237772cfb9380a160195e8f3bafa76f9.ppt
- Количество слайдов: 27

DESERT (Drug-Eluting Stent Event Registry of Thrombosis) The International FDA approved DESThrombosis Registry Ron Waksman, MD and Martin Leon, MD On Behalf of the DESERT Investigators

Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship • Grant/Research Support • Consulting Fees/Honoraria Company • Volcano • Medtronic Vascular • Abbott Vascular • Boston Scientific • Biotronik • Medtronic • Abbott Vascular • Boston Scientific • Lilly Daiichi • Astra Zeneca This is an investigator initiated trial sponsored in part by Medtronic Vascular

Background DES Thrombosis • Acute and subacute DES thrombosis are the most devastating complication of coronary stent implantation • Late DES thrombosis is a relatively new and poorly studied phenomenon • Cohort-based analyses require years of follow-up, and very large numbers of patients

Background Limitations of Prior Analyses • Most have not included angiographic and procedural data • Limited number of stent thrombosis events (frequency of 2% or less): ¡ Ability to assess only very strong correlates ¡ Wide confidence intervals around risk estimates ¡ A study of 5000 patients would only have approximately 100 events, with ability to reliably assess 5 -10 predictors

DESERT: Study Objectives • To describe the correlates (clinical, procedural, and angiographic) of late (FDA approved) DES thrombosis (>30 days from stent implantation) using an adequately sized and powered casecontrol study design ¡ 500 total late Stent thrombosis events • To describe the outcomes of patients with stent thrombosis in the DES era

DESERT Investigator Sponsored Study Organization Study Role Name Principal Investigators Ron Waksman, MD & Martin Leon, MD Executive Committee Ron Waksman, MD Jeffrey Popma, MD David Cohen, MD Renu. Virmani, MD Study Management/ CRO Project Management Cardiovascular Research Institute Aisha Wellington Rebecca Torguson, MPH Data Coordinating Center Cardiovascular Research Institute Kimberly Kaneshige Zhenyi. Xue, MS Anne Hassell Martin Leon, MD Ajay Kirtane, MD Alan Yeung, MD Donald Cutlip, MD Core Angiographic Laboratory Cardiovascular Research Foundation
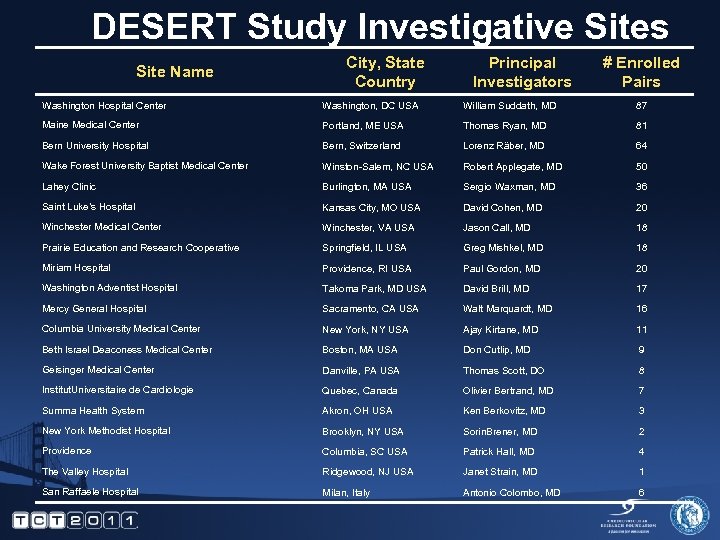
DESERT Study Investigative Sites Site Name City, State Country Principal Investigators # Enrolled Pairs Washington Hospital Center Washington, DC USA William Suddath, MD 87 Maine Medical Center Portland, ME USA Thomas Ryan, MD 81 Bern University Hospital Bern, Switzerland Lorenz Räber, MD 64 Wake Forest University Baptist Medical Center Winston-Salem, NC USA Robert Applegate, MD 50 Lahey Clinic Burlington, MA USA Sergio Waxman, MD 36 Saint Luke’s Hospital Kansas City, MO USA David Cohen, MD 20 Winchester Medical Center Winchester, VA USA Jason Call, MD 18 Prairie Education and Research Cooperative Springfield, IL USA Greg Mishkel, MD 18 Miriam Hospital Providence, RI USA Paul Gordon, MD 20 Washington Adventist Hospital Takoma Park, MD USA David Brill, MD 17 Mercy General Hospital Sacramento, CA USA Walt Marquardt, MD 16 Columbia University Medical Center New York, NY USA Ajay Kirtane, MD 11 Beth Israel Deaconess Medical Center Boston, MA USA Don Cutlip, MD 9 Geisinger Medical Center Danville, PA USA Thomas Scott, DO 8 Institut. Universitaire de Cardiologie Quebec, Canada Olivier Bertrand, MD 7 Summa Health System Akron, OH USA Ken Berkovitz, MD 3 New York Methodist Hospital Brooklyn, NY USA Sorin. Brener, MD 2 Providence Columbia, SC USA Patrick Hall, MD 4 The Valley Hospital Ridgewood, NJ USA Janet Strain, MD 1 San Raffaele Hospital Milan, Italy Antonio Colombo, MD 6
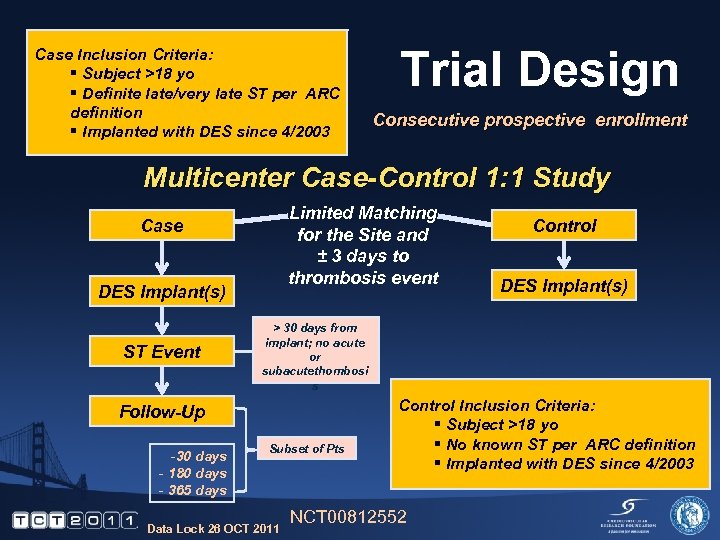
Case Inclusion Criteria: § Subject >18 yo § Definite late/very late ST per ARC definition § Implanted with DES since 4/2003 Trial Design Consecutive prospective enrollment Multicenter Case-Control 1: 1 Study Limited Matching for the Site and ± 3 days to thrombosis event Case DES Implant(s) ST Event DES Implant(s) > 30 days from implant; no acute or subacutethombosi s Follow-Up -30 days - 180 days - 365 days Control Subset of Pts Data Lock 26 OCT 2011 Control Inclusion Criteria: § Subject >18 yo § No known ST per ARC definition § Implanted with DES since 4/2003 NCT 00812552
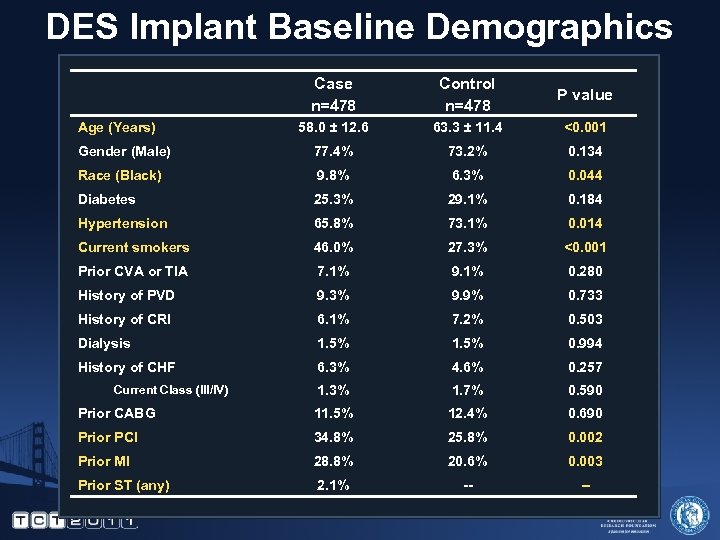
DES Implant Baseline Demographics Case n=478 Control n=478 P value 58. 0 ± 12. 6 63. 3 ± 11. 4 <0. 001 Gender (Male) 77. 4% 73. 2% 0. 134 Race (Black) 9. 8% 6. 3% 0. 044 Diabetes 25. 3% 29. 1% 0. 184 Hypertension 65. 8% 73. 1% 0. 014 Current smokers 46. 0% 27. 3% <0. 001 Prior CVA or TIA 7. 1% 9. 1% 0. 280 History of PVD 9. 3% 9. 9% 0. 733 History of CRI 6. 1% 7. 2% 0. 503 Dialysis 1. 5% 0. 994 History of CHF 6. 3% 4. 6% 0. 257 1. 3% 1. 7% 0. 590 Prior CABG 11. 5% 12. 4% 0. 690 Prior PCI 34. 8% 25. 8% 0. 002 Prior MI 28. 8% 20. 6% 0. 003 Prior ST (any) 2. 1% -- -- Age (Years) Current Class (III/IV)
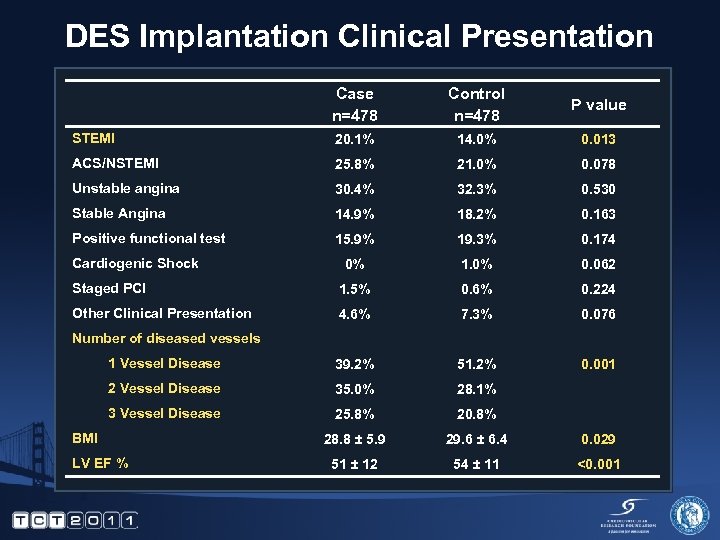
DES Implantation Clinical Presentation Case n=478 Control n=478 P value STEMI 20. 1% 14. 0% 0. 013 ACS/NSTEMI 25. 8% 21. 0% 0. 078 Unstable angina 30. 4% 32. 3% 0. 530 Stable Angina 14. 9% 18. 2% 0. 163 Positive functional test 15. 9% 19. 3% 0. 174 0% 1. 0% 0. 062 Staged PCI 1. 5% 0. 6% 0. 224 Other Clinical Presentation 4. 6% 7. 3% 0. 076 1 Vessel Disease 39. 2% 51. 2% 0. 001 2 Vessel Disease 35. 0% 28. 1% 3 Vessel Disease 25. 8% 20. 8% 28. 8 ± 5. 9 29. 6 ± 6. 4 0. 029 51 ± 12 54 ± 11 <0. 001 Cardiogenic Shock Number of diseased vessels BMI LV EF %
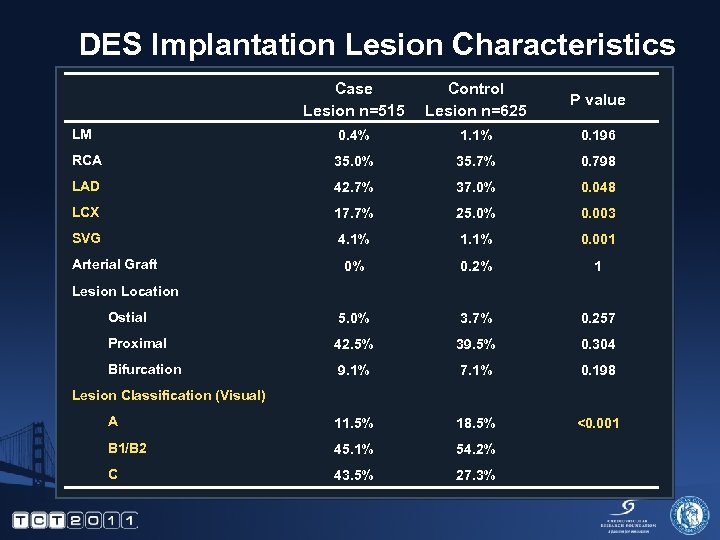
DES Implantation Lesion Characteristics Case Lesion n=515 Control Lesion n=625 P value LM 0. 4% 1. 1% 0. 196 RCA 35. 0% 35. 7% 0. 798 LAD 42. 7% 37. 0% 0. 048 LCX 17. 7% 25. 0% 0. 003 SVG 4. 1% 1. 1% 0. 001 0% 0. 2% 1 Ostial 5. 0% 3. 7% 0. 257 Proximal 42. 5% 39. 5% 0. 304 Bifurcation 9. 1% 7. 1% 0. 198 A 11. 5% 18. 5% <0. 001 B 1/B 2 45. 1% 54. 2% C 43. 5% 27. 3% Arterial Graft Lesion Location Lesion Classification (Visual)
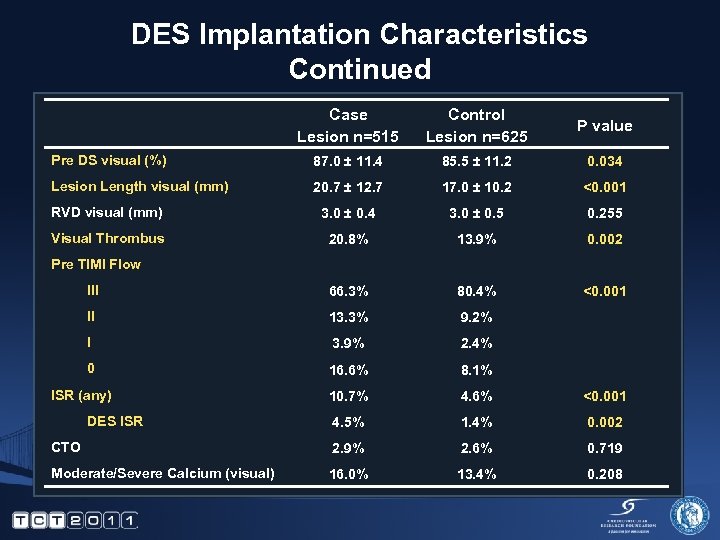
DES Implantation Characteristics Continued Case Lesion n=515 Control Lesion n=625 P value Pre DS visual (%) 87. 0 ± 11. 4 85. 5 ± 11. 2 0. 034 Lesion Length visual (mm) 20. 7 ± 12. 7 17. 0 ± 10. 2 <0. 001 RVD visual (mm) 3. 0 ± 0. 4 3. 0 ± 0. 5 0. 255 Visual Thrombus 20. 8% 13. 9% 0. 002 III 66. 3% 80. 4% <0. 001 II 13. 3% 9. 2% I 3. 9% 2. 4% 0 16. 6% 8. 1% 10. 7% 4. 6% <0. 001 4. 5% 1. 4% 0. 002 CTO 2. 9% 2. 6% 0. 719 Moderate/Severe Calcium (visual) 16. 0% 13. 4% 0. 208 Pre TIMI Flow ISR (any) DES ISR
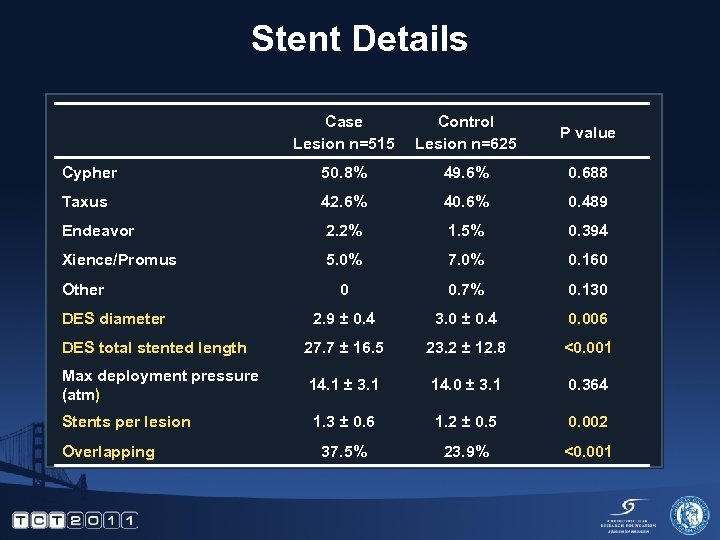
Stent Details Case Lesion n=515 Control Lesion n=625 P value Cypher 50. 8% 49. 6% 0. 688 Taxus 42. 6% 40. 6% 0. 489 Endeavor 2. 2% 1. 5% 0. 394 Xience/Promus 5. 0% 7. 0% 0. 160 0 0. 7% 0. 130 2. 9 ± 0. 4 3. 0 ± 0. 4 0. 006 DES total stented length 27. 7 ± 16. 5 23. 2 ± 12. 8 <0. 001 Max deployment pressure (atm) 14. 1 ± 3. 1 14. 0 ± 3. 1 0. 364 Stents per lesion 1. 3 ± 0. 6 1. 2 ± 0. 5 0. 002 37. 5% 23. 9% <0. 001 Other DES diameter Overlapping
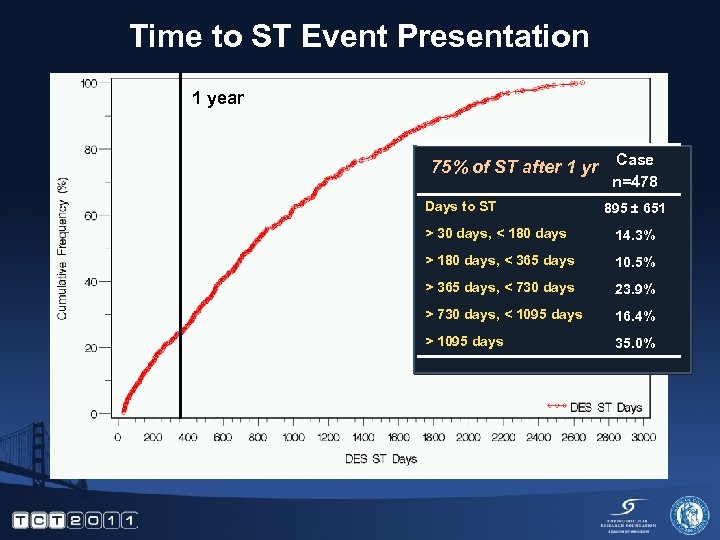
Time to ST Event Presentation 1 year 75% of ST after 1 yr Case n=478 Days to ST 895 ± 651 > 30 days, < 180 days 14. 3% > 180 days, < 365 days 10. 5% > 365 days, < 730 days 23. 9% > 730 days, < 1095 days 16. 4% > 1095 days 35. 0%

ST Event Clinical Presentation Case n=478 STEMI 66. 9% ACS/NSTEMI 22. 0% Unstable angina 7. 7% Stable Angina 1. 7% Positive functional test 1. 7% Cardiogenic Shock 4. 8% Other Clinical Presentation 1. 9% ST identified by autopsy 0. 2% LV EF % 51 ± 12 Length of Hospitalization (days) 4. 3 ± 4. 6 ICU stay required Length of ICU Stay (days) 72. 6% 2. 5 ± 3. 5
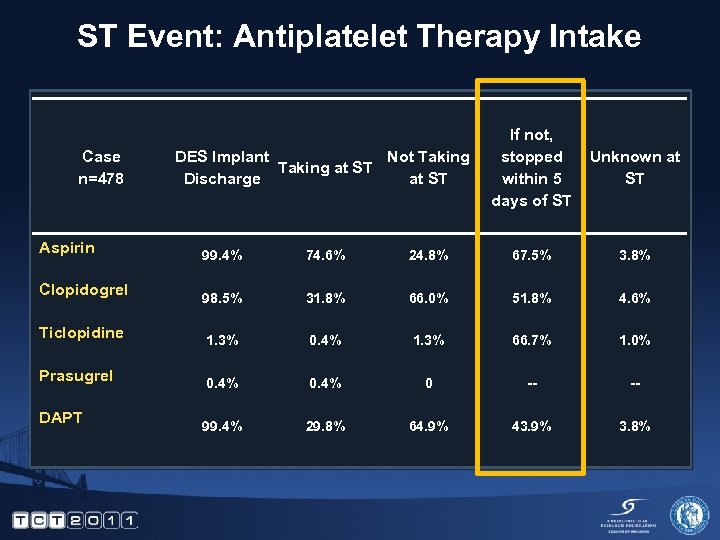
ST Event: Antiplatelet Therapy Intake Case n=478 Aspirin Clopidogrel Ticlopidine Prasugrel DAPT DES Implant Not Taking at ST Discharge at ST If not, stopped within 5 days of ST Unknown at ST 99. 4% 74. 6% 24. 8% 67. 5% 3. 8% 98. 5% 31. 8% 66. 0% 51. 8% 4. 6% 1. 3% 0. 4% 1. 3% 66. 7% 1. 0% 0. 4% 0 -- -- 99. 4% 29. 8% 64. 9% 43. 9% 3. 8%

ST Event Lesion Characteristics Case Lesion n=514 Visual Thrombus 99. 6% Pre DS visual (%) 96. 5 ± 10. 5 Pre TIMI Flow III 10. 9% I 3. 8% 0 74. 5% Malapposition By IVUS (Investigator determination) 4. 9% By Angiography (Investigator determination) 5. 8% Revascularization any 96. 3% CABG 1. 0% PCI 95. 3%
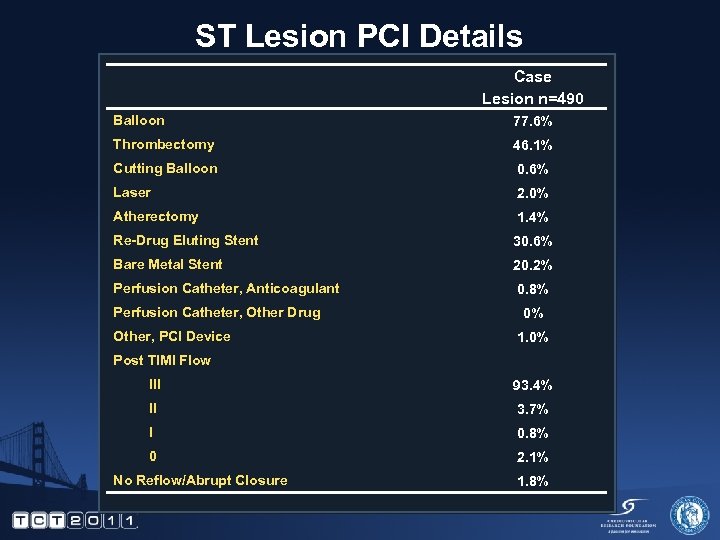
ST Lesion PCI Details Case Lesion n=490 Balloon 77. 6% Thrombectomy 46. 1% Cutting Balloon 0. 6% Laser 2. 0% Atherectomy 1. 4% Re-Drug Eluting Stent 30. 6% Bare Metal Stent 20. 2% Perfusion Catheter, Anticoagulant 0. 8% Perfusion Catheter, Other Drug Other, PCI Device 0% 1. 0% Post TIMI Flow III 93. 4% II 3. 7% I 0. 8% 0 2. 1% No Reflow/Abrupt Closure 1. 8%
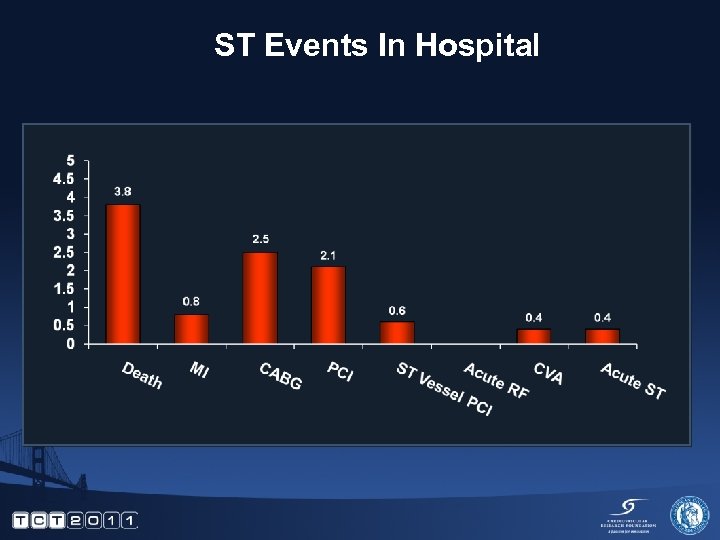
ST Events In Hospital
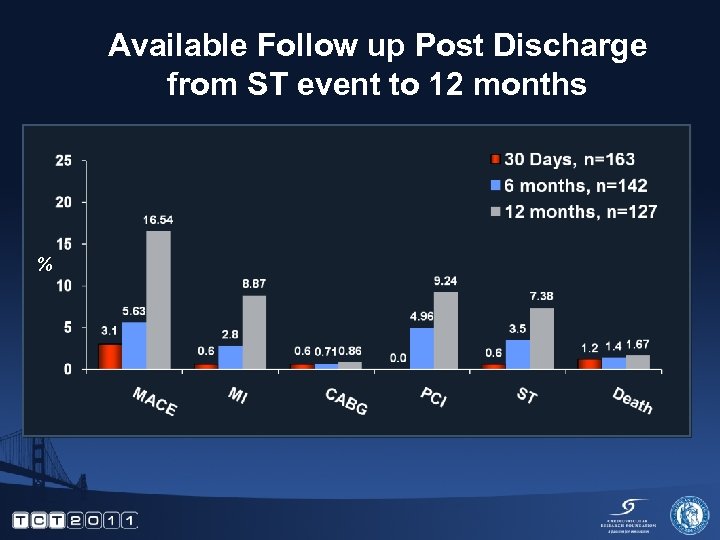
Available Follow up Post Discharge from ST event to 12 months %
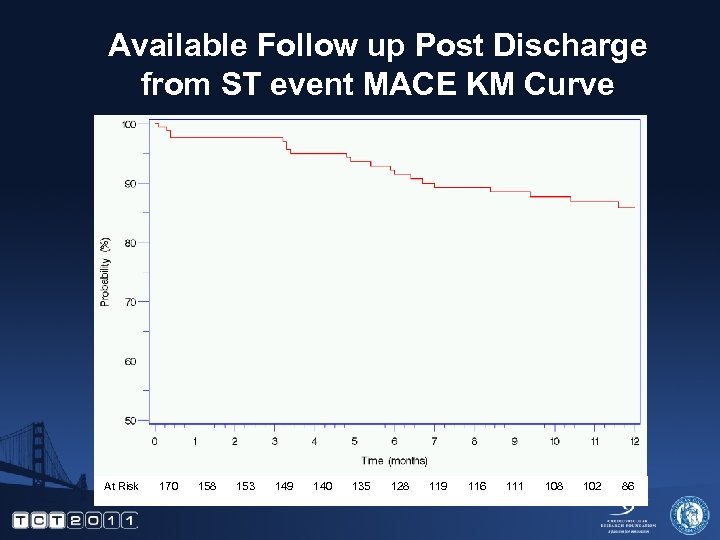
Available Follow up Post Discharge from ST event MACE KM Curve At Risk 170 158 153 149 140 135 128 119 116 111 108 102 86
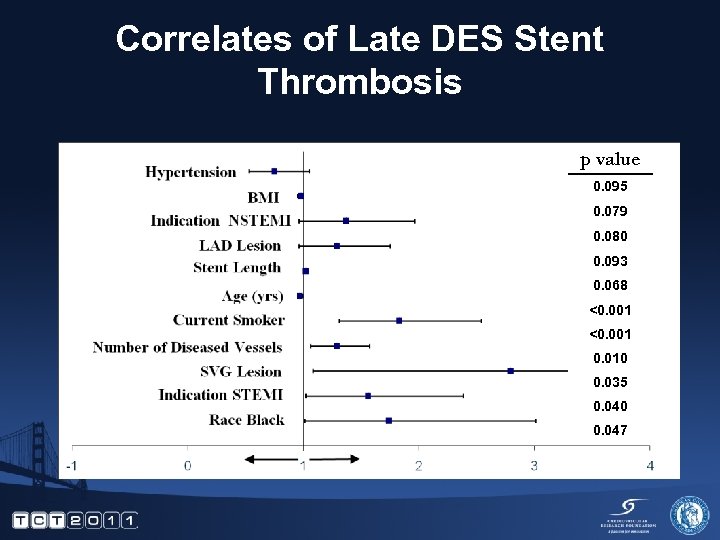
Correlates of Late DES Stent Thrombosis p value 0. 095 0. 079 0. 080 0. 093 0. 068 <0. 001 0. 010 0. 035 0. 040 0. 047
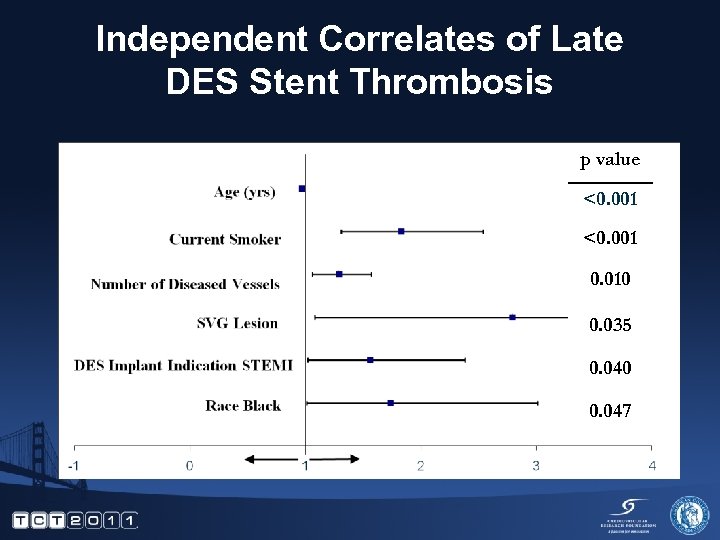
Independent Correlates of Late DES Stent Thrombosis p value <0. 001 0. 010 0. 035 0. 040 0. 047

Limitations • Prevalence of Late and Very Late DES ST cannot be assessed • In DESERT 90% of the patients in both groups had first generation DES • This analysis only identifies patients who survived the acute event of ST and presented for an angiogram • Angiographic data is currently being analyzed

Summary • DESERT is the largest case-control registry of late and very late DES Stent Thrombosis • In DESERT, the majority of the Late ST occurred after one year (~75%) and continued to occur up to 7. 3 years • Theclinical presentation of late ST was mainly MI (66. 9% STEMI and 22% NSTEMI) • Nearly 30% of the patients with L ate ST were on DAPT at the time of the event • In hospital mortality of patients who presented with late ST was 3. 8% and 1. 67% at one year

Conclusions ¡ Patients who had first generation DES continue to be at risk for late stent thrombosis up to 7 years ¡ Younger patients, smokers, black ethnicity, patients with multi vessel disease, STEMI, or SVG lesions are at higher risk of developing late ST and should be reconsider for DES, or for a potent or longer DAPT regimen ¡ Mortality with late ST is lower when compared with historically reported acute and subacute ST. This suggest a different pathological mechanism for late ST: (late restenosis and/or neo-atherosclerosis)

Thank You for your attention
237772cfb9380a160195e8f3bafa76f9.ppt