 DISCONTINUATION OR NO DISCONTIUNATION: COMPARISON OF SINGLE UNIT HIV ANTIGEN TESTING VS. POOLED NAT TESTING Gerald Schochetman, Ph. D. Abbott Laboratories Diagnostics Division BPAC Meeting September 14, 2000
DISCONTINUATION OR NO DISCONTIUNATION: COMPARISON OF SINGLE UNIT HIV ANTIGEN TESTING VS. POOLED NAT TESTING Gerald Schochetman, Ph. D. Abbott Laboratories Diagnostics Division BPAC Meeting September 14, 2000
 OBJECTIVE To develop more sensitive HIV antigen assays Short term: Comparable sensitivity to pooled NAT testing Long term: Sensitivity equivalent to single unit NAT testing
OBJECTIVE To develop more sensitive HIV antigen assays Short term: Comparable sensitivity to pooled NAT testing Long term: Sensitivity equivalent to single unit NAT testing
 SENSITIVITY OF RESEARCH PRISM HIV Ag VS. CURRENT HIV ANTIGEN ASSAY p 24 ANTIGEN pg/ml (est. ) Estimated RNA Copies/ml @ Dilution of copies/ml 1: 96 1: 1200 4 4 Research Prism. HIV Ag 1 -2 1 x 10 -2 x 10 104 -208 8 -16 HIVAG-1 MC 7 -10 7 x 104 -1 x 105 729 -1041 58 -83 1 pg = 1 x 104 copies/ml
SENSITIVITY OF RESEARCH PRISM HIV Ag VS. CURRENT HIV ANTIGEN ASSAY p 24 ANTIGEN pg/ml (est. ) Estimated RNA Copies/ml @ Dilution of copies/ml 1: 96 1: 1200 4 4 Research Prism. HIV Ag 1 -2 1 x 10 -2 x 10 104 -208 8 -16 HIVAG-1 MC 7 -10 7 x 104 -1 x 105 729 -1041 58 -83 1 pg = 1 x 104 copies/ml
 Comparison Between Sensitive HIV Antigen Testing and NAT • Sensitivity of antigen assay is ~1. 0 pg or ~10, 000 copies of viral RNA • Even at a claimed 50 copies/ml sensitivity for NAT: - a sample must have at least 4800 copies of viral RNA/ml to be detected in a pool of 96, or - 60, 000 copies of viral RNA/ml in a pool of 1200
Comparison Between Sensitive HIV Antigen Testing and NAT • Sensitivity of antigen assay is ~1. 0 pg or ~10, 000 copies of viral RNA • Even at a claimed 50 copies/ml sensitivity for NAT: - a sample must have at least 4800 copies of viral RNA/ml to be detected in a pool of 96, or - 60, 000 copies of viral RNA/ml in a pool of 1200
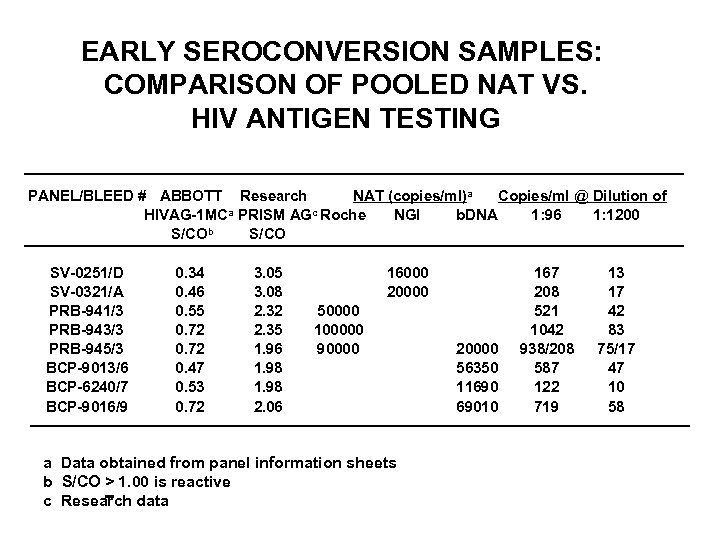 EARLY SEROCONVERSION SAMPLES: COMPARISON OF POOLED NAT VS. HIV ANTIGEN TESTING PANEL/BLEED # ABBOTT Research NAT (copies/ml)a Copies/ml @ Dilution of a PRISM AGc Roche HIVAG-1 MC NGI b. DNA 1: 96 1: 1200 b S/CO SV-0251/D SV-0321/A PRB-941/3 PRB-943/3 PRB-945/3 BCP-9013/6 BCP-6240/7 BCP-9016/9 0. 34 0. 46 0. 55 0. 72 0. 47 0. 53 0. 72 3. 05 3. 08 2. 32 2. 35 1. 96 1. 98 2. 06 16000 20000 50000 100000 90000 a Data obtained from panel information sheets b S/CO > 1. 00 is reactive c Research data 20000 56350 11690 69010 167 208 521 1042 938/208 587 122 719 13 17 42 83 75/17 47 10 58
EARLY SEROCONVERSION SAMPLES: COMPARISON OF POOLED NAT VS. HIV ANTIGEN TESTING PANEL/BLEED # ABBOTT Research NAT (copies/ml)a Copies/ml @ Dilution of a PRISM AGc Roche HIVAG-1 MC NGI b. DNA 1: 96 1: 1200 b S/CO SV-0251/D SV-0321/A PRB-941/3 PRB-943/3 PRB-945/3 BCP-9013/6 BCP-6240/7 BCP-9016/9 0. 34 0. 46 0. 55 0. 72 0. 47 0. 53 0. 72 3. 05 3. 08 2. 32 2. 35 1. 96 1. 98 2. 06 16000 20000 50000 100000 90000 a Data obtained from panel information sheets b S/CO > 1. 00 is reactive c Research data 20000 56350 11690 69010 167 208 521 1042 938/208 587 122 719 13 17 42 83 75/17 47 10 58
 Advantages of Individual HIV Antigen Testing vs Pooled NAT Testing • Fully automated system for antigen testing • Rapid test results • Process controls for enhanced GMP compliance • No sample preparation/contamination issues • Ability to confirm using neutralization test • Simplicity for implementation No pools to dilute sensitivity No dissection of pools No shipping of specimens
Advantages of Individual HIV Antigen Testing vs Pooled NAT Testing • Fully automated system for antigen testing • Rapid test results • Process controls for enhanced GMP compliance • No sample preparation/contamination issues • Ability to confirm using neutralization test • Simplicity for implementation No pools to dilute sensitivity No dissection of pools No shipping of specimens
 CONCLUSIONS • Gap between individual antigen testing using a sensitive assay for HIV antigen and NAT of pooled samples may not be significant • As opposed to discontinuation: Manufacturers should be encouraged to develop ultra-sensitive HIV antigen assays with sensitivities equal to or greater than single unit NAT
CONCLUSIONS • Gap between individual antigen testing using a sensitive assay for HIV antigen and NAT of pooled samples may not be significant • As opposed to discontinuation: Manufacturers should be encouraged to develop ultra-sensitive HIV antigen assays with sensitivities equal to or greater than single unit NAT