424811e367ed8279cc52b889a646e835.ppt
- Количество слайдов: 15
 Dormancy of cells and organisms – strategies for survival and preservation Cyanobacteria Dormancy Forms in an Aquatic environment Ora Hadas, Assaf Sukenik, Ruth Kaplan-Levi Diti Viner-Mozzini, Merav Hadary Kinneret Limnological Laboratory Israel Oceanographic & Limnological Research
Dormancy of cells and organisms – strategies for survival and preservation Cyanobacteria Dormancy Forms in an Aquatic environment Ora Hadas, Assaf Sukenik, Ruth Kaplan-Levi Diti Viner-Mozzini, Merav Hadary Kinneret Limnological Laboratory Israel Oceanographic & Limnological Research
 Task: Determine conditions for the induction of akinetes Ø K deficiency triggers akinete formation in a yet unexplained process. Ø 50% deficiency in K triggers the formation of akinetes but only slightly affect growth Ø P deficiency and high light have additional effect on the formation of akinetes K>HL>P
Task: Determine conditions for the induction of akinetes Ø K deficiency triggers akinete formation in a yet unexplained process. Ø 50% deficiency in K triggers the formation of akinetes but only slightly affect growth Ø P deficiency and high light have additional effect on the formation of akinetes K>HL>P
 Task: Physiological processes involved in the induction of the dormant stage Ø Young akinetes maintain photosynthetic capacity at a similar manner as found for their adjacent vegetative cells in filaments grown in akineteinducing medium. Ø Mature akinetes maintain residual photosynthetic activity. Ø Some components of the photosynthetic apparatus appear to remain intact in akinetes. Ø In mature akinetes Photosystem I (PSI) and Photosystem II (PSII) complexes are kept apparently at a slightly higher molar ratio then in vegetative young cells (less PSII). Ø The phycobilisome pool is reduced in akinetes and disattached from the core antenna complexes. Sukenik A. , Beardall J. and Hadas O. (2006) Photosynthetic characterization of developing and mature akinetes of Aphanizomenon ovalisporum (Cyanoprokaryota). J. Phycol. (accepted)
Task: Physiological processes involved in the induction of the dormant stage Ø Young akinetes maintain photosynthetic capacity at a similar manner as found for their adjacent vegetative cells in filaments grown in akineteinducing medium. Ø Mature akinetes maintain residual photosynthetic activity. Ø Some components of the photosynthetic apparatus appear to remain intact in akinetes. Ø In mature akinetes Photosystem I (PSI) and Photosystem II (PSII) complexes are kept apparently at a slightly higher molar ratio then in vegetative young cells (less PSII). Ø The phycobilisome pool is reduced in akinetes and disattached from the core antenna complexes. Sukenik A. , Beardall J. and Hadas O. (2006) Photosynthetic characterization of developing and mature akinetes of Aphanizomenon ovalisporum (Cyanoprokaryota). J. Phycol. (accepted)
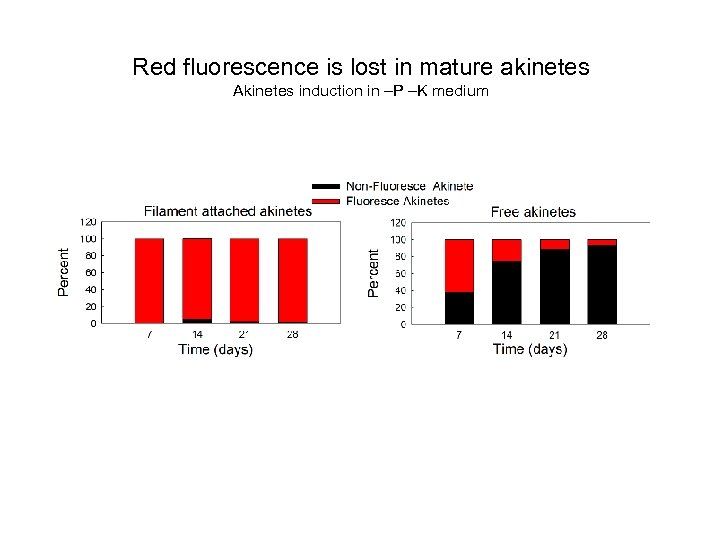 Red fluorescence is lost in mature akinetes Akinetes induction in –P –K medium
Red fluorescence is lost in mature akinetes Akinetes induction in –P –K medium
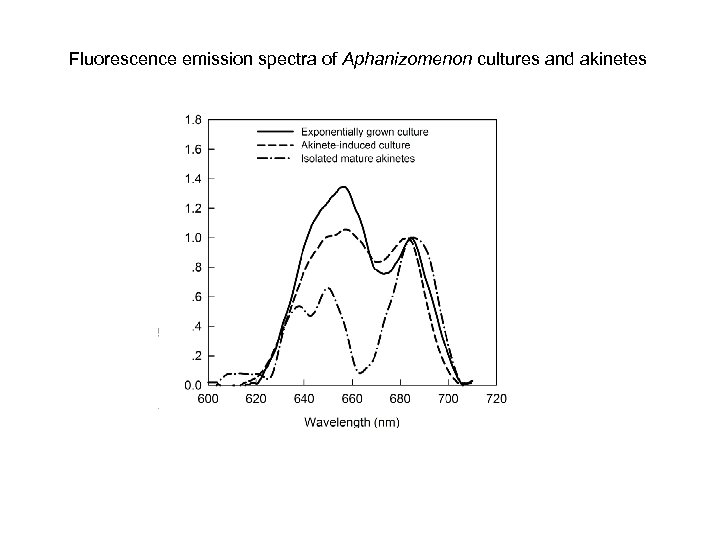 Fluorescence emission spectra of Aphanizomenon cultures and akinetes
Fluorescence emission spectra of Aphanizomenon cultures and akinetes
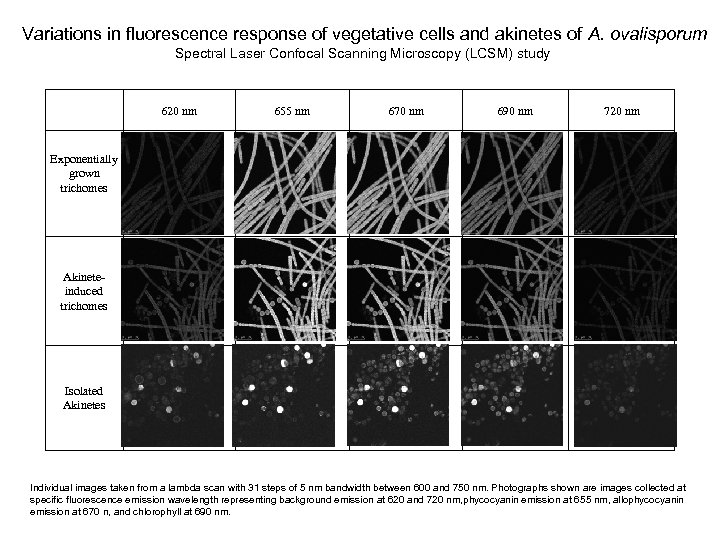 Variations in fluorescence response of vegetative cells and akinetes of A. ovalisporum Spectral Laser Confocal Scanning Microscopy (LCSM) study 620 nm 655 nm 670 nm 690 nm 720 nm Exponentially grown trichomes Akineteinduced trichomes Isolated Akinetes Individual images taken from a lambda scan with 31 steps of 5 nm bandwidth between 600 and 750 nm. Photographs shown are images collected at specific fluorescence emission wavelength representing background emission at 620 and 720 nm, phycocyanin emission at 655 nm, allophycocyanin emission at 670 n, and chlorophyll at 690 nm.
Variations in fluorescence response of vegetative cells and akinetes of A. ovalisporum Spectral Laser Confocal Scanning Microscopy (LCSM) study 620 nm 655 nm 670 nm 690 nm 720 nm Exponentially grown trichomes Akineteinduced trichomes Isolated Akinetes Individual images taken from a lambda scan with 31 steps of 5 nm bandwidth between 600 and 750 nm. Photographs shown are images collected at specific fluorescence emission wavelength representing background emission at 620 and 720 nm, phycocyanin emission at 655 nm, allophycocyanin emission at 670 n, and chlorophyll at 690 nm.
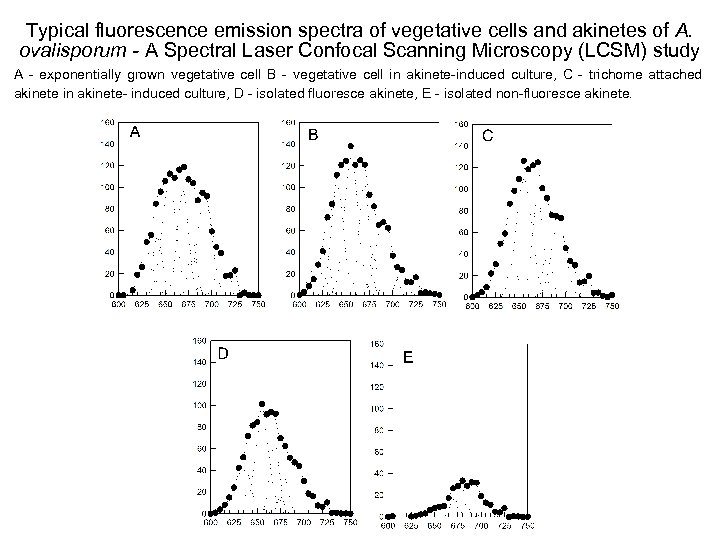 Typical fluorescence emission spectra of vegetative cells and akinetes of A. ovalisporum - A Spectral Laser Confocal Scanning Microscopy (LCSM) study A - exponentially grown vegetative cell B - vegetative cell in akinete-induced culture, C - trichome attached akinete in akinete- induced culture, D - isolated fluoresce akinete, E - isolated non-fluoresce akinete.
Typical fluorescence emission spectra of vegetative cells and akinetes of A. ovalisporum - A Spectral Laser Confocal Scanning Microscopy (LCSM) study A - exponentially grown vegetative cell B - vegetative cell in akinete-induced culture, C - trichome attached akinete in akinete- induced culture, D - isolated fluoresce akinete, E - isolated non-fluoresce akinete.
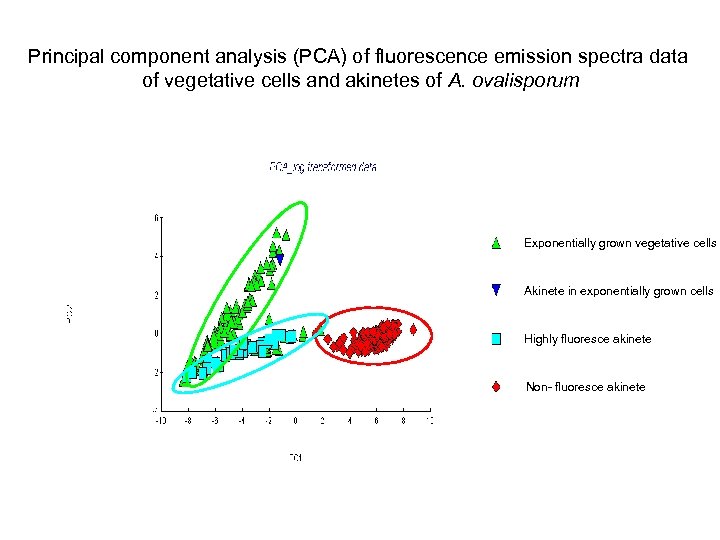 Principal component analysis (PCA) of fluorescence emission spectra data of vegetative cells and akinetes of A. ovalisporum Exponentially grown vegetative cells Akinete in exponentially grown cells Highly fluoresce akinete Non- fluoresce akinete
Principal component analysis (PCA) of fluorescence emission spectra data of vegetative cells and akinetes of A. ovalisporum Exponentially grown vegetative cells Akinete in exponentially grown cells Highly fluoresce akinete Non- fluoresce akinete
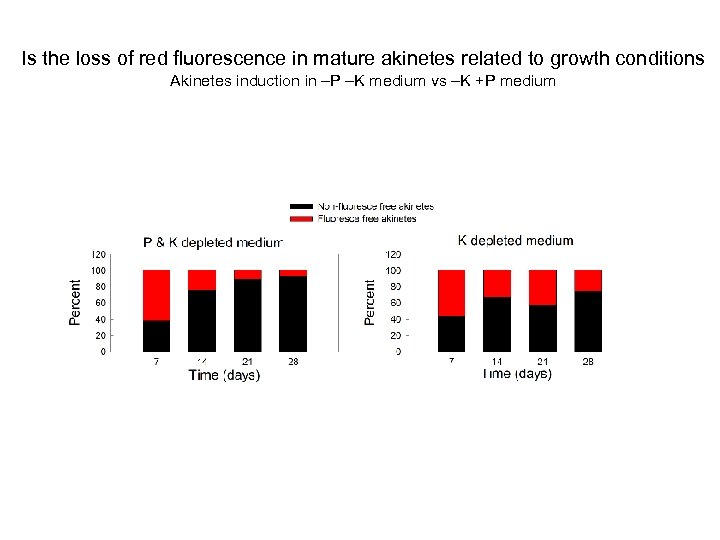 Is the loss of red fluorescence in mature akinetes related to growth conditions Akinetes induction in –P –K medium vs –K +P medium
Is the loss of red fluorescence in mature akinetes related to growth conditions Akinetes induction in –P –K medium vs –K +P medium
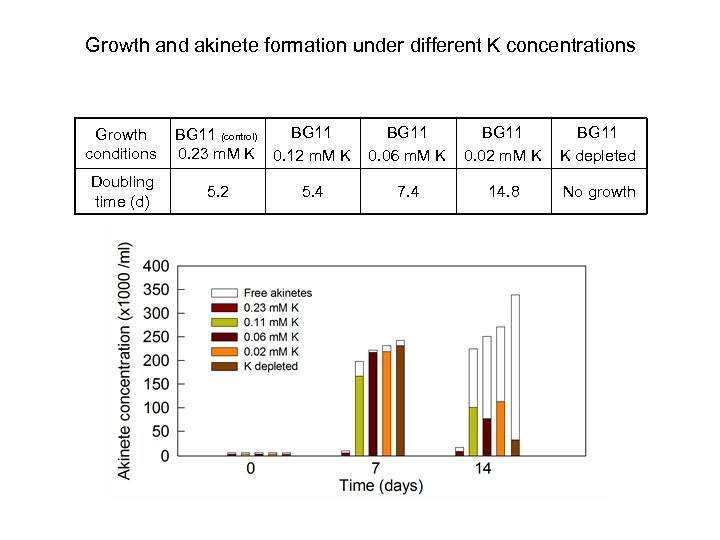 Growth and akinete formation under different K concentrations Growth conditions Doubling time (d) BG 11 (control) 0. 23 m. M K 0. 12 m. M K 5. 2 5. 4 BG 11 0. 06 m. M K BG 11 0. 02 m. M K BG 11 K depleted 7. 4 14. 8 No growth
Growth and akinete formation under different K concentrations Growth conditions Doubling time (d) BG 11 (control) 0. 23 m. M K 0. 12 m. M K 5. 2 5. 4 BG 11 0. 06 m. M K BG 11 0. 02 m. M K BG 11 K depleted 7. 4 14. 8 No growth
 Task: Determine environmental variables/stimuli responsible for germination of akinetes Ø Induction and isolation of akinetes Ø Germination experiments in multi-well plates Ø Experimental parameters: P concentration, p. H, temperature (10 – 30 C), light intensity and quality, L/D regime Preliminary results (Effect of P concentration on germination rate) P m. M 0 100 200 400 % germination (STD) 0 19 (6) 14 (3) 34 (10)
Task: Determine environmental variables/stimuli responsible for germination of akinetes Ø Induction and isolation of akinetes Ø Germination experiments in multi-well plates Ø Experimental parameters: P concentration, p. H, temperature (10 – 30 C), light intensity and quality, L/D regime Preliminary results (Effect of P concentration on germination rate) P m. M 0 100 200 400 % germination (STD) 0 19 (6) 14 (3) 34 (10)
 Task: Development of molecular tools to study the development and germination of akinetes - Progress, obstacles and plans Genomic Library construction Two cultures of Aphanizomenon ovalisporum were provided to Richard Reinhardt MPI Molecular Genetics Berlin-Dahlem, to create genomic libraries: 1. KLL strain grown in rich medium (BG 11) 2. HUJI strain grown in rich medium (BG 11) The genomic libraries were cloned into p. CC 1 Fos. The number of clones per culture: 1. 13, 056 fosmids 2. 15, 360 fosmids A total of 3481 sequencing reads were made from both library fosmids, using primers T 7 and M 13(-28)
Task: Development of molecular tools to study the development and germination of akinetes - Progress, obstacles and plans Genomic Library construction Two cultures of Aphanizomenon ovalisporum were provided to Richard Reinhardt MPI Molecular Genetics Berlin-Dahlem, to create genomic libraries: 1. KLL strain grown in rich medium (BG 11) 2. HUJI strain grown in rich medium (BG 11) The genomic libraries were cloned into p. CC 1 Fos. The number of clones per culture: 1. 13, 056 fosmids 2. 15, 360 fosmids A total of 3481 sequencing reads were made from both library fosmids, using primers T 7 and M 13(-28)
 Contig assembly of Aphanizomenon ovalisporum genome Anabaena variabillis ATCC 29413 Genome size ~ 6. 4 Mbp Aphanizomenon ovalisporum ~ 3500 sequencing fragments from the genomic libraries The sequencing results of the Aphanizomenon ovalisporum genomic libraries were submitted to the assemble program Seq. Man. TMII 5. 03, DNASTAR package. Anabaena variabillis ATCC 29413 genome was included in the contig assembly, as both Cyanobacteria are members of the Nostoceae family, it is expected to share some similarities in various gene loci. A. ovalisporum nucleotides database – http: //est. molgen. mpg. de/Sleeping Beauty/ A. variabillis ATCC 29413 genome - ftp: //ftp. ncbi. nih. gov/genomes/Bacteria/
Contig assembly of Aphanizomenon ovalisporum genome Anabaena variabillis ATCC 29413 Genome size ~ 6. 4 Mbp Aphanizomenon ovalisporum ~ 3500 sequencing fragments from the genomic libraries The sequencing results of the Aphanizomenon ovalisporum genomic libraries were submitted to the assemble program Seq. Man. TMII 5. 03, DNASTAR package. Anabaena variabillis ATCC 29413 genome was included in the contig assembly, as both Cyanobacteria are members of the Nostoceae family, it is expected to share some similarities in various gene loci. A. ovalisporum nucleotides database – http: //est. molgen. mpg. de/Sleeping Beauty/ A. variabillis ATCC 29413 genome - ftp: //ftp. ncbi. nih. gov/genomes/Bacteria/
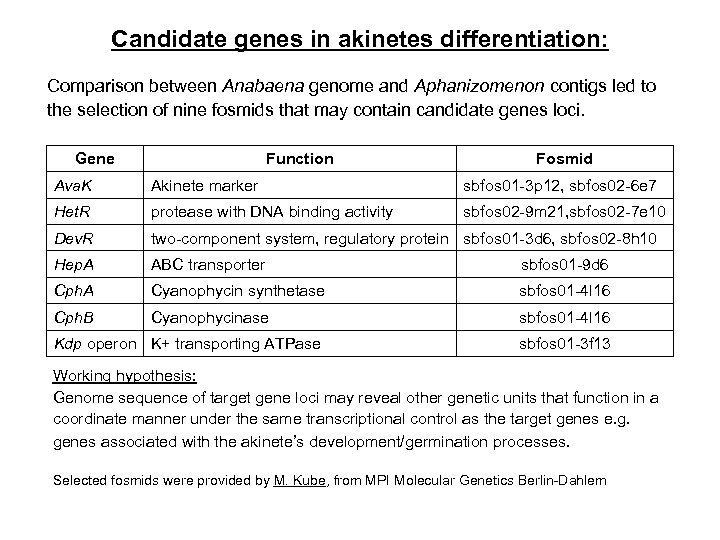 Candidate genes in akinetes differentiation: Comparison between Anabaena genome and Aphanizomenon contigs led to the selection of nine fosmids that may contain candidate genes loci. Gene Function Fosmid Ava. K Akinete marker sbfos 01 -3 p 12, sbfos 02 -6 e 7 Het. R protease with DNA binding activity sbfos 02 -9 m 21, sbfos 02 -7 e 10 Dev. R two-component system, regulatory protein sbfos 01 -3 d 6, sbfos 02 -8 h 10 Hep. A ABC transporter sbfos 01 -9 d 6 Cph. A Cyanophycin synthetase sbfos 01 -4 l 16 Cph. B Cyanophycinase sbfos 01 -4 l 16 Kdp operon K+ transporting ATPase sbfos 01 -3 f 13 Working hypothesis: Genome sequence of target gene loci may reveal other genetic units that function in a coordinate manner under the same transcriptional control as the target genes e. g. genes associated with the akinete’s development/germination processes. Selected fosmids were provided by M. Kube, from MPI Molecular Genetics Berlin-Dahlem
Candidate genes in akinetes differentiation: Comparison between Anabaena genome and Aphanizomenon contigs led to the selection of nine fosmids that may contain candidate genes loci. Gene Function Fosmid Ava. K Akinete marker sbfos 01 -3 p 12, sbfos 02 -6 e 7 Het. R protease with DNA binding activity sbfos 02 -9 m 21, sbfos 02 -7 e 10 Dev. R two-component system, regulatory protein sbfos 01 -3 d 6, sbfos 02 -8 h 10 Hep. A ABC transporter sbfos 01 -9 d 6 Cph. A Cyanophycin synthetase sbfos 01 -4 l 16 Cph. B Cyanophycinase sbfos 01 -4 l 16 Kdp operon K+ transporting ATPase sbfos 01 -3 f 13 Working hypothesis: Genome sequence of target gene loci may reveal other genetic units that function in a coordinate manner under the same transcriptional control as the target genes e. g. genes associated with the akinete’s development/germination processes. Selected fosmids were provided by M. Kube, from MPI Molecular Genetics Berlin-Dahlem
 Working Plan: l Following up several candidate genes: cloning, sequencing, expression , etc. l Total RNA extraction and m. RNA isolation from various developmental stages (Exponentially grown culture, akinete induced culture, mature isolated akinetes, germinating akinetes). In cooperation with MPI l Preparation of normalized combined c. DNA libraries (by MPI) to be further used for microarray slides. l ESTs of a c. DNA library l Microarray analysis to test genes expression from different developmental stages. l Candidate genes selected from the microarray experiments will be further analyzed by Real-Time PCR. l Genes sequences will be deposited in SB gene database and analyzed by bioinformatic tools (i. e. protein structure, comparison to genes expressed in other organisms, etc). l Establishment of a transformation system, in order to follow gene expression in vivo, by GFP (green fluorescence protein) fusions.
Working Plan: l Following up several candidate genes: cloning, sequencing, expression , etc. l Total RNA extraction and m. RNA isolation from various developmental stages (Exponentially grown culture, akinete induced culture, mature isolated akinetes, germinating akinetes). In cooperation with MPI l Preparation of normalized combined c. DNA libraries (by MPI) to be further used for microarray slides. l ESTs of a c. DNA library l Microarray analysis to test genes expression from different developmental stages. l Candidate genes selected from the microarray experiments will be further analyzed by Real-Time PCR. l Genes sequences will be deposited in SB gene database and analyzed by bioinformatic tools (i. e. protein structure, comparison to genes expressed in other organisms, etc). l Establishment of a transformation system, in order to follow gene expression in vivo, by GFP (green fluorescence protein) fusions.


