dac6784af98f6ce251fe28bf625173db.ppt
- Количество слайдов: 69

Deliver More … Deliver Excellence! Pittsburgh, PA. USA

About Caliber þ Caliber is a company focused on IT solutions for Laboratories Specially GLP/GAMP compliant. þ Established in the year 2001 þ Global Marketing HQ at Pittsburgh USA þ ISO 9001 -2000 Certified þ Well organized Development process þ Absolute leadership in Indian Pharma Industry þ Best Product Award winner--- from HYSEA þ CIO 100 Asia, best project award winner

Caliber. LIMS® Industry Verticals Ø Ø Ø Ø Pharmaceuticals Manufacturing QC Research & Drug Development Petrochemicals Water & Environment Food Testing Public health Training Management in GMP


Recognition þ þ Product — Hysea & Site approvals Project Management- CIO Asia Award Company- Deliotte Asia Award Team -- Entrepreneur of the year More important than all the above: Repeat orders

A Few of Our Valued Customers… Caliber. LIMS® þ þ þ þ Wyeth Mylan India (Matrix) Fresenius Dr. Reddy’s Ltd (5) Aurobindo (3) Unichem Medicap - Thailand Panacea BPCL (25) Dabur UK JBCPL (8 Plants) Zydus Hospira Sanofi Aventis Divis Laboratories Ltd Ajanta Pharma Repeat Orders Speak Volumes!!

Pharma Industry ……. The Journey
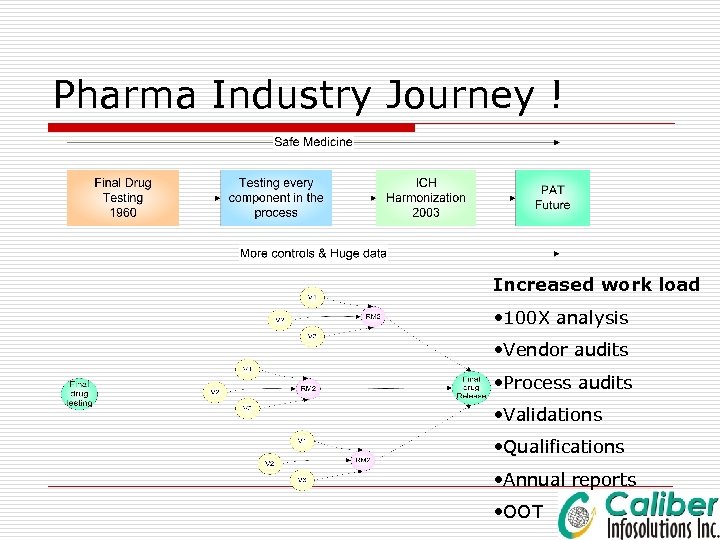
Pharma Industry Journey ! Increased work load • 100 X analysis • Vendor audits • Process audits • Validations • Qualifications • Annual reports • OOT
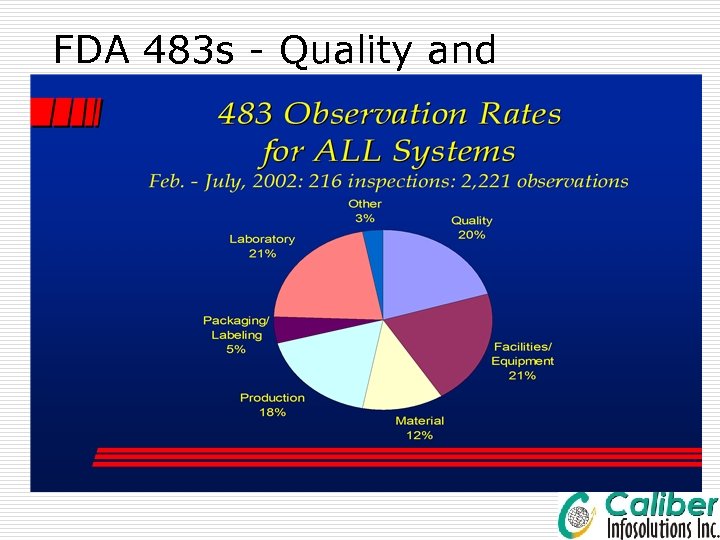
FDA 483 s - Quality and Laboratory Issues

Common Lab Errors, Common Root Cause o Transcription Errors-- Major Contributor o Calculation errors o Lack of Information o Insufficient Training o Improper Review o Lack of Controls
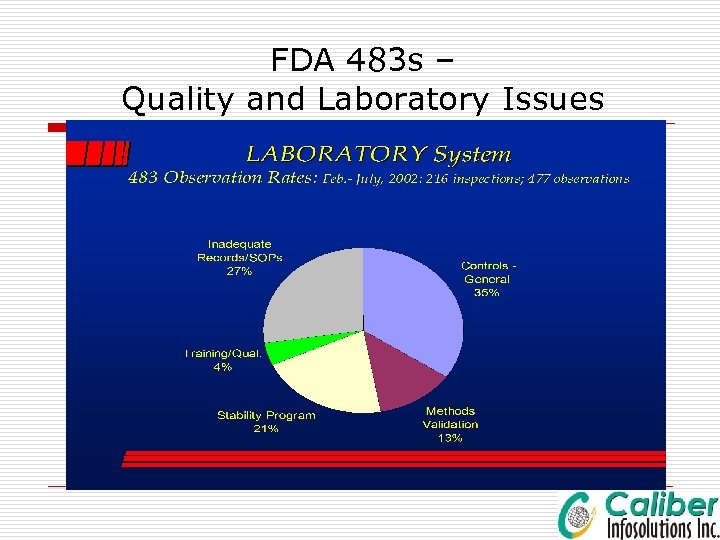
FDA 483 s – Quality and Laboratory Issues

How will LIMS help you!!

What is in it for Me? ? ü Clear job Description ü Great reduction in errors, LIMS is Error prevention system !! ü Quicker completion of the job. . ü - Deliver More! ü Automatic Adherence to the Quality System like ISO 17025 ü - Deliver Excellence!
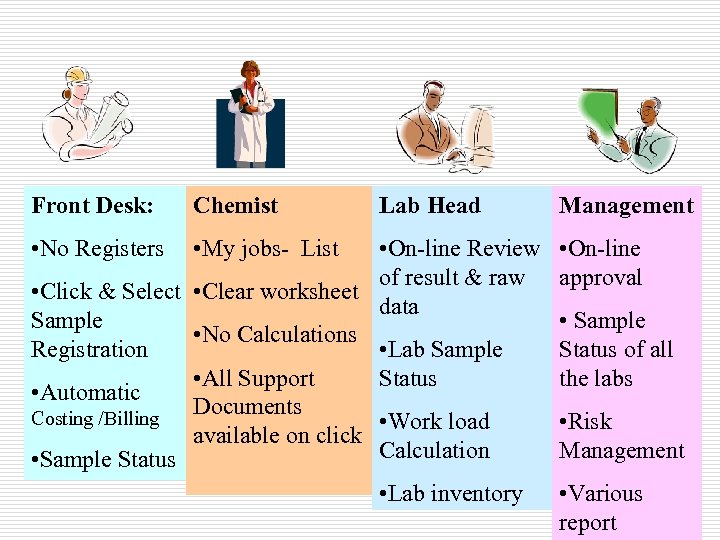
Front Desk: Chemist • No Registers • My jobs- List Lab Head Management • On-line Review • On-line of result & raw approval • Click & Select • Clear worksheet data Sample • No Calculations Registration • Lab Sample Status of all • All Support Status the labs • Automatic Documents Costing /Billing • Work load • Risk available on click Calculation Management • Sample Status • Lab inventory • Various report

Solution … Caliber. LIMS ü Fully web-based, . NET technology ü N-tier architecture, modular design ü Easy integration with ERP (certified SAP integration) ü Easy to implement, Easy to use ü Requires minimal customization, out of the box ü Model based: Requires minimal training, ü Meets/exceeds Regulatory requirements of FDA/ MHRA ü Webservices & XML based engines
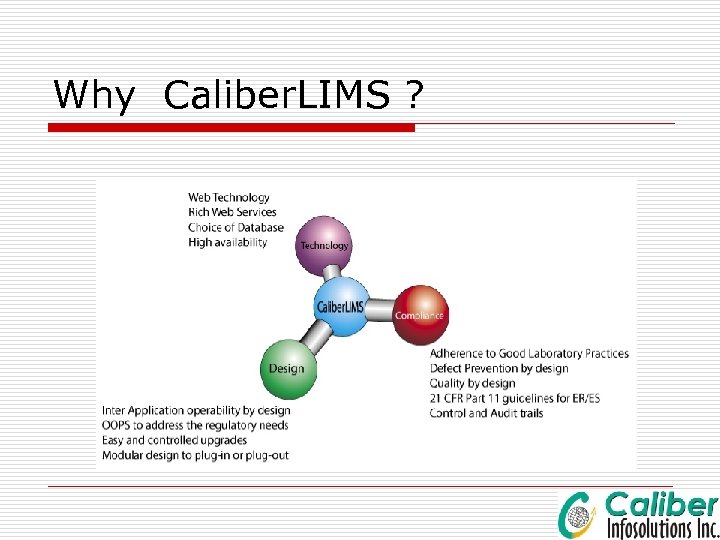
Why Caliber. LIMS ?

Why Caliber LIMS ü Technology: On web technology since Year 2001 ü Good value proposition ü Robust Framework ü GAMP compliant implementation process ü Short Learning Curve ü Proven Track record through successful international regulatory audits

Product Technology - Benefits þ Caliber. LIMS 3. X. X is built on Microsoft. Net Technology þ Built on n-tier Technology web Technology. þ Choice of DB MS SQL or Oracle þ 100 % Browser based client operations þ Specially designed OOPS model for regulated industry. þ Highly stable and leading Microsoft Architecture.

Technology Benefits þ þ þ þ Highly scalable application. Lesser maintenance. Lesser Validation burden. Lower cost of ownership. Easier Upgrades. Shorter Learning curve. Quick ROI. …… Hence it is a preferred product!!

Caliber. LIMS Features End- to - End Lab Management Solution Easy to Master configurable workflows Shorter Learning Curve Highly modular, hence implementation can be staggered, if desired. þ Comprehensive functionality to address all common lab practices, see it to believe it ! þ Configurable enforcement of GLP/FDA norms þ Reports to address all needs of Lab Management þ þ
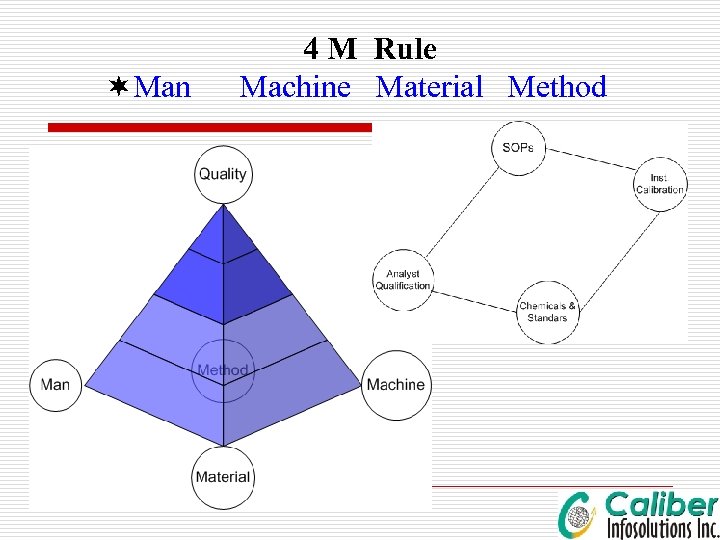
¬Man 4 M Rule Machine Material Method

Sample Manager ……. . Workflow
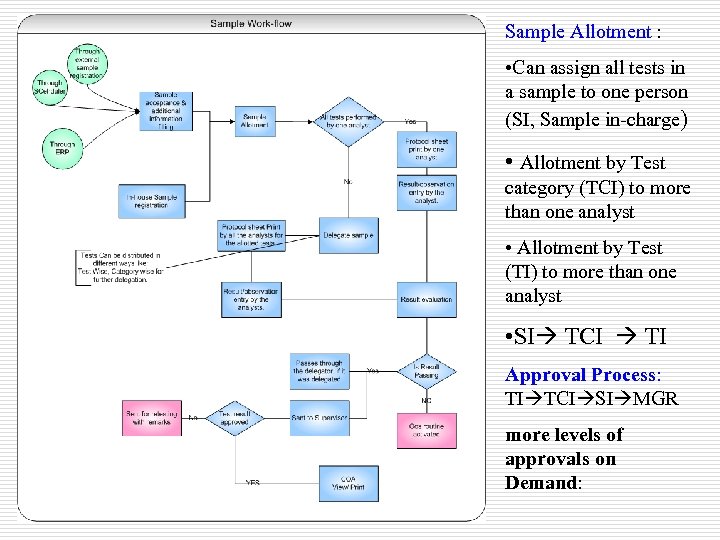
Sample Allotment : • Can assign all tests in a sample to one person (SI, Sample in-charge) • Allotment by Test category (TCI) to more than one analyst • Allotment by Test (TI) to more than one analyst • SI TCI TI Approval Process: TI TCI SI MGR more levels of approvals on Demand:
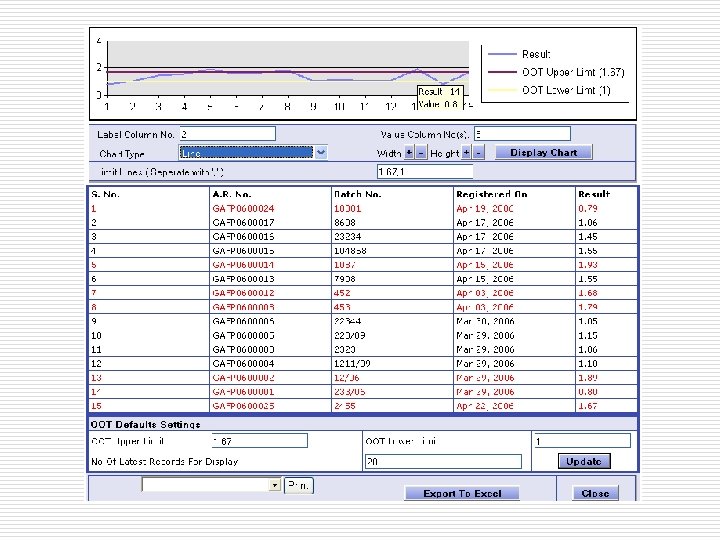
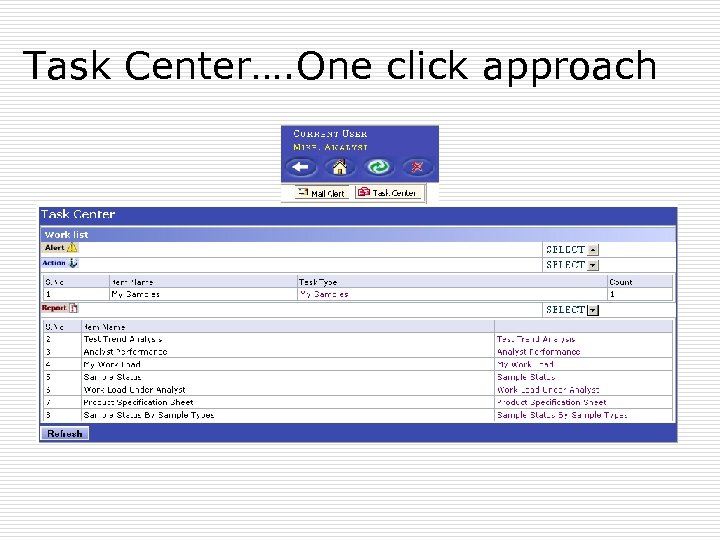
Task Center…. One click approach
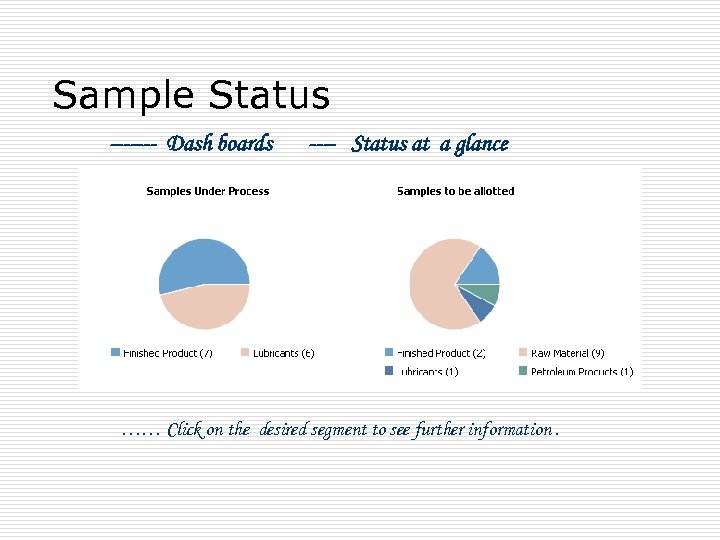
Sample Status ------- Dash boards ---- Status at a glance …… Click on the desired segment to see further information.
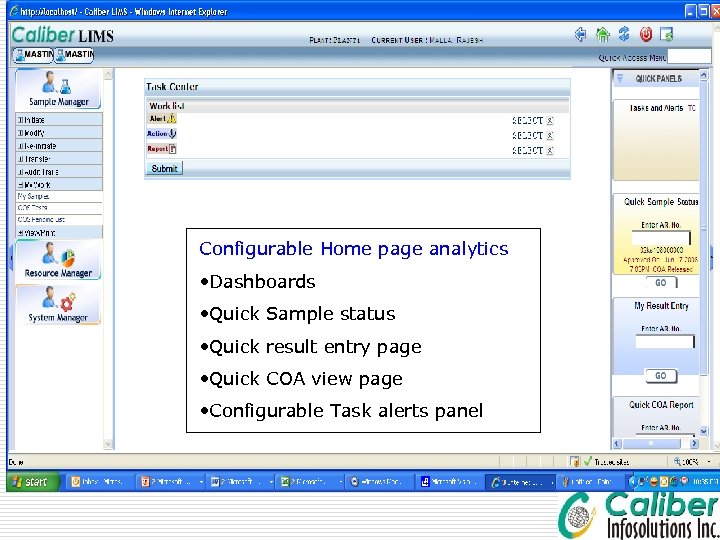
Configurable Home page analytics • Dashboards • Quick Sample status • Quick result entry page • Quick COA view page • Configurable Task alerts panel
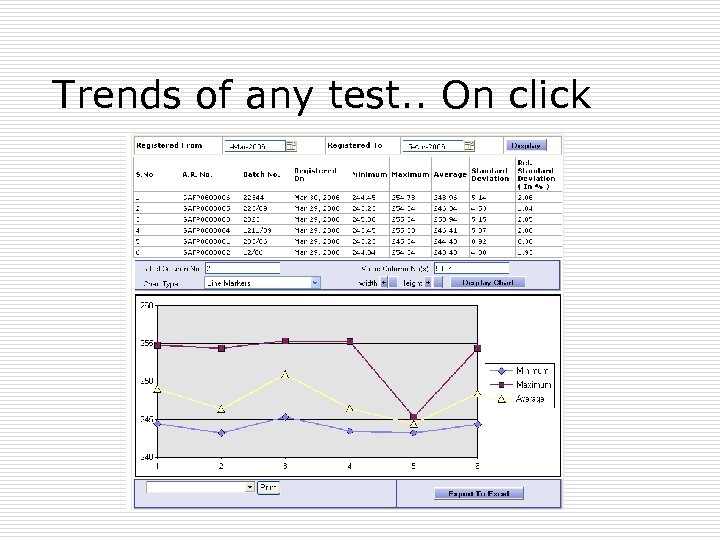
Trends of any test. . On click
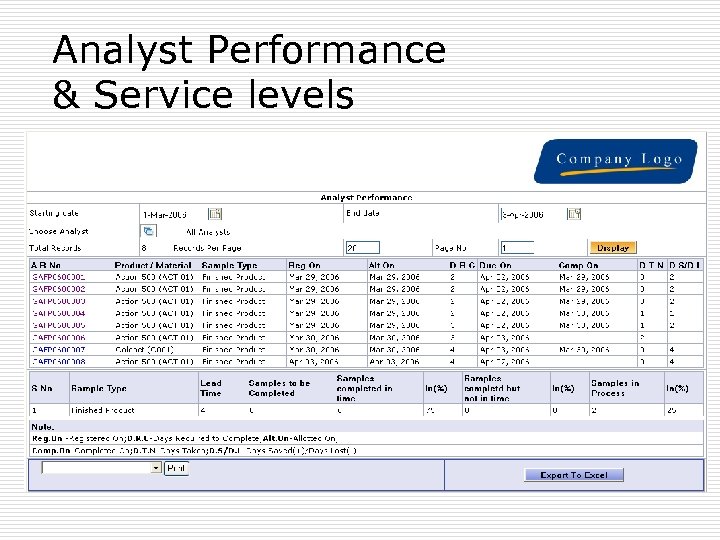
Analyst Performance & Service levels

Sample Manager Demo
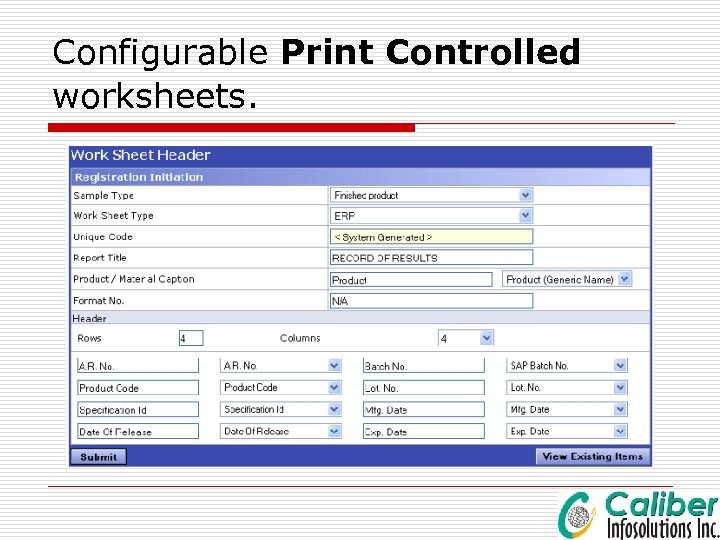
Configurable Print Controlled worksheets.
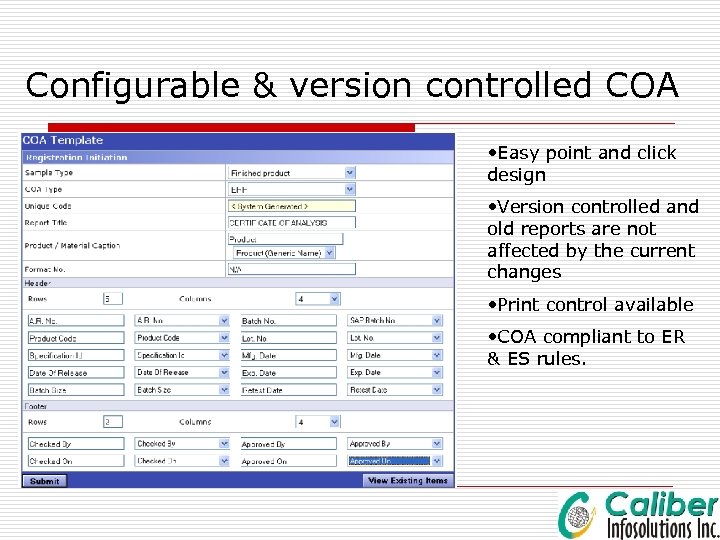
Configurable & version controlled COA • Easy point and click design • Version controlled and old reports are not affected by the current changes • Print control available • COA compliant to ER & ES rules.

Technical Benefits Caliber. LIMS Framework þ Caliber. LIMS has been repositioned as an Enterprise Quality Management Frame work rather than just a LIMS þ It is a one place to manage all laboratory functions sitting right at one desktop þ Application integration is an important aspect of LIMS 3. x. x
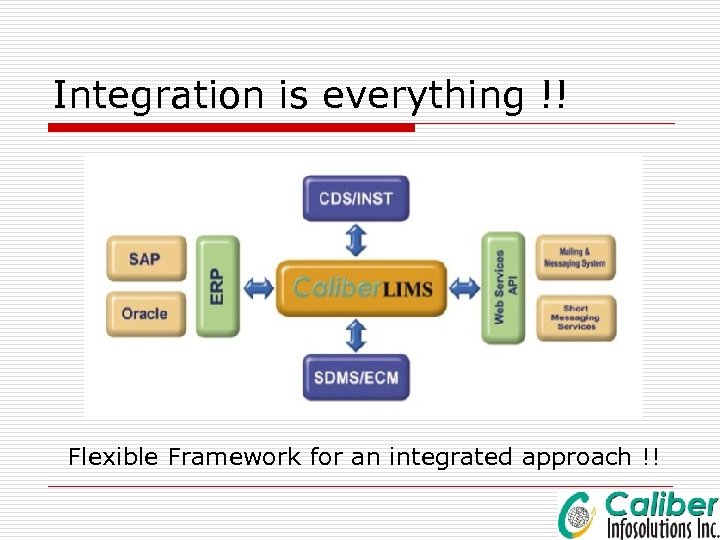
Integration is everything !! Flexible Framework for an integrated approach !!

Application integration o Caliber. LIMS can be integrated with applications like: ERP like SAP MES Historians Instrument Interface tools Report Generators

Functional Benefits High Value addition optional Modules 1. Chemicals Management 2. Working Standards 3. Ref. Standards 4. Volumetric Solutions 5. **Analyst Qualification 6. Training Management 7. Column Management 8. **Instrument Calibration Mgmt. 9. **Stability Management 9. Microbiology Culture Management. 10. Media Management 11. Label Printing. 12. Lab DMS. 13. Market Complaints. 14. Change Management. 15. Deviation Management. 16. **Outsourcing/ Reduced testing. 17. Lab Costing Module

Just Not Inventory Modules… …. Much more than that !! Working Standards 1. Get the material Get it analyzed by 3 -4 analysts check RSD if OK approve the material 2. Distribute the material in to bottles (may be one every month) 3. Use bottle after bottle 4. Usage tight integration. i. e unless the usage is recorded, result confirmation is not possible 5. Maintain consumption records for all the bottles (stock cards)

Instrument Management : þ þ þ þ þ Asset information like. . History Calibration scheduling, Calendar Calibration Certificate generation Maintenance tracking, trend analysis. Problem reporting Service logs Internal, external calibration tracking Calibration data storage Hyperlinks to Validation or SOP docs

Volumetric Solutions: þ Prepare- by using Templates for the variable quantity þ Standardize þ Schedule a re-standardization time þ Get alerts þ Record Usage þ Usage throuh tight Integration

Chemical Reagent Preparation and Inventory þ Template for Reagent Preparation for a standard volume of reagent þ Prepare reagent within tolerance limits þ Record the usage þ Get Stock cards, inventory alerts and validity alerts

Configured COA: Unique Feature ! Problem: You have tested a material batch with in-house specifications and you want to release the COA with a different specifications with the same test results. Solution: Release differently configured COA without altering the results and AR no under strict regulatory guidelines o Analyze the batch (Bulk FP) o Release with n Different Spec Description n Different Customers n Different Packing types n May be Different Language …. . Everything Under strict regulatory guidelines and audit trails.

Compiled COA : Unique Feature !! Problem: You have analyzed a batch earlier with 10 tests, now customer wants one more test to be conducted and a new COA released. Solution: Just conduct the new test and combine the earlier test results and the new one and release COA within regulatory guidelines þ þ Do additional testing Add additional testing to the existing analysis Release the COA Keep track of all such releases

Control Sample Management þ Keep track of the samples which need to stored. þ Record: Storage place, Quantity, conditions. þ Pull the sample through a Requestapproval process þ Re-keep the un-used sample þ Get alerts for Sample disposing þ Reports: Stock, withdrawal, distruction

Stability Studies Management

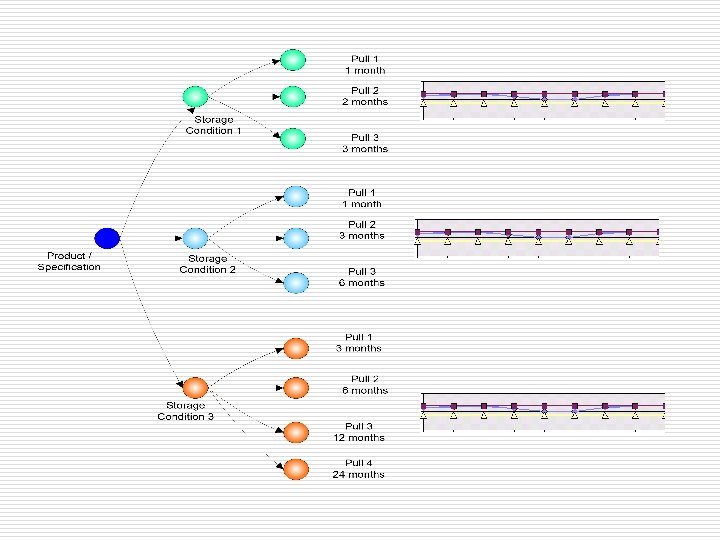
Stability Protocol þ Dedicated Module to Manage complete Stability Studies. þ A stability Protocol can be registered with all the necessary details like: þ Different Conditions with independent set of tests. þ No. of Pulls per condition. þ Associated details of a condition and study. þ Can be implemented as Stand alone stability Management System

Stability Protocol Registration þ A Product/ Material can have multiple storage conditions þ Each Storage Condition can have multiple Sample pull frequencies registered þ For Each condition tests to be carried out can be selected. þ Up on completion of the sample approval, trends can be obtained

Stability Protocol þ Stability protocol registration is highly configurable to have various input fields relevant to the study. þ User configurable to all types of products/Materials like: Finished Products. Raw Material, Packing Material þ Dynamic conditions and pulls at the time of Protocol registration þ Configurable Protocol Approval Process þ Once a protocol is approved, changes can be made as per the present stage of the protocol. þ All such changes are tracked and maintained in a audit trail.
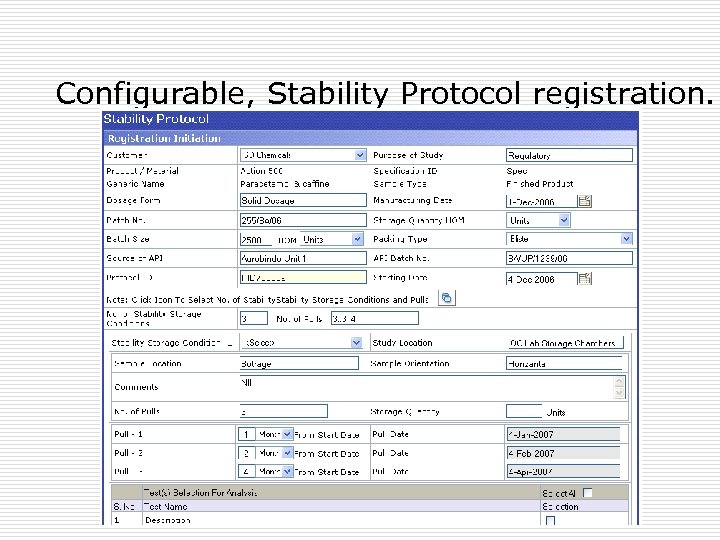
Configurable, Stability Protocol registration.

Other Supporting features þ Stability study specific Worksheet generation. þ Copy control of the worksheets þ Stability specific COA header and footer configuration þ Stability specific test trends
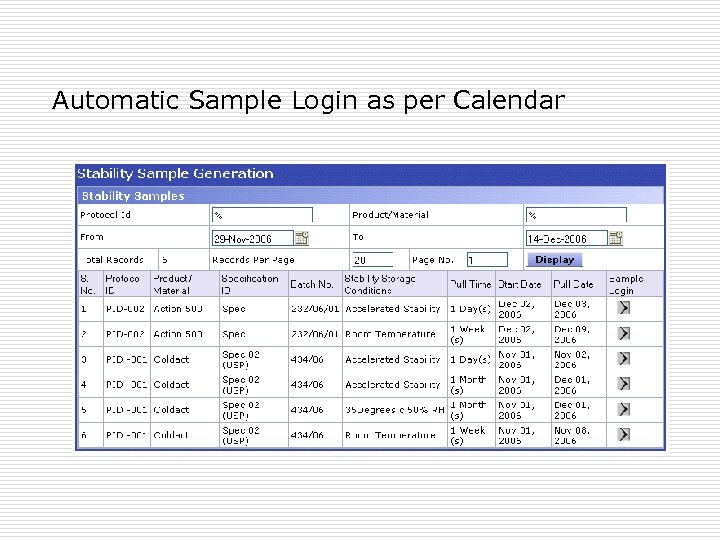
Automatic Sample Login as per Calendar

Stability Sample Chambers (Optional) þ Define a chamber with all attributes with Storage positions defined. þ Stability chamber is assigned to Condition þ Sample storage along with Quantity stored þ Sample withdrawal with Quantity withdrawn þ Stock inventory reports
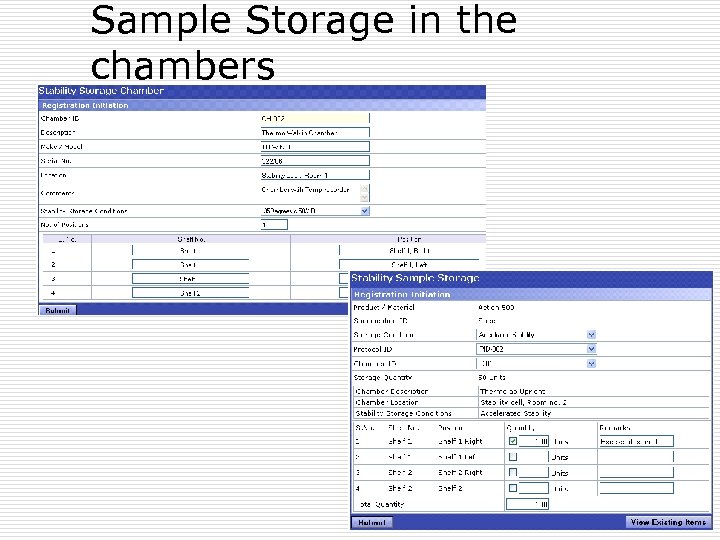
Sample Storage in the chambers

Stability Studies reports þ Reports on click of the mouse: þ Sample Analytical Test report (COA) for each sample þ Very comprehensive , comparative report for one product, different batches, Selected condition, selected Tests. þ Trend graphs þ Annual calendar þ Reports as per protocols þ Result compilation
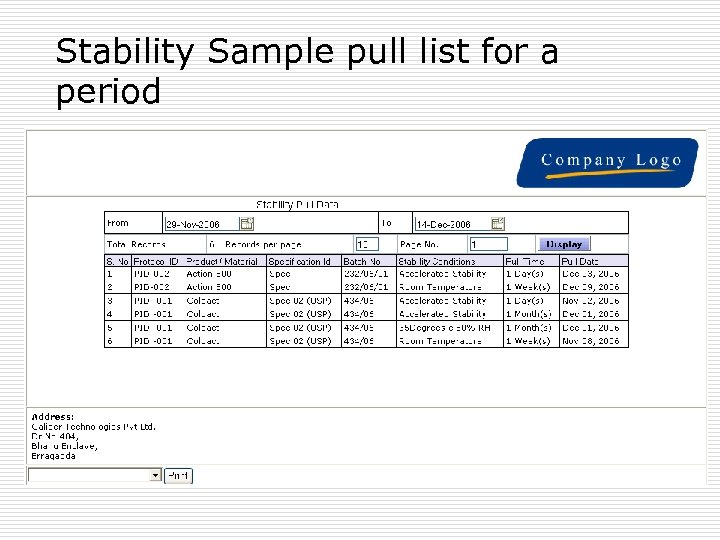
Stability Sample pull list for a period
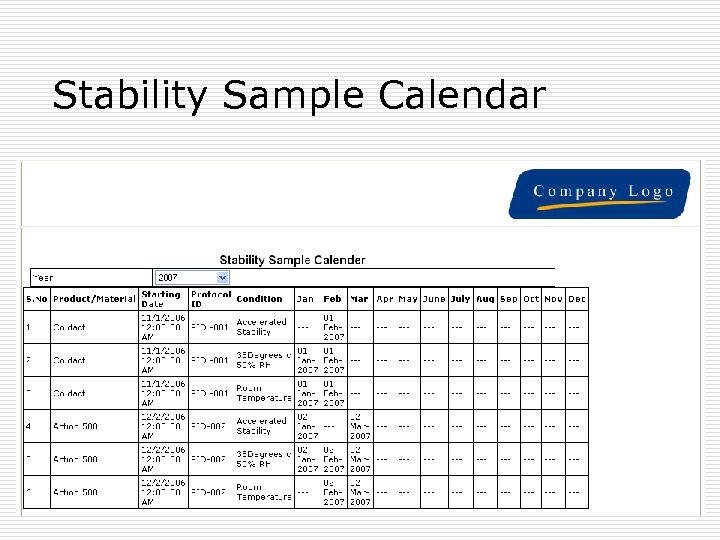
Stability Sample Calendar

LIMS Configuration

Configuration o Objects o There are in excess of 100 Objects in Caliber. LIMS 3. 0 and every object has inherent capability / configurability to include activities like: Initiation, Approval and E-sign challenge and generation of Audit Trails.
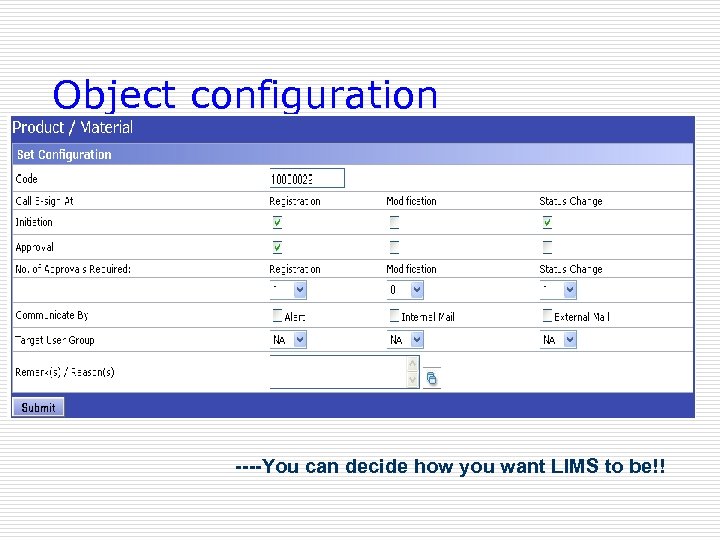
Object configuration ----You can decide how you want LIMS to be!!

Architectural details !

Application Architecture þ Developed on MS ASP. net þ Caliber. LIMS 3. XX versions are developed on specially designed OOPS model to suite the regulated industry practices. (GLP) þ More than 100 objects to make the system highly configurable. þ Increased configurability of the objects
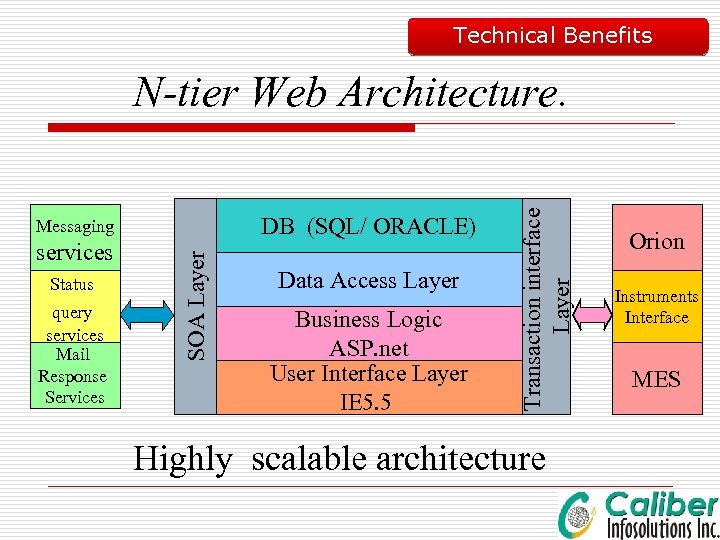
Technical Benefits DB (SQL/ ORACLE) services Status query services Mail Response Services SOA Layer Messaging Data Access Layer Business Logic ASP. net User Interface Layer IE 5. 5 Transaction interface Layer N-tier Web Architecture. Highly scalable architecture Orion Instruments Interface MES

Software Environment Application server: Windows 2003 Front End: User Interface: IE 5. 5 Back end: 1. SQL 2000 Above 2. Oracle 9 i & above Web server: IIS 6. 0
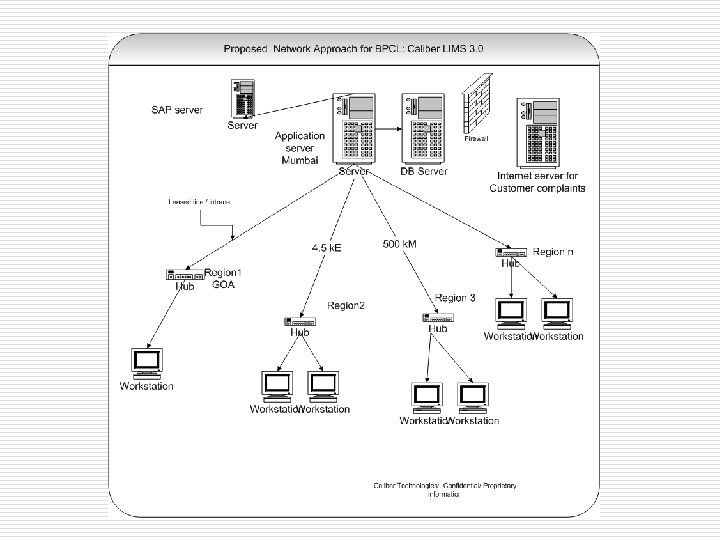

Regulatory Compliance

Regulatory/ Quality Standards þ 21 CFR Part 11 Code for Electronic Signatures and Electronic Records þ GLP Good Laboratory Practices þ ISO 17025 þ NABL þ GAMP compliant documentation
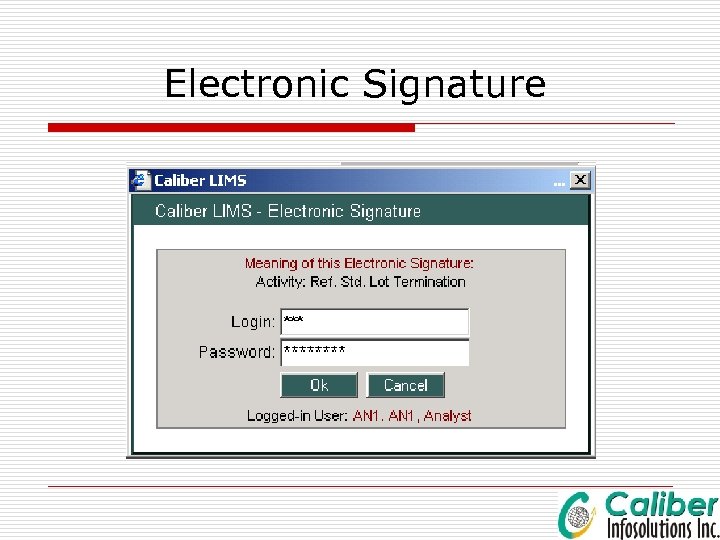
Electronic Signature

Deliver More… deliver excellence !! Caliber Infosolutions, Inc. 100, Technology Dr. Suite 400 Pittsburgh. PA 15219 sekhar@caliberinfosolutions. com www. caliberinfosolutions. com Caliber Technologies Pvt. Ltd. 401, 406, Bhanu Enclave, Model Colony, Hyderabad-500038, INDIA Tel : 91 40 23811322, sekhar@caliberindia. com URL: www. caliberindia. com
dac6784af98f6ce251fe28bf625173db.ppt