57be29d5accd179464aa12f5ac9a0411.ppt
- Количество слайдов: 39
 Director’s Report to the National Advisory Council on Drug Abuse May 16, 2007
Director’s Report to the National Advisory Council on Drug Abuse May 16, 2007
 Director’s Report to the National Advisory Council on Drug Abuse Budget Update What’s New @ NIH? Recent NIDA Activities
Director’s Report to the National Advisory Council on Drug Abuse Budget Update What’s New @ NIH? Recent NIDA Activities
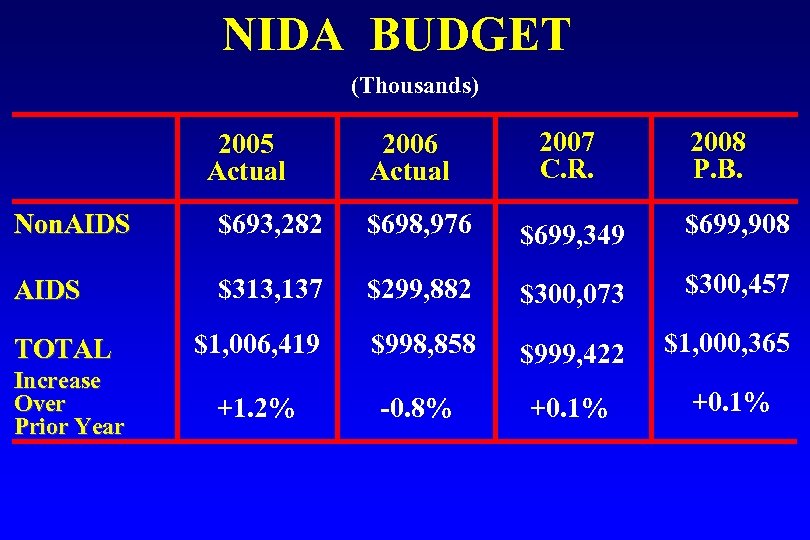 NIDA BUDGET (Thousands) 2005 Actual 2006 Actual 2007 C. R. 2008 P. B. Non. AIDS $693, 282 $698, 976 $699, 349 $699, 908 AIDS $313, 137 $299, 882 $300, 073 $300, 457 TOTAL $1, 006, 419 $998, 858 $1, 000, 365 Increase Over Prior Year $999, 422 +1. 2% -0. 8% +0. 1%
NIDA BUDGET (Thousands) 2005 Actual 2006 Actual 2007 C. R. 2008 P. B. Non. AIDS $693, 282 $698, 976 $699, 349 $699, 908 AIDS $313, 137 $299, 882 $300, 073 $300, 457 TOTAL $1, 006, 419 $998, 858 $1, 000, 365 Increase Over Prior Year $999, 422 +1. 2% -0. 8% +0. 1%
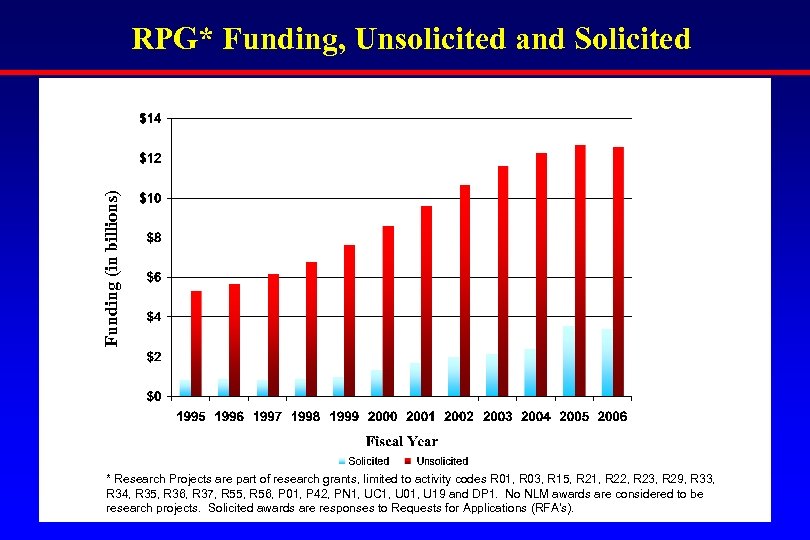 Funding (in billions) RPG* Funding, Unsolicited and Solicited * Research Projects are part of research grants, limited to activity codes R 01, R 03, R 15, R 21, R 22, R 23, R 29, R 33, R 34, R 35, R 36, R 37, R 55, R 56, P 01, P 42, PN 1, UC 1, U 01, U 19 and DP 1. No NLM awards are considered to be research projects. Solicited awards are responses to Requests for Applications (RFA’s).
Funding (in billions) RPG* Funding, Unsolicited and Solicited * Research Projects are part of research grants, limited to activity codes R 01, R 03, R 15, R 21, R 22, R 23, R 29, R 33, R 34, R 35, R 36, R 37, R 55, R 56, P 01, P 42, PN 1, UC 1, U 01, U 19 and DP 1. No NLM awards are considered to be research projects. Solicited awards are responses to Requests for Applications (RFA’s).
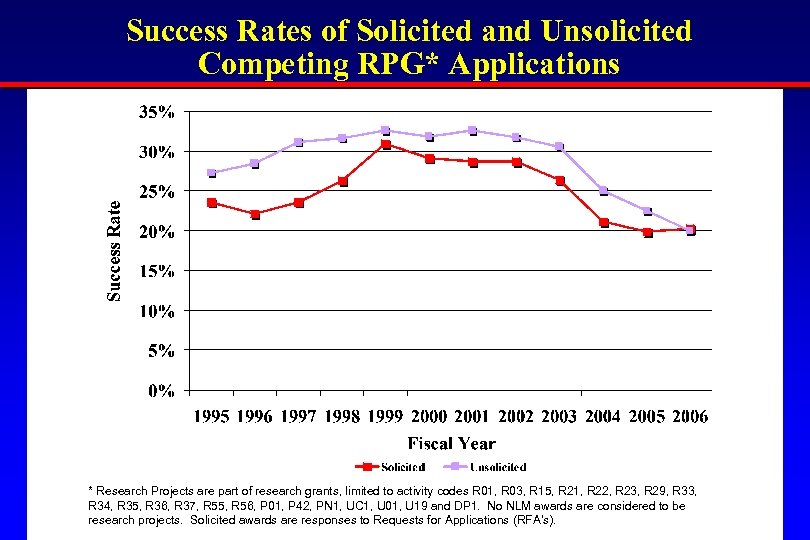 Success Rates of Solicited and Unsolicited Competing RPG* Applications * Research Projects are part of research grants, limited to activity codes R 01, R 03, R 15, R 21, R 22, R 23, R 29, R 33, R 34, R 35, R 36, R 37, R 55, R 56, P 01, P 42, PN 1, UC 1, U 01, U 19 and DP 1. No NLM awards are considered to be research projects. Solicited awards are responses to Requests for Applications (RFA’s).
Success Rates of Solicited and Unsolicited Competing RPG* Applications * Research Projects are part of research grants, limited to activity codes R 01, R 03, R 15, R 21, R 22, R 23, R 29, R 33, R 34, R 35, R 36, R 37, R 55, R 56, P 01, P 42, PN 1, UC 1, U 01, U 19 and DP 1. No NLM awards are considered to be research projects. Solicited awards are responses to Requests for Applications (RFA’s).
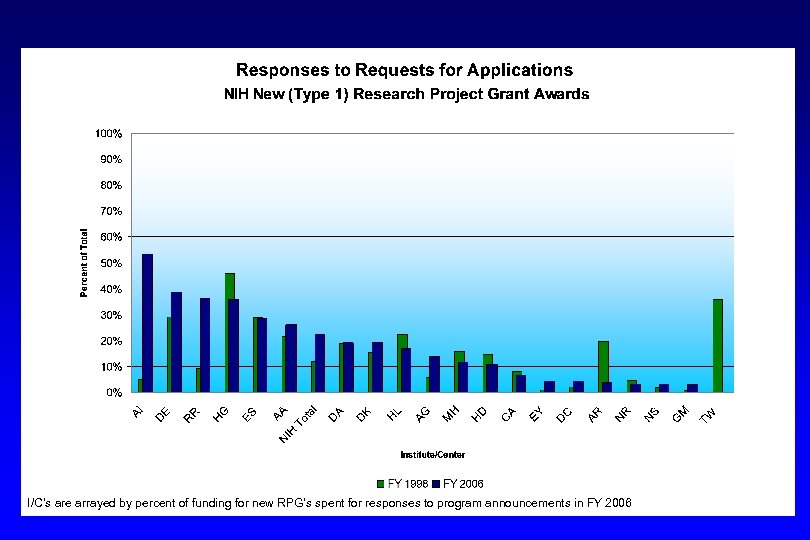 I/C’s are arrayed by percent of funding for new RPG’s spent for responses to program announcements in FY 2006
I/C’s are arrayed by percent of funding for new RPG’s spent for responses to program announcements in FY 2006
 NIDA Dashboard ? Medicinal Chemists -- Recruitment -- Training New PIs Collaborations ? ?
NIDA Dashboard ? Medicinal Chemists -- Recruitment -- Training New PIs Collaborations ? ?
 FY 2008 Budget Hearings • March 1, 2007 -- House Appropriations Subcommittee on Labor, HHS and Education Hearing on FY 2008 President’s Budget Request for SAMHSA, NIDA, NIAAA and NIMH • March 6, 2007 -- House Appropriations Subcommittee on Labor, HHS and Education Hearing on FY 2008 President’s Budget Request for NIH • March 19, 2007 – Senate Appropriations Subcommittee on Labor, HHS and Education Hearing on FY 2008 President’s Budget Request for NIH • March 26, 2007 – Senate Appropriations Subcommittee on Labor, HHS, and Education FY 2008 Budget for Mind, Brain and Behavioral Research
FY 2008 Budget Hearings • March 1, 2007 -- House Appropriations Subcommittee on Labor, HHS and Education Hearing on FY 2008 President’s Budget Request for SAMHSA, NIDA, NIAAA and NIMH • March 6, 2007 -- House Appropriations Subcommittee on Labor, HHS and Education Hearing on FY 2008 President’s Budget Request for NIH • March 19, 2007 – Senate Appropriations Subcommittee on Labor, HHS and Education Hearing on FY 2008 President’s Budget Request for NIH • March 26, 2007 – Senate Appropriations Subcommittee on Labor, HHS, and Education FY 2008 Budget for Mind, Brain and Behavioral Research
 IC Directors’ Retreat – May 17 -18, 2007 Roadmap II Top 5 Topics Strategic Planning Groups 1) Microbiome 2) Protein Capture/Proteome Tools 3) Phenotyping Services, Tools 4) Inflammation 5) Epigenetics a) Training/Careers b) Health Disparities c) Science of Science d) Pharmacogenomics
IC Directors’ Retreat – May 17 -18, 2007 Roadmap II Top 5 Topics Strategic Planning Groups 1) Microbiome 2) Protein Capture/Proteome Tools 3) Phenotyping Services, Tools 4) Inflammation 5) Epigenetics a) Training/Careers b) Health Disparities c) Science of Science d) Pharmacogenomics
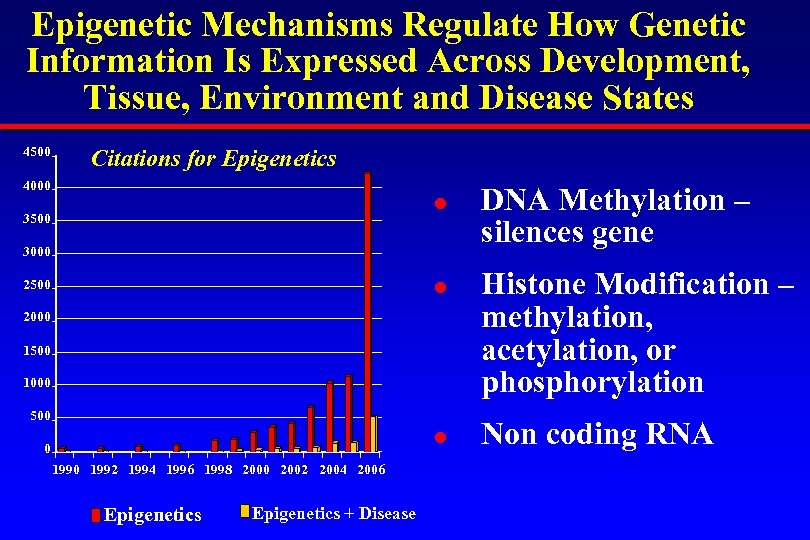 Epigenetic Mechanisms Regulate How Genetic Information Is Expressed Across Development, Tissue, Environment and Disease States 4500 Citations for Epigenetics 4000 l 3500 3000 l 2500 2000 1500 1000 500 l 0 1992 1994 1996 1998 2000 2002 2004 2006 Epigenetics + Disease DNA Methylation – silences gene Histone Modification – methylation, acetylation, or phosphorylation Non coding RNA
Epigenetic Mechanisms Regulate How Genetic Information Is Expressed Across Development, Tissue, Environment and Disease States 4500 Citations for Epigenetics 4000 l 3500 3000 l 2500 2000 1500 1000 500 l 0 1992 1994 1996 1998 2000 2002 2004 2006 Epigenetics + Disease DNA Methylation – silences gene Histone Modification – methylation, acetylation, or phosphorylation Non coding RNA
 Epigenetic Marks Are Altered in Common Brain Disorders Including Addiction
Epigenetic Marks Are Altered in Common Brain Disorders Including Addiction
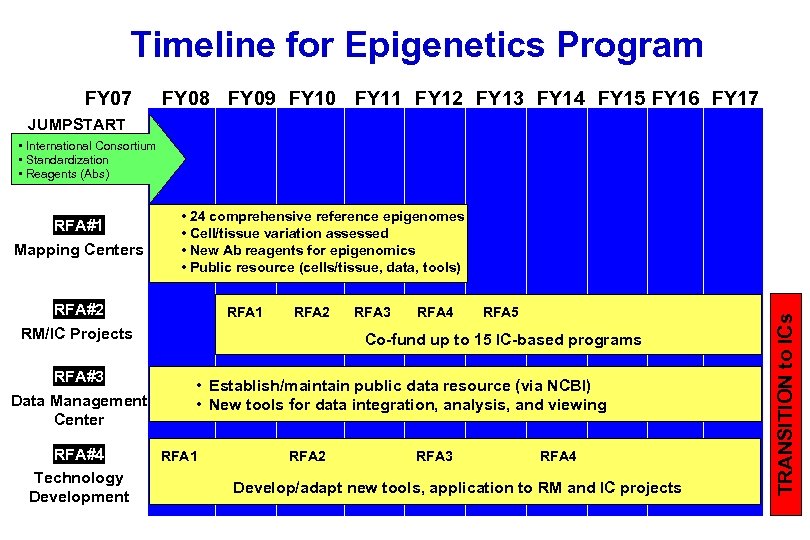 Timeline for Epigenetics Program FY 07 FY 08 FY 09 FY 10 FY 11 FY 12 FY 13 FY 14 FY 15 FY 16 FY 17 JUMPSTART • International Consortium • Standardization • Reagents (Abs) • 24 comprehensive reference epigenomes • Cell/tissue variation assessed • New Ab reagents for epigenomics • Public resource (cells/tissue, data, tools) RFA#2 RM/IC Projects RFA#3 Data Management Center RFA#4 Technology Development RFA 1 RFA 2 RFA 3 RFA 4 RFA 5 Co-fund up to 15 IC-based programs • Establish/maintain public data resource (via NCBI) • New tools for data integration, analysis, and viewing RFA 1 RFA 2 RFA 3 RFA 4 Develop/adapt new tools, application to RM and IC projects TRANSITION to ICs RFA#1 Mapping Centers
Timeline for Epigenetics Program FY 07 FY 08 FY 09 FY 10 FY 11 FY 12 FY 13 FY 14 FY 15 FY 16 FY 17 JUMPSTART • International Consortium • Standardization • Reagents (Abs) • 24 comprehensive reference epigenomes • Cell/tissue variation assessed • New Ab reagents for epigenomics • Public resource (cells/tissue, data, tools) RFA#2 RM/IC Projects RFA#3 Data Management Center RFA#4 Technology Development RFA 1 RFA 2 RFA 3 RFA 4 RFA 5 Co-fund up to 15 IC-based programs • Establish/maintain public data resource (via NCBI) • New tools for data integration, analysis, and viewing RFA 1 RFA 2 RFA 3 RFA 4 Develop/adapt new tools, application to RM and IC projects TRANSITION to ICs RFA#1 Mapping Centers
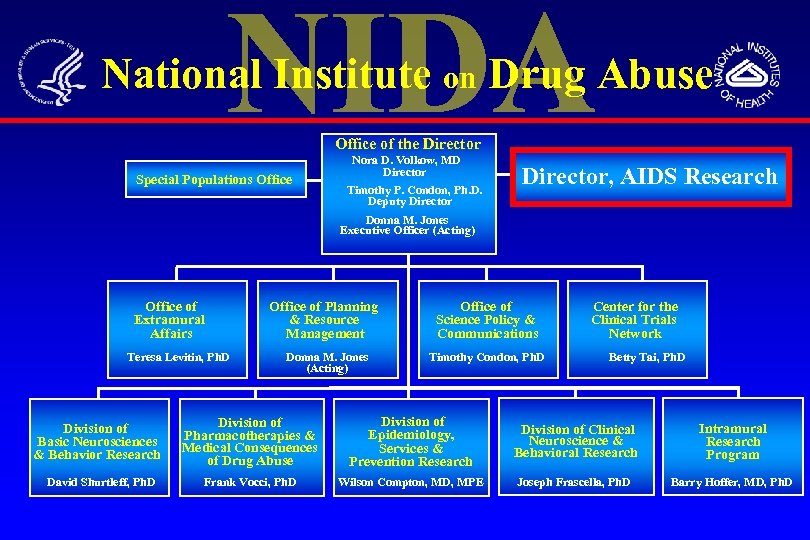 National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D
National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D
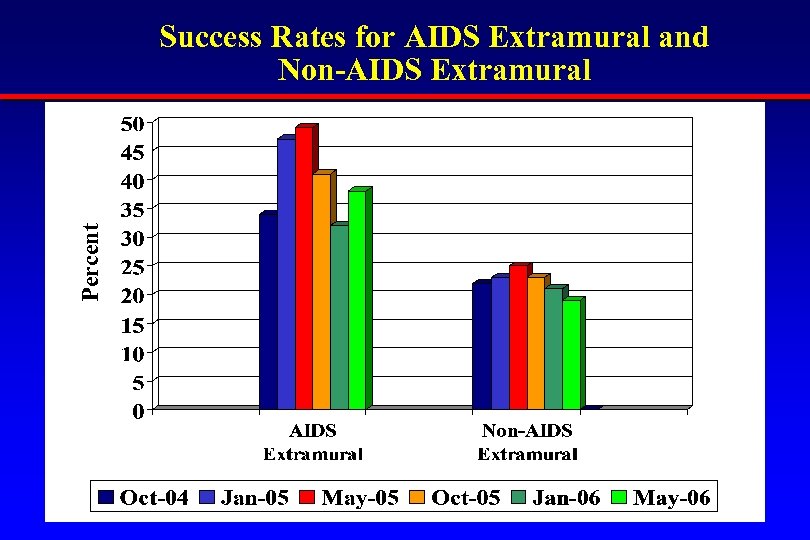 Percent Success Rates for AIDS Extramural and Non-AIDS Extramural
Percent Success Rates for AIDS Extramural and Non-AIDS Extramural
 Strategies of NIDA’s Investment on AIDS FY 06 $40 M Uncommitted Funds 1. NIDA Staff Meetings to identify priority areas • • March 12, 2007 April 9, 2007 2. Scientific Meeting of Experts May 8 -9, 2007 FY 07 $58 M FY 08 FY 09 FY 10 $65 M ~$60 M
Strategies of NIDA’s Investment on AIDS FY 06 $40 M Uncommitted Funds 1. NIDA Staff Meetings to identify priority areas • • March 12, 2007 April 9, 2007 2. Scientific Meeting of Experts May 8 -9, 2007 FY 07 $58 M FY 08 FY 09 FY 10 $65 M ~$60 M
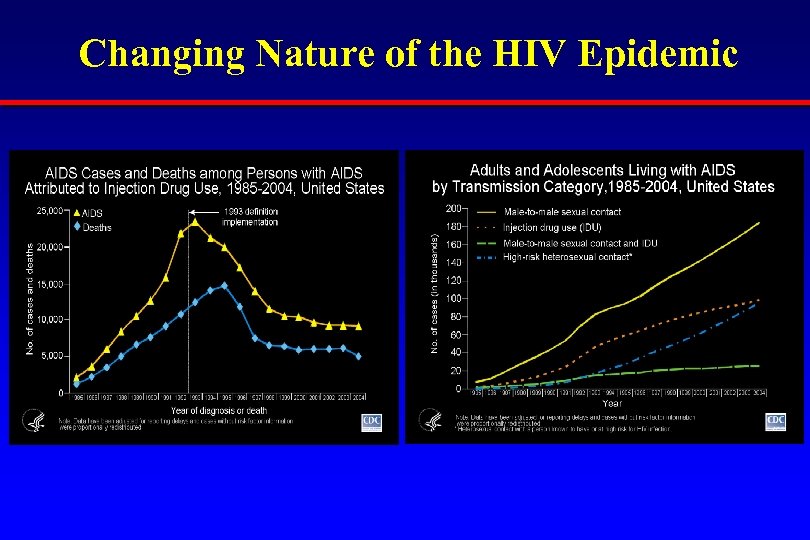 Changing Nature of the HIV Epidemic
Changing Nature of the HIV Epidemic
 HIV/AIDS Mechanisms • Infrastructure: facilitate access to existing cohorts (ALIVE, MLS) proteomic analysis animal models • Innovative research: • International: Avant-Garde Award (equivalent to Pioneer Award) Create clinical and basic research networks Training: e. g. , Invest Fellows/China/CTN Integrate research with NIAID international networks
HIV/AIDS Mechanisms • Infrastructure: facilitate access to existing cohorts (ALIVE, MLS) proteomic analysis animal models • Innovative research: • International: Avant-Garde Award (equivalent to Pioneer Award) Create clinical and basic research networks Training: e. g. , Invest Fellows/China/CTN Integrate research with NIAID international networks
 National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D DESPR
National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D DESPR
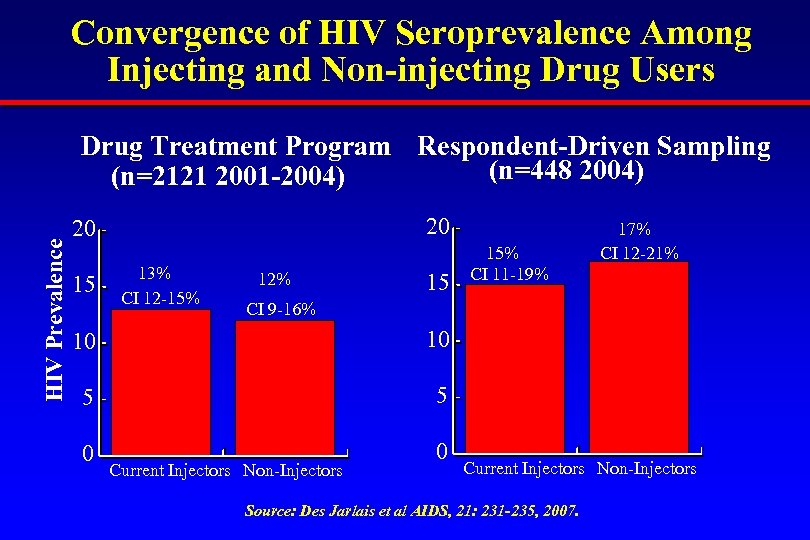 Convergence of HIV Seroprevalence Among Injecting and Non-injecting Drug Users HIV Prevalence Drug Treatment Program Respondent-Driven Sampling (n=448 2004) (n=2121 2001 -2004) 20 20 15 13% CI 12 -15% 12% 15 15% CI 11 -19% 17% CI 12 -21% CI 9 -16% 10 10 5 5 0 0 Current Injectors Non-Injectors Source: Des Jarlais et al AIDS, 21: 231 -235, 2007.
Convergence of HIV Seroprevalence Among Injecting and Non-injecting Drug Users HIV Prevalence Drug Treatment Program Respondent-Driven Sampling (n=448 2004) (n=2121 2001 -2004) 20 20 15 13% CI 12 -15% 12% 15 15% CI 11 -19% 17% CI 12 -21% CI 9 -16% 10 10 5 5 0 0 Current Injectors Non-Injectors Source: Des Jarlais et al AIDS, 21: 231 -235, 2007.
 National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D DCNBR
National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D DCNBR
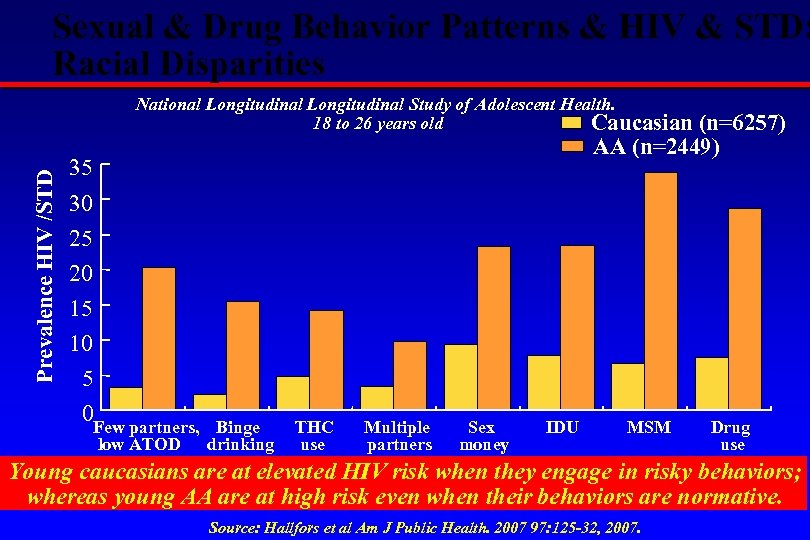 Sexual & Drug Behavior Patterns & HIV & STD: Racial Disparities Prevalence HIV /STD National Longitudinal Study of Adolescent Health. 18 to 26 years old Caucasian (n=6257) AA (n=2449) 35 30 25 20 15 10 5 0 Few partners, low ATOD Binge drinking THC use Multiple partners Sex money IDU MSM Drug use Young caucasians are at elevated HIV risk when they engage in risky behaviors; whereas young AA are at high risk even when their behaviors are normative. Source: Hallfors et al Am J Public Health. 2007 97: 125 -32, 2007.
Sexual & Drug Behavior Patterns & HIV & STD: Racial Disparities Prevalence HIV /STD National Longitudinal Study of Adolescent Health. 18 to 26 years old Caucasian (n=6257) AA (n=2449) 35 30 25 20 15 10 5 0 Few partners, low ATOD Binge drinking THC use Multiple partners Sex money IDU MSM Drug use Young caucasians are at elevated HIV risk when they engage in risky behaviors; whereas young AA are at high risk even when their behaviors are normative. Source: Hallfors et al Am J Public Health. 2007 97: 125 -32, 2007.
 National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research DBNBR David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D
National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research DBNBR David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D
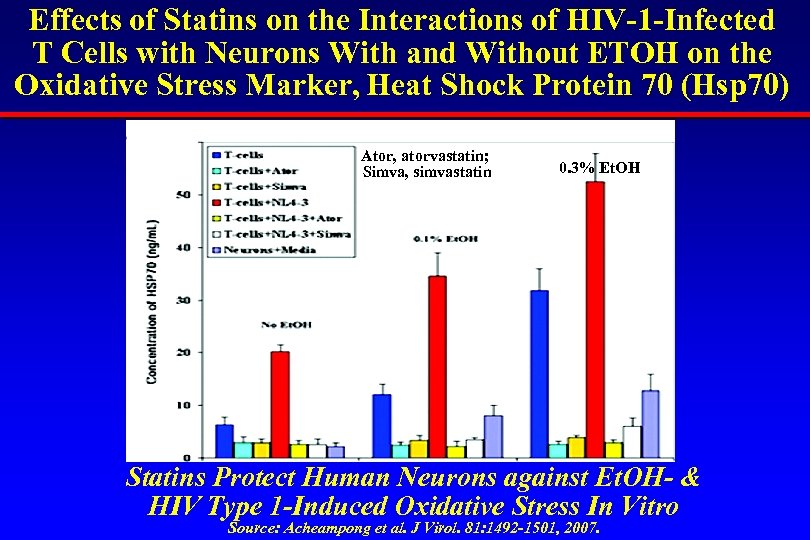 Effects of Statins on the Interactions of HIV-1 -Infected T Cells with Neurons With and Without ETOH on the Oxidative Stress Marker, Heat Shock Protein 70 (Hsp 70) Ator, atorvastatin; Simva, simvastatin 0. 3% Et. OH Statins Protect Human Neurons against Et. OH- & HIV Type 1 -Induced Oxidative Stress In Vitro Source: Acheampong et al. J Virol. 81: 1492 -1501, 2007.
Effects of Statins on the Interactions of HIV-1 -Infected T Cells with Neurons With and Without ETOH on the Oxidative Stress Marker, Heat Shock Protein 70 (Hsp 70) Ator, atorvastatin; Simva, simvastatin 0. 3% Et. OH Statins Protect Human Neurons against Et. OH- & HIV Type 1 -Induced Oxidative Stress In Vitro Source: Acheampong et al. J Virol. 81: 1492 -1501, 2007.
 National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Update from Dr. Betty Tai this morning Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network CCTN Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D
National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Update from Dr. Betty Tai this morning Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network CCTN Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D
 Integrating HIV Rapid Testing and Counseling into Drug Treatment • • 1, 000 people in USA are infected with HIV – 40, 000 new cases each year – 25 % of persons with HIV are unaware they are infected – ¼ of persons who test HIV+ fail to return to receive results CDC now recommends offering routine HIV testing to persons at risk for HIV Rapid Test • • • FDA approved blood from a finger stick or oral fluid 20 minutes $12 -15/kit can be done by counselors
Integrating HIV Rapid Testing and Counseling into Drug Treatment • • 1, 000 people in USA are infected with HIV – 40, 000 new cases each year – 25 % of persons with HIV are unaware they are infected – ¼ of persons who test HIV+ fail to return to receive results CDC now recommends offering routine HIV testing to persons at risk for HIV Rapid Test • • • FDA approved blood from a finger stick or oral fluid 20 minutes $12 -15/kit can be done by counselors
 National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D DPMCDA
National Institute on Drug Abuse Office of the Director Special Populations Office Nora D. Volkow, MD Director Timothy P. Condon, Ph. D. Deputy Director, AIDS Research Donna M. Jones Executive Officer (Acting) Office of Extramural Affairs Teresa Levitin, Ph. D Division of Basic Neurosciences & Behavior Research David Shurtleff, Ph. D Office of Planning & Resource Management Donna M. Jones (Acting) Office of Science Policy & Communications Timothy Condon, Ph. D Center for the Clinical Trials Network Betty Tai, Ph. D Division of Pharmacotherapies & Medical Consequences of Drug Abuse Division of Epidemiology, Services & Prevention Research Division of Clinical Neuroscience & Behavioral Research Intramural Research Program Frank Vocci, Ph. D Wilson Compton, MD, MPE Joseph Frascella, Ph. D Barry Hoffer, MD, Ph. D DPMCDA
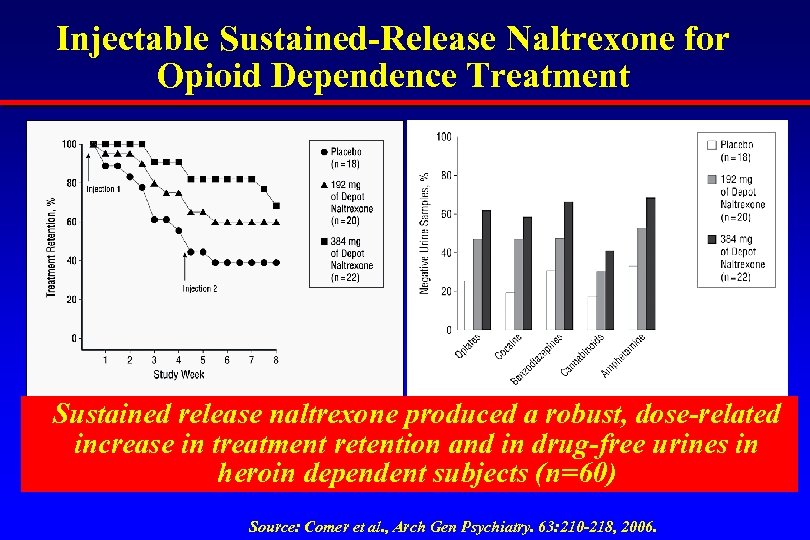 Injectable Sustained-Release Naltrexone for Opioid Dependence Treatment Sustained release naltrexone produced a robust, dose-related increase in treatment retention and in drug-free urines in heroin dependent subjects (n=60) Source: Comer et al. , Arch Gen Psychiatry. 63: 210 -218, 2006.
Injectable Sustained-Release Naltrexone for Opioid Dependence Treatment Sustained release naltrexone produced a robust, dose-related increase in treatment retention and in drug-free urines in heroin dependent subjects (n=60) Source: Comer et al. , Arch Gen Psychiatry. 63: 210 -218, 2006.
 Brain Imaging Drug Use Prevention Messages (R 21) (RFA-DA-07 -007) – 16 Applications DCNBR and DESPR
Brain Imaging Drug Use Prevention Messages (R 21) (RFA-DA-07 -007) – 16 Applications DCNBR and DESPR
 Extinction and Pharmacotherapies for Drug Addiction (R 01 --RFA-DA-07 -010) -- 15 Applications (R 03 --RFA-DA-07 -011) -- 12 Applications Mechanisms of Drug Abuse Interactions with HIV Neuropathogenesis (R 01 --RFA-DA-07 -002) – 14 Applications (R 21 --RFA-DA-07 -003) – 13 Applications
Extinction and Pharmacotherapies for Drug Addiction (R 01 --RFA-DA-07 -010) -- 15 Applications (R 03 --RFA-DA-07 -011) -- 12 Applications Mechanisms of Drug Abuse Interactions with HIV Neuropathogenesis (R 01 --RFA-DA-07 -002) – 14 Applications (R 21 --RFA-DA-07 -003) – 13 Applications
 Development of Immunotherapeutic Products for the Treatment of Methamphetamine Addiction (U 01) (RFA-DA-07 -004) – 5 Applications Design, Synthesis, and Preclinical Testing of Potential Treatment Agents for Drug Addiction (R 01) (RFA-DA-07 -006) – 30 Applications
Development of Immunotherapeutic Products for the Treatment of Methamphetamine Addiction (U 01) (RFA-DA-07 -004) – 5 Applications Design, Synthesis, and Preclinical Testing of Potential Treatment Agents for Drug Addiction (R 01) (RFA-DA-07 -006) – 30 Applications
 Recent and Upcoming Meetings, Conferences and Events
Recent and Upcoming Meetings, Conferences and Events
 The Addiction Project’s Audience is estimated to be over 50 million impressions…and counting • • • Including… HBO Channels – over 13 million viewers HBO on Demand – nearly 1 million orders DVD – more than 30, 000 sold Companion Book – over 7, 000 sold Podcasts – over 380, 000 downloads Online – hbo. com/addiction, AOL & cable affiliate websites – over 600, 000 video streams & 2. 9 million page views Local & National Outreach Parties & Screenings – over 32, 000 people attended TV Media Coverage: 35 million viewers (including The Oprah Winfrey Show, Good Morning America, The View, Lou Dobbs, Larry King Live…)
The Addiction Project’s Audience is estimated to be over 50 million impressions…and counting • • • Including… HBO Channels – over 13 million viewers HBO on Demand – nearly 1 million orders DVD – more than 30, 000 sold Companion Book – over 7, 000 sold Podcasts – over 380, 000 downloads Online – hbo. com/addiction, AOL & cable affiliate websites – over 600, 000 video streams & 2. 9 million page views Local & National Outreach Parties & Screenings – over 32, 000 people attended TV Media Coverage: 35 million viewers (including The Oprah Winfrey Show, Good Morning America, The View, Lou Dobbs, Larry King Live…)
 Timothy P. Condon, Ph. D. Deputy Director, NIDA Gregory Brigham, Ph. D. CRO, Maryhaven Dennis Mc. Carty, Ph. D. Oregon Health Sciences University
Timothy P. Condon, Ph. D. Deputy Director, NIDA Gregory Brigham, Ph. D. CRO, Maryhaven Dennis Mc. Carty, Ph. D. Oregon Health Sciences University
 ACNP American College of Neuropsychopharmacology Liaison Committee Opportunities and Impediments to Drug Development for Alcoholism and Other Substance Abuse The National Press Club Washington, D. C. May 20, 2007
ACNP American College of Neuropsychopharmacology Liaison Committee Opportunities and Impediments to Drug Development for Alcoholism and Other Substance Abuse The National Press Club Washington, D. C. May 20, 2007

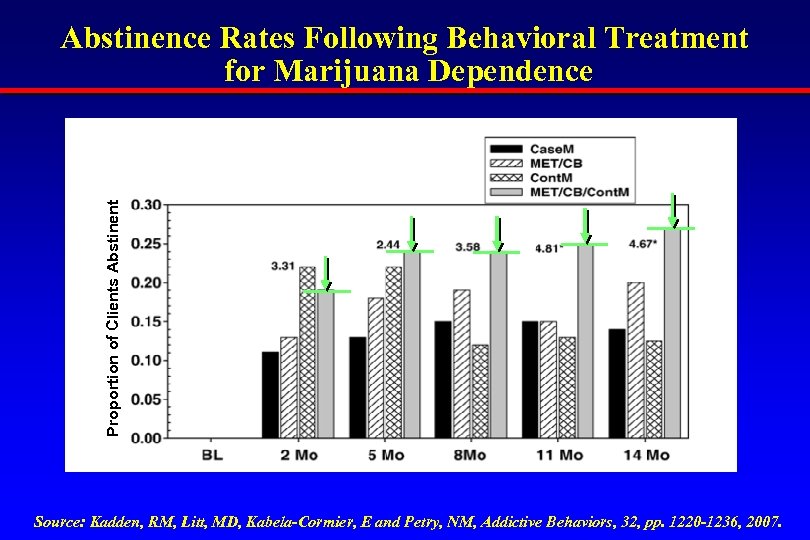 Proportion of Clients Abstinent Abstinence Rates Following Behavioral Treatment for Marijuana Dependence Source: Kadden, RM, Litt, MD, Kabela-Cormier, E and Petry, NM, Addictive Behaviors, 32, pp. 1220 -1236, 2007.
Proportion of Clients Abstinent Abstinence Rates Following Behavioral Treatment for Marijuana Dependence Source: Kadden, RM, Litt, MD, Kabela-Cormier, E and Petry, NM, Addictive Behaviors, 32, pp. 1220 -1236, 2007.
 NABI Biopharmaceuticals Recently Announced Positive Results of It’s Phase IIb Trial of Nic. Vax® • Double-blind, placebo-controlled and dose ranging study: 2 antigen doses (200 mcg and 400 mcg per injection) Nic. VAX® (Nicotine Conjugate Vaccine) • Patients who showed continuous abstinence between weeks 19 -26 had significantly higher antibody levels than those who did not quit at both the beginning and at the end of the assessment period • • 25 Among the top 30% of antibody responders who received the drug 24. 6% showed continuous abstinence from smoking between weeks 19 -26 compared to 13. 0% of controls 20 15 10 5 0 Nic. VAX Controls high-antibody responders This Phase IIb trial is continuing after all patients received a booster at six months; secondary endpoints will be assessed at 12 months
NABI Biopharmaceuticals Recently Announced Positive Results of It’s Phase IIb Trial of Nic. Vax® • Double-blind, placebo-controlled and dose ranging study: 2 antigen doses (200 mcg and 400 mcg per injection) Nic. VAX® (Nicotine Conjugate Vaccine) • Patients who showed continuous abstinence between weeks 19 -26 had significantly higher antibody levels than those who did not quit at both the beginning and at the end of the assessment period • • 25 Among the top 30% of antibody responders who received the drug 24. 6% showed continuous abstinence from smoking between weeks 19 -26 compared to 13. 0% of controls 20 15 10 5 0 Nic. VAX Controls high-antibody responders This Phase IIb trial is continuing after all patients received a booster at six months; secondary endpoints will be assessed at 12 months
 NIDA HIV/AIDS PAs, PAS/PARs & RFAs PAS/PARs PAs 2005 -2007 PA-07 -349 PA-07 -307 PA-07 -308 PA-06 -069 PA-07 -116 PA-07 -289 PA-07 -290 PA-06 -309 PA-06 -310 AIDS-Science Track Award for Research Transition (R 03) Drug Abuse Aspects of HIV/AIDS (R 01) Drug Abuse Aspects of HIV/AIDS (R 03) Health Disparities in HIV/AIDS: Focus on African Americans (R 01) Health Disparities in HIV/AIDS: Focus on African Americans (R 21) Health Disparities in HIV/AIDS: Focus on African Americans (R 03) Drug Abuse Aspects of HIV/AIDS and Other Infections (R 21) Drug Abuse Aspects of HIV/AIDS and Other Infections (R 03) PAS-07 -324 PAS-07 -325 PAS-07 -326 PAS-07 -115 PAS-07 -261 PAS-07 -262 PAS-06 -064 Drug Abuse, Risky Decision Making and HIV/AIDS (R 01) Drug Abuse, Risky Decision Making and HIV/AIDS (R 21) Drug Abuse, Risky Decision Making and HIV/AIDS (R 03) Non-Injection Drug Abuse and HIV/AIDS (R 01) Non-Injection Drug Abuse and HIV/AIDS (R 03) Non-Injection Drug Abuse and HIV/AIDS (R 21) Non-Injection Drug Abuse and HIV/AIDS (R 01) RFA-DA-07 -002 Mechanisms of Drug Abuse Interactions with HIV Neuropathogenesis (R 01) RFA-DA-07 -003 Mechanisms of Drug Abuse Interactions with HIV Neuropathogenesis (R 21) RFA-DA-05 -008 HIV and Drug Abuse Interventions Among Pregnant Women in Drug Abuse Treatment (R 01, R 03 and R 21)
NIDA HIV/AIDS PAs, PAS/PARs & RFAs PAS/PARs PAs 2005 -2007 PA-07 -349 PA-07 -307 PA-07 -308 PA-06 -069 PA-07 -116 PA-07 -289 PA-07 -290 PA-06 -309 PA-06 -310 AIDS-Science Track Award for Research Transition (R 03) Drug Abuse Aspects of HIV/AIDS (R 01) Drug Abuse Aspects of HIV/AIDS (R 03) Health Disparities in HIV/AIDS: Focus on African Americans (R 01) Health Disparities in HIV/AIDS: Focus on African Americans (R 21) Health Disparities in HIV/AIDS: Focus on African Americans (R 03) Drug Abuse Aspects of HIV/AIDS and Other Infections (R 21) Drug Abuse Aspects of HIV/AIDS and Other Infections (R 03) PAS-07 -324 PAS-07 -325 PAS-07 -326 PAS-07 -115 PAS-07 -261 PAS-07 -262 PAS-06 -064 Drug Abuse, Risky Decision Making and HIV/AIDS (R 01) Drug Abuse, Risky Decision Making and HIV/AIDS (R 21) Drug Abuse, Risky Decision Making and HIV/AIDS (R 03) Non-Injection Drug Abuse and HIV/AIDS (R 01) Non-Injection Drug Abuse and HIV/AIDS (R 03) Non-Injection Drug Abuse and HIV/AIDS (R 21) Non-Injection Drug Abuse and HIV/AIDS (R 01) RFA-DA-07 -002 Mechanisms of Drug Abuse Interactions with HIV Neuropathogenesis (R 01) RFA-DA-07 -003 Mechanisms of Drug Abuse Interactions with HIV Neuropathogenesis (R 21) RFA-DA-05 -008 HIV and Drug Abuse Interventions Among Pregnant Women in Drug Abuse Treatment (R 01, R 03 and R 21)
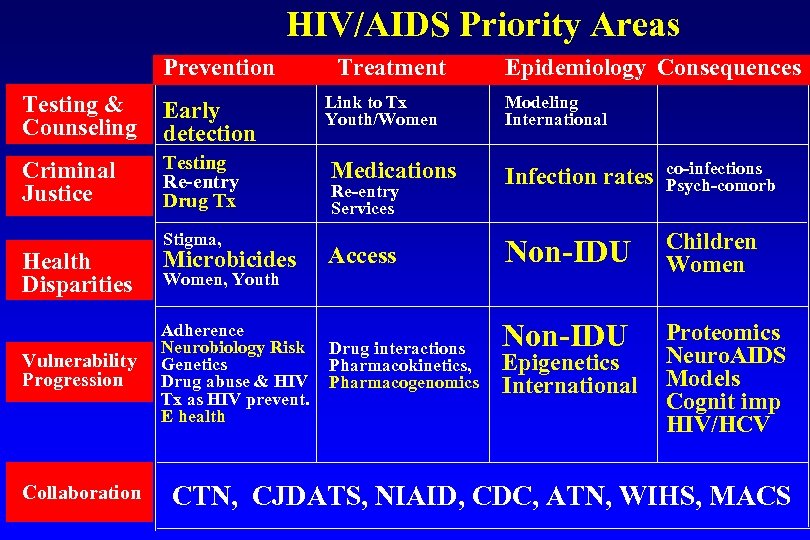 HIV/AIDS Priority Areas Prevention Testing & Counseling Early detection Criminal Justice Testing Re-entry Drug Tx Health Disparities Vulnerability Progression Collaboration Treatment Link to Tx Youth/Women Epidemiology Consequences Modeling International Medications Infection rates co-infections Psych-comorb Microbicides Access Non-IDU Children Women Adherence Neurobiology Risk Genetics Drug abuse & HIV Tx as HIV prevent. E health Drug interactions Pharmacokinetics, Pharmacogenomics Non-IDU Proteomics Neuro. AIDS Models Cognit imp HIV/HCV Stigma, Re-entry Services Women, Youth Epigenetics International CTN, CJDATS, NIAID, CDC, ATN, WIHS, MACS
HIV/AIDS Priority Areas Prevention Testing & Counseling Early detection Criminal Justice Testing Re-entry Drug Tx Health Disparities Vulnerability Progression Collaboration Treatment Link to Tx Youth/Women Epidemiology Consequences Modeling International Medications Infection rates co-infections Psych-comorb Microbicides Access Non-IDU Children Women Adherence Neurobiology Risk Genetics Drug abuse & HIV Tx as HIV prevent. E health Drug interactions Pharmacokinetics, Pharmacogenomics Non-IDU Proteomics Neuro. AIDS Models Cognit imp HIV/HCV Stigma, Re-entry Services Women, Youth Epigenetics International CTN, CJDATS, NIAID, CDC, ATN, WIHS, MACS


