225e99f039be6d9aebbb5e8c0eee41e4.ppt
- Количество слайдов: 24
 DEPARTMENT OF AYURVEDA, YOGA & NATUROPATHY, UNANI, SIDDHA AND HOMOEOPATHY (AYUSH), , MINISTRY OF HEALTH & F. W. , GOVERNMENT OF INDIA WELCOMES THE DELEGATES OF CIS COUNTRIES
DEPARTMENT OF AYURVEDA, YOGA & NATUROPATHY, UNANI, SIDDHA AND HOMOEOPATHY (AYUSH), , MINISTRY OF HEALTH & F. W. , GOVERNMENT OF INDIA WELCOMES THE DELEGATES OF CIS COUNTRIES
 Schedule of Presentation i) Status of - Shri Bala Prasad, Traditional Systems Director, Department of Medicine in India AYUSH ii) Life-Style Disorders - Dr. G. S. Lavekar, and Ayurveda Director, CCRAS iii) Standardization - Shri Ranjit Puranik, and Quality Control C. E. O. , of Ayurveda Dhootpapeshwar Ltd. Medicines iv) Unani System of Dr. Mohd. Khalid Medicine in India Siddiqui, Director, CCRUM
Schedule of Presentation i) Status of - Shri Bala Prasad, Traditional Systems Director, Department of Medicine in India AYUSH ii) Life-Style Disorders - Dr. G. S. Lavekar, and Ayurveda Director, CCRAS iii) Standardization - Shri Ranjit Puranik, and Quality Control C. E. O. , of Ayurveda Dhootpapeshwar Ltd. Medicines iv) Unani System of Dr. Mohd. Khalid Medicine in India Siddiqui, Director, CCRUM
 TRADITIONAL INDIAN HEALTH KNOWLEDGE s s s Rich bio-diversity. 12 agro-climatic zones (out of total 16 the world over). Wide range of medicinal plant species (about 8000). Long history of use of medicinal plants and health practices (right from prehistoric period). Codified, organized, documented and officially recognized health systems (e. g. Ayurveda, Unani)
TRADITIONAL INDIAN HEALTH KNOWLEDGE s s s Rich bio-diversity. 12 agro-climatic zones (out of total 16 the world over). Wide range of medicinal plant species (about 8000). Long history of use of medicinal plants and health practices (right from prehistoric period). Codified, organized, documented and officially recognized health systems (e. g. Ayurveda, Unani)
 OFFICIALLY RECOGNIZED SYSTEMS s s Ayurveda – DOMINANT & OLDEST Yoga – THE OLDEST REFERENCE IN VEDIC LTD. s s Naturopathy Unani Siddha Homeopathy
OFFICIALLY RECOGNIZED SYSTEMS s s Ayurveda – DOMINANT & OLDEST Yoga – THE OLDEST REFERENCE IN VEDIC LTD. s s Naturopathy Unani Siddha Homeopathy
 TRADITIONAL MEDICINE IN INDIA –Thrust Areas 1. 2. 3. 4. 5. 6. Improvement and upgradation of standards of education in ISM&H; Standardisation of drugs; Ensuring sustained availability of raw materials, i. e. , medicinal plants, metals, minerals and materials of animal origin etc. ; Research and Development; Participation of ISM&H in the National Health Care Delivery System, National Health and Family Welfare Programmes; Information, Education and Communication
TRADITIONAL MEDICINE IN INDIA –Thrust Areas 1. 2. 3. 4. 5. 6. Improvement and upgradation of standards of education in ISM&H; Standardisation of drugs; Ensuring sustained availability of raw materials, i. e. , medicinal plants, metals, minerals and materials of animal origin etc. ; Research and Development; Participation of ISM&H in the National Health Care Delivery System, National Health and Family Welfare Programmes; Information, Education and Communication
 ORGANISATIONAL SET UP (Central Level) s s s Central Department under Ministry of Health & Family Welfare. Central Research Councils – four research councils, viz. Central Council for Research in Ayurveda and Shiddha (CCRAS), Central Council for Research in Unani (CCRUM), Central Council for Research in Homoeopathy (CCRH) and Central Council for Research in Yoga and Naturopathy (CCRYN), Central Regulatory Bodies. National Institutes – One for each system National Medicinal Plant Board. Central Pharmacopoeia Laboratories.
ORGANISATIONAL SET UP (Central Level) s s s Central Department under Ministry of Health & Family Welfare. Central Research Councils – four research councils, viz. Central Council for Research in Ayurveda and Shiddha (CCRAS), Central Council for Research in Unani (CCRUM), Central Council for Research in Homoeopathy (CCRH) and Central Council for Research in Yoga and Naturopathy (CCRYN), Central Regulatory Bodies. National Institutes – One for each system National Medicinal Plant Board. Central Pharmacopoeia Laboratories.
 ORGANISATIONAL SET UP (State level) s s s s AYUSH Department or Ministry. State Directorates State Boards/Councils for registration of practitioners. State Licensing/Drug Control Authorities. State colleges/institutions. State Pharmacies State Drug Testing Laboratories. State Medicinal Plant Boards
ORGANISATIONAL SET UP (State level) s s s s AYUSH Department or Ministry. State Directorates State Boards/Councils for registration of practitioners. State Licensing/Drug Control Authorities. State colleges/institutions. State Pharmacies State Drug Testing Laboratories. State Medicinal Plant Boards
 RECOGNISED COURSES OF STUDY s Bachelor Degree course : - 5 and ½ year. s Postgraduate degree course : - 3 years M. D. ; M. S. (Ayurveda/Siddha/Unani) s Doctorate (Ph. D. ) course : - s Short & midterm courses : - 3 months - 2 years. 2 -5 years.
RECOGNISED COURSES OF STUDY s Bachelor Degree course : - 5 and ½ year. s Postgraduate degree course : - 3 years M. D. ; M. S. (Ayurveda/Siddha/Unani) s Doctorate (Ph. D. ) course : - s Short & midterm courses : - 3 months - 2 years. 2 -5 years.
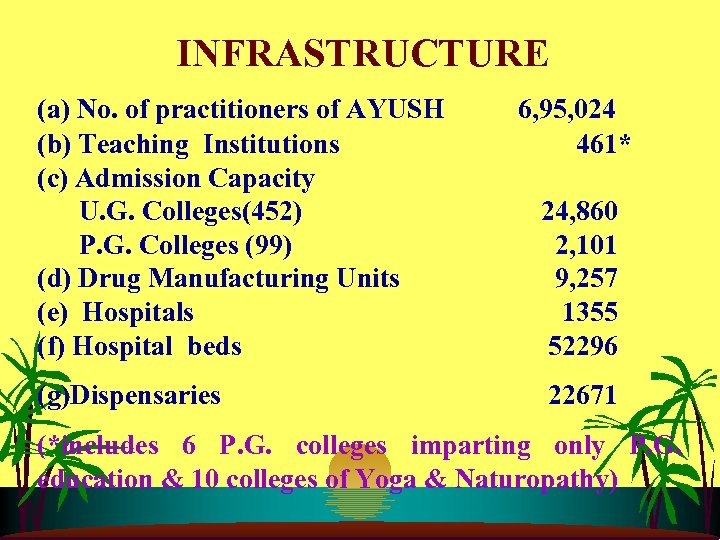 INFRASTRUCTURE (a) No. of practitioners of AYUSH 6, 95, 024 (b) Teaching Institutions 461* (c) Admission Capacity U. G. Colleges(452) 24, 860 P. G. Colleges (99) 2, 101 (d) Drug Manufacturing Units 9, 257 (e) Hospitals 1355 (f) Hospital beds 52296 (g)Dispensaries 22671 (*includes 6 P. G. colleges imparting only P. G. education & 10 colleges of Yoga & Naturopathy)
INFRASTRUCTURE (a) No. of practitioners of AYUSH 6, 95, 024 (b) Teaching Institutions 461* (c) Admission Capacity U. G. Colleges(452) 24, 860 P. G. Colleges (99) 2, 101 (d) Drug Manufacturing Units 9, 257 (e) Hospitals 1355 (f) Hospital beds 52296 (g)Dispensaries 22671 (*includes 6 P. G. colleges imparting only P. G. education & 10 colleges of Yoga & Naturopathy)
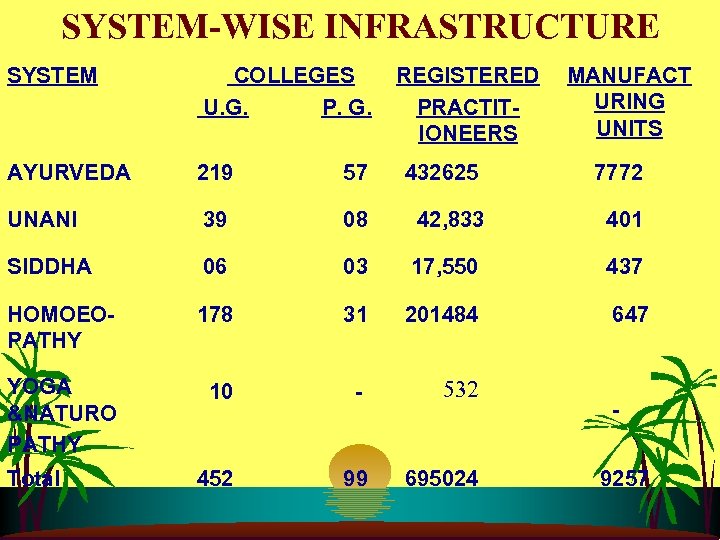 SYSTEM-WISE INFRASTRUCTURE SYSTEM COLLEGES U. G. P. G. AYURVEDA 219 57 432625 7772 UNANI 39 08 42, 833 401 SIDDHA 06 03 17, 550 437 HOMOEOPATHY 178 31 201484 647 YOGA &NATURO PATHY 10 - 532 452 99 695024 Total REGISTERED PRACTITIONEERS MANUFACT URING UNITS 9257
SYSTEM-WISE INFRASTRUCTURE SYSTEM COLLEGES U. G. P. G. AYURVEDA 219 57 432625 7772 UNANI 39 08 42, 833 401 SIDDHA 06 03 17, 550 437 HOMOEOPATHY 178 31 201484 647 YOGA &NATURO PATHY 10 - 532 452 99 695024 Total REGISTERED PRACTITIONEERS MANUFACT URING UNITS 9257
 NATIONAL INSTITUTES SET UP BY THE GOVERNMENT OF INDIA ¨ National Institute of Ayurveda, Jaipur ¨ National Institute of Unani Medicine, Bangalore ¨ National Institute of Homoeopathy, Calcutta ¨ National Institute of Naturopathy, Pune ¨ Morarji Desai National Institute of Yoga, New Delhi ¨ National Institute of Siddha, Chennai ¨ Rashtriya Ayurveda Vidyapeeth, New Delhi ¨ Institute of Post-graduate teaching & Research, Gujarat Ayurved University, Jamnagar ·
NATIONAL INSTITUTES SET UP BY THE GOVERNMENT OF INDIA ¨ National Institute of Ayurveda, Jaipur ¨ National Institute of Unani Medicine, Bangalore ¨ National Institute of Homoeopathy, Calcutta ¨ National Institute of Naturopathy, Pune ¨ Morarji Desai National Institute of Yoga, New Delhi ¨ National Institute of Siddha, Chennai ¨ Rashtriya Ayurveda Vidyapeeth, New Delhi ¨ Institute of Post-graduate teaching & Research, Gujarat Ayurved University, Jamnagar ·
 RESEARCH COUNCILS. . s 1. 2. 3. 4. 5. 6. The Research Councils are engaged in the following areas of Health Care Research and Development: Clinical Research Drug Research including standardization Survey and Cultivation of Medicinal Plants Tribal health Research Literary Research Family Welfare Research
RESEARCH COUNCILS. . s 1. 2. 3. 4. 5. 6. The Research Councils are engaged in the following areas of Health Care Research and Development: Clinical Research Drug Research including standardization Survey and Cultivation of Medicinal Plants Tribal health Research Literary Research Family Welfare Research
 PHARMACOPOEIA COMMITTEES FOR INDIAN SYSTEMS OF MEDICINE & HOMOEOPATHY ¨ Pharmacopoeia Committees notified by Central Government for laying down Pharmacopoeial Standards - Ayurveda Pharmacopoeia Committee - Siddha Pharmacopoeia Committee - Unani Pharmacopoeia Committee - Homoeopathic Pharmacopoeia Committee
PHARMACOPOEIA COMMITTEES FOR INDIAN SYSTEMS OF MEDICINE & HOMOEOPATHY ¨ Pharmacopoeia Committees notified by Central Government for laying down Pharmacopoeial Standards - Ayurveda Pharmacopoeia Committee - Siddha Pharmacopoeia Committee - Unani Pharmacopoeia Committee - Homoeopathic Pharmacopoeia Committee
 APEX LEVEL PHARMACOPOEIAL TESTING FACILITIES Pharmacopoeial Laboratory for Indian Medicine (For evolving Pharmacopoeial standards of Ayurveda, Siddha and Unani drugs) Homoeopathic Pharmacopoeia Laboratory (For evolving Pharmacopoeial Standards for Homoeopathic drugs These Laboratories are also Appellate Laboratories for Drug Testing and Quality Control) 21 State Laboratories have been upgraded Private laboratories are being recognised as Public Test Laboratories Good Manufacturing Practices are being implemented.
APEX LEVEL PHARMACOPOEIAL TESTING FACILITIES Pharmacopoeial Laboratory for Indian Medicine (For evolving Pharmacopoeial standards of Ayurveda, Siddha and Unani drugs) Homoeopathic Pharmacopoeia Laboratory (For evolving Pharmacopoeial Standards for Homoeopathic drugs These Laboratories are also Appellate Laboratories for Drug Testing and Quality Control) 21 State Laboratories have been upgraded Private laboratories are being recognised as Public Test Laboratories Good Manufacturing Practices are being implemented.
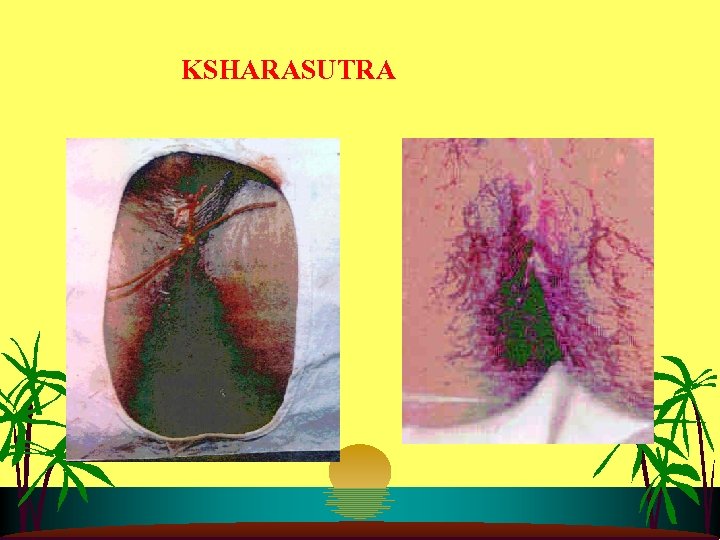
 STRENGTH OF AYURVEDA s s s Science of Holistic Health caring Body, Mind and Soul. Preventive, Promotive & Curative aspects Source of Ayurvedic Medicine is Natural. Panchakarma – Body cleansing therapy, eliminating accumulated toxic metabolites. Rasayana – Immuno modulator, adoptogenic, cognitive, memory enhancer etc. Minimal invasive parasurgical measures like Ksharsootra – an ambulatory treatment in Fistula in ano; Leech therapy – Bio apparatus
STRENGTH OF AYURVEDA s s s Science of Holistic Health caring Body, Mind and Soul. Preventive, Promotive & Curative aspects Source of Ayurvedic Medicine is Natural. Panchakarma – Body cleansing therapy, eliminating accumulated toxic metabolites. Rasayana – Immuno modulator, adoptogenic, cognitive, memory enhancer etc. Minimal invasive parasurgical measures like Ksharsootra – an ambulatory treatment in Fistula in ano; Leech therapy – Bio apparatus
 KSHARASUTRA
KSHARASUTRA
 Important Reseach Achievments of CCRAS s s s s 777 oil for psoriasis Standardisation of Ksharsutra 20 patents obtained and 11 filed Treatment of Rhemotoid arthiritis Standardisation of Panchkarma Development of anti malaria drugs Management of diabetes, cancer, hypertension thruogh Ayurveda
Important Reseach Achievments of CCRAS s s s s 777 oil for psoriasis Standardisation of Ksharsutra 20 patents obtained and 11 filed Treatment of Rhemotoid arthiritis Standardisation of Panchkarma Development of anti malaria drugs Management of diabetes, cancer, hypertension thruogh Ayurveda
 Important Reseach Achievments of CCRUM s s s s Treatment of Vitiligo Treatment of Eczema Treatment of Sinusitis Filariasis Management of hypertension Herbal tea Database of medicinal plants
Important Reseach Achievments of CCRUM s s s s Treatment of Vitiligo Treatment of Eczema Treatment of Sinusitis Filariasis Management of hypertension Herbal tea Database of medicinal plants
 RECENT INITIATIVE s s s Traditional Knowledge Digital Library – to ensure patent is not taken by others Golden Triangle Project – to validate classical drugs and develop new drugs with modern scientific parameter Improvement of educational standards Strict compliance of Good Manufacturing Practices Emphasis on Development of Pharmacopoeial standards and drug testing facilities
RECENT INITIATIVE s s s Traditional Knowledge Digital Library – to ensure patent is not taken by others Golden Triangle Project – to validate classical drugs and develop new drugs with modern scientific parameter Improvement of educational standards Strict compliance of Good Manufacturing Practices Emphasis on Development of Pharmacopoeial standards and drug testing facilities
![Traditional Knowledge Digital Library[TKDL] I ¨ ¨ s 60, 000 formulations described in Ayurvedic Traditional Knowledge Digital Library[TKDL] I ¨ ¨ s 60, 000 formulations described in Ayurvedic](https://present5.com/presentation/225e99f039be6d9aebbb5e8c0eee41e4/image-20.jpg) Traditional Knowledge Digital Library[TKDL] I ¨ ¨ s 60, 000 formulations described in Ayurvedic & Unani texts. The library is available in English, Hindi, Spanish, French, German and Japanese languages in international patent compatible format, which is easily accessible for patent examination. The objective is to prevent bio-piracy and grant of wrong patents based on Indian Traditional Knowledge. TKDLs for Siddha & Yoga taken up. Folk medicine is being revitalized, validated, documented and published. The objective is to prevent patent claims on traditional medical knowledge available in public domain.
Traditional Knowledge Digital Library[TKDL] I ¨ ¨ s 60, 000 formulations described in Ayurvedic & Unani texts. The library is available in English, Hindi, Spanish, French, German and Japanese languages in international patent compatible format, which is easily accessible for patent examination. The objective is to prevent bio-piracy and grant of wrong patents based on Indian Traditional Knowledge. TKDLs for Siddha & Yoga taken up. Folk medicine is being revitalized, validated, documented and published. The objective is to prevent patent claims on traditional medical knowledge available in public domain.
 NATIONAL MEDICINAL PLANTS BOARD ¨ ¨ ¨ Government has setup a Medicinal Plants Board under the Chairmanship of Union Minister of Health & Family Welfare on 24 th Nov. 2000 to coordinate all activities relating to conservation, cultivation, marketing and augmenting supply of medicinal plants both for domestic consumption and export of medicinal plants. 32 States/UTs have set up state level Medicinal Plants Board. Other states are in the process of setting up of Medicinal Plants Board 72000 Acres have been brought under cultivation of medicinal plants to supply quality raw material for Ayurvedic products.
NATIONAL MEDICINAL PLANTS BOARD ¨ ¨ ¨ Government has setup a Medicinal Plants Board under the Chairmanship of Union Minister of Health & Family Welfare on 24 th Nov. 2000 to coordinate all activities relating to conservation, cultivation, marketing and augmenting supply of medicinal plants both for domestic consumption and export of medicinal plants. 32 States/UTs have set up state level Medicinal Plants Board. Other states are in the process of setting up of Medicinal Plants Board 72000 Acres have been brought under cultivation of medicinal plants to supply quality raw material for Ayurvedic products.
 POSSIBILITIES OF INTERNATIONAL CO-OPERATION s s s Survey & Documentation of traditionally used medicinal plants, remedies and practices. Exchange of experts and strategies on pharmaceutical technology, databasing & digitization, pharmacopoeias, formularies, regulatory mechanism and standardization & quality control measures. Import and export of raw materials & finished TM products as per mutual agreement
POSSIBILITIES OF INTERNATIONAL CO-OPERATION s s s Survey & Documentation of traditionally used medicinal plants, remedies and practices. Exchange of experts and strategies on pharmaceutical technology, databasing & digitization, pharmacopoeias, formularies, regulatory mechanism and standardization & quality control measures. Import and export of raw materials & finished TM products as per mutual agreement
 Contd…. . s s s Training programmes on agro-technology, manufacturing technology, quality control & standardization, R&D and collaborative scientific validation studies on TM. To organise bilateral and multilateral programmes on themes of common interest for development of TM. Formulation of common strategy for protection of TM against misappropriation and international market authorization of Traditional Herbal Medicinal Products.
Contd…. . s s s Training programmes on agro-technology, manufacturing technology, quality control & standardization, R&D and collaborative scientific validation studies on TM. To organise bilateral and multilateral programmes on themes of common interest for development of TM. Formulation of common strategy for protection of TM against misappropriation and international market authorization of Traditional Herbal Medicinal Products.
 Conclusion s Time has come to co-operate with each other. Hopefully this conference will help us to develop and grow together in the field of TM to serve our people better.
Conclusion s Time has come to co-operate with each other. Hopefully this conference will help us to develop and grow together in the field of TM to serve our people better.


