaccine schedule • Age for initiation of vaccination is 10 - 12 years. – Catch up vaccination is permitted up to 45 yrs • Three doses at 0, 2 and 6 months with quadrivalent vaccine (Gardasil) • Three doses 0, 1 and 6 months with bivalent vaccine (Cerverix) Intra muscular – Deltoid reason
 fogsi recommendations need for booster • At present there is no data to support use of boosters www. fogsi. org/hpv vaccine
fogsi recommendations need for booster • At present there is no data to support use of boosters www. fogsi. org/hpv vaccine
 fogsi recommendations: pregnancy & lactation • Not recommended for use in pregnancy • If patient becomes pregnant - Delay remaining doses till delivery • If vaccinated during pregnancy - No intervention (MTP) needed • Lactating women can receive the HPV vaccine and still continue breastfeeding as it is a vaccine without live viral DNA www. fogsi. org/hpv vaccine
fogsi recommendations: pregnancy & lactation • Not recommended for use in pregnancy • If patient becomes pregnant - Delay remaining doses till delivery • If vaccinated during pregnancy - No intervention (MTP) needed • Lactating women can receive the HPV vaccine and still continue breastfeeding as it is a vaccine without live viral DNA www. fogsi. org/hpv vaccine
 do we need to screen before vaccination? • No! • The results of screening will not influence to decision to vaccinate because: – Sexually active women continue to be at risk of new infections – Hence, vaccination will protect women from future infections regardless of an on going infection – NOTE: Vaccination will have NO effect on the ongoing infection or lesion.
do we need to screen before vaccination? • No! • The results of screening will not influence to decision to vaccinate because: – Sexually active women continue to be at risk of new infections – Hence, vaccination will protect women from future infections regardless of an on going infection – NOTE: Vaccination will have NO effect on the ongoing infection or lesion.


