6235377b878404dd21f21313101beb20.ppt
- Количество слайдов: 141
 Disease-Specific Treatment of Relapse after Allogeneic Transplantation
Disease-Specific Treatment of Relapse after Allogeneic Transplantation
 Treatment of Relapse • Cellular Immunotherapy – – – • Withdrawal of immune suppression DLI ( + chemotherapy) Second allogeneic SCT Non-Cellular therapies – – – Supportive care Conventional chemotherapy or radiation Novel cytotoxic agents Biological agents Immunotherapy approaches
Treatment of Relapse • Cellular Immunotherapy – – – • Withdrawal of immune suppression DLI ( + chemotherapy) Second allogeneic SCT Non-Cellular therapies – – – Supportive care Conventional chemotherapy or radiation Novel cytotoxic agents Biological agents Immunotherapy approaches
 Treatment of Relapse • Cellular therapy – Withdraw IS – Second BMT with same or different donor – DLI – Activated DLI – Manipulated DLI • CD 8 depleted, CD 4 enriched, antigen-specific. – Dose titrated DLI – NK cells – Modified T cells (CARs) • Otherapies – Conventional chemotherapy or radiation – Antibodies • CD 20, CD 33, CD 30, CTLA -4, etc… • Bispecific antibodies – Biological therapy • Lenalidomide, thalidomide, bortezomib, azacytadine, decitabine, m. Tor inhibitors, others. • Interferon, IL-2, etc… • Vaccines – Others
Treatment of Relapse • Cellular therapy – Withdraw IS – Second BMT with same or different donor – DLI – Activated DLI – Manipulated DLI • CD 8 depleted, CD 4 enriched, antigen-specific. – Dose titrated DLI – NK cells – Modified T cells (CARs) • Otherapies – Conventional chemotherapy or radiation – Antibodies • CD 20, CD 33, CD 30, CTLA -4, etc… • Bispecific antibodies – Biological therapy • Lenalidomide, thalidomide, bortezomib, azacytadine, decitabine, m. Tor inhibitors, others. • Interferon, IL-2, etc… • Vaccines – Others
 Available data for treatment of relapse • CML-CP: DLI restores durable CR in 80% of pts with CP relapse. – Dose, schedule, toxicity well defined. – Role of TKI’s? • AML, ALL, NHL, CLL, MM, CML-AP/BC – DLI response rate, most effective approach, and long term outcomes? – Second SCT ? – Otherapies ? An embarrassing lack of data
Available data for treatment of relapse • CML-CP: DLI restores durable CR in 80% of pts with CP relapse. – Dose, schedule, toxicity well defined. – Role of TKI’s? • AML, ALL, NHL, CLL, MM, CML-AP/BC – DLI response rate, most effective approach, and long term outcomes? – Second SCT ? – Otherapies ? An embarrassing lack of data
 Excuses (good ones) • Patients are heterogeneous – – Age Impact of prior transplant • • – Myeloablative vs RIC GVHD prophylaxis (TCD, alemtuzumab, sirolimus, etc…) Different grafts • • • BM vs PBSC TCD, other manipulation Donor source and availability (sibling/family, URD, UCB, matched or mismatched) – – Clinical complications and co-morbidities after allogeneic SCT • – Different risks, timing to intervention, treatment choice, etc… May not tolerate therapies well Active GVHD? Acute or chronic? • Use of immune suppression
Excuses (good ones) • Patients are heterogeneous – – Age Impact of prior transplant • • – Myeloablative vs RIC GVHD prophylaxis (TCD, alemtuzumab, sirolimus, etc…) Different grafts • • • BM vs PBSC TCD, other manipulation Donor source and availability (sibling/family, URD, UCB, matched or mismatched) – – Clinical complications and co-morbidities after allogeneic SCT • – Different risks, timing to intervention, treatment choice, etc… May not tolerate therapies well Active GVHD? Acute or chronic? • Use of immune suppression
 Excuses (good ones) • Disease-related issues are heterogeneous – – – Timing of relapse: early vs late relapse may be very different Histology (particularly in NHL) Disease burden and pace of progression • – • • • In indolent diseases residual disease may not require intervention Influence of prior therapies and likely drug resistance Small numbers of patients studied with some diseases. No central database or sample repository to assess interventions. Reticence for clinical trials in this population (sponsors, cooperative group, FDA, investigators. – Limits access to new compounds
Excuses (good ones) • Disease-related issues are heterogeneous – – – Timing of relapse: early vs late relapse may be very different Histology (particularly in NHL) Disease burden and pace of progression • – • • • In indolent diseases residual disease may not require intervention Influence of prior therapies and likely drug resistance Small numbers of patients studied with some diseases. No central database or sample repository to assess interventions. Reticence for clinical trials in this population (sponsors, cooperative group, FDA, investigators. – Limits access to new compounds
 Excuses (good ones) • Lack of insurance coverage for clinical trials (and in some cases standard DLI or otherapy) Bias for: • – Treatment selection • – Patient selection • – Depends on patient, disease activity, donor availability, prior therapies… Age, co-morbidity, past and present transplant-related complications. Reporting of outcomes
Excuses (good ones) • Lack of insurance coverage for clinical trials (and in some cases standard DLI or otherapy) Bias for: • – Treatment selection • – Patient selection • – Depends on patient, disease activity, donor availability, prior therapies… Age, co-morbidity, past and present transplant-related complications. Reporting of outcomes
 Many issues are common to treatment of relapse of multiple diseases
Many issues are common to treatment of relapse of multiple diseases
 Cell Dose • Is there a dose: response relationship? – In CML, low dose DLI (1 x 10^7/kg) with dose escalation may preserve GVL with limited GVHD. • • • Is this effective for other indolent relapses (NHL, MM, CLL? ) Not useful for rapidly progressive acute leukemia Is there a dose: toxicity relationship? – – Minimal threshold for GVHD may vary by donor source (haplo vs well matched) Sibling vs unrelated donor • Often dosed differently but not supported by comparative trials
Cell Dose • Is there a dose: response relationship? – In CML, low dose DLI (1 x 10^7/kg) with dose escalation may preserve GVL with limited GVHD. • • • Is this effective for other indolent relapses (NHL, MM, CLL? ) Not useful for rapidly progressive acute leukemia Is there a dose: toxicity relationship? – – Minimal threshold for GVHD may vary by donor source (haplo vs well matched) Sibling vs unrelated donor • Often dosed differently but not supported by comparative trials
 Role of Chimerism • Does mixed or changing chimerism signify relapse? – – For CML, mixed chimerism predicts relapse. Role for intervention depends on timing, pace of change, and other factors Significance in other diseases less clear May depend on the cellular compartment evaluated (T, NK, myeloid) Role of intervention for chimerism and influence on outcome not well defined for most diseases. • “Treatment” of mixed chimerism with DLI may prevent relapse in some cases, but not others.
Role of Chimerism • Does mixed or changing chimerism signify relapse? – – For CML, mixed chimerism predicts relapse. Role for intervention depends on timing, pace of change, and other factors Significance in other diseases less clear May depend on the cellular compartment evaluated (T, NK, myeloid) Role of intervention for chimerism and influence on outcome not well defined for most diseases. • “Treatment” of mixed chimerism with DLI may prevent relapse in some cases, but not others.
 Role for Second SCT • • • Available data is limited and should be reassessed in the “modern” era. Conventional vs RIC conditioning? Influence of disease type and extent of relapse? Same vs alternate donor? Influence of timing of relapse? Manipulations to enhance GVT activity of second SCT?
Role for Second SCT • • • Available data is limited and should be reassessed in the “modern” era. Conventional vs RIC conditioning? Influence of disease type and extent of relapse? Same vs alternate donor? Influence of timing of relapse? Manipulations to enhance GVT activity of second SCT?
 Overcoming Limitations • • • NCI Sponsored Workshop on Relapse after Allogeneic SCT! The first step…… Define critical issues relating to relapse therapy. Multicenter and international collaborations to rapidly test and analyze new therapies for relapse.
Overcoming Limitations • • • NCI Sponsored Workshop on Relapse after Allogeneic SCT! The first step…… Define critical issues relating to relapse therapy. Multicenter and international collaborations to rapidly test and analyze new therapies for relapse.
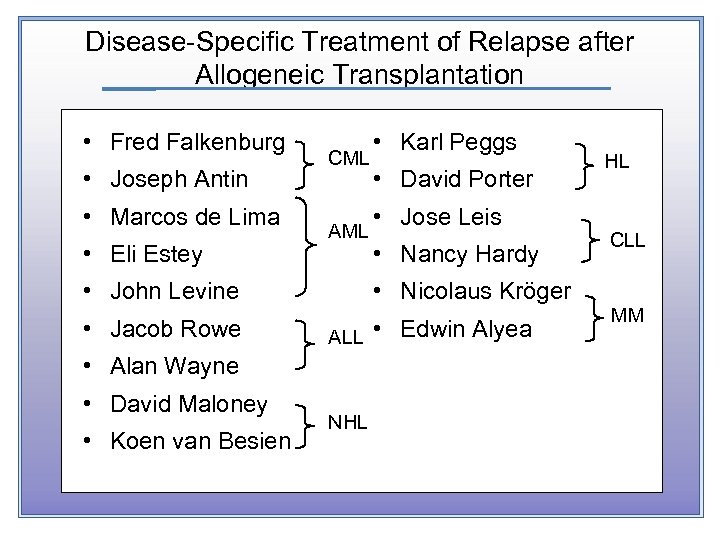 Disease-Specific Treatment of Relapse after Allogeneic Transplantation • Fred Falkenburg • Joseph Antin • Marcos de Lima • Eli Estey CML AML • John Levine • Jacob Rowe • Koen van Besien • David Porter • Jose Leis • Nancy Hardy • Nicolaus Kröger ALL • Alan Wayne • David Maloney • Karl Peggs NHL • Edwin Alyea HL CLL MM
Disease-Specific Treatment of Relapse after Allogeneic Transplantation • Fred Falkenburg • Joseph Antin • Marcos de Lima • Eli Estey CML AML • John Levine • Jacob Rowe • Koen van Besien • David Porter • Jose Leis • Nancy Hardy • Nicolaus Kröger ALL • Alan Wayne • David Maloney • Karl Peggs NHL • Edwin Alyea HL CLL MM

 Treatment of CML relapse after allogeneic SCT Fred Falkenburg Joseph Antin
Treatment of CML relapse after allogeneic SCT Fred Falkenburg Joseph Antin
 Treatment of relapsed Chronic Phase CML with DLI after Allogeneic Hematopoietic Stem Cell Transplantation (SCT) · Complete remissions in 80 -90 % of cases · Relatively low doses of DLI required · Time interval between treatment and response is dose and disease state dependent · Interferon may potentiate onset and effect · Target recognition may determine balance between GVHD and GVL
Treatment of relapsed Chronic Phase CML with DLI after Allogeneic Hematopoietic Stem Cell Transplantation (SCT) · Complete remissions in 80 -90 % of cases · Relatively low doses of DLI required · Time interval between treatment and response is dose and disease state dependent · Interferon may potentiate onset and effect · Target recognition may determine balance between GVHD and GVL
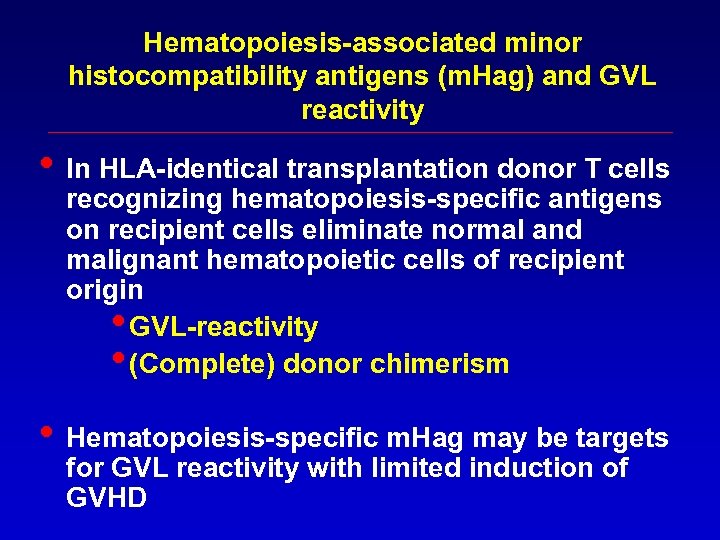 Hematopoiesis-associated minor histocompatibility antigens (m. Hag) and GVL reactivity • In HLA-identical transplantation donor T cells recognizing hematopoiesis-specific antigens on recipient cells eliminate normal and malignant hematopoietic cells of recipient origin • GVL-reactivity • (Complete) donor chimerism • Hematopoiesis-specific m. Hag may be targets for GVL reactivity with limited induction of GVHD
Hematopoiesis-associated minor histocompatibility antigens (m. Hag) and GVL reactivity • In HLA-identical transplantation donor T cells recognizing hematopoiesis-specific antigens on recipient cells eliminate normal and malignant hematopoietic cells of recipient origin • GVL-reactivity • (Complete) donor chimerism • Hematopoiesis-specific m. Hag may be targets for GVL reactivity with limited induction of GVHD
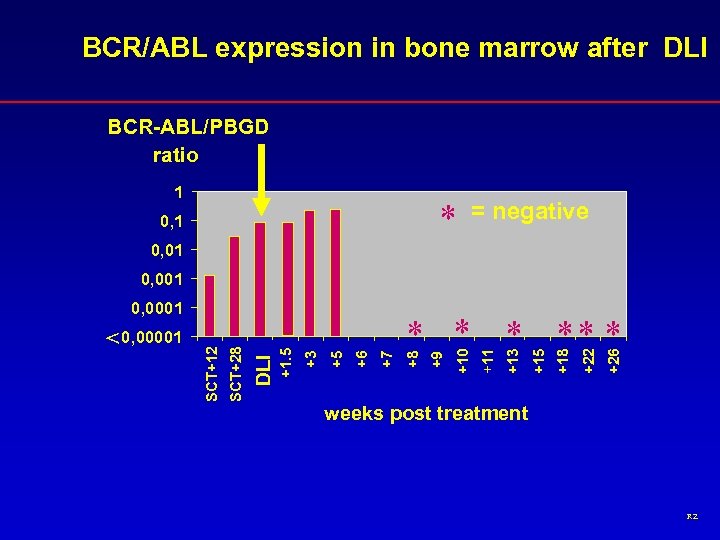 BCR/ABL expression in bone marrow after DLI BCR-ABL/PBGD ratio 1 * 0, 1 = negative 0, 01 0, 001 +26 +22 +18 +15 +13 +11 +10 +9 * ** * +8 +7 +6 +5 +3 +1. 5 DLI SCT+28 < 0, 00001 SCT+12 0, 0001 weeks post treatment RZ
BCR/ABL expression in bone marrow after DLI BCR-ABL/PBGD ratio 1 * 0, 1 = negative 0, 01 0, 001 +26 +22 +18 +15 +13 +11 +10 +9 * ** * +8 +7 +6 +5 +3 +1. 5 DLI SCT+28 < 0, 00001 SCT+12 0, 0001 weeks post treatment RZ
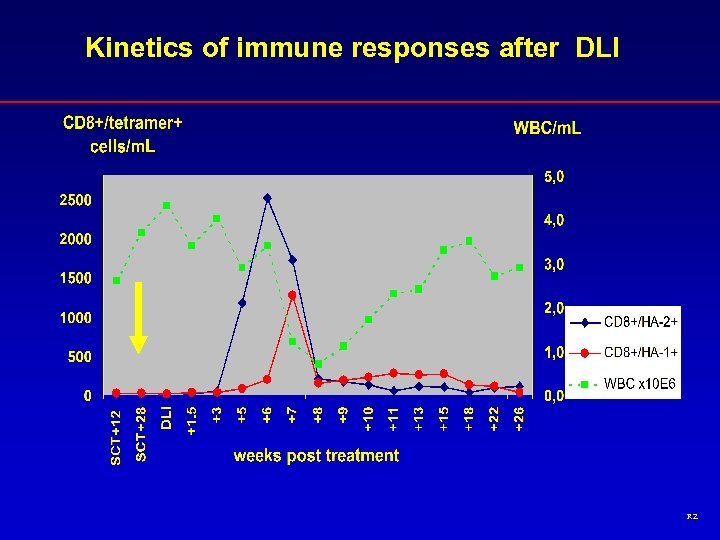 Kinetics of immune responses after DLI RZ
Kinetics of immune responses after DLI RZ
 Treatment of relapsed Chronic Phase CML with DLI after Allogeneic Hematopoietic Stem Cell Transplantation (SCT) · Which antigens need to be targeted to provoke a GVL response without GVHD · Targeting hematopoiesis restricted antigens results in specific GVL? · Are over-expressed self antigens targets of high avidity T cell responses?
Treatment of relapsed Chronic Phase CML with DLI after Allogeneic Hematopoietic Stem Cell Transplantation (SCT) · Which antigens need to be targeted to provoke a GVL response without GVHD · Targeting hematopoiesis restricted antigens results in specific GVL? · Are over-expressed self antigens targets of high avidity T cell responses?
 Persistance of BCR/ABL despite repeated DLI CML chronic phase, allogeneic SCT from HLA identical brother Hematological relapse: DLI 3 x 10 E 7 T cells/kg: hematological remission, molecular persistence Molecular persistence of disease despite escalating doses of DLI: Localized myeloid blast crise in epidural space: Systemic chemotherapy Radiotherapy + DLI 1. 5 x 10 E 8 T cells/kg,
Persistance of BCR/ABL despite repeated DLI CML chronic phase, allogeneic SCT from HLA identical brother Hematological relapse: DLI 3 x 10 E 7 T cells/kg: hematological remission, molecular persistence Molecular persistence of disease despite escalating doses of DLI: Localized myeloid blast crise in epidural space: Systemic chemotherapy Radiotherapy + DLI 1. 5 x 10 E 8 T cells/kg,
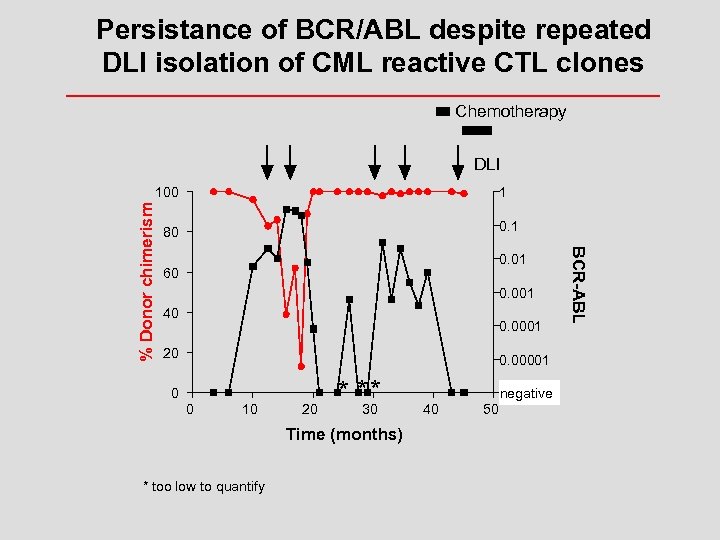 Persistance of BCR/ABL despite repeated DLI isolation of CML reactive CTL clones Chemotherapy DLI 1 0. 1 80 0. 01 60 0. 001 40 0. 0001 20 0 0. 00001 0 10 20 * * *30 Time (months) * too low to quantify 40 50 0. 000001 negative BCR-ABL % Donor chimerism 100
Persistance of BCR/ABL despite repeated DLI isolation of CML reactive CTL clones Chemotherapy DLI 1 0. 1 80 0. 01 60 0. 001 40 0. 0001 20 0 0. 00001 0 10 20 * * *30 Time (months) * too low to quantify 40 50 0. 000001 negative BCR-ABL % Donor chimerism 100
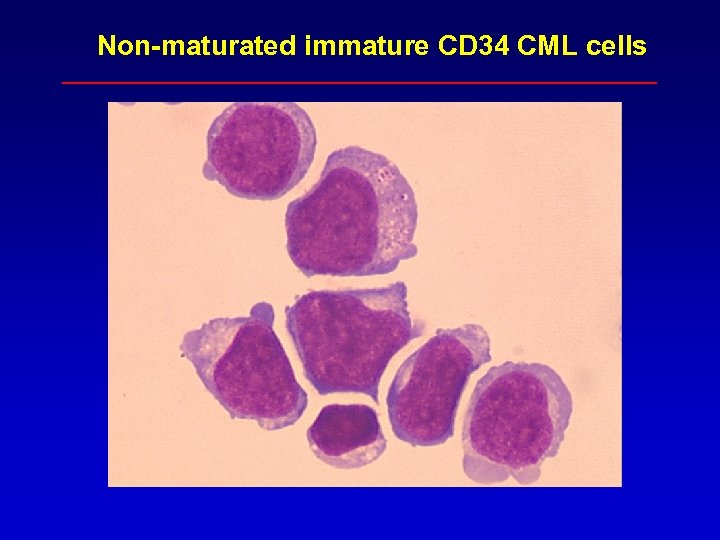 Non-maturated immature CD 34 CML cells
Non-maturated immature CD 34 CML cells
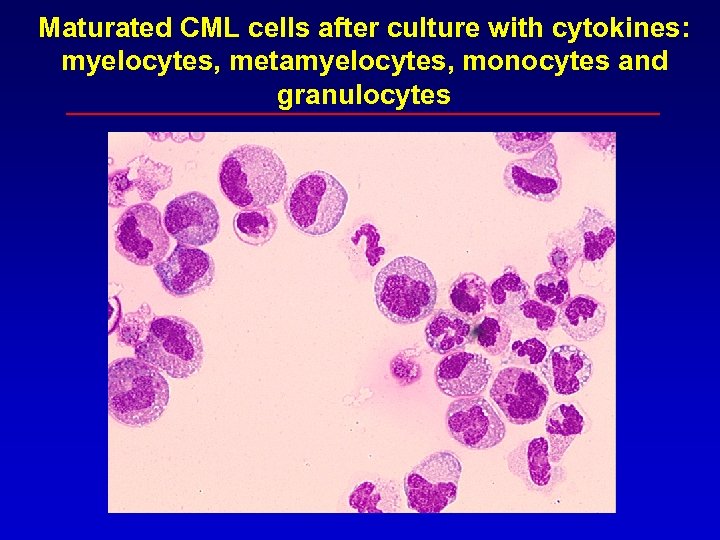 Maturated CML cells after culture with cytokines: myelocytes, metamyelocytes, monocytes and granulocytes
Maturated CML cells after culture with cytokines: myelocytes, metamyelocytes, monocytes and granulocytes
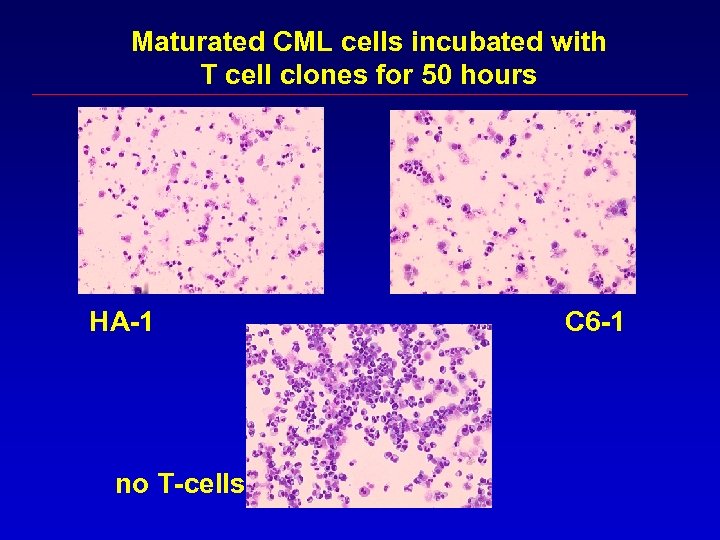 Maturated CML cells incubated with T cell clones for 50 hours HA-1 no T-cells C 6 -1
Maturated CML cells incubated with T cell clones for 50 hours HA-1 no T-cells C 6 -1
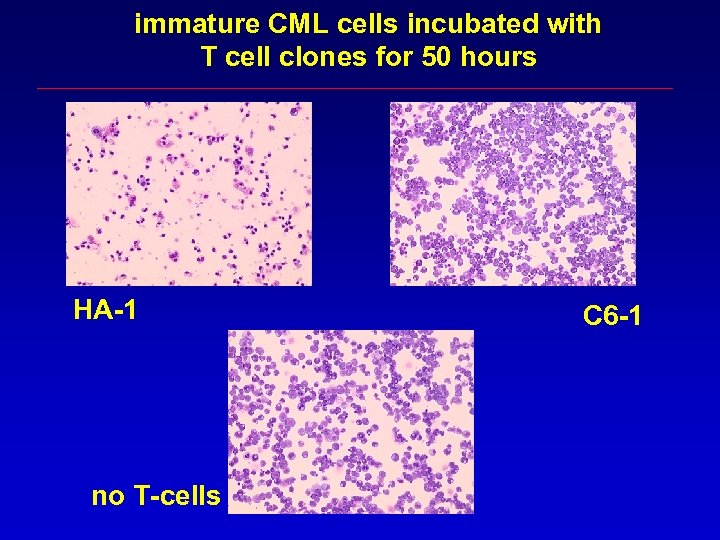 immature CML cells incubated with T cell clones for 50 hours HA-1 no T-cells C 6 -1
immature CML cells incubated with T cell clones for 50 hours HA-1 no T-cells C 6 -1
 Conclusions CD 8+ m. Hag specific CTL recognizing only maturation associated antigens not present on CD 34 positive stem or early progenitor cells may result in persistence of disease Should CML stem cells be targeted?
Conclusions CD 8+ m. Hag specific CTL recognizing only maturation associated antigens not present on CD 34 positive stem or early progenitor cells may result in persistence of disease Should CML stem cells be targeted?
 Cellular immunotherapy and TKI · Are leukemic stem cells residing after imatinib treatment susceptible targets for cellular immunotherapeutic interventions? · Should allo-SCT performed for persistent CML be combined with continuous treatment with tyrosine kinase inhibitors?
Cellular immunotherapy and TKI · Are leukemic stem cells residing after imatinib treatment susceptible targets for cellular immunotherapeutic interventions? · Should allo-SCT performed for persistent CML be combined with continuous treatment with tyrosine kinase inhibitors?
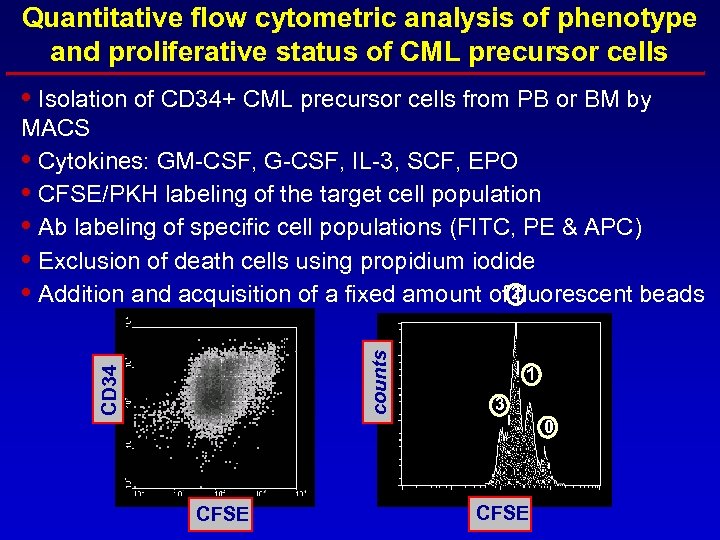 Quantitative flow cytometric analysis of phenotype and proliferative status of CML precursor cells • Isolation of CD 34+ CML precursor cells from PB or BM by CD 34 counts MACS • Cytokines: GM-CSF, G-CSF, IL-3, SCF, EPO • CFSE/PKH labeling of the target cell population • Ab labeling of specific cell populations (FITC, PE & APC) • Exclusion of death cells using propidium iodide 2 • Addition and acquisition of a fixed amount of fluorescent beads 1 3 0 CFSE
Quantitative flow cytometric analysis of phenotype and proliferative status of CML precursor cells • Isolation of CD 34+ CML precursor cells from PB or BM by CD 34 counts MACS • Cytokines: GM-CSF, G-CSF, IL-3, SCF, EPO • CFSE/PKH labeling of the target cell population • Ab labeling of specific cell populations (FITC, PE & APC) • Exclusion of death cells using propidium iodide 2 • Addition and acquisition of a fixed amount of fluorescent beads 1 3 0 CFSE
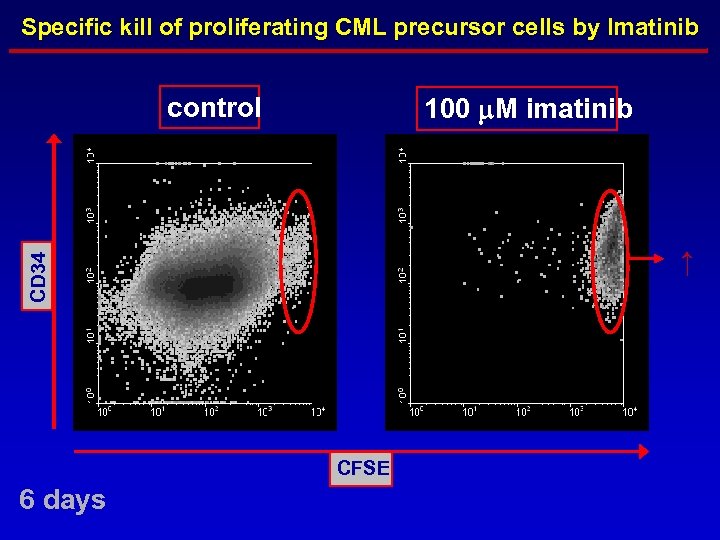 Specific kill of proliferating CML precursor cells by Imatinib control 100 m. M imatinib CD 34 ↑ CFSE 6 days
Specific kill of proliferating CML precursor cells by Imatinib control 100 m. M imatinib CD 34 ↑ CFSE 6 days
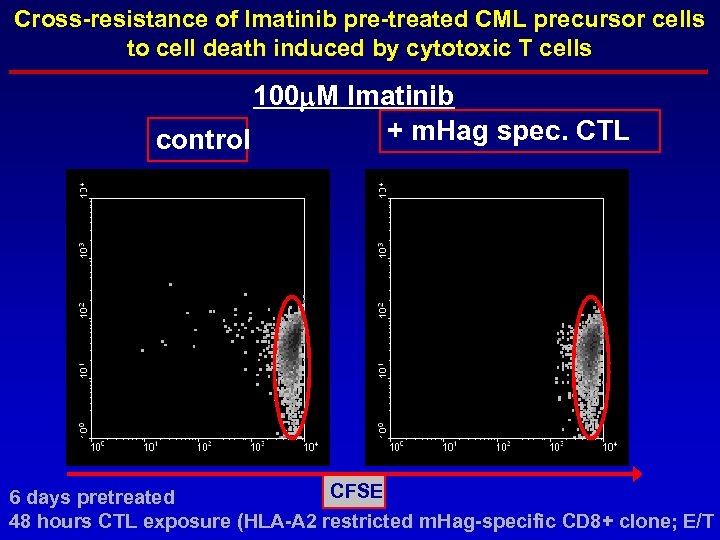 Cross-resistance of Imatinib pre-treated CML precursor cells to cell death induced by cytotoxic T cells 100 m. M Imatinib + m. Hag spec. CTL control CFSE 6 days pretreated 48 hours CTL exposure (HLA-A 2 restricted m. Hag-specific CD 8+ clone; E/T 3
Cross-resistance of Imatinib pre-treated CML precursor cells to cell death induced by cytotoxic T cells 100 m. M Imatinib + m. Hag spec. CTL control CFSE 6 days pretreated 48 hours CTL exposure (HLA-A 2 restricted m. Hag-specific CD 8+ clone; E/T 3
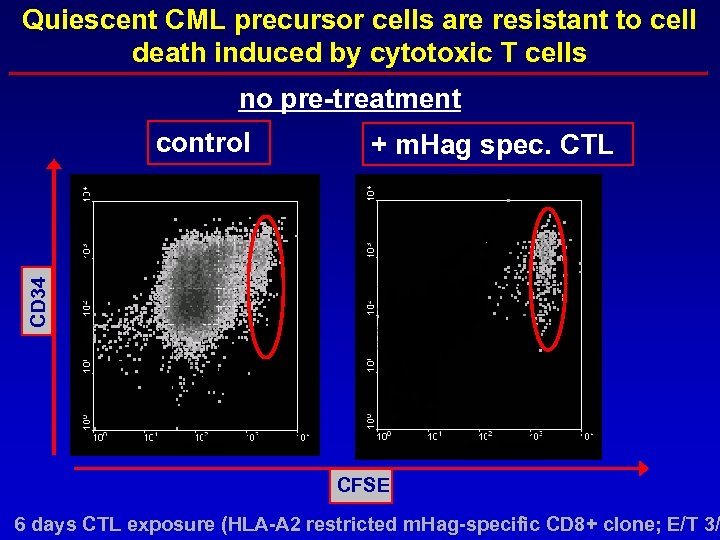 Quiescent CML precursor cells are resistant to cell death induced by cytotoxic T cells no pre-treatment + m. Hag spec. CTL CD 34 control CFSE 6 days CTL exposure (HLA-A 2 restricted m. Hag-specific CD 8+ clone; E/T 3/
Quiescent CML precursor cells are resistant to cell death induced by cytotoxic T cells no pre-treatment + m. Hag spec. CTL CD 34 control CFSE 6 days CTL exposure (HLA-A 2 restricted m. Hag-specific CD 8+ clone; E/T 3/
 Conclusions · Quiescent leukemic stem cells are protected from the cytotoxic effect of tyrosine kinase inhibitors · This population of quiescent leukemic stem cells shows cross-resistance to cytotoxic T cells involved in the GVL effect after allo-SCT, · The anti-proliferative effect of tyrosine kinase inhibitors on both the leukemic cells and the T cells may potentially hamper the potentially curative immune response after allo-SCT · If cellular immunotherapy is combined with TKI, should treatment be intermittent?
Conclusions · Quiescent leukemic stem cells are protected from the cytotoxic effect of tyrosine kinase inhibitors · This population of quiescent leukemic stem cells shows cross-resistance to cytotoxic T cells involved in the GVL effect after allo-SCT, · The anti-proliferative effect of tyrosine kinase inhibitors on both the leukemic cells and the T cells may potentially hamper the potentially curative immune response after allo-SCT · If cellular immunotherapy is combined with TKI, should treatment be intermittent?
 Treatment of relapsed CML after Allogeneic Hematopoietic Stem Cell Transplantation (SCT) · · · Identification of target cells and/or antigens to be targeted Separation of DLI into fractions (CD 4 T cells) Targeting minor histocompatiblity antigens or leukemia associated antigens by adoptive transfer if purified T cells · · · Vaccination of patient with m. Hag, APC. Vaccination of donor with m. Hag Interferon may potentiate onset and effect Are TKI useful, or harmful? How to treat extramedullary relapses
Treatment of relapsed CML after Allogeneic Hematopoietic Stem Cell Transplantation (SCT) · · · Identification of target cells and/or antigens to be targeted Separation of DLI into fractions (CD 4 T cells) Targeting minor histocompatiblity antigens or leukemia associated antigens by adoptive transfer if purified T cells · · · Vaccination of patient with m. Hag, APC. Vaccination of donor with m. Hag Interferon may potentiate onset and effect Are TKI useful, or harmful? How to treat extramedullary relapses

 Treatment of relapsed acute myelogenous leukemia after allogeneic stem cell transplantation Marcos de Lima, MD M. D Anderson Cancer Center John Levine, MD University of Michigan Elihu Estey, MD Fred Hutchinson Cancer Research Center
Treatment of relapsed acute myelogenous leukemia after allogeneic stem cell transplantation Marcos de Lima, MD M. D Anderson Cancer Center John Levine, MD University of Michigan Elihu Estey, MD Fred Hutchinson Cancer Research Center
 AML Probability of relapse: 20% - 60% - definition of relapse is key. All results reflect (to a great extent) patient selection. Major co-variates predicting relapse: - disease stage / cytogenetics - preparative regimen intensity Ringdén et al. J Clin Oncol; 27; 2009: 4570 -4577 Trends affecting comparison with historic data: - better prognostication for diploid patients (FLT 3, NPM) - treatment of older patients - use of reduced-intensity regimens Oran et al. Leukemia (2007) 21, 2540– 2544. Eapen et al. BMT 2004; 34: 721 -727
AML Probability of relapse: 20% - 60% - definition of relapse is key. All results reflect (to a great extent) patient selection. Major co-variates predicting relapse: - disease stage / cytogenetics - preparative regimen intensity Ringdén et al. J Clin Oncol; 27; 2009: 4570 -4577 Trends affecting comparison with historic data: - better prognostication for diploid patients (FLT 3, NPM) - treatment of older patients - use of reduced-intensity regimens Oran et al. Leukemia (2007) 21, 2540– 2544. Eapen et al. BMT 2004; 34: 721 -727
 Donor Lymphocyte Infusions Addition of chemotherapy improves response rate but not longterm disease control. GVHD in 10 -60% of patients Marrow aplasia in 5 -20%. TRM: 0 -50% Most series : adults, using mostly related donors. Responses frequently do not translate into long-term survival, due to GVHD, pancytopenia, infections, and disease relapse. Donor availability and presence of GVHD are major impediments.
Donor Lymphocyte Infusions Addition of chemotherapy improves response rate but not longterm disease control. GVHD in 10 -60% of patients Marrow aplasia in 5 -20%. TRM: 0 -50% Most series : adults, using mostly related donors. Responses frequently do not translate into long-term survival, due to GVHD, pancytopenia, infections, and disease relapse. Donor availability and presence of GVHD are major impediments.
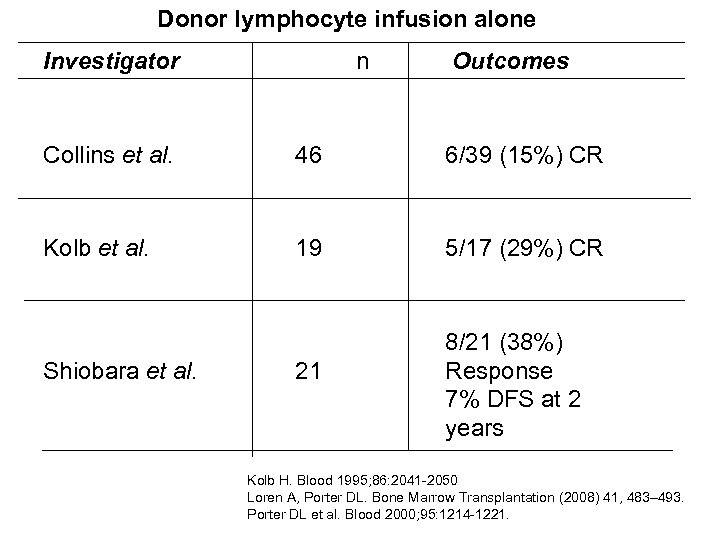 Donor lymphocyte infusion alone Investigator n Outcomes Collins et al. 46 6/39 (15%) CR Kolb et al. 19 5/17 (29%) CR Shiobara et al. 21 8/21 (38%) Response 7% DFS at 2 years Kolb H. Blood 1995; 86: 2041 -2050 Loren A, Porter DL. Bone Marrow Transplantation (2008) 41, 483– 493. Porter DL et al. Blood 2000; 95: 1214 -1221.
Donor lymphocyte infusion alone Investigator n Outcomes Collins et al. 46 6/39 (15%) CR Kolb et al. 19 5/17 (29%) CR Shiobara et al. 21 8/21 (38%) Response 7% DFS at 2 years Kolb H. Blood 1995; 86: 2041 -2050 Loren A, Porter DL. Bone Marrow Transplantation (2008) 41, 483– 493. Porter DL et al. Blood 2000; 95: 1214 -1221.
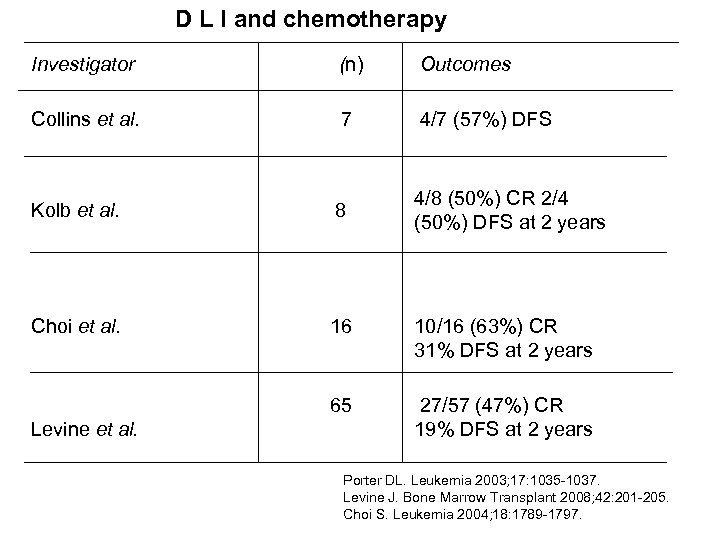 D L I and chemotherapy Investigator Collins et al. (n) 7 4/7 (57%) DFS Kolb et al. Outcomes 8 4/8 (50%) CR 2/4 (50%) DFS at 2 years Choi et al. 16 65 Levine et al. 10/16 (63%) CR 31% DFS at 2 years 27/57 (47%) CR 19% DFS at 2 years Porter DL. Leukemia 2003; 17: 1035 -1037. Levine J. Bone Marrow Transplant 2008; 42: 201 -205. Choi S. Leukemia 2004; 18: 1789 -1797.
D L I and chemotherapy Investigator Collins et al. (n) 7 4/7 (57%) DFS Kolb et al. Outcomes 8 4/8 (50%) CR 2/4 (50%) DFS at 2 years Choi et al. 16 65 Levine et al. 10/16 (63%) CR 31% DFS at 2 years 27/57 (47%) CR 19% DFS at 2 years Porter DL. Leukemia 2003; 17: 1035 -1037. Levine J. Bone Marrow Transplant 2008; 42: 201 -205. Choi S. Leukemia 2004; 18: 1789 -1797.
 Donor lymphocyte infusion EBMT analysis 399 patients with AML in first hematological relapse after HSCT DLI = n=171 versus no DLI (n = 228) Median follow-up was 27 and 40 months Schmid, C. et al. J Clin Oncol; 25: 4938 -4945 2007
Donor lymphocyte infusion EBMT analysis 399 patients with AML in first hematological relapse after HSCT DLI = n=171 versus no DLI (n = 228) Median follow-up was 27 and 40 months Schmid, C. et al. J Clin Oncol; 25: 4938 -4945 2007
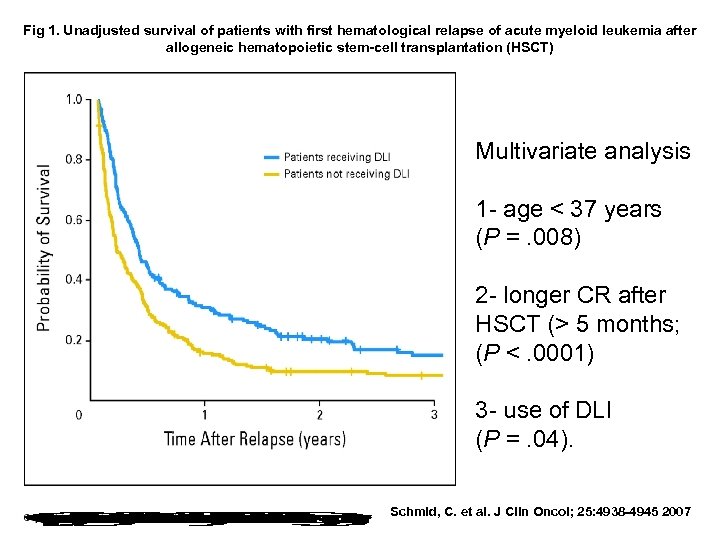 Fig 1. Unadjusted survival of patients with first hematological relapse of acute myeloid leukemia after allogeneic hematopoietic stem-cell transplantation (HSCT) Multivariate analysis 1 - age < 37 years (P =. 008) 2 - longer CR after HSCT (> 5 months; (P <. 0001) 3 - use of DLI (P =. 04). Schmid, C. et al. J Clin Oncol; 25: 4938 -4945 2007
Fig 1. Unadjusted survival of patients with first hematological relapse of acute myeloid leukemia after allogeneic hematopoietic stem-cell transplantation (HSCT) Multivariate analysis 1 - age < 37 years (P =. 008) 2 - longer CR after HSCT (> 5 months; (P <. 0001) 3 - use of DLI (P =. 04). Schmid, C. et al. J Clin Oncol; 25: 4938 -4945 2007
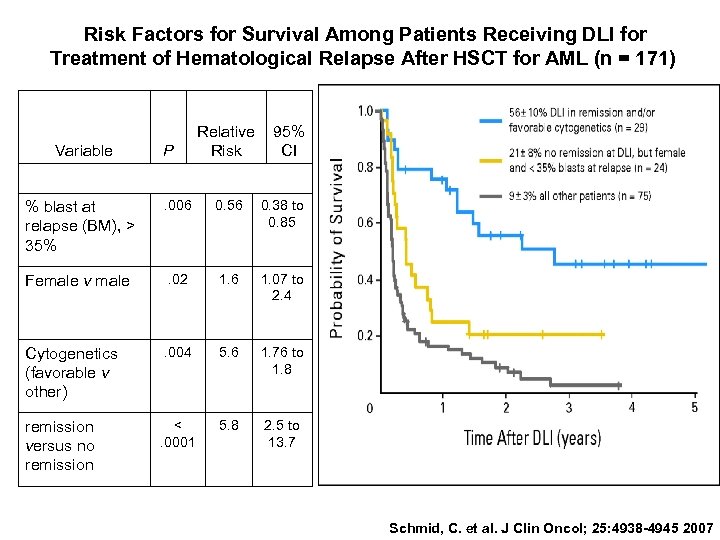 Risk Factors for Survival Among Patients Receiving DLI for Treatment of Hematological Relapse After HSCT for AML (n = 171) Variable P Relative 95% Risk CI % blast at relapse (BM), > 35% . 006 0. 56 0. 38 to 0. 85 Female v male . 02 1. 6 1. 07 to 2. 4 Cytogenetics (favorable v other) . 004 5. 6 1. 76 to 1. 8 remission versus no remission < . 0001 5. 8 2. 5 to 13. 7 Schmid, C. et al. J Clin Oncol; 25: 4938 -4945 2007
Risk Factors for Survival Among Patients Receiving DLI for Treatment of Hematological Relapse After HSCT for AML (n = 171) Variable P Relative 95% Risk CI % blast at relapse (BM), > 35% . 006 0. 56 0. 38 to 0. 85 Female v male . 02 1. 6 1. 07 to 2. 4 Cytogenetics (favorable v other) . 004 5. 6 1. 76 to 1. 8 remission versus no remission < . 0001 5. 8 2. 5 to 13. 7 Schmid, C. et al. J Clin Oncol; 25: 4938 -4945 2007
 Second transplant
Second transplant
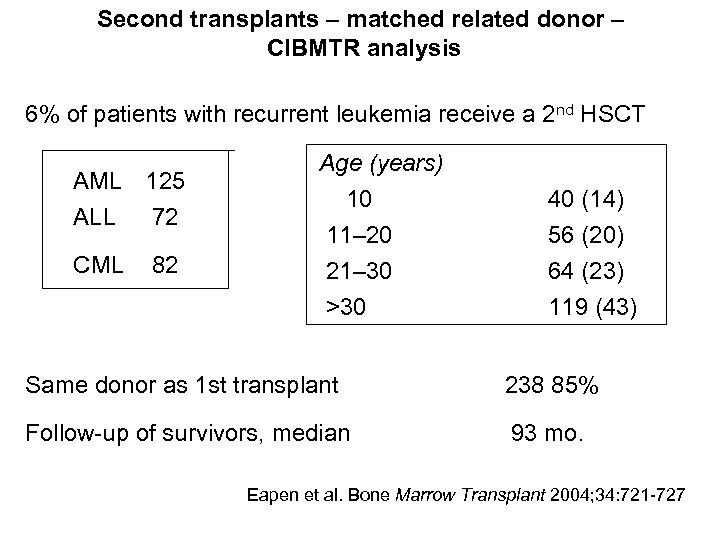 Second transplants – matched related donor – CIBMTR analysis 6% of patients with recurrent leukemia receive a 2 nd HSCT CML 82 40 (14) 56 (20) 64 (23) >30 AML 125 ALL 72 Age (years) 10 11– 20 21– 30 119 (43) Same donor as 1 st transplant Follow-up of survivors, median 238 85% 93 mo. Eapen et al. Bone Marrow Transplant 2004; 34: 721 -727
Second transplants – matched related donor – CIBMTR analysis 6% of patients with recurrent leukemia receive a 2 nd HSCT CML 82 40 (14) 56 (20) 64 (23) >30 AML 125 ALL 72 Age (years) 10 11– 20 21– 30 119 (43) Same donor as 1 st transplant Follow-up of survivors, median 238 85% 93 mo. Eapen et al. Bone Marrow Transplant 2004; 34: 721 -727
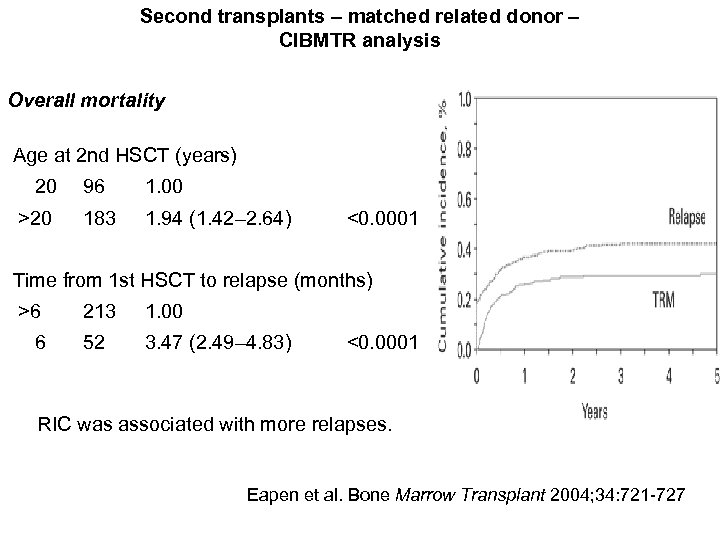 Second transplants – matched related donor – CIBMTR analysis Overall mortality Age at 2 nd HSCT (years) 20 96 1. 00 >20 183 1. 94 (1. 42– 2. 64) <0. 0001 Time from 1 st HSCT to relapse (months) >6 213 1. 00 6 52 3. 47 (2. 49– 4. 83) <0. 0001 RIC was associated with more relapses. Eapen et al. Bone Marrow Transplant 2004; 34: 721 -727
Second transplants – matched related donor – CIBMTR analysis Overall mortality Age at 2 nd HSCT (years) 20 96 1. 00 >20 183 1. 94 (1. 42– 2. 64) <0. 0001 Time from 1 st HSCT to relapse (months) >6 213 1. 00 6 52 3. 47 (2. 49– 4. 83) <0. 0001 RIC was associated with more relapses. Eapen et al. Bone Marrow Transplant 2004; 34: 721 -727
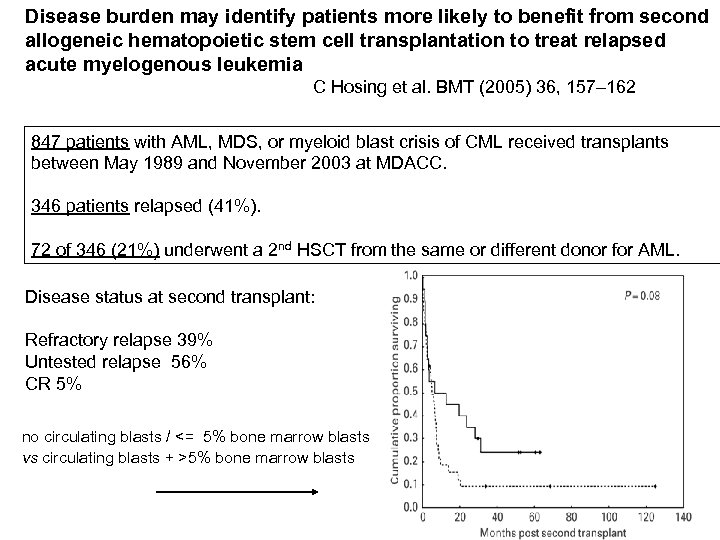 Disease burden may identify patients more likely to benefit from second allogeneic hematopoietic stem cell transplantation to treat relapsed acute myelogenous leukemia C Hosing et al. BMT (2005) 36, 157– 162 847 patients with AML, MDS, or myeloid blast crisis of CML received transplants between May 1989 and November 2003 at MDACC. 346 patients relapsed (41%). 72 of 346 (21%) underwent a 2 nd HSCT from the same or different donor for AML. Disease status at second transplant: Refractory relapse 39% Untested relapse 56% CR 5% no circulating blasts / <= 5% bone marrow blasts vs circulating blasts + >5% bone marrow blasts
Disease burden may identify patients more likely to benefit from second allogeneic hematopoietic stem cell transplantation to treat relapsed acute myelogenous leukemia C Hosing et al. BMT (2005) 36, 157– 162 847 patients with AML, MDS, or myeloid blast crisis of CML received transplants between May 1989 and November 2003 at MDACC. 346 patients relapsed (41%). 72 of 346 (21%) underwent a 2 nd HSCT from the same or different donor for AML. Disease status at second transplant: Refractory relapse 39% Untested relapse 56% CR 5% no circulating blasts / <= 5% bone marrow blasts vs circulating blasts + >5% bone marrow blasts
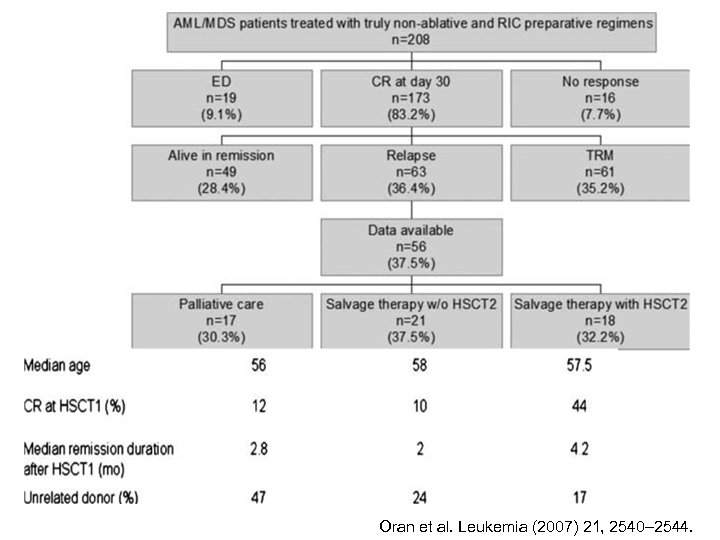 Oran et al. Leukemia (2007) 21, 2540– 2544.
Oran et al. Leukemia (2007) 21, 2540– 2544.
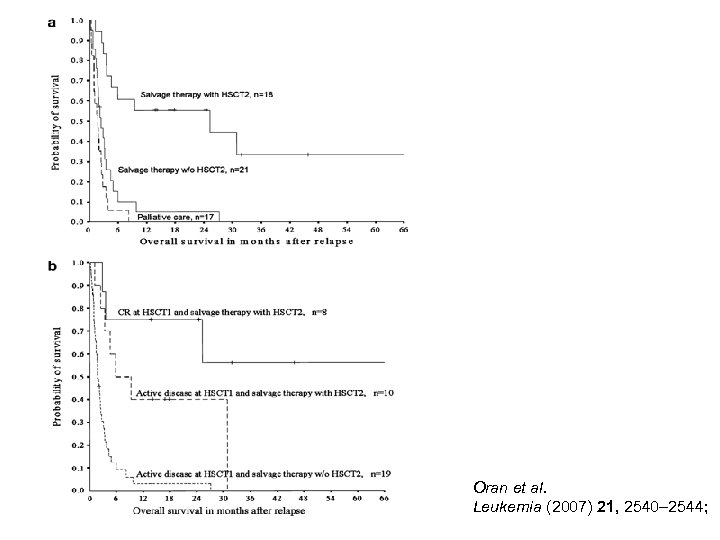 Oran et al. Leukemia (2007) 21, 2540– 2544;
Oran et al. Leukemia (2007) 21, 2540– 2544;
 Salvage chemotherapy
Salvage chemotherapy
 Chemotherapy Response rates are a function of : - chemosensitivity - remission duration - tempo (and disease “bulk”? ) of relapse No evidence that any particular regimen is superior to any other. FHCRC data (years 1977 -1984) 95 patients relapsed - 55 received chemotherapy Response rates: CR rate with cytarabine (+/- adriamycin): 32% of 34 patients median DFS of 9. 7 months.
Chemotherapy Response rates are a function of : - chemosensitivity - remission duration - tempo (and disease “bulk”? ) of relapse No evidence that any particular regimen is superior to any other. FHCRC data (years 1977 -1984) 95 patients relapsed - 55 received chemotherapy Response rates: CR rate with cytarabine (+/- adriamycin): 32% of 34 patients median DFS of 9. 7 months.
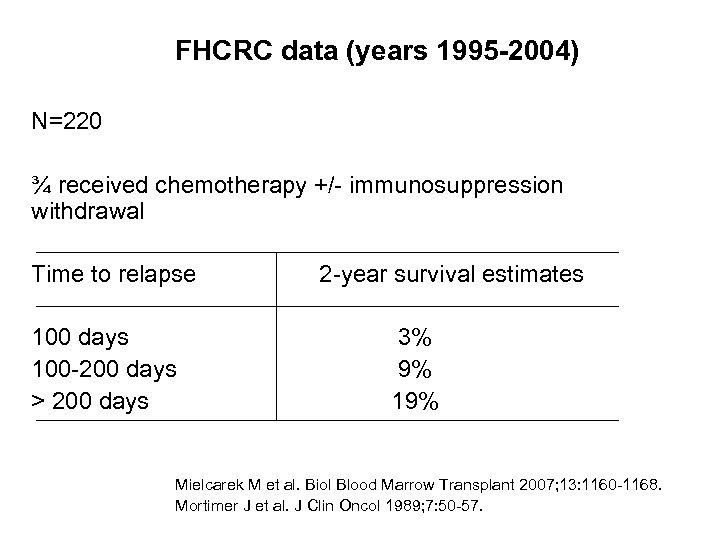 FHCRC data (years 1995 -2004) N=220 ¾ received chemotherapy +/- immunosuppression withdrawal Time to relapse 100 days 100 -200 days > 200 days 2 -year survival estimates 3% 9% 19% Mielcarek M et al. Biol Blood Marrow Transplant 2007; 13: 1160 -1168. Mortimer J et al. J Clin Oncol 1989; 7: 50 -57.
FHCRC data (years 1995 -2004) N=220 ¾ received chemotherapy +/- immunosuppression withdrawal Time to relapse 100 days 100 -200 days > 200 days 2 -year survival estimates 3% 9% 19% Mielcarek M et al. Biol Blood Marrow Transplant 2007; 13: 1160 -1168. Mortimer J et al. J Clin Oncol 1989; 7: 50 -57.
 Novel agents -A variety of ‘targeted’ agents are under investigation – role unclear in this setting. -sorafenib – FLT 3 positive patients Metzelder S et al. Blood 2009; 113: 6567 -6571. -5 -azacitidine. Jabbour et al. Cancer, 2009; 115: 1899 -1905 Lubbert et al. Bone Marrow Transplant 2009
Novel agents -A variety of ‘targeted’ agents are under investigation – role unclear in this setting. -sorafenib – FLT 3 positive patients Metzelder S et al. Blood 2009; 113: 6567 -6571. -5 -azacitidine. Jabbour et al. Cancer, 2009; 115: 1899 -1905 Lubbert et al. Bone Marrow Transplant 2009
 Unanswered questions Potentially answerable with a large database: 1 - Better definition of subgroups more likely to benefit from interventions (versus palliative care only). 2 - DLI after alternative donor transplants. 3 – Salvage therapy for children.
Unanswered questions Potentially answerable with a large database: 1 - Better definition of subgroups more likely to benefit from interventions (versus palliative care only). 2 - DLI after alternative donor transplants. 3 – Salvage therapy for children.
 Unanswered questions Potentially answerable with innovative, multicenter clinical trials: - how to incorporate newer drugs and/or cell therapy approaches AND prove that they work.
Unanswered questions Potentially answerable with innovative, multicenter clinical trials: - how to incorporate newer drugs and/or cell therapy approaches AND prove that they work.
 Conclusions Current therapies benefit a small minority of patients. This is clearly a ‘phase I’ scenario.
Conclusions Current therapies benefit a small minority of patients. This is clearly a ‘phase I’ scenario.
 Key obstacles for development of large, randomized, prospective clinical studies Lack of large, multicenter prospective phase I and II studies to define experimental arms in a randomized study. Lack of large databases dealing specifically with relapse information. Lack of a broad discussion and consensus that should ideally involve drug companies and the FDA on the need to enroll patients in phase I, II or III clinical trials for the treatment of AML relapsing after allogeneic HSCT.
Key obstacles for development of large, randomized, prospective clinical studies Lack of large, multicenter prospective phase I and II studies to define experimental arms in a randomized study. Lack of large databases dealing specifically with relapse information. Lack of a broad discussion and consensus that should ideally involve drug companies and the FDA on the need to enroll patients in phase I, II or III clinical trials for the treatment of AML relapsing after allogeneic HSCT.
 Proposed initiatives - Creation of a sample repository. - Development (or improvement of current available databases) of systems for detailed data collection in the relapse setting. - Multicenter approach! -Identification of target antigens, modification of DLI, targeting antigens by adoptive transfer, vaccines etc - NK cells - Maintenance therapy : azacitidine, decitabine, sorafenib etc
Proposed initiatives - Creation of a sample repository. - Development (or improvement of current available databases) of systems for detailed data collection in the relapse setting. - Multicenter approach! -Identification of target antigens, modification of DLI, targeting antigens by adoptive transfer, vaccines etc - NK cells - Maintenance therapy : azacitidine, decitabine, sorafenib etc

 Treatment of ALL that has relapsed after allogeneic Transplantation Alan S Wayne and Jacob M. Rowe
Treatment of ALL that has relapsed after allogeneic Transplantation Alan S Wayne and Jacob M. Rowe
 Relapsed ALL • Early diagnosis is probably crucial Early diagnosis Conventional and investigational therapies likely to be greater if intervention is prior to florid relapse
Relapsed ALL • Early diagnosis is probably crucial Early diagnosis Conventional and investigational therapies likely to be greater if intervention is prior to florid relapse
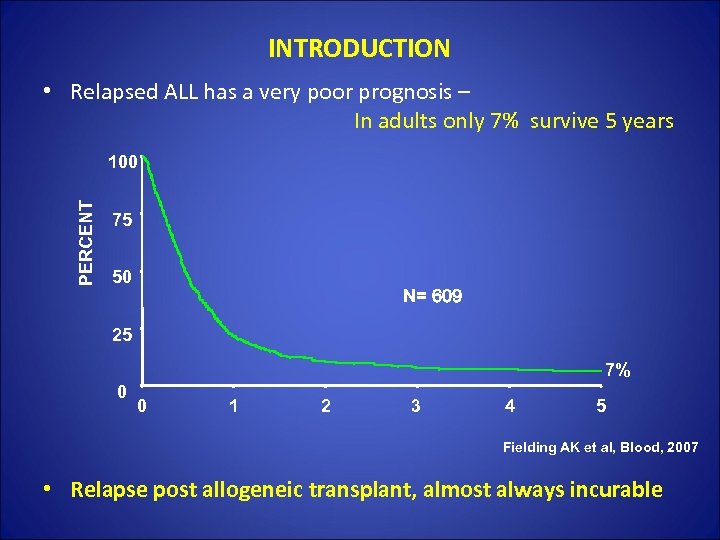 INTRODUCTION • Relapsed ALL has a very poor prognosis – In adults only 7% survive 5 years PERCENT 100 75 50 N= 609 25 7% 0 0 1 2 3 4 5 Fielding AK et al, Blood, 2007 • Relapse post allogeneic transplant, almost always incurable
INTRODUCTION • Relapsed ALL has a very poor prognosis – In adults only 7% survive 5 years PERCENT 100 75 50 N= 609 25 7% 0 0 1 2 3 4 5 Fielding AK et al, Blood, 2007 • Relapse post allogeneic transplant, almost always incurable
 Relapsed ALL Post Allogeneic HSCT While cures are rare, INDUCTION OF REMISSION and / or PROLONGATION OF RESPONSE is an important endpoint
Relapsed ALL Post Allogeneic HSCT While cures are rare, INDUCTION OF REMISSION and / or PROLONGATION OF RESPONSE is an important endpoint
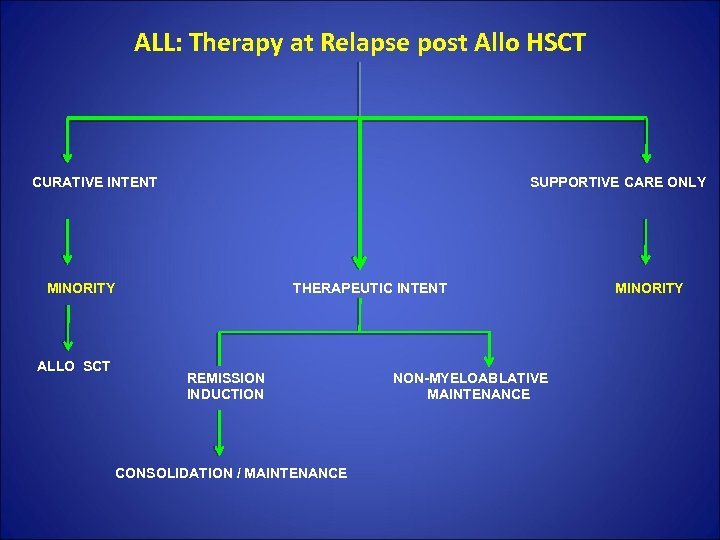 ALL: Therapy at Relapse post Allo HSCT CURATIVE INTENT SUPPORTIVE CARE ONLY MINORITY THERAPEUTIC INTENT MINORITY ALLO SCT REMISSION NON-MYELOABLATIVE INDUCTION MAINTENANCE CONSOLIDATION / MAINTENANCE
ALL: Therapy at Relapse post Allo HSCT CURATIVE INTENT SUPPORTIVE CARE ONLY MINORITY THERAPEUTIC INTENT MINORITY ALLO SCT REMISSION NON-MYELOABLATIVE INDUCTION MAINTENANCE CONSOLIDATION / MAINTENANCE
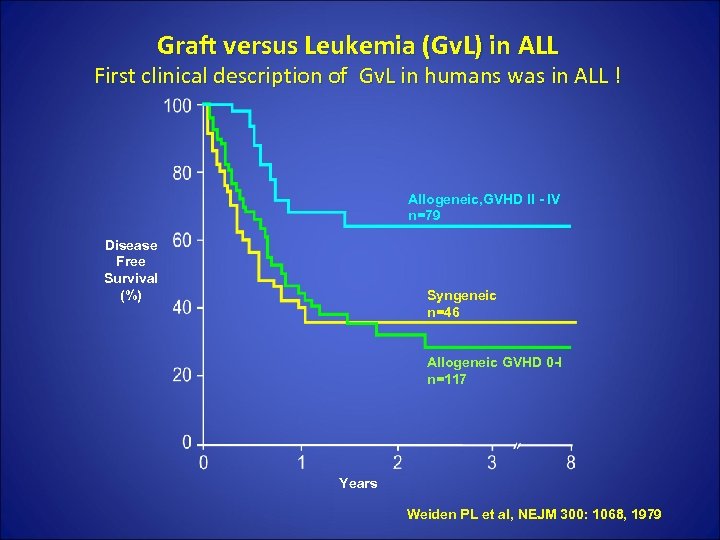 Graft versus Leukemia (Gv. L) in ALL First clinical description of Gv. L in humans was in ALL ! Allogeneic, GVHD II - IV n=79 Disease Free Survival (%) Syngeneic n=46 Allogeneic GVHD 0 -I n=117 Years Weiden PL et al, NEJM 300: 1068, 1979
Graft versus Leukemia (Gv. L) in ALL First clinical description of Gv. L in humans was in ALL ! Allogeneic, GVHD II - IV n=79 Disease Free Survival (%) Syngeneic n=46 Allogeneic GVHD 0 -I n=117 Years Weiden PL et al, NEJM 300: 1068, 1979
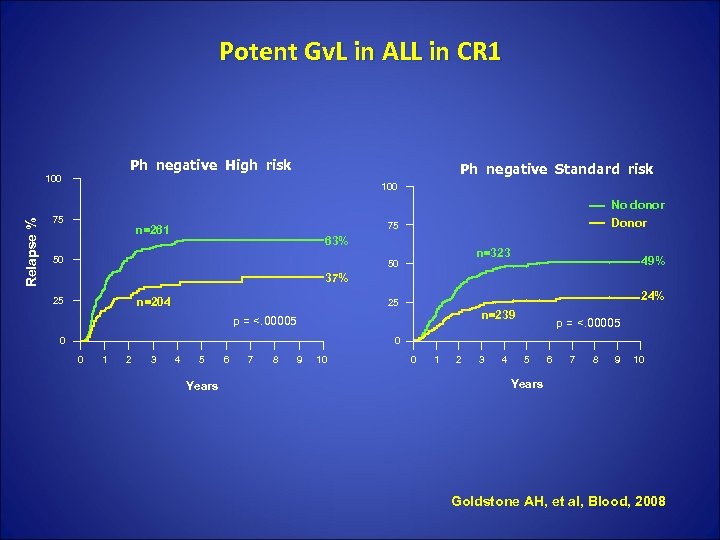 Potent Gv. L in ALL in CR 1 Ph negative High risk Relapse % 100 Ph negative Standard risk 100 75 No donor Donor 75 63% n=323 49% 50 37% n=261 50 25 n=204 24% 25 n=239 p = <. 00005 0 0 1 2 3 4 5 Years 6 7 8 9 10 0 1 2 3 4 5 6 7 8 9 10 Years Goldstone AH, et al, Blood, 2008
Potent Gv. L in ALL in CR 1 Ph negative High risk Relapse % 100 Ph negative Standard risk 100 75 No donor Donor 75 63% n=323 49% 50 37% n=261 50 25 n=204 24% 25 n=239 p = <. 00005 0 0 1 2 3 4 5 Years 6 7 8 9 10 0 1 2 3 4 5 6 7 8 9 10 Years Goldstone AH, et al, Blood, 2008
 Graft versus Leukemia (Gv. L) in ALL REDUCED-INTENSITY CONDITIONING FOR HIGH-RISK ALL CIBMTR STUDY OF ALL IN CR 1 OR CR 2 RIC (n= 92) vs myeloablative (n= 1421) Median Age, yrs, Median Age 45 28 p= <. 0001 OS @ 3 yrs, % OS 38 43 p= . 39 TRM @ 3 yrs, % TRM 32 33 p= . 86 Marks, DI et al, ASH 2009
Graft versus Leukemia (Gv. L) in ALL REDUCED-INTENSITY CONDITIONING FOR HIGH-RISK ALL CIBMTR STUDY OF ALL IN CR 1 OR CR 2 RIC (n= 92) vs myeloablative (n= 1421) Median Age, yrs, Median Age 45 28 p= <. 0001 OS @ 3 yrs, % OS 38 43 p= . 39 TRM @ 3 yrs, % TRM 32 33 p= . 86 Marks, DI et al, ASH 2009
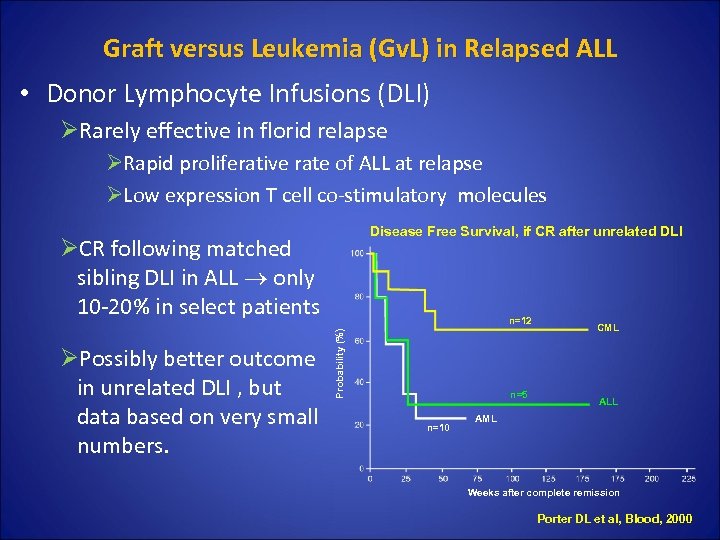 Graft versus Leukemia (Gv. L) in Relapsed ALL • Donor Lymphocyte Infusions (DLI) ØRarely effective in florid relapse ØRapid proliferative rate of ALL at relapse ØLow expression T cell co-stimulatory molecules Disease Free Survival, if CR after unrelated DLI ØCR following matched sibling DLI in ALL only 10 -20% in select patients Probability (%) ØPossibly better outcome in unrelated DLI , but data based on very small numbers. n=12 n=5 n=10 CML ALL AML Weeks after complete remission Porter DL et al, Blood, 2000
Graft versus Leukemia (Gv. L) in Relapsed ALL • Donor Lymphocyte Infusions (DLI) ØRarely effective in florid relapse ØRapid proliferative rate of ALL at relapse ØLow expression T cell co-stimulatory molecules Disease Free Survival, if CR after unrelated DLI ØCR following matched sibling DLI in ALL only 10 -20% in select patients Probability (%) ØPossibly better outcome in unrelated DLI , but data based on very small numbers. n=12 n=5 n=10 CML ALL AML Weeks after complete remission Porter DL et al, Blood, 2000
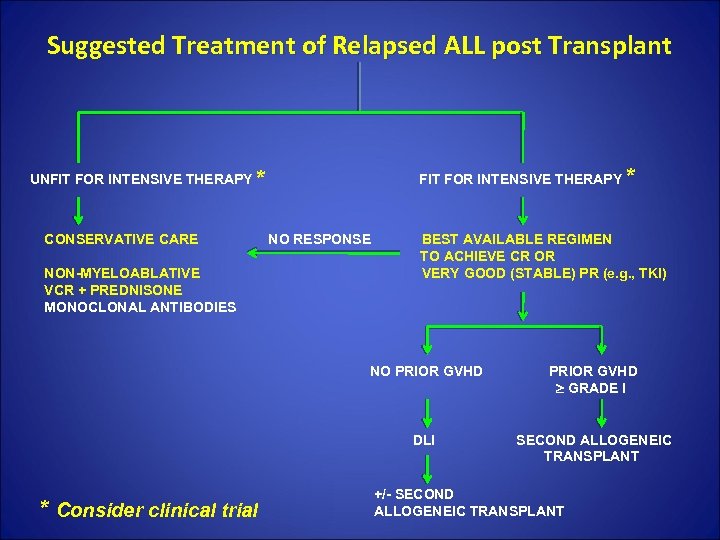 Suggested Treatment of Relapsed ALL post Transplant * UNFIT FOR INTENSIVE THERAPY * CONSERVATIVE CARE NO RESPONSE BEST AVAILABLE REGIMEN TO ACHIEVE CR OR NON-MYELOABLATIVE VERY GOOD (STABLE) PR (e. g. , TKI) VCR + PREDNISONE MONOCLONAL ANTIBODIES NO PRIOR GVHD GRADE I DLI SECOND ALLOGENEIC TRANSPLANT * Consider clinical trial +/- SECOND ALLOGENEIC TRANSPLANT
Suggested Treatment of Relapsed ALL post Transplant * UNFIT FOR INTENSIVE THERAPY * CONSERVATIVE CARE NO RESPONSE BEST AVAILABLE REGIMEN TO ACHIEVE CR OR NON-MYELOABLATIVE VERY GOOD (STABLE) PR (e. g. , TKI) VCR + PREDNISONE MONOCLONAL ANTIBODIES NO PRIOR GVHD GRADE I DLI SECOND ALLOGENEIC TRANSPLANT * Consider clinical trial +/- SECOND ALLOGENEIC TRANSPLANT
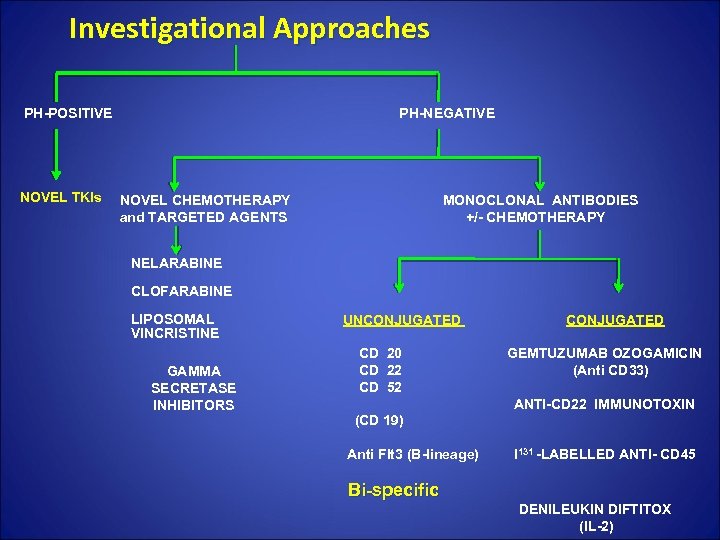 Investigational Approaches PH-POSITIVE PH-NEGATIVE NOVEL TKIs NOVEL CHEMOTHERAPY MONOCLONAL ANTIBODIES and TARGETED AGENTS +/- CHEMOTHERAPY NELARABINE CLOFARABINE LIPOSOMAL VINCRISTINE GAMMA SECRETASE INHIBITORS UNCONJUGATED CONJUGATED CD 20 GEMTUZUMAB OZOGAMICIN CD 22 (Anti CD 33) CD 52 ANTI-CD 22 IMMUNOTOXIN (CD 19) Anti Flt 3 (B-lineage) I 131 -LABELLED ANTI- CD 45 Bi-specific DENILEUKIN DIFTITOX (IL-2)
Investigational Approaches PH-POSITIVE PH-NEGATIVE NOVEL TKIs NOVEL CHEMOTHERAPY MONOCLONAL ANTIBODIES and TARGETED AGENTS +/- CHEMOTHERAPY NELARABINE CLOFARABINE LIPOSOMAL VINCRISTINE GAMMA SECRETASE INHIBITORS UNCONJUGATED CONJUGATED CD 20 GEMTUZUMAB OZOGAMICIN CD 22 (Anti CD 33) CD 52 ANTI-CD 22 IMMUNOTOXIN (CD 19) Anti Flt 3 (B-lineage) I 131 -LABELLED ANTI- CD 45 Bi-specific DENILEUKIN DIFTITOX (IL-2)
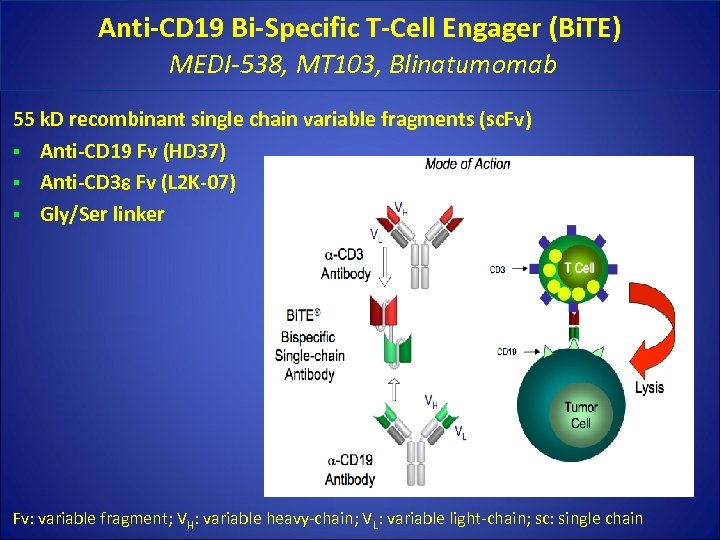 Anti-CD 19 Bi-Specific T-Cell Engager (Bi. TE) MEDI-538, MT 103, Blinatumomab 55 k. D recombinant single chain variable fragments (sc. Fv) § Anti-CD 19 Fv (HD 37) § Anti-CD 3 Fv (L 2 K-07) § Gly/Ser linker Fv: variable fragment; VH: variable heavy-chain; VL: variable light-chain; sc: single chain
Anti-CD 19 Bi-Specific T-Cell Engager (Bi. TE) MEDI-538, MT 103, Blinatumomab 55 k. D recombinant single chain variable fragments (sc. Fv) § Anti-CD 19 Fv (HD 37) § Anti-CD 3 Fv (L 2 K-07) § Gly/Ser linker Fv: variable fragment; VH: variable heavy-chain; VL: variable light-chain; sc: single chain
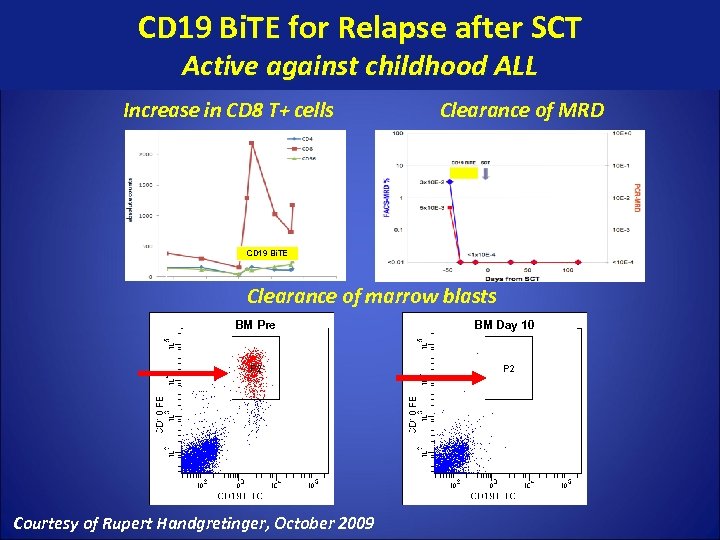 CD 19 Bi. TE for Relapse after SCT Active against childhood ALL Increase in CD 8 T+ cells Clearance of MRD CD 19 Bi. TE Clearance of marrow blasts BM Pre Courtesy of Rupert Handgretinger, October 2009 BM Day 10
CD 19 Bi. TE for Relapse after SCT Active against childhood ALL Increase in CD 8 T+ cells Clearance of MRD CD 19 Bi. TE Clearance of marrow blasts BM Pre Courtesy of Rupert Handgretinger, October 2009 BM Day 10
 Future Directions - Cancer Vaccines I • Attractive conceptually in this patient population due to low toxicity – Efficacy more likely in states of minimal residual disease – More data in AML, but similar rationale may be applicable in ALL
Future Directions - Cancer Vaccines I • Attractive conceptually in this patient population due to low toxicity – Efficacy more likely in states of minimal residual disease – More data in AML, but similar rationale may be applicable in ALL
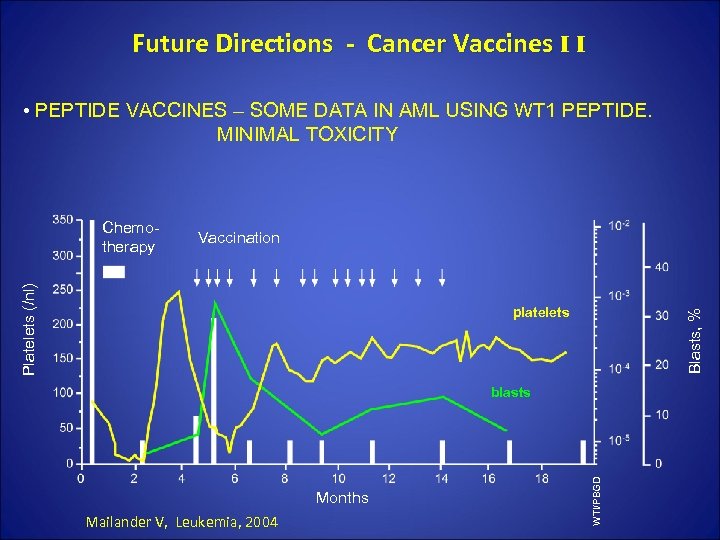 Future Directions - Cancer Vaccines I I • PEPTIDE VACCINES – SOME DATA IN AML USING WT 1 PEPTIDE. MINIMAL TOXICITY Vaccination Platelets (/nl) Mailander V, Leukemia, 2004 Blasts, % platelets blasts Months WTI/PBGD Chemotherapy
Future Directions - Cancer Vaccines I I • PEPTIDE VACCINES – SOME DATA IN AML USING WT 1 PEPTIDE. MINIMAL TOXICITY Vaccination Platelets (/nl) Mailander V, Leukemia, 2004 Blasts, % platelets blasts Months WTI/PBGD Chemotherapy
 Future Directions - Cancer Vaccines III • PR 1, a leukemia-associated antigen, studied for vaccination in AML, with some success [HESLOP HE, STVENSON FK AND MOLLDDREM JJ, HEMATOLOGY, ASH EDUCATION 2003] • Dendritic cells and antigen presenting cells utilized to improve the immune response to tumor associated antigen in multiple myeloma [AVIGAN D ET AL, ASH 2008 AND 2009] • Rationale applicable to other malignancies including ALL SUHOSKI MM , MOL THER, 2007
Future Directions - Cancer Vaccines III • PR 1, a leukemia-associated antigen, studied for vaccination in AML, with some success [HESLOP HE, STVENSON FK AND MOLLDDREM JJ, HEMATOLOGY, ASH EDUCATION 2003] • Dendritic cells and antigen presenting cells utilized to improve the immune response to tumor associated antigen in multiple myeloma [AVIGAN D ET AL, ASH 2008 AND 2009] • Rationale applicable to other malignancies including ALL SUHOSKI MM , MOL THER, 2007

 Disease-Specific Interventions for Relapse of non-Hodgkin Lymphoma After Allogeneic Transplantation David Maloney MD, Ph. D Koen van Besien, MD
Disease-Specific Interventions for Relapse of non-Hodgkin Lymphoma After Allogeneic Transplantation David Maloney MD, Ph. D Koen van Besien, MD
 NHL Relapse Following Allogeneic HCT: A Wide Range of Issues to Consider • Many different histologic subtypes of NHL – indolent to aggressive behavior • Patients often treated with Allogeneic HCT as “last resort” – after failed high-dose therapy and autologous HCT – with chemotherapy refractory disease • Transplanted with a variety of conditioning intensities – myeloablative, reduced intensity, nonmyeloablative – T depleted or T replete grafts – HLA matched or mismatched, related, unrelated, cord blood or haploidentical stem cell sources
NHL Relapse Following Allogeneic HCT: A Wide Range of Issues to Consider • Many different histologic subtypes of NHL – indolent to aggressive behavior • Patients often treated with Allogeneic HCT as “last resort” – after failed high-dose therapy and autologous HCT – with chemotherapy refractory disease • Transplanted with a variety of conditioning intensities – myeloablative, reduced intensity, nonmyeloablative – T depleted or T replete grafts – HLA matched or mismatched, related, unrelated, cord blood or haploidentical stem cell sources
 Factors that Influence the Outcome of NHL Relapse post Allogeneic HCT • • Histology Presence or absence of GHVD T-replete vs T-depleted grafts Disease status at HCT (chemorefractory vs sensitive) • Transplant conditioning intensity • Timing of relapse (early vs late)
Factors that Influence the Outcome of NHL Relapse post Allogeneic HCT • • Histology Presence or absence of GHVD T-replete vs T-depleted grafts Disease status at HCT (chemorefractory vs sensitive) • Transplant conditioning intensity • Timing of relapse (early vs late)
 Approach to the Treatment of NHL Relapse post Allogeneic HCT • In the absence of GHVD – Withdrawal of immunosuppression (IS) – Donor Lymphocyte Infusions (DLI) • Monoclonal antibody therapy • Chemotherapy +/- DLI • Radiotherapy +/- DLI • Immune stimulants (IL-2 etc) • Second allogeneic HCT
Approach to the Treatment of NHL Relapse post Allogeneic HCT • In the absence of GHVD – Withdrawal of immunosuppression (IS) – Donor Lymphocyte Infusions (DLI) • Monoclonal antibody therapy • Chemotherapy +/- DLI • Radiotherapy +/- DLI • Immune stimulants (IL-2 etc) • Second allogeneic HCT
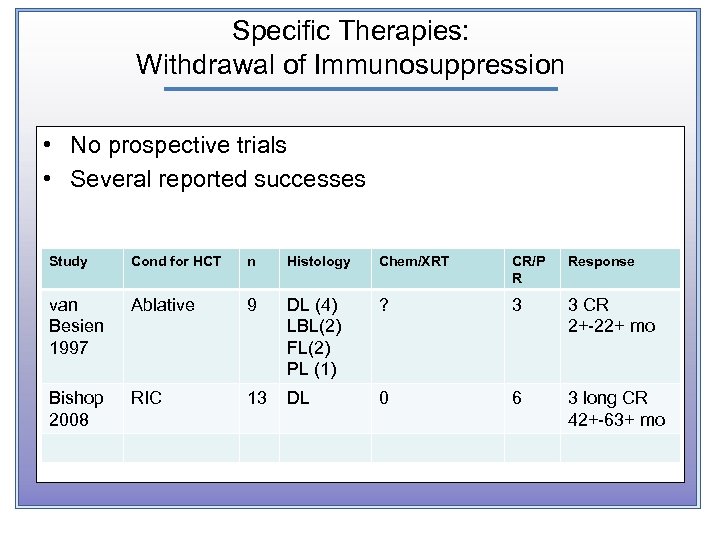 Specific Therapies: Withdrawal of Immunosuppression • No prospective trials • Several reported successes Study Cond for HCT n Histology Chem/XRT CR/P R Response van Besien 1997 Ablative 9 DL (4) LBL(2) FL(2) PL (1) ? 3 3 CR 2+-22+ mo Bishop 2008 RIC 13 DL 0 6 3 long CR 42+-63+ mo
Specific Therapies: Withdrawal of Immunosuppression • No prospective trials • Several reported successes Study Cond for HCT n Histology Chem/XRT CR/P R Response van Besien 1997 Ablative 9 DL (4) LBL(2) FL(2) PL (1) ? 3 3 CR 2+-22+ mo Bishop 2008 RIC 13 DL 0 6 3 long CR 42+-63+ mo
 Specific Therapies: Donor Lymphocyte Infusions (DLI) • An option in the absence of GVHD • More commonly reported following T-depleted HCT • Response often correlated with disease histology and pace of progression • Some risk of subsequent GVHD • A wide range of CD 3 cell doses used
Specific Therapies: Donor Lymphocyte Infusions (DLI) • An option in the absence of GVHD • More commonly reported following T-depleted HCT • Response often correlated with disease histology and pace of progression • Some risk of subsequent GVHD • A wide range of CD 3 cell doses used
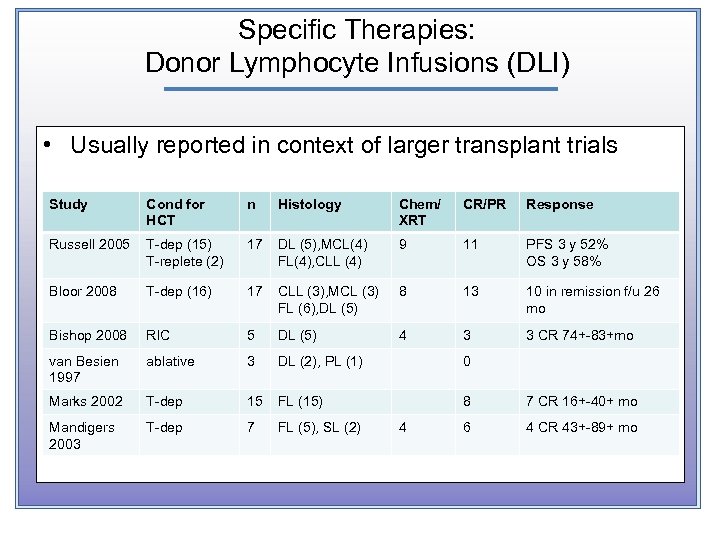 Specific Therapies: Donor Lymphocyte Infusions (DLI) • Usually reported in context of larger transplant trials Study Cond for HCT n Histology Chem/ XRT CR/PR Response Russell 2005 T-dep (15) T-replete (2) 17 DL (5), MCL(4) FL(4), CLL (4) 9 11 PFS 3 y 52% OS 3 y 58% Bloor 2008 T-dep (16) 17 CLL (3), MCL (3) FL (6), DL (5) 8 13 10 in remission f/u 26 mo Bishop 2008 RIC 5 DL (5) 4 3 3 CR 74+-83+mo van Besien 1997 ablative 3 DL (2), PL (1) 0 Marks 2002 T-dep 15 FL (15) 8 7 CR 16+-40+ mo Mandigers 2003 T-dep 7 FL (5), SL (2) 6 4 CR 43+-89+ mo 4
Specific Therapies: Donor Lymphocyte Infusions (DLI) • Usually reported in context of larger transplant trials Study Cond for HCT n Histology Chem/ XRT CR/PR Response Russell 2005 T-dep (15) T-replete (2) 17 DL (5), MCL(4) FL(4), CLL (4) 9 11 PFS 3 y 52% OS 3 y 58% Bloor 2008 T-dep (16) 17 CLL (3), MCL (3) FL (6), DL (5) 8 13 10 in remission f/u 26 mo Bishop 2008 RIC 5 DL (5) 4 3 3 CR 74+-83+mo van Besien 1997 ablative 3 DL (2), PL (1) 0 Marks 2002 T-dep 15 FL (15) 8 7 CR 16+-40+ mo Mandigers 2003 T-dep 7 FL (5), SL (2) 6 4 CR 43+-89+ mo 4
 Specific Therapies: Chemotherapy +/- DLI • Anecdotal reports of chemotherapy +/- DLI generally within clinical trial results – Occasionally successful – Support the concept of ongoing graft-vs-tumor effects that may be capable of controlling minimal residual disease – No clear choice of agents – Concern of “ablating” graft appears low, but needs to be studied
Specific Therapies: Chemotherapy +/- DLI • Anecdotal reports of chemotherapy +/- DLI generally within clinical trial results – Occasionally successful – Support the concept of ongoing graft-vs-tumor effects that may be capable of controlling minimal residual disease – No clear choice of agents – Concern of “ablating” graft appears low, but needs to be studied
 Specific Therapies: Monoclonal Antibody Therapy • Frequently utilized for B cell NHL – Low hematologic toxicity – May promote antigen priming and graft-vs-tumor effects • Tumor cell lines exposed to rituximab were more effective at alloantigen presentation (Selenko 2002) • Clinical trials from MD Anderson suggest improved outcome following the addition of high-dose rituximab (FL and CLL) • Reasonable option for most patients
Specific Therapies: Monoclonal Antibody Therapy • Frequently utilized for B cell NHL – Low hematologic toxicity – May promote antigen priming and graft-vs-tumor effects • Tumor cell lines exposed to rituximab were more effective at alloantigen presentation (Selenko 2002) • Clinical trials from MD Anderson suggest improved outcome following the addition of high-dose rituximab (FL and CLL) • Reasonable option for most patients
 Specific Therapies: Other Immune Modulatory Agents • Interleukin-2 (Kiss 2003) – CR in low grade NHL pt (flare GVHD) • Interferon alpha (Kawano 2004) – CR in DLBCL with IFN + DLI • Thalidomide (Tueger 2006) – 1 pt with DLBCL and CR • CTLA-4 blockade with ipilimumab (Bashey 2009) – 3 pts with HD or MCL had response
Specific Therapies: Other Immune Modulatory Agents • Interleukin-2 (Kiss 2003) – CR in low grade NHL pt (flare GVHD) • Interferon alpha (Kawano 2004) – CR in DLBCL with IFN + DLI • Thalidomide (Tueger 2006) – 1 pt with DLBCL and CR • CTLA-4 blockade with ipilimumab (Bashey 2009) – 3 pts with HD or MCL had response
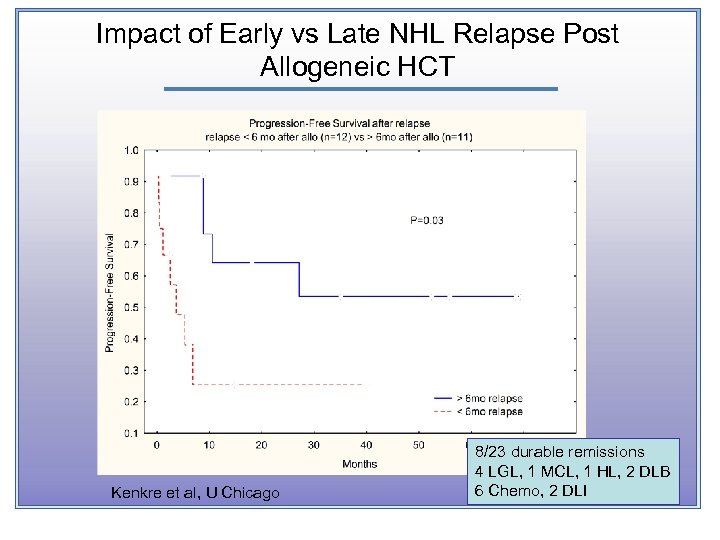 Impact of Early vs Late NHL Relapse Post Allogeneic HCT Kenkre et al, U Chicago 8/23 durable remissions 4 LGL, 1 MCL, 1 HL, 2 DLB 6 Chemo, 2 DLI
Impact of Early vs Late NHL Relapse Post Allogeneic HCT Kenkre et al, U Chicago 8/23 durable remissions 4 LGL, 1 MCL, 1 HL, 2 DLB 6 Chemo, 2 DLI
 Specific Interventions: Indolent NHL (FL) • MD Anderson (Khouri 2008) – Flu/Cy/Rituximab N=47 – All achieved CR, 2 relapses treated with rituximab +/- DLI to CR – OS 85% at 5 years • Seattle flu(2 Gy TBI) n=46 indolent (Rezvani 2008) – Relapse rate indolent 14%, 2 treated with IS/rituximab +/- DLI • UK (alemtuzumab containing) n=41 (Morris 2004) – Relapse at 3 years 44% – 6/10 responded to DLI leading to 65% current PFS at 3 years • UK (BEAM-alemtuzumab) n=44 (Ingram 2008) – Relapse rate of 20% – 4/6 treated with DLI had CR
Specific Interventions: Indolent NHL (FL) • MD Anderson (Khouri 2008) – Flu/Cy/Rituximab N=47 – All achieved CR, 2 relapses treated with rituximab +/- DLI to CR – OS 85% at 5 years • Seattle flu(2 Gy TBI) n=46 indolent (Rezvani 2008) – Relapse rate indolent 14%, 2 treated with IS/rituximab +/- DLI • UK (alemtuzumab containing) n=41 (Morris 2004) – Relapse at 3 years 44% – 6/10 responded to DLI leading to 65% current PFS at 3 years • UK (BEAM-alemtuzumab) n=44 (Ingram 2008) – Relapse rate of 20% – 4/6 treated with DLI had CR
 Specific Interventions: Indolent NHL • Generally very sensitive to graft-vs-NHL effects with low relapse rate following T replete HCT • Prototype follicular NHL – Sensitive to IS withdrawal – Monoclonal antibody therapy (rituximab) – DLI • In the absence of GVHD generally start with above, then consider chemotherapy +/- above • Long term survival possible post relapse
Specific Interventions: Indolent NHL • Generally very sensitive to graft-vs-NHL effects with low relapse rate following T replete HCT • Prototype follicular NHL – Sensitive to IS withdrawal – Monoclonal antibody therapy (rituximab) – DLI • In the absence of GVHD generally start with above, then consider chemotherapy +/- above • Long term survival possible post relapse
 Specific Interventions: Aggressive NHL • Seattle flu(2 Gy TBI) (Rezvani 2008) – DLBCL n=32, 41% disease progression • • • 1 DLI x 3 no effect 1 IS withdrawal- no effect 1 second myeloablative HCT died TRM 1 successful IS withdrawal-rituximab-XRT (alive in CR 54+ mo) 1 successful second RIC (same donor) (alive CR 34+ mo) 1 response IS withdrawal/ DLI • UK flu-mel-alemtuzumab (Thomson 2009) – DLBCL or transformed (n=48) – 15/48 (33%) relapsed • 5/12 CR to DLI +/- chemotherapy
Specific Interventions: Aggressive NHL • Seattle flu(2 Gy TBI) (Rezvani 2008) – DLBCL n=32, 41% disease progression • • • 1 DLI x 3 no effect 1 IS withdrawal- no effect 1 second myeloablative HCT died TRM 1 successful IS withdrawal-rituximab-XRT (alive in CR 54+ mo) 1 successful second RIC (same donor) (alive CR 34+ mo) 1 response IS withdrawal/ DLI • UK flu-mel-alemtuzumab (Thomson 2009) – DLBCL or transformed (n=48) – 15/48 (33%) relapsed • 5/12 CR to DLI +/- chemotherapy
 Specific Interventions: Aggressive NHL • French Registry (Sirvent 2009) – – DLBCL n=68 Relapse at 4 years 41% 20/26 relapses died of disease 5 in CR after chemo +/- XRT +/- DLI • Vancouver, BC (Doocey 2005) – Myeloablative conditioning for DLBCL n=44 – 13 progressed (32%) – 3 received DLI, all 13 died of disease • NCI (Bishop 2008) – – N=15 with aggressive NHL with relapse/ persistent disease post HCT 6/11 treated with IS withdrawal or DLI responded 3 / 4 treated with Chemotherapy/ DLI responded 6 remain in long term CR
Specific Interventions: Aggressive NHL • French Registry (Sirvent 2009) – – DLBCL n=68 Relapse at 4 years 41% 20/26 relapses died of disease 5 in CR after chemo +/- XRT +/- DLI • Vancouver, BC (Doocey 2005) – Myeloablative conditioning for DLBCL n=44 – 13 progressed (32%) – 3 received DLI, all 13 died of disease • NCI (Bishop 2008) – – N=15 with aggressive NHL with relapse/ persistent disease post HCT 6/11 treated with IS withdrawal or DLI responded 3 / 4 treated with Chemotherapy/ DLI responded 6 remain in long term CR
 Specific Interventions: Aggressive NHL • • Generally a kinetic problem post allogeneic HCT Requires disease control prior to HCT Early relapse difficult to manage Limited success with – IS withdrawal, DLI and antibody therapy • Long term benefit anecdotal – Aggressive chemotherapy +/- DLI – XRT +/- DLI – Worth considering if disease is sensitive
Specific Interventions: Aggressive NHL • • Generally a kinetic problem post allogeneic HCT Requires disease control prior to HCT Early relapse difficult to manage Limited success with – IS withdrawal, DLI and antibody therapy • Long term benefit anecdotal – Aggressive chemotherapy +/- DLI – XRT +/- DLI – Worth considering if disease is sensitive
 Specific Interventions: Mantle-cell NHL • MD Anderson – Myeloablative 1/16 relapsed (Khouri 1999) – RIC 3/18 relapsed (1/3 responded DLI) (Khouri 2003) – RIC n=35, (Tam 2009) • 6 year OS 53%, PFS 46% (3 pts rituximab + DLI) • Seattle (Maris 2004) – Flu(2 Gy TBI), n=33 – Relapse rate 9%, none after 6 months • EBMT n=22 ( Robinson 2002) – Progression at 2 years = 100%, ~50% overall group had T depletion • UK (Morris, 2004) – Alemtuzumab containing RIC (n=10 mcl) – At 3 years 50% relapse (1/2 responded to DLI)
Specific Interventions: Mantle-cell NHL • MD Anderson – Myeloablative 1/16 relapsed (Khouri 1999) – RIC 3/18 relapsed (1/3 responded DLI) (Khouri 2003) – RIC n=35, (Tam 2009) • 6 year OS 53%, PFS 46% (3 pts rituximab + DLI) • Seattle (Maris 2004) – Flu(2 Gy TBI), n=33 – Relapse rate 9%, none after 6 months • EBMT n=22 ( Robinson 2002) – Progression at 2 years = 100%, ~50% overall group had T depletion • UK (Morris, 2004) – Alemtuzumab containing RIC (n=10 mcl) – At 3 years 50% relapse (1/2 responded to DLI)
 Specific Interventions: Mantle-cell NHL • Surprisingly sensitive to allogeneic GVT activity • Late relapses rare following T-replete transplants • Higher relapse rate following T cell depletion – Requiring DLI or T cell add back • Similar to indolent NHL for – response to IS withdrawal – DLI – chemotherapy +/- DLI • Relapsing patients have chance of long-term DFS with RX
Specific Interventions: Mantle-cell NHL • Surprisingly sensitive to allogeneic GVT activity • Late relapses rare following T-replete transplants • Higher relapse rate following T cell depletion – Requiring DLI or T cell add back • Similar to indolent NHL for – response to IS withdrawal – DLI – chemotherapy +/- DLI • Relapsing patients have chance of long-term DFS with RX
 Future: Conclusions • NHL often sensitive to GVT activity • Post transplant modulation is promising – DLI – Immune modulatory agents • Achieving a remission may allow re-establishment of GVT effects – Chemotherapy or other +/- DLI • Ultimately therapy should invoke specific GVT immune responses without GVHD • Registry studies may be a first step toward prospective studies in specific disease/risk categories. • Prospective investigation of existing strategies in favorable groups • Radically novel approaches in unfavorable groups (i. e. aggressive histology/early relapse)
Future: Conclusions • NHL often sensitive to GVT activity • Post transplant modulation is promising – DLI – Immune modulatory agents • Achieving a remission may allow re-establishment of GVT effects – Chemotherapy or other +/- DLI • Ultimately therapy should invoke specific GVT immune responses without GVHD • Registry studies may be a first step toward prospective studies in specific disease/risk categories. • Prospective investigation of existing strategies in favorable groups • Radically novel approaches in unfavorable groups (i. e. aggressive histology/early relapse)

 Salvage Options: Hodgkin Lymphoma Dr Karl Peggs University College London Dr David Porter University of Pennsylvania Medical Center
Salvage Options: Hodgkin Lymphoma Dr Karl Peggs University College London Dr David Porter University of Pennsylvania Medical Center
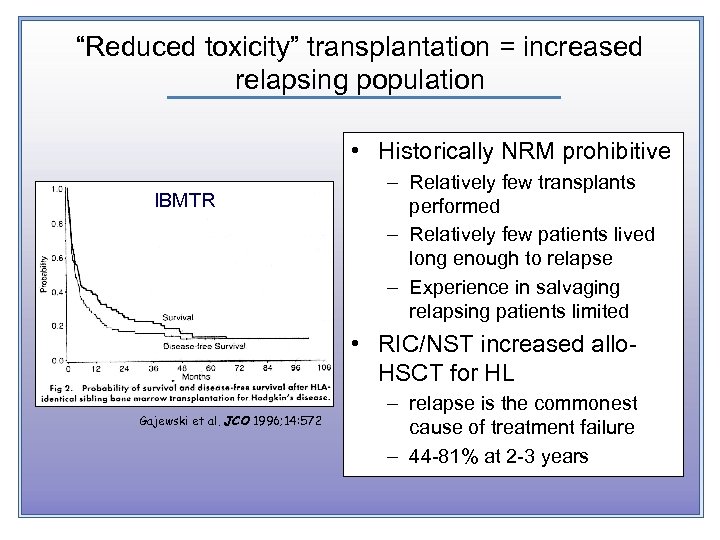 “Reduced toxicity” transplantation = increased relapsing population • Historically NRM prohibitive IBMTR – Relatively few transplants performed – Relatively few patients lived long enough to relapse – Experience in salvaging relapsing patients limited • RIC/NST increased allo. HSCT for HL Gajewski et al. JCO 1996; 14: 572 – relapse is the commonest cause of treatment failure – 44 -81% at 2 -3 years
“Reduced toxicity” transplantation = increased relapsing population • Historically NRM prohibitive IBMTR – Relatively few transplants performed – Relatively few patients lived long enough to relapse – Experience in salvaging relapsing patients limited • RIC/NST increased allo. HSCT for HL Gajewski et al. JCO 1996; 14: 572 – relapse is the commonest cause of treatment failure – 44 -81% at 2 -3 years
 Salvage chemotherapy • Most series demonstrate relapse risk higher in those with refractory disease at transplant – Most have received multiple lines of salvage pre-transplant so limited options to explore agents to which patients are naïve • No published series appear prescriptive re salvage; often the precise regimens used are not described • Regimens including IVE, ESHAP, mini-BEAM, irradiation, gemcitibine have been used in small numbers, often differing within series
Salvage chemotherapy • Most series demonstrate relapse risk higher in those with refractory disease at transplant – Most have received multiple lines of salvage pre-transplant so limited options to explore agents to which patients are naïve • No published series appear prescriptive re salvage; often the precise regimens used are not described • Regimens including IVE, ESHAP, mini-BEAM, irradiation, gemcitibine have been used in small numbers, often differing within series
 Salvage chemotherapy • • Response rates likely reflect disease-related features e. g. chemosensitivity at transplant, time to relapse, tempo of relapse No evidence that any particular regimen is a) likely to affect a cure b) superior to any other • • Anecdotal reports suggest that occasional patients achieve durable responses Most relapses occur early - the role of 2 nd transplants has not been explored
Salvage chemotherapy • • Response rates likely reflect disease-related features e. g. chemosensitivity at transplant, time to relapse, tempo of relapse No evidence that any particular regimen is a) likely to affect a cure b) superior to any other • • Anecdotal reports suggest that occasional patients achieve durable responses Most relapses occur early - the role of 2 nd transplants has not been explored
 Novel agents • Monoclonal antibodies (m. Ab) are of interest (might augment DLI) – anti-CD 20: CD 20+ nodular lymphocyte predominant • – – anti-CD 25: anti-CD 30: • • relatively few of these cases are transplanted due to the rarity of NLP HL and high cure rates with conventional approaches both may be more effective if used as vectors for delivery of radio -conjugates or cytotoxics (monomethyl auristatin E) Immunostimulatory m. Ab: – – anti-CTLA-4, anti-PD 1, anti-PDL 1 (antagonistic) anti-4 -1 BB, anti-OX 40 (agonistic)
Novel agents • Monoclonal antibodies (m. Ab) are of interest (might augment DLI) – anti-CD 20: CD 20+ nodular lymphocyte predominant • – – anti-CD 25: anti-CD 30: • • relatively few of these cases are transplanted due to the rarity of NLP HL and high cure rates with conventional approaches both may be more effective if used as vectors for delivery of radio -conjugates or cytotoxics (monomethyl auristatin E) Immunostimulatory m. Ab: – – anti-CTLA-4, anti-PD 1, anti-PDL 1 (antagonistic) anti-4 -1 BB, anti-OX 40 (agonistic)
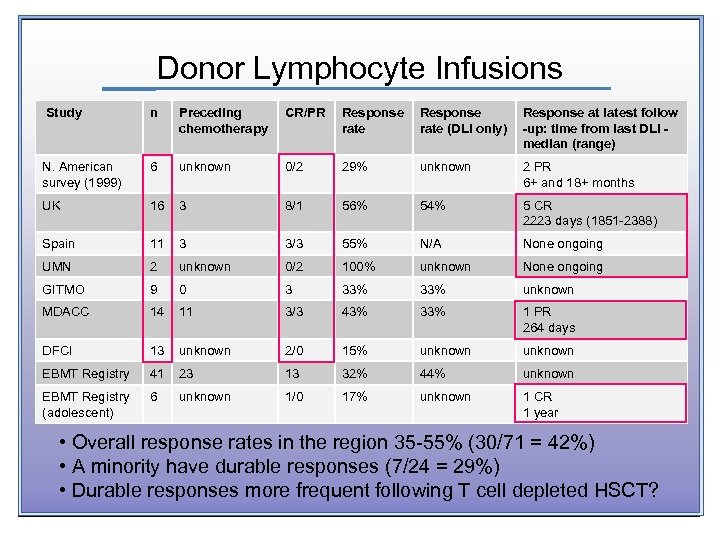 Donor Lymphocyte Infusions Study n Preceding chemotherapy CR/PR Response rate (DLI only) Response at latest follow -up: time from last DLI - median (range) N. American survey (1999) 6 unknown 0/2 29% unknown 2 PR 6+ and 18+ months UK 16 3 8/1 56% 54% 5 CR 2223 days (1851 -2388) Spain 11 3 3/3 55% N/A None ongoing UMN 2 unknown 0/2 100% unknown None ongoing GITMO 9 0 3 33% unknown MDACC 14 11 3/3 43% 33% 1 PR 264 days DFCI 13 unknown 2/0 15% unknown EBMT Registry 41 23 13 32% 44% unknown EBMT Registry (adolescent) 6 unknown 1/0 17% unknown 1 CR 1 year • Overall response rates in the region 35 -55% (30/71 = 42%) • A minority have durable responses (7/24 = 29%) • Durable responses more frequent following T cell depleted HSCT?
Donor Lymphocyte Infusions Study n Preceding chemotherapy CR/PR Response rate (DLI only) Response at latest follow -up: time from last DLI - median (range) N. American survey (1999) 6 unknown 0/2 29% unknown 2 PR 6+ and 18+ months UK 16 3 8/1 56% 54% 5 CR 2223 days (1851 -2388) Spain 11 3 3/3 55% N/A None ongoing UMN 2 unknown 0/2 100% unknown None ongoing GITMO 9 0 3 33% unknown MDACC 14 11 3/3 43% 33% 1 PR 264 days DFCI 13 unknown 2/0 15% unknown EBMT Registry 41 23 13 32% 44% unknown EBMT Registry (adolescent) 6 unknown 1/0 17% unknown 1 CR 1 year • Overall response rates in the region 35 -55% (30/71 = 42%) • A minority have durable responses (7/24 = 29%) • Durable responses more frequent following T cell depleted HSCT?
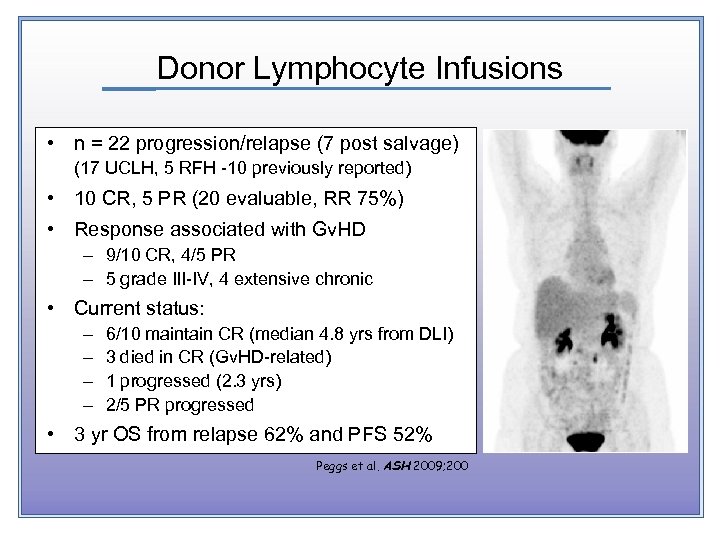 Donor Lymphocyte Infusions • n = 22 progression/relapse (7 post salvage) (17 UCLH, 5 RFH -10 previously reported) • 10 CR, 5 PR (20 evaluable, RR 75%) • Response associated with Gv. HD – 9/10 CR, 4/5 PR – 5 grade III-IV, 4 extensive chronic • Current status: – – 6/10 maintain CR (median 4. 8 yrs from DLI) 3 died in CR (Gv. HD-related) 1 progressed (2. 3 yrs) 2/5 PR progressed • 3 yr OS from relapse 62% and PFS 52% Peggs et al. ASH 2009; 200
Donor Lymphocyte Infusions • n = 22 progression/relapse (7 post salvage) (17 UCLH, 5 RFH -10 previously reported) • 10 CR, 5 PR (20 evaluable, RR 75%) • Response associated with Gv. HD – 9/10 CR, 4/5 PR – 5 grade III-IV, 4 extensive chronic • Current status: – – 6/10 maintain CR (median 4. 8 yrs from DLI) 3 died in CR (Gv. HD-related) 1 progressed (2. 3 yrs) 2/5 PR progressed • 3 yr OS from relapse 62% and PFS 52% Peggs et al. ASH 2009; 200
 Unanswered questions Largely overlap with those raised for other diseases: • Nature of the target antigens – EBV antigens? • Biomarkers of responsiveness: histology, immune profiling • Role of dose escalation, optimal dosing • Role of manipulated DLI e. g. CD 8 -depleted DLI, LMP-specific T cells, ex-vivo activation • Possible efficacy of genetically re-targeted T cells – CAR or TCR gene therapies e. g. to CD 30
Unanswered questions Largely overlap with those raised for other diseases: • Nature of the target antigens – EBV antigens? • Biomarkers of responsiveness: histology, immune profiling • Role of dose escalation, optimal dosing • Role of manipulated DLI e. g. CD 8 -depleted DLI, LMP-specific T cells, ex-vivo activation • Possible efficacy of genetically re-targeted T cells – CAR or TCR gene therapies e. g. to CD 30
 Conclusions • Relapsed HL following allogeneic HSCT is increasingly common • To date there have been no reports of systematic evaluation of salvage chemotherapy • There is no published evidence on the use of 2 nd transplants • Increasing experience with DLI confirms graft versus HL, though optimal strategies remain undefined
Conclusions • Relapsed HL following allogeneic HSCT is increasingly common • To date there have been no reports of systematic evaluation of salvage chemotherapy • There is no published evidence on the use of 2 nd transplants • Increasing experience with DLI confirms graft versus HL, though optimal strategies remain undefined
 Proposed Initiatives • Many of the issues regarding DLI overlap with those in other diseases and could be addressed across disease types. – Dose, schedule, manipulation, timing, MRD, significance of mixed chimerism • An international collaborative network would facilitate our ability to address disease-specific issues – Rapid testing of new interventions and early adoption of uniform treatment strategies.
Proposed Initiatives • Many of the issues regarding DLI overlap with those in other diseases and could be addressed across disease types. – Dose, schedule, manipulation, timing, MRD, significance of mixed chimerism • An international collaborative network would facilitate our ability to address disease-specific issues – Rapid testing of new interventions and early adoption of uniform treatment strategies.

 Treatment of Relapsed CLL after Allogeneic Stem Cell Transplantation Jose F. Leis, M. D. , Ph. D. Mayo Clinic & Nancy M. Hardy, M. D. NCI Experimental Transplantation and Immunology Branch
Treatment of Relapsed CLL after Allogeneic Stem Cell Transplantation Jose F. Leis, M. D. , Ph. D. Mayo Clinic & Nancy M. Hardy, M. D. NCI Experimental Transplantation and Immunology Branch
 Introduction • Relapse remains a major cause of treatment failure after allogeneic transplantation for CLL • PFS rates 34 -67% and relapse rates 20 -48% reported • Risk factors for relapse include bulky disease, chemotherapy refractory disease at SCT, T-cell depletion, donor selection, marrow involvement at SCT, increased number prior therapies, etc. • Both early and late relapses occur • R/O donor derived CLL in MRD SCT – Monoclonal B lymphocytosis in 13. 5 -18% of sibs
Introduction • Relapse remains a major cause of treatment failure after allogeneic transplantation for CLL • PFS rates 34 -67% and relapse rates 20 -48% reported • Risk factors for relapse include bulky disease, chemotherapy refractory disease at SCT, T-cell depletion, donor selection, marrow involvement at SCT, increased number prior therapies, etc. • Both early and late relapses occur • R/O donor derived CLL in MRD SCT – Monoclonal B lymphocytosis in 13. 5 -18% of sibs
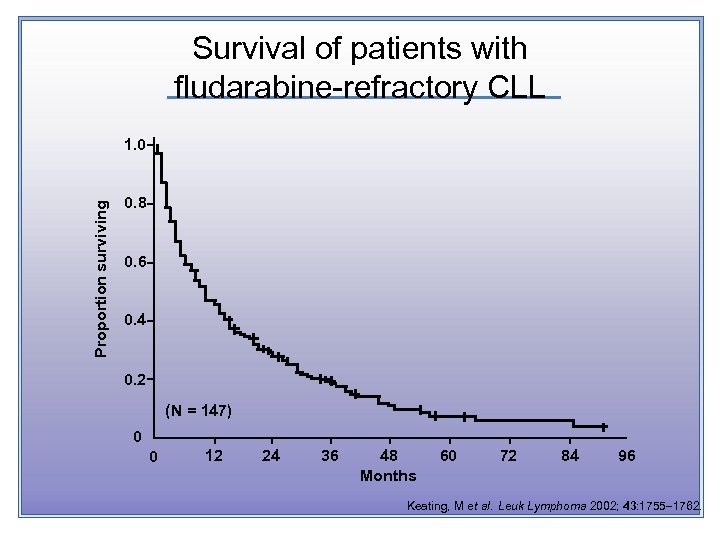 Survival of patients with fludarabine-refractory CLL Proportion surviving 1. 0 0. 8 0. 6 0. 4 0. 2 (N = 147) 0 0 12 24 36 48 Months 60 72 84 96 Keating, M et al. Leuk Lymphoma 2002; 43: 1755 1762.
Survival of patients with fludarabine-refractory CLL Proportion surviving 1. 0 0. 8 0. 6 0. 4 0. 2 (N = 147) 0 0 12 24 36 48 Months 60 72 84 96 Keating, M et al. Leuk Lymphoma 2002; 43: 1755 1762.
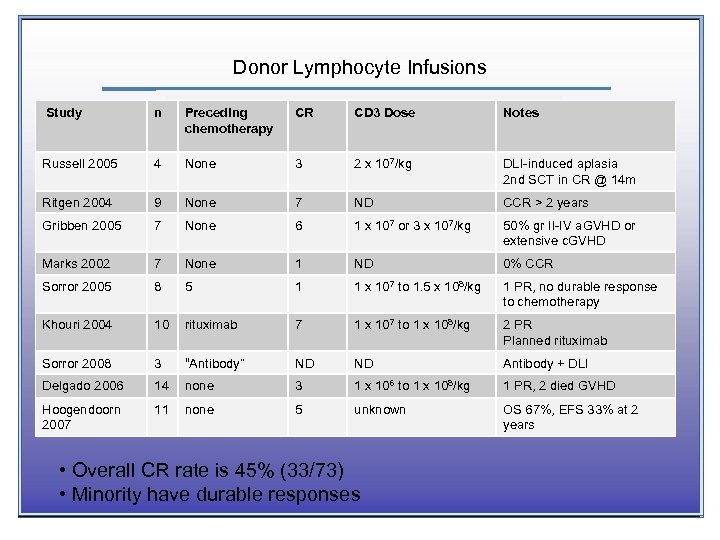 Donor Lymphocyte Infusions Study n Preceding chemotherapy CR CD 3 Dose Notes Russell 2005 4 None 3 2 x 107/kg DLI-induced aplasia 2 nd SCT in CR @ 14 m Ritgen 2004 9 None 7 ND CCR > 2 years Gribben 2005 7 None 6 1 x 107 or 3 x 107/kg 50% gr II-IV a. GVHD or extensive c. GVHD Marks 2002 7 None 1 ND 0% CCR Sorror 2005 8 5 1 1 x 107 to 1. 5 x 108/kg 1 PR, no durable response to chemotherapy Khouri 2004 10 rituximab 7 1 x 107 to 1 x 108/kg 2 PR Planned rituximab Sorror 2008 3 “Antibody” ND ND Antibody + DLI Delgado 2006 14 none 3 1 x 106 to 1 x 108/kg 1 PR, 2 died GVHD Hoogendoorn 2007 11 none 5 unknown OS 67%, EFS 33% at 2 years • Overall CR rate is 45% (33/73) • Minority have durable responses
Donor Lymphocyte Infusions Study n Preceding chemotherapy CR CD 3 Dose Notes Russell 2005 4 None 3 2 x 107/kg DLI-induced aplasia 2 nd SCT in CR @ 14 m Ritgen 2004 9 None 7 ND CCR > 2 years Gribben 2005 7 None 6 1 x 107 or 3 x 107/kg 50% gr II-IV a. GVHD or extensive c. GVHD Marks 2002 7 None 1 ND 0% CCR Sorror 2005 8 5 1 1 x 107 to 1. 5 x 108/kg 1 PR, no durable response to chemotherapy Khouri 2004 10 rituximab 7 1 x 107 to 1 x 108/kg 2 PR Planned rituximab Sorror 2008 3 “Antibody” ND ND Antibody + DLI Delgado 2006 14 none 3 1 x 106 to 1 x 108/kg 1 PR, 2 died GVHD Hoogendoorn 2007 11 none 5 unknown OS 67%, EFS 33% at 2 years • Overall CR rate is 45% (33/73) • Minority have durable responses
 Risk Factors for Failure of DLI “secondary graft-vs. -CLL resistance” • • GCLLSG CLL 3 X Used real-time PCR and/or flow-based MRD monitoring Identified 5 distinct patterns of MRD kinetics One pattern: GVL response but failed to reach complete MRD then relapsed despite extensive c. GVHD • Possible mechanisms: – – clonal evolution survival of clonogeneic cells at GVL sactuary sites (LNs) developed tolerance presence of tumor stem cell Ritgen, Leukemia, 2008
Risk Factors for Failure of DLI “secondary graft-vs. -CLL resistance” • • GCLLSG CLL 3 X Used real-time PCR and/or flow-based MRD monitoring Identified 5 distinct patterns of MRD kinetics One pattern: GVL response but failed to reach complete MRD then relapsed despite extensive c. GVHD • Possible mechanisms: – – clonal evolution survival of clonogeneic cells at GVL sactuary sites (LNs) developed tolerance presence of tumor stem cell Ritgen, Leukemia, 2008
 Augmenting DLI • Donor T-cells may not be appropriately activated to induce GVL response • Ex-vivo co-stimulation donor T-cells with magnetic beads coated with OKT 3 & anti-CD 28 • Conventional DLI --> escalating a. DLI (1 x 106 to 1 x 108/kg in 5 dose levels) 12 days later • 18 patients: 8 CR (4/7 ALL, 2/4 AML, 1/1 CLL, 1/2 NHL) – CLL patient in CR at 6 years post a. DLI – 7 a. GVHD (5 grade I-II, 2 with grade III) – 4 c. GVHD Porter, Blood 2006
Augmenting DLI • Donor T-cells may not be appropriately activated to induce GVL response • Ex-vivo co-stimulation donor T-cells with magnetic beads coated with OKT 3 & anti-CD 28 • Conventional DLI --> escalating a. DLI (1 x 106 to 1 x 108/kg in 5 dose levels) 12 days later • 18 patients: 8 CR (4/7 ALL, 2/4 AML, 1/1 CLL, 1/2 NHL) – CLL patient in CR at 6 years post a. DLI – 7 a. GVHD (5 grade I-II, 2 with grade III) – 4 c. GVHD Porter, Blood 2006
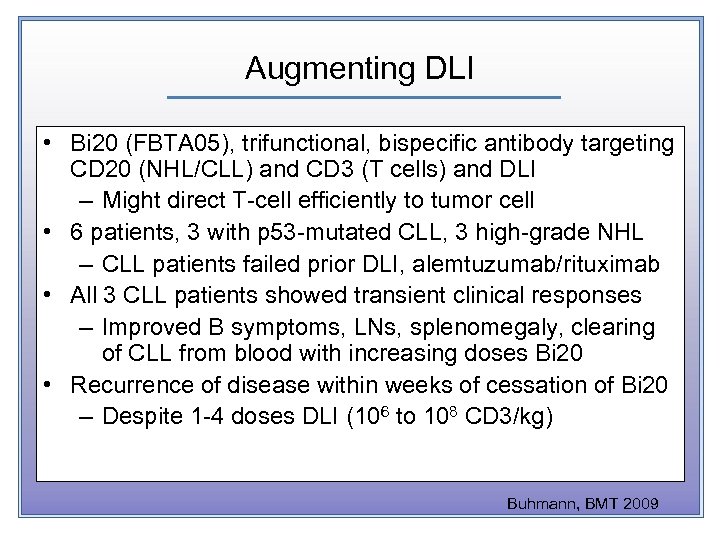 Augmenting DLI • Bi 20 (FBTA 05), trifunctional, bispecific antibody targeting CD 20 (NHL/CLL) and CD 3 (T cells) and DLI – Might direct T-cell efficiently to tumor cell • 6 patients, 3 with p 53 -mutated CLL, 3 high-grade NHL – CLL patients failed prior DLI, alemtuzumab/rituximab • All 3 CLL patients showed transient clinical responses – Improved B symptoms, LNs, splenomegaly, clearing of CLL from blood with increasing doses Bi 20 • Recurrence of disease within weeks of cessation of Bi 20 – Despite 1 -4 doses DLI (106 to 108 CD 3/kg) Buhmann, BMT 2009
Augmenting DLI • Bi 20 (FBTA 05), trifunctional, bispecific antibody targeting CD 20 (NHL/CLL) and CD 3 (T cells) and DLI – Might direct T-cell efficiently to tumor cell • 6 patients, 3 with p 53 -mutated CLL, 3 high-grade NHL – CLL patients failed prior DLI, alemtuzumab/rituximab • All 3 CLL patients showed transient clinical responses – Improved B symptoms, LNs, splenomegaly, clearing of CLL from blood with increasing doses Bi 20 • Recurrence of disease within weeks of cessation of Bi 20 – Despite 1 -4 doses DLI (106 to 108 CD 3/kg) Buhmann, BMT 2009
 Chemotherapy Approaches • Obstacles: – 33 -90% reported to be fludarabine-refractory – High-frequency of p 53 mutations (up to 50%) – Factors predict for failure to alkylating agents, purine analogues, and rituximab • Limited data available for salvage chemotherapy – Usually followed by DLI – Sorror: 5 patients (Flu+rit, CHOP, pentostatin, VCR/pred)- no durable responses – Delgado: 6 patients (2 CHOP, CMOP=rit/alem, alemtuz+chl, flu/chl) • 1 patient alive in CR at 40+ months after CHOP and 2 DLI
Chemotherapy Approaches • Obstacles: – 33 -90% reported to be fludarabine-refractory – High-frequency of p 53 mutations (up to 50%) – Factors predict for failure to alkylating agents, purine analogues, and rituximab • Limited data available for salvage chemotherapy – Usually followed by DLI – Sorror: 5 patients (Flu+rit, CHOP, pentostatin, VCR/pred)- no durable responses – Delgado: 6 patients (2 CHOP, CMOP=rit/alem, alemtuz+chl, flu/chl) • 1 patient alive in CR at 40+ months after CHOP and 2 DLI
 Agents with activity against fludarabine-refractory, p 53 mutated CLL • Flavopiridol • CDK inhibitor • 45% response rate, 42% in p 53 deleted, 72% in 11 q deleted • Major toxicity: hyperacute tumor lysis • Bendamustine • Bifunctional alkylating agent with purine-like structure • Active regardless of p 53 or ZAP-70 status • ORR > 50% • Alemtuzumab • ORR 33%, equivalent for p 53 deleted • Cytopenias and infection risk, ? Effect on GVL Byrd, Blood, 2007; Bergmann, Haematologica, 2005
Agents with activity against fludarabine-refractory, p 53 mutated CLL • Flavopiridol • CDK inhibitor • 45% response rate, 42% in p 53 deleted, 72% in 11 q deleted • Major toxicity: hyperacute tumor lysis • Bendamustine • Bifunctional alkylating agent with purine-like structure • Active regardless of p 53 or ZAP-70 status • ORR > 50% • Alemtuzumab • ORR 33%, equivalent for p 53 deleted • Cytopenias and infection risk, ? Effect on GVL Byrd, Blood, 2007; Bergmann, Haematologica, 2005
 Agents with activity against fludarabine-refractory, p 53 mutated CLL • High-dose methylprednisolone – 1 gram/m 2/day x 5 days + rituximab – ORR 78% including 5/9 p 53 -deleted with 1 cycle – Infection in 1/3, Effect on GVL? • Ofatumumab – Humanized anti-CD 20 antibody – Impressive activity in relapsed/refractory CLL (ORR 50%) – Effects on acute and chronic GVHD unknown • Lenalidomide – Immunomodulatory effects: T-cell activation via CD 28, NK cytotoxicity, increased IL-2 expression – 30% RR in 11 q or 17 p deletion CLL Bowen, Leuk Lymph, 2007; Coiffier, Blood, 2008: Ferrajoli, Blood, 2008
Agents with activity against fludarabine-refractory, p 53 mutated CLL • High-dose methylprednisolone – 1 gram/m 2/day x 5 days + rituximab – ORR 78% including 5/9 p 53 -deleted with 1 cycle – Infection in 1/3, Effect on GVL? • Ofatumumab – Humanized anti-CD 20 antibody – Impressive activity in relapsed/refractory CLL (ORR 50%) – Effects on acute and chronic GVHD unknown • Lenalidomide – Immunomodulatory effects: T-cell activation via CD 28, NK cytotoxicity, increased IL-2 expression – 30% RR in 11 q or 17 p deletion CLL Bowen, Leuk Lymph, 2007; Coiffier, Blood, 2008: Ferrajoli, Blood, 2008
 Conclusions • There is an absence of evidenced-based therapeutic options for treatment of relapsed CLL after allogeneic transplantation • Response rates to standard salvage chemotherapy regimens has been disappointing • Reports on the use of DLI are limited and response rates have been highly variable (0% to 60% CCR) and questions regarding the durability of response raised • Methods to augment DLI and novel agents active against fludarabine-refractory CLL are promising
Conclusions • There is an absence of evidenced-based therapeutic options for treatment of relapsed CLL after allogeneic transplantation • Response rates to standard salvage chemotherapy regimens has been disappointing • Reports on the use of DLI are limited and response rates have been highly variable (0% to 60% CCR) and questions regarding the durability of response raised • Methods to augment DLI and novel agents active against fludarabine-refractory CLL are promising
 Proposed Initiatives • Development of a national/international consortium to evaluated the optimal use of DLI in treatment of relapsed CLL • Initiation of multi-center clinical trails to evaluate efficacy of promising novel agents for treatment of relapsed CLL
Proposed Initiatives • Development of a national/international consortium to evaluated the optimal use of DLI in treatment of relapsed CLL • Initiation of multi-center clinical trails to evaluate efficacy of promising novel agents for treatment of relapsed CLL

 Disease specific Treatment of Relapse after Allogeneic Hematopoietic Cell Transplantation Multiple Myeloma NCI Workshop 1/2 -11. 2009 Nicolaus Kröger, Ted Alyea
Disease specific Treatment of Relapse after Allogeneic Hematopoietic Cell Transplantation Multiple Myeloma NCI Workshop 1/2 -11. 2009 Nicolaus Kröger, Ted Alyea
 Specific feature of allografting in Multiple Myeloma 1. Only about 50% will achieve complete remission 2. Despite CR, relapse rate is higher than in other diseases and is about 50% at 5 years 3. For patients without CR after allografting the progression-rate is even higher (>80%) 4. Extramedullary relapse seems to be increased after allogeneic SCT
Specific feature of allografting in Multiple Myeloma 1. Only about 50% will achieve complete remission 2. Despite CR, relapse rate is higher than in other diseases and is about 50% at 5 years 3. For patients without CR after allografting the progression-rate is even higher (>80%) 4. Extramedullary relapse seems to be increased after allogeneic SCT
 Donor lymphocyte infusion for relapse After standard conditioning • Studies for relapsed patients: ORR: 40 -67% and CR: 19 -30% • Acute Gv. HD II-IV: 52 -56% and c. Gv. HD: 26 -44% • Strong correlation between response and occurrence of Gv. HD • No clear correlation between CD 3 cell dose and response (Lokhorst et al 1997, Verdonck et al 1996, Salama 2000, Ayuk et al. , 2004, Tricot 1996, Bertz et al. , 1997)
Donor lymphocyte infusion for relapse After standard conditioning • Studies for relapsed patients: ORR: 40 -67% and CR: 19 -30% • Acute Gv. HD II-IV: 52 -56% and c. Gv. HD: 26 -44% • Strong correlation between response and occurrence of Gv. HD • No clear correlation between CD 3 cell dose and response (Lokhorst et al 1997, Verdonck et al 1996, Salama 2000, Ayuk et al. , 2004, Tricot 1996, Bertz et al. , 1997)
 Donor lymphocyte infusion for relapse After RIC Overall response rate: 38%: PR: 19% and CR 19% Acute Gv. HD II-IV: 38% and c Gv. HD: 42% (extensive c. Gv. HD: 10%) Response according Gv. HD: acute Gv. HD : no: 21% ORR yes: 67% ORR chronic Gv. HD: no: 19% ORR yes: 63% ORR van de Donk et al. , 2006
Donor lymphocyte infusion for relapse After RIC Overall response rate: 38%: PR: 19% and CR 19% Acute Gv. HD II-IV: 38% and c Gv. HD: 42% (extensive c. Gv. HD: 10%) Response according Gv. HD: acute Gv. HD : no: 21% ORR yes: 67% ORR chronic Gv. HD: no: 19% ORR yes: 63% ORR van de Donk et al. , 2006
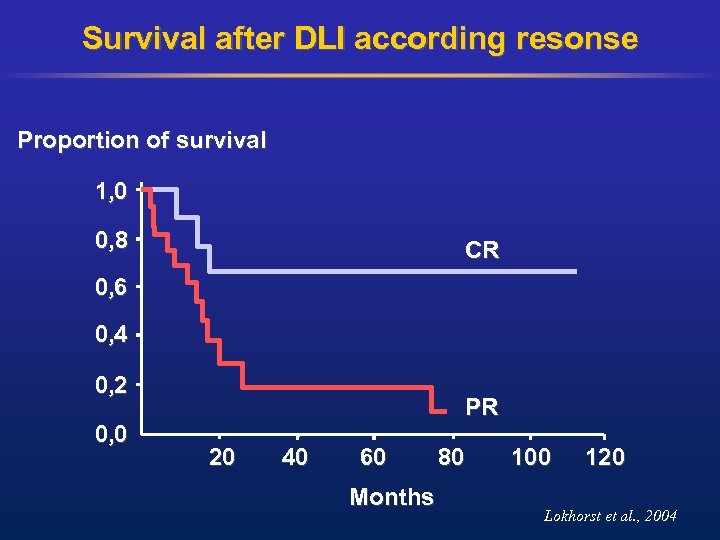 Survival after DLI according resonse Proportion of survival 1, 0 0, 8 CR 0, 6 0, 4 0, 2 0, 0 PR 20 40 60 Months 80 100 120 Lokhorst et al. , 2004
Survival after DLI according resonse Proportion of survival 1, 0 0, 8 CR 0, 6 0, 4 0, 2 0, 0 PR 20 40 60 Months 80 100 120 Lokhorst et al. , 2004
 Novel agents as salvage post allogeneic SCT Rationales Bortezomib: 1. highly active against myeloma cells 2. in animal model: reduced Gv. HD but retain Graft versus leukemia effect (Sun et al. , PNAS 2004) and lead to a decreased T-helper 1 response among allreactive T-lymphocytes (Blanco et al. , Blood 2006) Lenalidomide and Thalidomide: 1. Activate T-cells and NK-cells which might augment the graft versus myeloma effect (Lioznov et al. , BMT 2009) 2. Low dose thalidomide more immunosuppressive properties
Novel agents as salvage post allogeneic SCT Rationales Bortezomib: 1. highly active against myeloma cells 2. in animal model: reduced Gv. HD but retain Graft versus leukemia effect (Sun et al. , PNAS 2004) and lead to a decreased T-helper 1 response among allreactive T-lymphocytes (Blanco et al. , Blood 2006) Lenalidomide and Thalidomide: 1. Activate T-cells and NK-cells which might augment the graft versus myeloma effect (Lioznov et al. , BMT 2009) 2. Low dose thalidomide more immunosuppressive properties
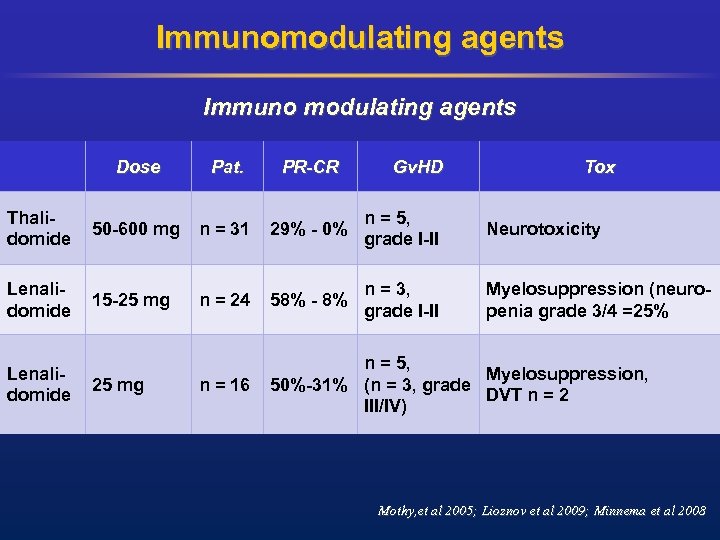 Immunomodulating agents Immuno modulating agents Dose Pat. PR-CR Thalidomide 50 -600 mg n = 31 29% - 0% n = 5, grade I-II Neurotoxicity Lenalidomide 15 -25 mg n = 24 58% - 8% n = 3, grade I-II Myelosuppression (neuropenia grade 3/4 =25% n = 16 n = 5, Myelosuppression, 50%-31% (n = 3, grade DVT n = 2 III/IV) Lenalidomide 25 mg Gv. HD Tox Mothy, et al 2005; Lioznov et al 2009; Minnema et al 2008
Immunomodulating agents Immuno modulating agents Dose Pat. PR-CR Thalidomide 50 -600 mg n = 31 29% - 0% n = 5, grade I-II Neurotoxicity Lenalidomide 15 -25 mg n = 24 58% - 8% n = 3, grade I-II Myelosuppression (neuropenia grade 3/4 =25% n = 16 n = 5, Myelosuppression, 50%-31% (n = 3, grade DVT n = 2 III/IV) Lenalidomide 25 mg Gv. HD Tox Mothy, et al 2005; Lioznov et al 2009; Minnema et al 2008
 Proteasome Inhibitor Dose Bortezomib 1 -1. 3 mg/m 2 day 1, 4, 8 + 11 Pat. PR - CR n = 37 34% - 19% Gv. HD none Tox Neurotoxicity, grade 1 -2: 85% El-Cheikh, 2008
Proteasome Inhibitor Dose Bortezomib 1 -1. 3 mg/m 2 day 1, 4, 8 + 11 Pat. PR - CR n = 37 34% - 19% Gv. HD none Tox Neurotoxicity, grade 1 -2: 85% El-Cheikh, 2008
 Donor lymphocyte infusion in combination with novel agents Treatment plan Thalidomide 100 mg 14 days DLI: 1 x 106 CD 3+/kg (MUD) or: 5 x 106 CD 3+/kg (related) No response: escalating Thalidomide (200 → 300 → 400 mg) And/or: further DLI Kröger et al. , Blood 2004
Donor lymphocyte infusion in combination with novel agents Treatment plan Thalidomide 100 mg 14 days DLI: 1 x 106 CD 3+/kg (MUD) or: 5 x 106 CD 3+/kg (related) No response: escalating Thalidomide (200 → 300 → 400 mg) And/or: further DLI Kröger et al. , Blood 2004
 Low dose thalidomide (100 mg) and escalating DLI Response rate CR: 6/18 33% PR: 4/18 22% MR: 2/18 12% SD/NC: 5/18 28% PD: 1/18 5% 67% (ORR) Med. time to response: 108 days (36 – 266)
Low dose thalidomide (100 mg) and escalating DLI Response rate CR: 6/18 33% PR: 4/18 22% MR: 2/18 12% SD/NC: 5/18 28% PD: 1/18 5% 67% (ORR) Med. time to response: 108 days (36 – 266)
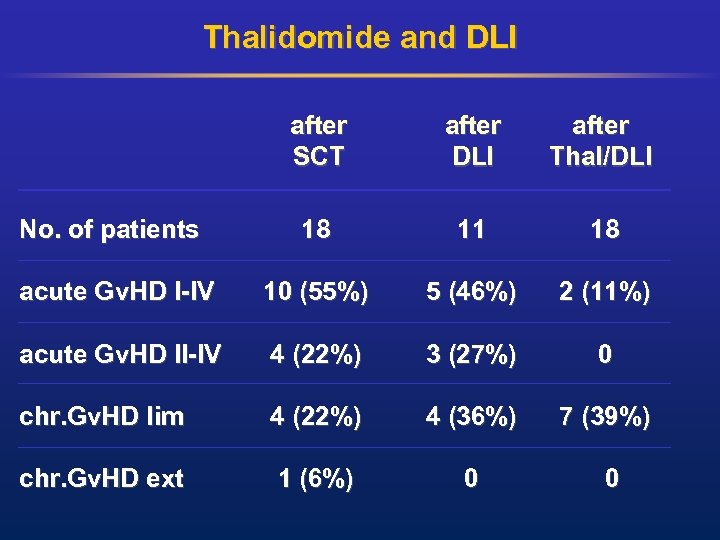 Thalidomide and DLI after SCT after DLI after Thal/DLI No. of patients 18 11 18 acute Gv. HD I-IV 10 (55%) 5 (46%) 2 (11%) acute Gv. HD II-IV 4 (22%) 3 (27%) 0 chr. Gv. HD lim 4 (22%) 4 (36%) 7 (39%) chr. Gv. HD ext 1 (6%) 0 0
Thalidomide and DLI after SCT after DLI after Thal/DLI No. of patients 18 11 18 acute Gv. HD I-IV 10 (55%) 5 (46%) 2 (11%) acute Gv. HD II-IV 4 (22%) 3 (27%) 0 chr. Gv. HD lim 4 (22%) 4 (36%) 7 (39%) chr. Gv. HD ext 1 (6%) 0 0
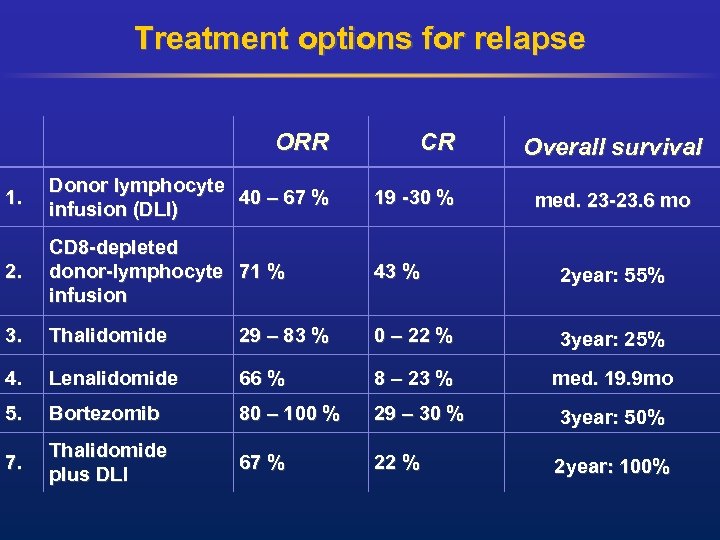 Treatment options for relapse ORR CR 1. Donor lymphocyte 40 – 67 % infusion (DLI) 19 -30 % 2. CD 8 -depleted donor-lymphocyte 71 % infusion 43 % 2 year: 55% 3. Thalidomide 29 – 83 % 0 – 22 % 3 year: 25% 4. Lenalidomide 66 % 8 – 23 % med. 19. 9 mo 5. Bortezomib 80 – 100 % 29 – 30 % 3 year: 50% 7. Thalidomide plus DLI 67 % 22 % 2 year: 100% Overall survival med. 23 -23. 6 mo
Treatment options for relapse ORR CR 1. Donor lymphocyte 40 – 67 % infusion (DLI) 19 -30 % 2. CD 8 -depleted donor-lymphocyte 71 % infusion 43 % 2 year: 55% 3. Thalidomide 29 – 83 % 0 – 22 % 3 year: 25% 4. Lenalidomide 66 % 8 – 23 % med. 19. 9 mo 5. Bortezomib 80 – 100 % 29 – 30 % 3 year: 50% 7. Thalidomide plus DLI 67 % 22 % 2 year: 100% Overall survival med. 23 -23. 6 mo
 Novel approaches and future research 1. Manipulation to reduce Gv. HD after DLI - CD 8 depleted T-cells - CD 4 enriched T-cells - Depleting alloreactive T-cells - Alloreactive NK-cells - Tumor specific T-cells (anti-idiotype, Cancer-tests antigen, HAG - Antibody mediated cytotoxicity (BCMA) - Donor vaccination (e. g. idiotype) 2. Enhance cell mediated cytotoxicity - DLI in combination with novel agents (thalidomide, Bortezomib, Lenalidomide) 3. Investigate novel drug combinations, e. g. Lenalidomide plus Bortezomib etc
Novel approaches and future research 1. Manipulation to reduce Gv. HD after DLI - CD 8 depleted T-cells - CD 4 enriched T-cells - Depleting alloreactive T-cells - Alloreactive NK-cells - Tumor specific T-cells (anti-idiotype, Cancer-tests antigen, HAG - Antibody mediated cytotoxicity (BCMA) - Donor vaccination (e. g. idiotype) 2. Enhance cell mediated cytotoxicity - DLI in combination with novel agents (thalidomide, Bortezomib, Lenalidomide) 3. Investigate novel drug combinations, e. g. Lenalidomide plus Bortezomib etc
 Proposed Initiatives for the subcommittee Treatment of Relapse after allo SCT 1. Dose-finding studies for novel agents after allogeneic SCT 2. (Lenalidomide, bortezomib, hypomethylating agents) 3. 2. Clinical trial investigating early (MRD based) vs late (clinical based 4. therapeutic intervention (DLI and/or novel agents) to treat relapse 5. after allogeneic SCT 6. 3. Investigate novel agents in combination with adoptive 7. immunotherapy (T-cells or NK-cells) as treatment for relapse 8. after allogeneic SCT 9. 4. Investigate in a prospective trial the value of a second allogeneic 10. SCT in those patient who relapsed to a RIC allograft
Proposed Initiatives for the subcommittee Treatment of Relapse after allo SCT 1. Dose-finding studies for novel agents after allogeneic SCT 2. (Lenalidomide, bortezomib, hypomethylating agents) 3. 2. Clinical trial investigating early (MRD based) vs late (clinical based 4. therapeutic intervention (DLI and/or novel agents) to treat relapse 5. after allogeneic SCT 6. 3. Investigate novel agents in combination with adoptive 7. immunotherapy (T-cells or NK-cells) as treatment for relapse 8. after allogeneic SCT 9. 4. Investigate in a prospective trial the value of a second allogeneic 10. SCT in those patient who relapsed to a RIC allograft

 Future Directions and Proposals • Better data collection and reporting. • Sample repository for relapse. • Identify target cells and/or antigens for GVT induction. • Define prognostic factors and use to develop and test appropriate strategies – Clinical – Biomarkers for response? An embarrassing lack of data
Future Directions and Proposals • Better data collection and reporting. • Sample repository for relapse. • Identify target cells and/or antigens for GVT induction. • Define prognostic factors and use to develop and test appropriate strategies – Clinical – Biomarkers for response? An embarrassing lack of data
 Future Directions and Proposals • Study novel agents – azacitidine, decitabine, lenalidomide, bortezomib, sorafenib, etc. . • Combination drug, antibody and/or cellular therapies. • Vaccination strategies of patient/donor – m. Hag, tumor-specific antigens, APCs • Modification of DLI – Tumor specific DLI through selection, genetic modification, etc. – Activation – Subset selection or depletion • Readdress role of second SCT, particularly after RIC SCT.
Future Directions and Proposals • Study novel agents – azacitidine, decitabine, lenalidomide, bortezomib, sorafenib, etc. . • Combination drug, antibody and/or cellular therapies. • Vaccination strategies of patient/donor – m. Hag, tumor-specific antigens, APCs • Modification of DLI – Tumor specific DLI through selection, genetic modification, etc. – Activation – Subset selection or depletion • Readdress role of second SCT, particularly after RIC SCT.
 Obstacles • Lack of comprehensive databases capturing relapse information. – Registry studies helpful “first step” • Lack of large multicenter prospective trials to define best therapies. – Difficulty setting up national and international collaborations. • Difficulty engaging sponsors, payors, regulators, physicians, patients in very high risk, novel therapies.
Obstacles • Lack of comprehensive databases capturing relapse information. – Registry studies helpful “first step” • Lack of large multicenter prospective trials to define best therapies. – Difficulty setting up national and international collaborations. • Difficulty engaging sponsors, payors, regulators, physicians, patients in very high risk, novel therapies.
 Moving Forward • International multicenter network to rapidly and definitively test and disseminate new treatment approaches for relapse. – Begin with the First International Workshop on the Biology, Prevention and Treatment of Relapse after Allogeneic SCT. – Maintain interactions and momentum through workshops, meetings, consortium-building – Develop trials and seek funding. • Consider trial design carefully for maximum and rapid impact • . Multiple small pilots vs randomized phase II or III?
Moving Forward • International multicenter network to rapidly and definitively test and disseminate new treatment approaches for relapse. – Begin with the First International Workshop on the Biology, Prevention and Treatment of Relapse after Allogeneic SCT. – Maintain interactions and momentum through workshops, meetings, consortium-building – Develop trials and seek funding. • Consider trial design carefully for maximum and rapid impact • . Multiple small pilots vs randomized phase II or III?
 PROUD GLOOMY
PROUD GLOOMY
 Disease-Specific Treatment of Relapse after Allogeneic Transplantation • Fred Falkenburg • Karl Peggs • Joseph Antin • David Porter • Marcos de Lima • Jose Leis • Eli Estey • Nancy Hardy • John Levine • Nicolaus Kröger • Jacob Rowe • Edwin Alyea • Alan Wayne • David Maloney • Koen van Besien
Disease-Specific Treatment of Relapse after Allogeneic Transplantation • Fred Falkenburg • Karl Peggs • Joseph Antin • David Porter • Marcos de Lima • Jose Leis • Eli Estey • Nancy Hardy • John Levine • Nicolaus Kröger • Jacob Rowe • Edwin Alyea • Alan Wayne • David Maloney • Koen van Besien


