d9526a878503470a6243ae42f6f417b1.ppt
- Количество слайдов: 1
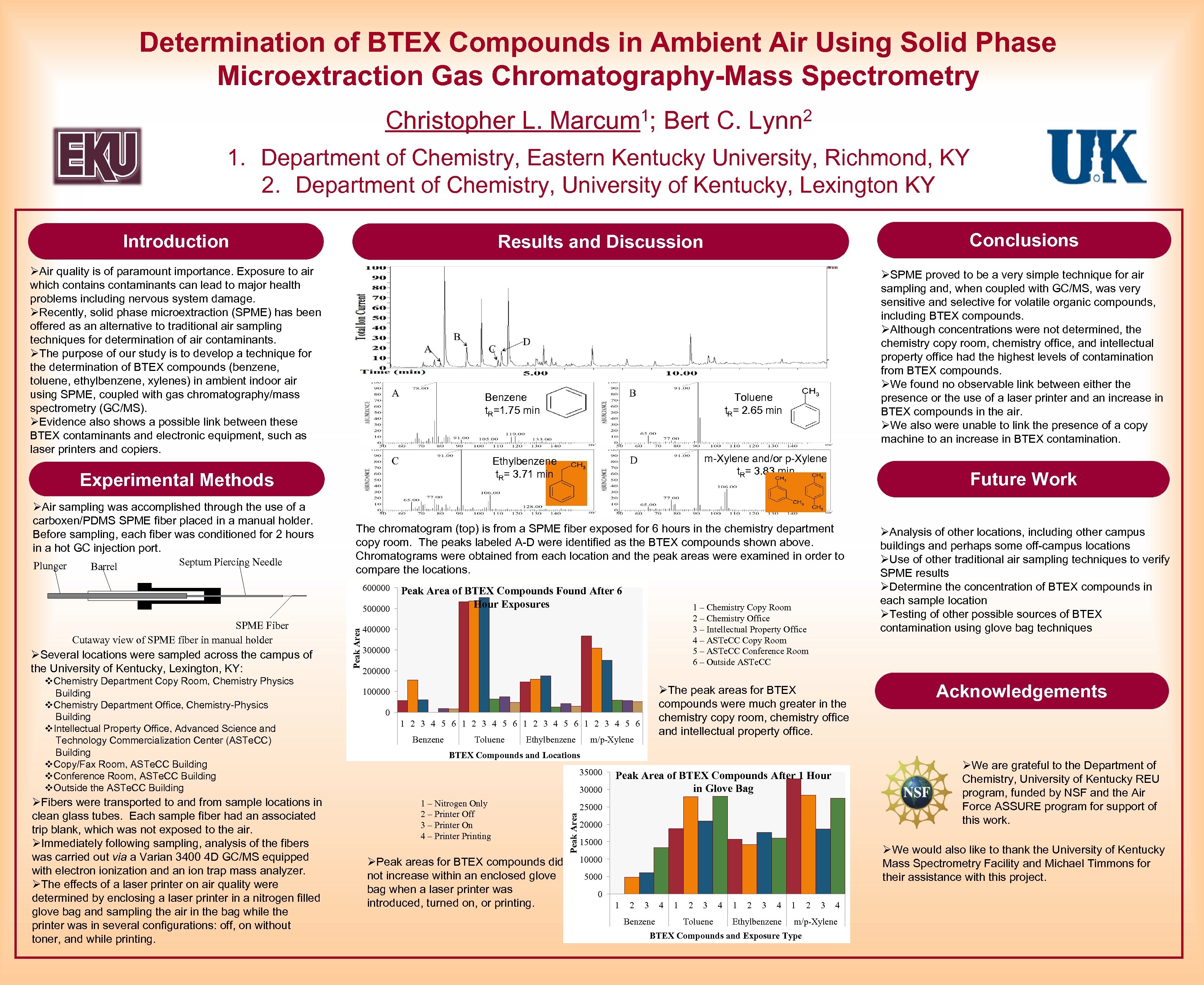
Determination of BTEX Compounds in Ambient Air Using Solid Phase Microextraction Gas Chromatography-Mass Spectrometry 1; Bert C. Lynn 2 Christopher L. Marcum 1. Department of Chemistry, Eastern Kentucky University, Richmond, KY 2. Department of Chemistry, University of Kentucky, Lexington KY Introduction ØAir quality is of paramount importance. Exposure to air which contains contaminants can lead to major health problems including nervous system damage. ØRecently, solid phase microextraction (SPME) has been offered as an alternative to traditional air sampling techniques for determination of air contaminants. ØThe purpose of our study is to develop a technique for the determination of BTEX compounds (benzene, toluene, ethylbenzene, xylenes) in ambient indoor air using SPME, coupled with gas chromatography/mass spectrometry (GC/MS). ØEvidence also shows a possible link between these BTEX contaminants and electronic equipment, such as laser printers and copiers. Benzene t. R=1. 75 min Plunger Barrel Septum Piercing Needle ØSPME proved to be a very simple technique for air sampling and, when coupled with GC/MS, was very sensitive and selective for volatile organic compounds, including BTEX compounds. ØAlthough concentrations were not determined, the chemistry copy room, chemistry office, and intellectual property office had the highest levels of contamination from BTEX compounds. ØWe found no observable link between either the presence or the use of a laser printer and an increase in BTEX compounds in the air. ØWe also were unable to link the presence of a copy machine to an increase in BTEX contamination. Toluene t. R= 2. 65 min m-Xylene and/or p-Xylene t. R= 3. 83 min Ethylbenzene t. R= 3. 71 min Experimental Methods ØAir sampling was accomplished through the use of a carboxen/PDMS SPME fiber placed in a manual holder. Before sampling, each fiber was conditioned for 2 hours in a hot GC injection port. Conclusions Results and Discussion Future Work The chromatogram (top) is from a SPME fiber exposed for 6 hours in the chemistry department copy room. The peaks labeled A-D were identified as the BTEX compounds shown above. Chromatograms were obtained from each location and the peak areas were examined in order to compare the locations. 600000 ØSeveral locations were sampled across the campus of the University of Kentucky, Lexington, KY: v. Chemistry Department Copy Room, Chemistry Physics Building v. Chemistry Department Office, Chemistry-Physics Building v. Intellectual Property Office, Advanced Science and Technology Commercialization Center (ASTe. CC) Building v. Copy/Fax Room, ASTe. CC Building v. Conference Room, ASTe. CC Building v. Outside the ASTe. CC Building ØFibers were transported to and from sample locations in clean glass tubes. Each sample fiber had an associated trip blank, which was not exposed to the air. ØImmediately following sampling, analysis of the fibers was carried out via a Varian 3400 4 D GC/MS equipped with electron ionization and an ion trap mass analyzer. ØThe effects of a laser printer on air quality were determined by enclosing a laser printer in a nitrogen filled glove bag and sampling the air in the bag while the printer was in several configurations: off, on without toner, and while printing. 1 – Chemistry Copy Room 2 – Chemistry Office 3 – Intellectual Property Office 4 – ASTe. CC Copy Room 5 – ASTe. CC Conference Room 6 – Outside ASTe. CC 400000 300000 200000 ØThe peak areas for BTEX compounds were much greater in the chemistry copy room, chemistry office and intellectual property office. 100000 0 1 2 3 4 5 6 Benzene Toluene Ethylbenzene m/p-Xylene BTEX Compounds and Locations 35000 30000 1 – Nitrogen Only 2 – Printer Off 3 – Printer On 4 – Printer Printing ØPeak areas for BTEX compounds did not increase within an enclosed glove bag when a laser printer was introduced, turned on, or printing. Acknowledgements ØWe are grateful to the Department of Chemistry, University of Kentucky REU program, funded by NSF and the Air Force ASSURE program for support of this work. Peak Area of BTEX Compounds After 1 Hour in Glove Bag 25000 Peak Area SPME Fiber Cutaway view of SPME fiber in manual holder Peak Area of BTEX Compounds Found After 6 Hour Exposures 500000 ØAnalysis of other locations, including other campus buildings and perhaps some off-campus locations ØUse of other traditional air sampling techniques to verify SPME results ØDetermine the concentration of BTEX compounds in each sample location ØTesting of other possible sources of BTEX contamination using glove bag techniques 20000 15000 ØWe would also like to thank the University of Kentucky Mass Spectrometry Facility and Michael Timmons for their assistance with this project. 10000 5000 0 1 2 3 4 Benzene 1 2 3 Toluene 4 1 2 3 4 Ethylbenzene 1 2 3 4 m/p-Xylene BTEX Compounds and Exposure Type
d9526a878503470a6243ae42f6f417b1.ppt