97362a880d3ae5f75d118451889c9144.ppt
- Количество слайдов: 21
 DACEHTA MTV Den europæiske udvikling på MTV – speciallægekursus i samfundsmedicin området - EUnet. HTA-projektet Nye veje for MTV, 14. september 2007 Finn Børlum Kristensen Enhed for MTV, Sundhedsstyrelsen
DACEHTA MTV Den europæiske udvikling på MTV – speciallægekursus i samfundsmedicin området - EUnet. HTA-projektet Nye veje for MTV, 14. september 2007 Finn Børlum Kristensen Enhed for MTV, Sundhedsstyrelsen
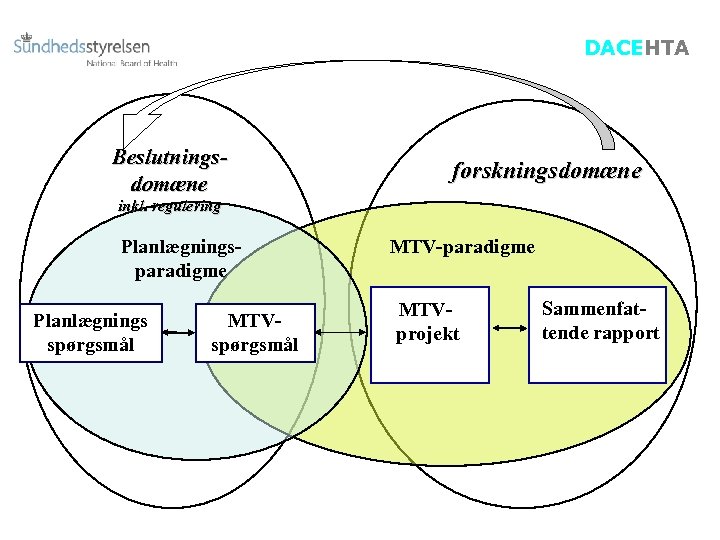 DACEHTA Beslutningsdomæne forskningsdomæne inkl. regulering Planlægningsparadigme Planlægnings spørgsmål MTV-paradigme MTVprojekt Sammenfattende rapport
DACEHTA Beslutningsdomæne forskningsdomæne inkl. regulering Planlægningsparadigme Planlægnings spørgsmål MTV-paradigme MTVprojekt Sammenfattende rapport
 DACEHTA Disposition • • • EUnet. HTA – baggrund EUnet. HTA – partnere Formål Opgaver Seneste udviklinger National nytte af EUnet. HTA
DACEHTA Disposition • • • EUnet. HTA – baggrund EUnet. HTA – partnere Formål Opgaver Seneste udviklinger National nytte af EUnet. HTA
 DACEHTA MTV er en international aktivitet • • undgå unødvendigt dobbeltarbejde bruge og udvikle ”best practice” dele de opgaver, der kan deles tilpasning til nationale/regionale sammenhænge • men…. . sundhedsplanlægning og politik er et national/regionalt anliggende
DACEHTA MTV er en international aktivitet • • undgå unødvendigt dobbeltarbejde bruge og udvikle ”best practice” dele de opgaver, der kan deles tilpasning til nationale/regionale sammenhænge • men…. . sundhedsplanlægning og politik er et national/regionalt anliggende
 DACEHTA EU High Level Group on Medical Services – november 2004 • Rapport fra EU kommissionens High level Group, 30. november 2004, konkluderede, at: • ”MTV er blevet en politisk prioritet, og der er et tvingende behov for etablering af et permanent europæisk netværk for MTV”
DACEHTA EU High Level Group on Medical Services – november 2004 • Rapport fra EU kommissionens High level Group, 30. november 2004, konkluderede, at: • ”MTV er blevet en politisk prioritet, og der er et tvingende behov for etablering af et permanent europæisk netværk for MTV”
 DACEHTA Udviklingen i 2005 • Januar: DG SANCO Public Health Work Plan 2005 (2006) MTV er et prioriteret område • EU Kommissær Kyprianou fremhæver, at : ”det er blevet anerkendt, at det er fornuftigt at etablere et permanent europæisk netværk for MTV…. Jeg håber at netværket etableres i år med støtte fra Public Health programmet”
DACEHTA Udviklingen i 2005 • Januar: DG SANCO Public Health Work Plan 2005 (2006) MTV er et prioriteret område • EU Kommissær Kyprianou fremhæver, at : ”det er blevet anerkendt, at det er fornuftigt at etablere et permanent europæisk netværk for MTV…. Jeg håber at netværket etableres i år med støtte fra Public Health programmet”
 European network for Health Technology Assessment European network for HTA, EUnet. HTA | www. eunethta. net
European network for Health Technology Assessment European network for HTA, EUnet. HTA | www. eunethta. net
 EUnet. HTA partnere • 59 partnere – 34 associated partners (nationale/regionale MTVinstitutioner, sundhedsministerier, forskningsinstitutioner, internationale institutioner – 25 collaborating partners (fx WHO, OECD, Europarådet) • • 24 EU lande (- Slovakiet) 2 EØS lande (Norge, Island) Schweiz 4 ikke-europæiske lande (Israel, Australien, Canada, USA) EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
EUnet. HTA partnere • 59 partnere – 34 associated partners (nationale/regionale MTVinstitutioner, sundhedsministerier, forskningsinstitutioner, internationale institutioner – 25 collaborating partners (fx WHO, OECD, Europarådet) • • 24 EU lande (- Slovakiet) 2 EØS lande (Norge, Island) Schweiz 4 ikke-europæiske lande (Israel, Australien, Canada, USA) EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
 Formål I Overordnet formål: • at etablere et effektivt og permanent europæisk netværk for MTV Strategiske formål: • at sikre bedre koordination af MTV-aktiviteter og at udgå dobbeltarbejde → mere effektiv brug af national MTVressourcer • at øge produktion af MTV og dermed input til beslutningstagning • at styrke koblingen mellem MTV og beslutningstagning • at støtte lande med begrænset erfaring med MTV EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
Formål I Overordnet formål: • at etablere et effektivt og permanent europæisk netværk for MTV Strategiske formål: • at sikre bedre koordination af MTV-aktiviteter og at udgå dobbeltarbejde → mere effektiv brug af national MTVressourcer • at øge produktion af MTV og dermed input til beslutningstagning • at styrke koblingen mellem MTV og beslutningstagning • at støtte lande med begrænset erfaring med MTV EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
 Formål II To praktiske formål: 1. at skabe en organisatorisk ramme for et permanent netværk for MTV 2. at udvikle praktiske redskaber der kan anvendes inden for rammerne EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
Formål II To praktiske formål: 1. at skabe en organisatorisk ramme for et permanent netværk for MTV 2. at udvikle praktiske redskaber der kan anvendes inden for rammerne EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
 Opgaver – EUnet. HTA Work Packages (WPs) WP 1 WP 2 WP 3 Koordination (EMTV, Danmark) Kommunikation (SBU, Sverige & DAHTA, Tyskland) Evaluering (NOKC, Norge) WP 4 WP 5 Kerne MTV (Fin. OHTA, Finland) Tilpasning af eksisterende MTV’er og Kerne MTV til andre kontekster (NCCHTA, England) WP 6 WP 7 Kobling mellem MTV og policy (EMTV, Danmark) Monitorering af kommende teknologier og prioritering af MTV (HAS, Frankrig & LBI@HTA, Østrig) MTV supportsystem i lande uden institutionaliseret MTV WP 8 EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
Opgaver – EUnet. HTA Work Packages (WPs) WP 1 WP 2 WP 3 Koordination (EMTV, Danmark) Kommunikation (SBU, Sverige & DAHTA, Tyskland) Evaluering (NOKC, Norge) WP 4 WP 5 Kerne MTV (Fin. OHTA, Finland) Tilpasning af eksisterende MTV’er og Kerne MTV til andre kontekster (NCCHTA, England) WP 6 WP 7 Kobling mellem MTV og policy (EMTV, Danmark) Monitorering af kommende teknologier og prioritering af MTV (HAS, Frankrig & LBI@HTA, Østrig) MTV supportsystem i lande uden institutionaliseret MTV WP 8 EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
 Starting points for Core Model work • HTA is implemented differently across European countries – Reduced applicability of foreign reports • HTA reports lack a standardised information structure – Extraction of data from reports may be difficult • • • Attempt to define and standardise elements of an HTA to facilitate shared understanding of HTA and promote the international use of HTA results Only a part of all possible health technologies can be addressed through EUnet. HTA models for two types of technologies. . . but it is a start Two types of core HTA are developed 1) an intervention (example: Drug Eluding Stents) 2) a diagnostic technology (example: Multislice CT of Coranary Arteries) EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
Starting points for Core Model work • HTA is implemented differently across European countries – Reduced applicability of foreign reports • HTA reports lack a standardised information structure – Extraction of data from reports may be difficult • • • Attempt to define and standardise elements of an HTA to facilitate shared understanding of HTA and promote the international use of HTA results Only a part of all possible health technologies can be addressed through EUnet. HTA models for two types of technologies. . . but it is a start Two types of core HTA are developed 1) an intervention (example: Drug Eluding Stents) 2) a diagnostic technology (example: Multislice CT of Coranary Arteries) EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
 The Common Core of evidence, concepts, and methods Common framework n tio Common taxonomy of HTA concepts Systematic assessment of • Clinical evidence • Safety • Organisation • Economy • Societal/ethical issues r o tf s g s re rk in o pr o W - using common generic HTA-methodological framework Common core of evidence that Question / evidence can be shared element cards EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu no , ita c
The Common Core of evidence, concepts, and methods Common framework n tio Common taxonomy of HTA concepts Systematic assessment of • Clinical evidence • Safety • Organisation • Economy • Societal/ethical issues r o tf s g s re rk in o pr o W - using common generic HTA-methodological framework Common core of evidence that Question / evidence can be shared element cards EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu no , ita c
 Domain • • • r t fo no ss, re rog p k in r Wo n atio cit Current use of the technology (implementation level) Description and technical characteristics of technology Safety Effectiveness Costs, economic evaluation Ethical aspects Organisational aspects Social aspects Legal aspects EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu
Domain • • • r t fo no ss, re rog p k in r Wo n atio cit Current use of the technology (implementation level) Description and technical characteristics of technology Safety Effectiveness Costs, economic evaluation Ethical aspects Organisational aspects Social aspects Legal aspects EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu
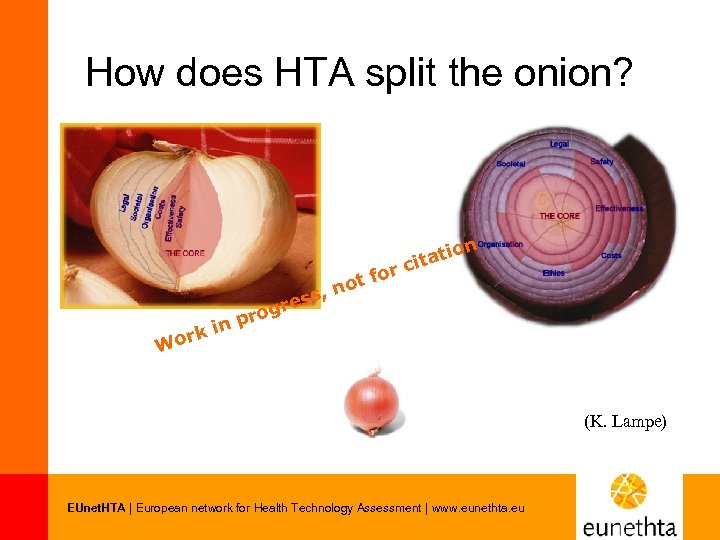 How does HTA split the onion? or ot f n ss, re rog p k in r Wo on tati ci (K. Lampe) EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu
How does HTA split the onion? or ot f n ss, re rog p k in r Wo on tati ci (K. Lampe) EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu
 n atio cit r Elements are described in further detail in t fo no ss, re element cards rog p n rk i o W The following information is available in each card: • Element ID • Domain • Topic • Issue • Clarification • Importance • Information sources • Transferability • Reference • Relations • Status EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu
n atio cit r Elements are described in further detail in t fo no ss, re element cards rog p n rk i o W The following information is available in each card: • Element ID • Domain • Topic • Issue • Clarification • Importance • Information sources • Transferability • Reference • Relations • Status EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu
 WP 5: Adapting existing and ‘common’ core HTA Develop a toolkit for adapting the “core” that may be shareable in existing HTA reports made for one country into advice appropriate to other contexts (social, political, economic and health system) Produce a glossary of ‘adaptation’ terms EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu
WP 5: Adapting existing and ‘common’ core HTA Develop a toolkit for adapting the “core” that may be shareable in existing HTA reports made for one country into advice appropriate to other contexts (social, political, economic and health system) Produce a glossary of ‘adaptation’ terms EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu
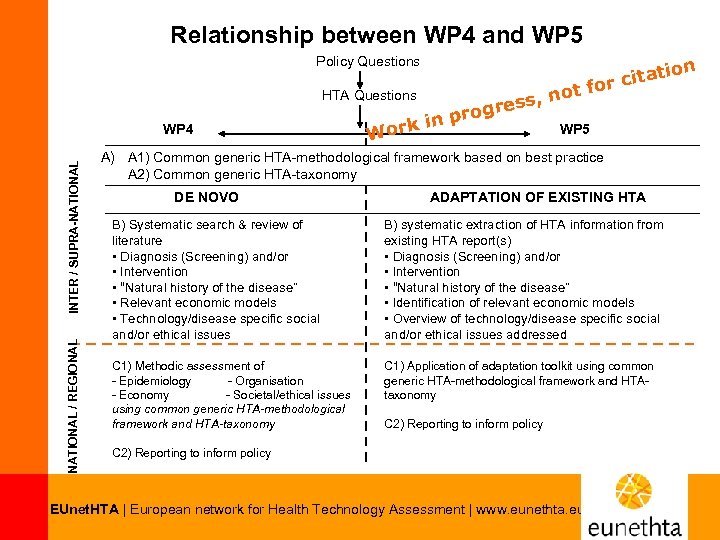 Relationship between WP 4 and WP 5 Policy Questions NATIONAL / REGIONAL INTER / SUPRA-NATIONAL WP 4 t r cita t fo HTA Questions s, no res prog n WP 5 ork i W A) A 1) Common generic HTA-methodological framework based on best practice A 2) Common generic HTA-taxonomy DE NOVO ADAPTATION OF EXISTING HTA B) Systematic search & review of literature • Diagnosis (Screening) and/or • Intervention • “Natural history of the disease” • Relevant economic models • Technology/disease specific social and/or ethical issues B) systematic extraction of HTA information from existing HTA report(s) • Diagnosis (Screening) and/or • Intervention • “Natural history of the disease” • Identification of relevant economic models • Overview of technology/disease specific social and/or ethical issues addressed C 1) Methodic assessment of - Epidemiology - Organisation - Economy - Societal/ethical issues using common generic HTA-methodological framework and HTA-taxonomy C 1) Application of adaptation toolkit using common generic HTA-methodological framework and HTAtaxonomy C 2) Reporting to inform policy EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu ion
Relationship between WP 4 and WP 5 Policy Questions NATIONAL / REGIONAL INTER / SUPRA-NATIONAL WP 4 t r cita t fo HTA Questions s, no res prog n WP 5 ork i W A) A 1) Common generic HTA-methodological framework based on best practice A 2) Common generic HTA-taxonomy DE NOVO ADAPTATION OF EXISTING HTA B) Systematic search & review of literature • Diagnosis (Screening) and/or • Intervention • “Natural history of the disease” • Relevant economic models • Technology/disease specific social and/or ethical issues B) systematic extraction of HTA information from existing HTA report(s) • Diagnosis (Screening) and/or • Intervention • “Natural history of the disease” • Identification of relevant economic models • Overview of technology/disease specific social and/or ethical issues addressed C 1) Methodic assessment of - Epidemiology - Organisation - Economy - Societal/ethical issues using common generic HTA-methodological framework and HTA-taxonomy C 1) Application of adaptation toolkit using common generic HTA-methodological framework and HTAtaxonomy C 2) Reporting to inform policy EUnet. HTA | European network for Health Technology Assessment | www. eunethta. eu ion
 EUnet. HTA Collaboration 2009+ • • Intern EUnet. HTA afklaring frem til oktober 2007 Offentlig høringsproces november – december 2007 Samstemning med EU Strategy for Health Services m. v. 2008 EUnet. HTA konference, Institute Pasteur, Paris 20. november 2008 EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
EUnet. HTA Collaboration 2009+ • • Intern EUnet. HTA afklaring frem til oktober 2007 Offentlig høringsproces november – december 2007 Samstemning med EU Strategy for Health Services m. v. 2008 EUnet. HTA konference, Institute Pasteur, Paris 20. november 2008 EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
 National nytte af EUnet. HTA • Transparente og sammenlignelige metoder på tværs af europæiske MTVrapporter • Øget produktion som følge af muligheden for at tilpasse udenlandske MTV-rapporter • Mulighed for at opnå øgede erfaringer med anvendelsen af MTV på tværs af Europa EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
National nytte af EUnet. HTA • Transparente og sammenlignelige metoder på tværs af europæiske MTVrapporter • Øget produktion som følge af muligheden for at tilpasse udenlandske MTV-rapporter • Mulighed for at opnå øgede erfaringer med anvendelsen af MTV på tværs af Europa EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
 WWW. eunethta. net EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net
WWW. eunethta. net EUnet. HTA | European network for Health Technology Assessment | www. eunethta. net


