6c2d17febe8658648f37b9d730cfe253.ppt
- Количество слайдов: 19
 DEVELOPMENT OF RAPID QPCR APPROACHES FOR MEASUREMENT OF E. COLI AND ENTEROCOCCUS IN ENVIRONMENTAL WATERS: THE FUTURE FOR ROUTINE MONITORING? Rachel T. Noble, A. Denene Blackwood, and Seth Yu National Monitoring Conference May 9, 2006 UNC Chapel Hill Institute of Marine Sciences Morehead City, NC
DEVELOPMENT OF RAPID QPCR APPROACHES FOR MEASUREMENT OF E. COLI AND ENTEROCOCCUS IN ENVIRONMENTAL WATERS: THE FUTURE FOR ROUTINE MONITORING? Rachel T. Noble, A. Denene Blackwood, and Seth Yu National Monitoring Conference May 9, 2006 UNC Chapel Hill Institute of Marine Sciences Morehead City, NC
 Bacterial indicator testing • Routine monitoring in US costs >10 M USD annually • Majority of money spent in sampling and analysis time • California alone responsible for more than half of the monitoring in the US • Aim: Protect public health • Impacts: Public perception, economy, tourism, recreational water usage • Need: Rapid, quantitative methods that can be used to accurately manage beaches and shellfish harvesting waters
Bacterial indicator testing • Routine monitoring in US costs >10 M USD annually • Majority of money spent in sampling and analysis time • California alone responsible for more than half of the monitoring in the US • Aim: Protect public health • Impacts: Public perception, economy, tourism, recreational water usage • Need: Rapid, quantitative methods that can be used to accurately manage beaches and shellfish harvesting waters
 Routine methods: bacterial indicators • Total coliforms (TC) heterogeneous group composed of Escherichia, Citrobacter, Enterobacter, Salmonella, Shigella, Yersinia, and Aeromonas genera • Fecal coliforms or E. coli: thermotolerant (44. 5 C) subset of TC (FC can include Klebsiella), freshwater • Enterococci, (> 20 species, e. g. faecalis, faecium, casseliflavus, durans, avium, gallinarum), marine waters • Membrane filtration, Chromogenic substrate (IDEXX), and Multiple Tube Fermentation • Require from 18 -96 hours for results • Inaccurate management of recreational and shellfish harvesting waters because of delay
Routine methods: bacterial indicators • Total coliforms (TC) heterogeneous group composed of Escherichia, Citrobacter, Enterobacter, Salmonella, Shigella, Yersinia, and Aeromonas genera • Fecal coliforms or E. coli: thermotolerant (44. 5 C) subset of TC (FC can include Klebsiella), freshwater • Enterococci, (> 20 species, e. g. faecalis, faecium, casseliflavus, durans, avium, gallinarum), marine waters • Membrane filtration, Chromogenic substrate (IDEXX), and Multiple Tube Fermentation • Require from 18 -96 hours for results • Inaccurate management of recreational and shellfish harvesting waters because of delay
 The need for faster results • Allow accurate management of beaches (keep open when clean and close immediately when not safe for swimming) • Faster results = better tracking down sources of contamination • Tracking sources down = reducing sources (or prioritizing reduction of human sources) = reduction of potential risks to the public
The need for faster results • Allow accurate management of beaches (keep open when clean and close immediately when not safe for swimming) • Faster results = better tracking down sources of contamination • Tracking sources down = reducing sources (or prioritizing reduction of human sources) = reduction of potential risks to the public
 Immediate future: real world example • 7: 00 AM: Beach water sample collected • 8: 00 AM: Return to lab, process using rapid method and routine method (MF, MTF, IDEXX) • 10: 00 AM: • 1) E. coli > 400/100 ml and/or Enterococcus >104/100 ml or = Return and close beach, sample on periodic basis until clean • 2) Enterococci <104/100 ml or E. coli < 400/100 ml= Keep beach open and sample again (or wait until next morning to repeat)
Immediate future: real world example • 7: 00 AM: Beach water sample collected • 8: 00 AM: Return to lab, process using rapid method and routine method (MF, MTF, IDEXX) • 10: 00 AM: • 1) E. coli > 400/100 ml and/or Enterococcus >104/100 ml or = Return and close beach, sample on periodic basis until clean • 2) Enterococci <104/100 ml or E. coli < 400/100 ml= Keep beach open and sample again (or wait until next morning to repeat)
 Longer term future • Same time-scale (or maybe more rapid) • Measure pathogens instead of indicators (i. e. conduct epidemiology studies) • Real time measurements on deployed systems could provide hourly indications of water quality • Technology applicable to shellfish harvesting waters, aquaculture effluents, stormwater runoff, NPDES permits etc.
Longer term future • Same time-scale (or maybe more rapid) • Measure pathogens instead of indicators (i. e. conduct epidemiology studies) • Real time measurements on deployed systems could provide hourly indications of water quality • Technology applicable to shellfish harvesting waters, aquaculture effluents, stormwater runoff, NPDES permits etc.
 Criteria for rapid methods • • • Time required for result Threshold exceedance Numerical result Accuracy, Variability, Reproducibility Portability Specificity and usefulness of result for mitigation or protecting public health Training required Data accessibility Maintenance required of system (Deployable in situ, unattended? ) Cost (initial buy in, and per sample) Equivalent results to ‘classical’ indicator bacteria data/historical data
Criteria for rapid methods • • • Time required for result Threshold exceedance Numerical result Accuracy, Variability, Reproducibility Portability Specificity and usefulness of result for mitigation or protecting public health Training required Data accessibility Maintenance required of system (Deployable in situ, unattended? ) Cost (initial buy in, and per sample) Equivalent results to ‘classical’ indicator bacteria data/historical data
 Developing Rapid Detection Technologies for Environmental Waters • Most currently developed technologies utilize sample collection and filtration approaches similar to routine methods • Capture is dependent upon approach • Detection: fluorescence, electrochemical, etc. • Data transfer and real-time access • Combination of available applications limited only by imagination and $$ • Noble and Weisberg 2005
Developing Rapid Detection Technologies for Environmental Waters • Most currently developed technologies utilize sample collection and filtration approaches similar to routine methods • Capture is dependent upon approach • Detection: fluorescence, electrochemical, etc. • Data transfer and real-time access • Combination of available applications limited only by imagination and $$ • Noble and Weisberg 2005
 Capture: nucleic acid priming • PCR Based methods • Transcription mediated amplification (TMA) • Microarrays • NASBA • Highly specific/sensitive • Based upon known sequence complementarity • Can be used to type specific types of pathogens
Capture: nucleic acid priming • PCR Based methods • Transcription mediated amplification (TMA) • Microarrays • NASBA • Highly specific/sensitive • Based upon known sequence complementarity • Can be used to type specific types of pathogens
 Rapid microbial detection assays • 1) 2) 3) 4) 5) Developing technology using QPCR in conjunction with Cepheid, Inc. QPCR- quantification of E. coli (~ 2. 0 hr) QPCR- quantification of Enterococcus sp. (~ 2. 0 hr) QRTPCR-quantification of human enteroviruses (~ 4 hr) QPCR and QRTPCR- Rapid assays for a wide range of other viral pathogens (noroviruses, Vibrio vulnificus) and fecal marker bacteria (Bacteroides, Enterococcus species) Apply technology in all types of water samples (estuarine, coastal, freshwater, brackish, wastewater, shellfish harvesting waters, shellfish harvesting meats)
Rapid microbial detection assays • 1) 2) 3) 4) 5) Developing technology using QPCR in conjunction with Cepheid, Inc. QPCR- quantification of E. coli (~ 2. 0 hr) QPCR- quantification of Enterococcus sp. (~ 2. 0 hr) QRTPCR-quantification of human enteroviruses (~ 4 hr) QPCR and QRTPCR- Rapid assays for a wide range of other viral pathogens (noroviruses, Vibrio vulnificus) and fecal marker bacteria (Bacteroides, Enterococcus species) Apply technology in all types of water samples (estuarine, coastal, freshwater, brackish, wastewater, shellfish harvesting waters, shellfish harvesting meats)
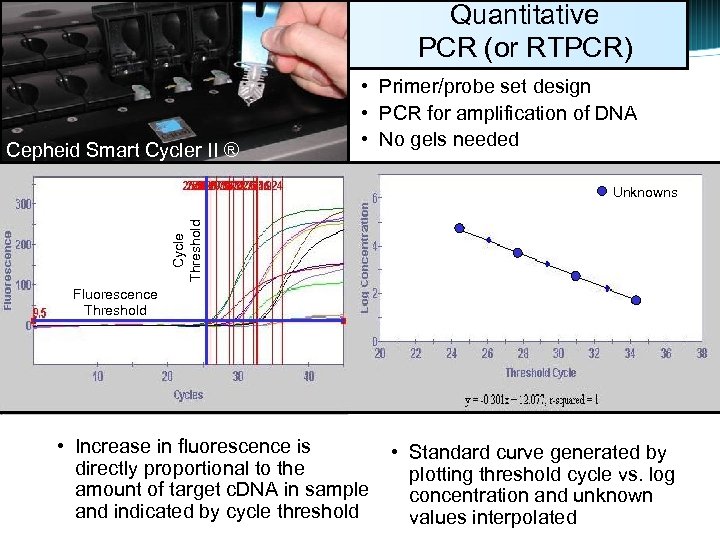 Quantitative PCR (or RTPCR) Cepheid Smart Cycler II ® • Primer/probe set design • PCR for amplification of DNA • No gels needed Cycle Threshold Unknowns Fluorescence Threshold • Increase in fluorescence is directly proportional to the amount of target c. DNA in sample and indicated by cycle threshold • Standard curve generated by plotting threshold cycle vs. log concentration and unknown values interpolated
Quantitative PCR (or RTPCR) Cepheid Smart Cycler II ® • Primer/probe set design • PCR for amplification of DNA • No gels needed Cycle Threshold Unknowns Fluorescence Threshold • Increase in fluorescence is directly proportional to the amount of target c. DNA in sample and indicated by cycle threshold • Standard curve generated by plotting threshold cycle vs. log concentration and unknown values interpolated
 QPCR Assay Details • 100 ml through PC filter to capture bacteria • 0. 45 µm pore size with vacuum needs same as for MF analysis • DNA purification (bead beating or full extraction) • Assay features Smart. Mix. TM Beads (all PCR reagents) Smart. Beads. TM contain primers, probes, and internal controls (lyophilized) • Reduces pipetting steps and errors, interanalyst variability, increases quality of standard curves • Assay also incorporates the use of innovative QPCR chemistry developed by Ds. X Limited (Manchester, UK) called SCORPIONS® • Scorpions give lower background, lower LOD • Licensing fees not prohibitive in cost to WQ agencies
QPCR Assay Details • 100 ml through PC filter to capture bacteria • 0. 45 µm pore size with vacuum needs same as for MF analysis • DNA purification (bead beating or full extraction) • Assay features Smart. Mix. TM Beads (all PCR reagents) Smart. Beads. TM contain primers, probes, and internal controls (lyophilized) • Reduces pipetting steps and errors, interanalyst variability, increases quality of standard curves • Assay also incorporates the use of innovative QPCR chemistry developed by Ds. X Limited (Manchester, UK) called SCORPIONS® • Scorpions give lower background, lower LOD • Licensing fees not prohibitive in cost to WQ agencies
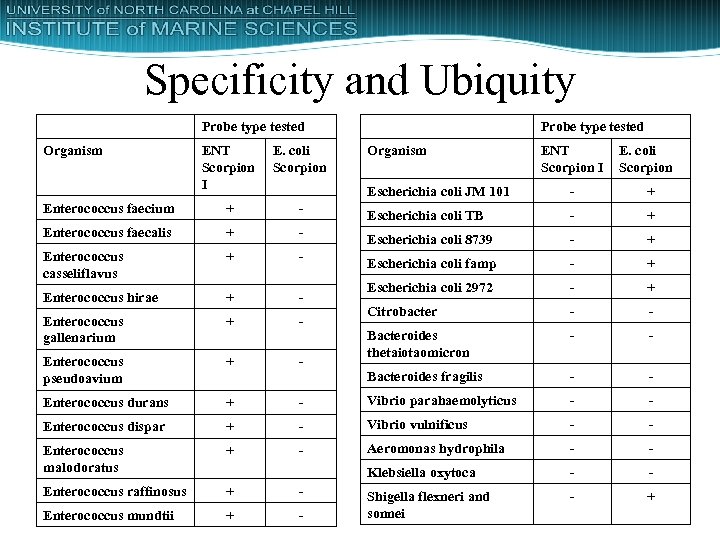 Specificity and Ubiquity Probe type tested Organism ENT Scorpion I E. coli Scorpion Enterococcus faecium + - Enterococcus faecalis + - Enterococcus casseliflavus + - Enterococcus hirae + - Enterococcus gallenarium + - Enterococcus pseudoavium + - Enterococcus durans + Enterococcus dispar Organism ENT Scorpion I E. coli Scorpion Escherichia coli JM 101 - + Escherichia coli TB - + Escherichia coli 8739 - + Escherichia coli famp - + Escherichia coli 2972 - + Citrobacter - - Bacteroides thetaiotaomicron - - Bacteroides fragilis - - - Vibrio parahaemolyticus - - + - Vibrio vulnificus - - Enterococcus malodoratus + - Aeromonas hydrophila - - Klebsiella oxytoca - - Enterococcus raffinosus + - - + Enterococcus mundtii + - Shigella flexneri and sonnei
Specificity and Ubiquity Probe type tested Organism ENT Scorpion I E. coli Scorpion Enterococcus faecium + - Enterococcus faecalis + - Enterococcus casseliflavus + - Enterococcus hirae + - Enterococcus gallenarium + - Enterococcus pseudoavium + - Enterococcus durans + Enterococcus dispar Organism ENT Scorpion I E. coli Scorpion Escherichia coli JM 101 - + Escherichia coli TB - + Escherichia coli 8739 - + Escherichia coli famp - + Escherichia coli 2972 - + Citrobacter - - Bacteroides thetaiotaomicron - - Bacteroides fragilis - - - Vibrio parahaemolyticus - - + - Vibrio vulnificus - - Enterococcus malodoratus + - Aeromonas hydrophila - - Klebsiella oxytoca - - Enterococcus raffinosus + - - + Enterococcus mundtii + - Shigella flexneri and sonnei
 SCCWRP Study • June 2005 study to compare rapid detection methods for Enterococcus and E. coli to routine methods (MF and either Enterolert™ or Colilert-18®) • 3 day study • 54 blind samples • Seawater, stormwater runoff, blanks, coastal water unseeded and seeded with cultured bacteria • Verification and speciation of isolates on plates and in quantitrays • QPCR and TMA-based assays both fared well in the comparison • Routine methods roughly 85 -90 percent agreement • QPCR Enterococcus roughly 80% accurate • QPCR E. coli results 90% accurate • QPCR had low variability compared to other methods
SCCWRP Study • June 2005 study to compare rapid detection methods for Enterococcus and E. coli to routine methods (MF and either Enterolert™ or Colilert-18®) • 3 day study • 54 blind samples • Seawater, stormwater runoff, blanks, coastal water unseeded and seeded with cultured bacteria • Verification and speciation of isolates on plates and in quantitrays • QPCR and TMA-based assays both fared well in the comparison • Routine methods roughly 85 -90 percent agreement • QPCR Enterococcus roughly 80% accurate • QPCR E. coli results 90% accurate • QPCR had low variability compared to other methods
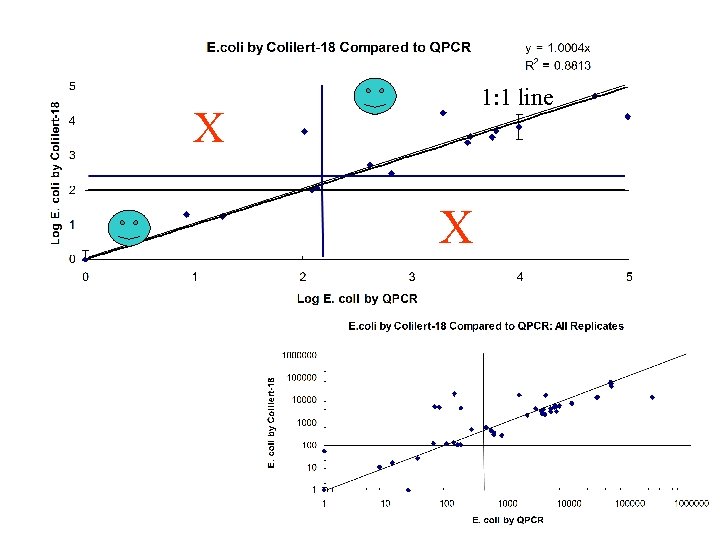 1: 1 line X X
1: 1 line X X
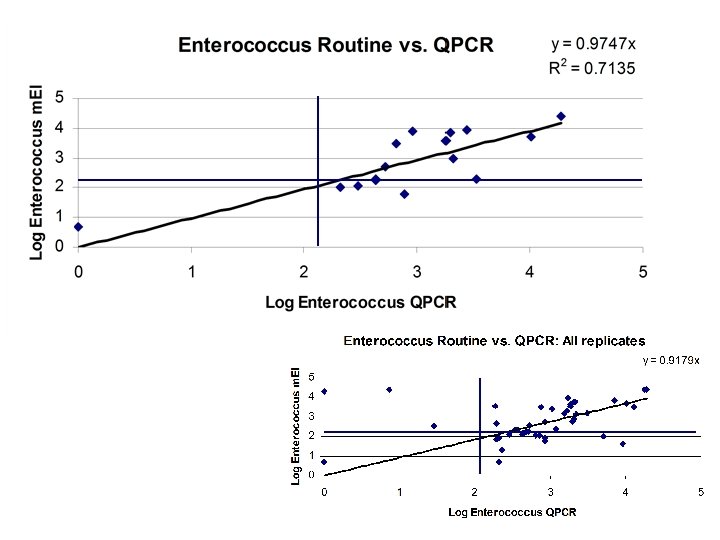
 Summary of results • Fully quantitative, wide dynamic range • LOD for both assays is ~ 1 cell per reaction, for assay with DNA extraction step is 10 cells/100 ml • 90% correct response rate for E. coli and 80 -85% correct response rate for Enterococcus • Strong correlation to routine methods (r 2 values range from 0. 70 to 0. 95 depending upon comparison to MF or DS) • ENT assay captures at least 10 known species of Enterococcus (including faecium, faecalis, caselliflavus, pseudoavium, gallinarum, etc. ) • Working towards ability to conduct filtration in the field • Internal controls and matrix controls in place for quantitative sample analysis • Currently beta-testing assay at OCSD and exploring use in drinking water (E. coli)
Summary of results • Fully quantitative, wide dynamic range • LOD for both assays is ~ 1 cell per reaction, for assay with DNA extraction step is 10 cells/100 ml • 90% correct response rate for E. coli and 80 -85% correct response rate for Enterococcus • Strong correlation to routine methods (r 2 values range from 0. 70 to 0. 95 depending upon comparison to MF or DS) • ENT assay captures at least 10 known species of Enterococcus (including faecium, faecalis, caselliflavus, pseudoavium, gallinarum, etc. ) • Working towards ability to conduct filtration in the field • Internal controls and matrix controls in place for quantitative sample analysis • Currently beta-testing assay at OCSD and exploring use in drinking water (E. coli)
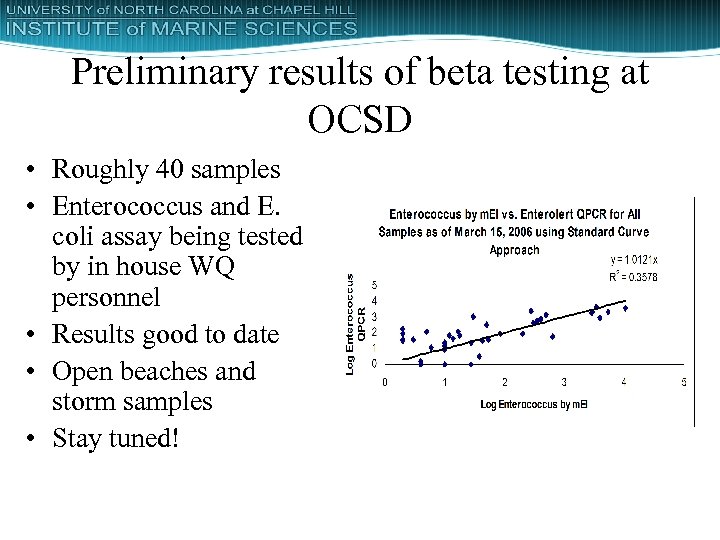 Preliminary results of beta testing at OCSD • Roughly 40 samples • Enterococcus and E. coli assay being tested by in house WQ personnel • Results good to date • Open beaches and storm samples • Stay tuned!
Preliminary results of beta testing at OCSD • Roughly 40 samples • Enterococcus and E. coli assay being tested by in house WQ personnel • Results good to date • Open beaches and storm samples • Stay tuned!
 Acknowledgements: Steve Weisberg and John Griffith, SCCWRP Denene Blackwood and Jason Gregory (UNC Chapel Hill) State of California, esp. Robin Mc. Graw and Shakoora Azimi-Gaylon United States Department of Agriculture Cepheid, Inc. : R. Schaller, C. Wilkins, N. Beckwith and S. Yu Orange County Sanitation District Rich Haugland, USEPA
Acknowledgements: Steve Weisberg and John Griffith, SCCWRP Denene Blackwood and Jason Gregory (UNC Chapel Hill) State of California, esp. Robin Mc. Graw and Shakoora Azimi-Gaylon United States Department of Agriculture Cepheid, Inc. : R. Schaller, C. Wilkins, N. Beckwith and S. Yu Orange County Sanitation District Rich Haugland, USEPA


