75702b0f890fedf3dfd7e0dd6347a7d7.ppt
- Количество слайдов: 31
 Did You Study For Your Test? Potency Over Time ≠ Stability Bob Light B. S. Pharm. , R. Ph. Senior Director, Humco Compounding Professional Services Humco Compounding
Did You Study For Your Test? Potency Over Time ≠ Stability Bob Light B. S. Pharm. , R. Ph. Senior Director, Humco Compounding Professional Services Humco Compounding
 Disclosures Bob Light “declare(s) no conflicts of interest, real or apparent, and no financial interests in any company, product, or service mentioned in this program, including grants, employment, gifts, stock holdings, and honoraria. ” The American College of Apothecaries is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education.
Disclosures Bob Light “declare(s) no conflicts of interest, real or apparent, and no financial interests in any company, product, or service mentioned in this program, including grants, employment, gifts, stock holdings, and honoraria. ” The American College of Apothecaries is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education.
 Learning Objectives At the conclusion of this program, the participating pharmacist/technician will be able to: • Define the components of potency and stability testing. • Explain the methodology used in conducting potency and stability testing for a compounded preparation. • Apply information gained from a stability test to determine an appropriate beyond-use-date. • Compare and contrast Potency Over Time (POT) test vs Stability Test
Learning Objectives At the conclusion of this program, the participating pharmacist/technician will be able to: • Define the components of potency and stability testing. • Explain the methodology used in conducting potency and stability testing for a compounded preparation. • Apply information gained from a stability test to determine an appropriate beyond-use-date. • Compare and contrast Potency Over Time (POT) test vs Stability Test
 What is Beyond Use Dating (BUD)? • Date assigned from time of compounding in which a compounded medication may be used • Determined by compounding personnel • Based on drug-specific, scientifically valid studies, specifically stability studies • Valid guidelines can be used in absence of studies
What is Beyond Use Dating (BUD)? • Date assigned from time of compounding in which a compounded medication may be used • Determined by compounding personnel • Based on drug-specific, scientifically valid studies, specifically stability studies • Valid guidelines can be used in absence of studies
 BUD vs Expiration Date • Compounded preparations are intended for immediate administration or following short term storage; therefore, BUD criteria differs from those applied to manufactured products • Manufacturers assign expiration dates based on studies measuring factors such as: forced degradation; acid and alkali degradation; oxidation; temperature stress; and photo stability
BUD vs Expiration Date • Compounded preparations are intended for immediate administration or following short term storage; therefore, BUD criteria differs from those applied to manufactured products • Manufacturers assign expiration dates based on studies measuring factors such as: forced degradation; acid and alkali degradation; oxidation; temperature stress; and photo stability
 Potency Vs Stability • Potency – Determination of the concentration of a drug in a sample at a given point in time – Most potency is measured by HPLC (highperformance liquid chromatography) because it is specific and efficient – Other methods include titration, microbial assay, and UV-Vis (ultraviolet-visible spectrophotometry – USP has established acceptable potency range to be 90%-110% (± 10%)
Potency Vs Stability • Potency – Determination of the concentration of a drug in a sample at a given point in time – Most potency is measured by HPLC (highperformance liquid chromatography) because it is specific and efficient – Other methods include titration, microbial assay, and UV-Vis (ultraviolet-visible spectrophotometry – USP has established acceptable potency range to be 90%-110% (± 10%)
 Potency Vs Stability • Stability – Extent to which a product retains, within specified limits, and throughout its period of storage and use, the same properties and characteristics that it possessed at the time of manufacture – USP defines 5 different types of stability • Chemical – each active ingredient retains its chemical integrity and labeled potency, within the specified limits • Physical – Appearance, palatability, uniformity, dissolution, and suspendability are retained • Microbiological – Sterility or resistance to microbial growth is maintained antimicrobial agents retain effectiveness • Therapeutic – Remains unchanged • Toxicological – No toxicity occurs • Stability indicating methods will assess a compound for each type of stability
Potency Vs Stability • Stability – Extent to which a product retains, within specified limits, and throughout its period of storage and use, the same properties and characteristics that it possessed at the time of manufacture – USP defines 5 different types of stability • Chemical – each active ingredient retains its chemical integrity and labeled potency, within the specified limits • Physical – Appearance, palatability, uniformity, dissolution, and suspendability are retained • Microbiological – Sterility or resistance to microbial growth is maintained antimicrobial agents retain effectiveness • Therapeutic – Remains unchanged • Toxicological – No toxicity occurs • Stability indicating methods will assess a compound for each type of stability
 Potency • Used to determine how much drug is in a sample at a particular point in time • Quantitative measure • High Performance Liquid Chromatography (HPLC) commonly used, testing is sensitive and specific • Does not necessarily take into account degradation factors such as temperature, p. H, humidity, etc.
Potency • Used to determine how much drug is in a sample at a particular point in time • Quantitative measure • High Performance Liquid Chromatography (HPLC) commonly used, testing is sensitive and specific • Does not necessarily take into account degradation factors such as temperature, p. H, humidity, etc.
 Stability • Defined in USP <795>: The extent to which a preparation retains, within specified limits, and throughout its period of storage and use, the same properties and characteristics that it possessed at the time of compounding.
Stability • Defined in USP <795>: The extent to which a preparation retains, within specified limits, and throughout its period of storage and use, the same properties and characteristics that it possessed at the time of compounding.
 What can effect the stability of a compound? – Type of container used – Addition of other drugs – Addition of excipients, preservatives, fillers, wetting agents, etc. – Temperature – Exposure to light – Exposure to air and humidity – Intended use and duration of therapy – Addition of water
What can effect the stability of a compound? – Type of container used – Addition of other drugs – Addition of excipients, preservatives, fillers, wetting agents, etc. – Temperature – Exposure to light – Exposure to air and humidity – Intended use and duration of therapy – Addition of water
 Assigning a BUD • Obtain general stability information from manufacturer • If no stability information is available, USP provides a general guideline • Based on compounded date, not dispensed date
Assigning a BUD • Obtain general stability information from manufacturer • If no stability information is available, USP provides a general guideline • Based on compounded date, not dispensed date
 Assigning a BUD • Establish and follow a written policy and procedure for all compounds that includes the following: – Copies of product specific stability studies based on stability indicating analytical procedures – Letters/documentation from manufacturers certifying BUDs – In-house quality control data and records collected from compounded preparations – Reference materials, information, any predictive material such as charts, tables, publications
Assigning a BUD • Establish and follow a written policy and procedure for all compounds that includes the following: – Copies of product specific stability studies based on stability indicating analytical procedures – Letters/documentation from manufacturers certifying BUDs – In-house quality control data and records collected from compounded preparations – Reference materials, information, any predictive material such as charts, tables, publications
 Assigning a BUD cont’d • In the absence of scientific data and/or information from the manufacturer, USP guidelines should be used
Assigning a BUD cont’d • In the absence of scientific data and/or information from the manufacturer, USP guidelines should be used
 PCAB Standard TCRX 6 -P Written policies and procedures are established and implemented to ensure that an appropriate BUD is assigned to each non-sterile preparation, which includes: 1. When the Pharmacy lacks stability information that is applicable to a specific drug and preparation, BUDs for non-sterile preparations are assigned using USP Chapter <795> "General Guidelines for Assigning Beyond-Use Dates": a. For Non-aqueous Formulations—The BUD is not later than the time remaining until the earliest expiration date of any API or six months, whichever is earlier b. For Water-Containing Oral Formulations—The BUD is not later than 14 days when stored at controlled cold temperatures c. For Water-Containing Topical/Dermal and Mucosal Liquid and Semisolid Formulations—The BUD is not later than 30 days
PCAB Standard TCRX 6 -P Written policies and procedures are established and implemented to ensure that an appropriate BUD is assigned to each non-sterile preparation, which includes: 1. When the Pharmacy lacks stability information that is applicable to a specific drug and preparation, BUDs for non-sterile preparations are assigned using USP Chapter <795> "General Guidelines for Assigning Beyond-Use Dates": a. For Non-aqueous Formulations—The BUD is not later than the time remaining until the earliest expiration date of any API or six months, whichever is earlier b. For Water-Containing Oral Formulations—The BUD is not later than 14 days when stored at controlled cold temperatures c. For Water-Containing Topical/Dermal and Mucosal Liquid and Semisolid Formulations—The BUD is not later than 30 days
 Standard TCRX 6 -P Cont’d 2. When BUDs are assigned that exceed USP Chapter <795> "General Guidelines for Assigning Beyond-Use Dates, " the rationale for the BUD assignment is based upon the following in order of priority: a. Stability information derived from validated testing of the specific preparation, conditions, and container b. USP/NF Monographs c. Published stability information for similar compounds and formulations with the specific container and conditions d. Stability studies published in literature (peer reviewed preferred) e. Manufacturer (if a manufactured product is involved) f. Professional judgment 3. Personnel should be aware that potency tests are designed to determine how much of the active drug is in the sample, whereas stability tests are used to determine a BUD for the preparation.
Standard TCRX 6 -P Cont’d 2. When BUDs are assigned that exceed USP Chapter <795> "General Guidelines for Assigning Beyond-Use Dates, " the rationale for the BUD assignment is based upon the following in order of priority: a. Stability information derived from validated testing of the specific preparation, conditions, and container b. USP/NF Monographs c. Published stability information for similar compounds and formulations with the specific container and conditions d. Stability studies published in literature (peer reviewed preferred) e. Manufacturer (if a manufactured product is involved) f. Professional judgment 3. Personnel should be aware that potency tests are designed to determine how much of the active drug is in the sample, whereas stability tests are used to determine a BUD for the preparation.
 State Requirements • New Jersey – Provides for pharmacist to use professional judgement but must have evidence of stability “The beyond-use date limits established in this section may be exceeded only when there is supporting valid scientific stability information that is directly applicable to the specific preparation (that is, the same drug concentration range, p. H, excipients, vehicle, water content, etc. ). ”
State Requirements • New Jersey – Provides for pharmacist to use professional judgement but must have evidence of stability “The beyond-use date limits established in this section may be exceeded only when there is supporting valid scientific stability information that is directly applicable to the specific preparation (that is, the same drug concentration range, p. H, excipients, vehicle, water content, etc. ). ”
 State Requirements • California – This “beyond use date” of the compounded drug product shall not exceed 180 days from preparation or the shortest expiration date of any component in the compounded drug product, unless a longer date is supported by stability studies of finished drugs or compounded drug products using the same components and packaging. Shorter dating than set forth in this subsection may be used if it is deemed appropriate in the professional judgment of the responsible pharmacist.
State Requirements • California – This “beyond use date” of the compounded drug product shall not exceed 180 days from preparation or the shortest expiration date of any component in the compounded drug product, unless a longer date is supported by stability studies of finished drugs or compounded drug products using the same components and packaging. Shorter dating than set forth in this subsection may be used if it is deemed appropriate in the professional judgment of the responsible pharmacist.
 State Requirements • Texas, Arizona – Both defer to USP standards BUD by Type of Formulation For Nonaqueous Formulations—The BUD is not later than the time remaining until the earliest expiration date of any API or 6 months, whichever is earlier. For Water-Containing Oral Formulations—The BUD is not later than 14 days when stored at controlled cold temperatures. For Water-Containing Topical/Dermal and Mucosal Liquid and Semisolid Formulations —The BUD is not later than 30 days. a These maximum BUDs are recommended for nonsterile compounded drug preparations in the absence of stability information that is applicable to a specific drug or preparation. The BUD shall not be later than the expiration date on the container of any component.
State Requirements • Texas, Arizona – Both defer to USP standards BUD by Type of Formulation For Nonaqueous Formulations—The BUD is not later than the time remaining until the earliest expiration date of any API or 6 months, whichever is earlier. For Water-Containing Oral Formulations—The BUD is not later than 14 days when stored at controlled cold temperatures. For Water-Containing Topical/Dermal and Mucosal Liquid and Semisolid Formulations —The BUD is not later than 30 days. a These maximum BUDs are recommended for nonsterile compounded drug preparations in the absence of stability information that is applicable to a specific drug or preparation. The BUD shall not be later than the expiration date on the container of any component.
 Common References • This is a list of some common references used by pharmacists to determine BUD: – – – – Trissel's Stability of Compounded Formulations AHFS Drug Information United States Pharmacopeia Remington: The Science and Practice of Pharmacy USP Dispensing Information Journal of Pharmaceutical Sciences American Journal of Health-System Pharmacy International Journal of Pharmaceutical Compounding
Common References • This is a list of some common references used by pharmacists to determine BUD: – – – – Trissel's Stability of Compounded Formulations AHFS Drug Information United States Pharmacopeia Remington: The Science and Practice of Pharmacy USP Dispensing Information Journal of Pharmaceutical Sciences American Journal of Health-System Pharmacy International Journal of Pharmaceutical Compounding
 Stability Study • The stability of compounded medications is often an unknown • Instabilities may or may not be visually observed but require instrumental analysis for confirmation • • Products are not identical Different drug concentration Different diluents Different packaging
Stability Study • The stability of compounded medications is often an unknown • Instabilities may or may not be visually observed but require instrumental analysis for confirmation • • Products are not identical Different drug concentration Different diluents Different packaging
 Stability Study Design Example • Must have quantity formulated for multiple containers • Storage conditions should include: room temperature; refrigeration; freezing • At a minimum, 3 sample containers should be at each temperature • 2 or 3 samples for analysis taken from each container at each time point for multiple data points • Time intervals should be based on potential storage time • Use standardized sample handling to eliminate degradation due to handling technique
Stability Study Design Example • Must have quantity formulated for multiple containers • Storage conditions should include: room temperature; refrigeration; freezing • At a minimum, 3 sample containers should be at each temperature • 2 or 3 samples for analysis taken from each container at each time point for multiple data points • Time intervals should be based on potential storage time • Use standardized sample handling to eliminate degradation due to handling technique
 Stability Testing • There are 3 components to stability testing: – 1. Method Development – 2. Method Validation – 3. Stability Study
Stability Testing • There are 3 components to stability testing: – 1. Method Development – 2. Method Validation – 3. Stability Study
 Stability Testing Method Development • Separates active ingredient(s) from degradants, excipients, and impurities • Involves forced degradation of the compound and ingredients to ensure no degradants interfere with analysis • Forced degradation is accomplished by exposing the compound to the following: – Temperature extremes (both high and low) – Humidity – UV radiation (exposure to light) – Acids and bases – Hydrogen peroxide (oxidation)
Stability Testing Method Development • Separates active ingredient(s) from degradants, excipients, and impurities • Involves forced degradation of the compound and ingredients to ensure no degradants interfere with analysis • Forced degradation is accomplished by exposing the compound to the following: – Temperature extremes (both high and low) – Humidity – UV radiation (exposure to light) – Acids and bases – Hydrogen peroxide (oxidation)
 Stability Testing Method Development – cont’d • Method of analysis will be decided • Most studies will use HPLC due to accuracy
Stability Testing Method Development – cont’d • Method of analysis will be decided • Most studies will use HPLC due to accuracy
 Stability Testing Method Validation • Confirm the method used meets the criteria outlined in USP <1163> – – – – Accuracy Precision Specificity – peak purity evaluation Detection limit Quantitation limit Linearity Range Ruggedness
Stability Testing Method Validation • Confirm the method used meets the criteria outlined in USP <1163> – – – – Accuracy Precision Specificity – peak purity evaluation Detection limit Quantitation limit Linearity Range Ruggedness
 Stability Testing Method Validation cont’d • HPLC typically utilized due to specificity and efficiency • Peak-purity evaluation – Validates that the method chosen can separate the API from degradation products and excipients by using a comparison to the reference standard – Part of the specificity test
Stability Testing Method Validation cont’d • HPLC typically utilized due to specificity and efficiency • Peak-purity evaluation – Validates that the method chosen can separate the API from degradation products and excipients by using a comparison to the reference standard – Part of the specificity test
 Stability Testing Stability Study • Samples are stored in stability chambers as determined in method development and validation • Samples are tested at predetermined points in time and stability is determined • Typically: 0, 7, 14, 21, 28, 45, 60, 90 and 180 days
Stability Testing Stability Study • Samples are stored in stability chambers as determined in method development and validation • Samples are tested at predetermined points in time and stability is determined • Typically: 0, 7, 14, 21, 28, 45, 60, 90 and 180 days
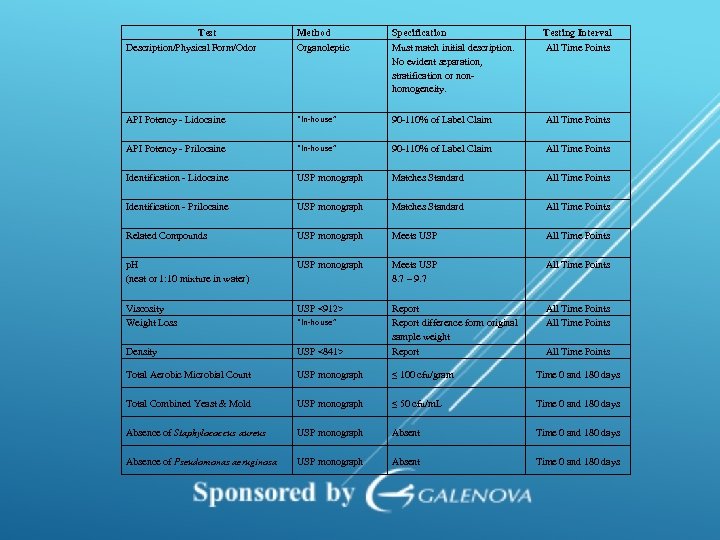 Test Description/Physical Form/Odor Method Organoleptic Specification Must match initial description. No evident separation, stratification or nonhomogeneity. Testing Interval All Time Points API Potency - Lidocaine “In-house” 90 -110% of Label Claim All Time Points API Potency - Prilocaine “In-house” 90 -110% of Label Claim All Time Points Identification - Lidocaine USP monograph Matches Standard All Time Points Identification - Prilocaine USP monograph Matches Standard All Time Points Related Compounds USP monograph Meets USP All Time Points p. H (neat or 1: 10 mixture in water) USP monograph Meets USP 8. 7 – 9. 7 All Time Points Viscosity Weight Loss USP <912> “In-house” Report difference form original sample weight All Time Points Density USP <841> Report All Time Points Total Aerobic Microbial Count USP monograph ≤ 100 cfu/gram Time 0 and 180 days Total Combined Yeast & Mold USP monograph ≤ 50 cfu/m. L Time 0 and 180 days Absence of Staphylococcus aureus USP monograph Absent Time 0 and 180 days Absence of Pseudomonas aeruginosa USP monograph Absent Time 0 and 180 days
Test Description/Physical Form/Odor Method Organoleptic Specification Must match initial description. No evident separation, stratification or nonhomogeneity. Testing Interval All Time Points API Potency - Lidocaine “In-house” 90 -110% of Label Claim All Time Points API Potency - Prilocaine “In-house” 90 -110% of Label Claim All Time Points Identification - Lidocaine USP monograph Matches Standard All Time Points Identification - Prilocaine USP monograph Matches Standard All Time Points Related Compounds USP monograph Meets USP All Time Points p. H (neat or 1: 10 mixture in water) USP monograph Meets USP 8. 7 – 9. 7 All Time Points Viscosity Weight Loss USP <912> “In-house” Report difference form original sample weight All Time Points Density USP <841> Report All Time Points Total Aerobic Microbial Count USP monograph ≤ 100 cfu/gram Time 0 and 180 days Total Combined Yeast & Mold USP monograph ≤ 50 cfu/m. L Time 0 and 180 days Absence of Staphylococcus aureus USP monograph Absent Time 0 and 180 days Absence of Pseudomonas aeruginosa USP monograph Absent Time 0 and 180 days
 Questions? Bob Light B. S. Pharm. , R. Ph. Senior Director, Humco Compounding Professional Services Humco Compounding blight@humco. com humcocompounding. com
Questions? Bob Light B. S. Pharm. , R. Ph. Senior Director, Humco Compounding Professional Services Humco Compounding blight@humco. com humcocompounding. com




