a8298b3408f2848a0b4d773007b1a751.ppt
- Количество слайдов: 28
 Current PFP Workgroups • Implementation & Communication • Information Technology • National Feed Assignment • PETNet Expansion • Training & Certification • Local Engagement • Response & Recall 1
Current PFP Workgroups • Implementation & Communication • Information Technology • National Feed Assignment • PETNet Expansion • Training & Certification • Local Engagement • Response & Recall 1
 Implementation & Communication WG Charge Current Projects • Develop an implementation plan to facilitate the use of IFSS best • practices across federal, state, local, tribal, & territorial governments. • WG Co-Chair: Tracey Forfa (CVM) WG Co-Chair: Mel Plaisier (ORA) WG Co-Chair: Roberta Wagner (CFSAN) Implement Local Work Planning Document from 50 -State Workshop Implement Joint Inspection framework from 50 -State Workshop Implement National Standards for Food/Feed Testing Guidance Document • Implement After Action Review process • Implement Performance Measures projects • Develop a Communication Strategy for PFP 8
Implementation & Communication WG Charge Current Projects • Develop an implementation plan to facilitate the use of IFSS best • practices across federal, state, local, tribal, & territorial governments. • WG Co-Chair: Tracey Forfa (CVM) WG Co-Chair: Mel Plaisier (ORA) WG Co-Chair: Roberta Wagner (CFSAN) Implement Local Work Planning Document from 50 -State Workshop Implement Joint Inspection framework from 50 -State Workshop Implement National Standards for Food/Feed Testing Guidance Document • Implement After Action Review process • Implement Performance Measures projects • Develop a Communication Strategy for PFP 8
 Implementation & Communication WG (FDA Reps) • • • Tracey Forfa (CVM) Mel Plaisier (ORA) Steve Solomon (ORA) Roberta Wagner (CFSAN) Kara Morgan (OC) Don Burr (CFSAN) Hitelia Castellanos (ORA) Tara Doran (ORA) Carie Jasperse (OCC) Andrew Becher (OCC) Cathy Mc. Dermott (OFVM) Sherri Mc. Garry (OFVM) • • • Tim Mc. Grath (ORA) Jenny Murphy (CVM) Eric Nelson (CVM) Deep Patel (OC) Sherry Purvis-Wynn (ORA) Shannon Rutherford (CVM) Pete Salsbury (CFSAN) Marianna Solomotis (CFSAN) Lawrence Sproul (ORA) Nadine Steinberg (CVM) Iris Valentin-Bon (OC) 3 Julianna Wittig (CVM)
Implementation & Communication WG (FDA Reps) • • • Tracey Forfa (CVM) Mel Plaisier (ORA) Steve Solomon (ORA) Roberta Wagner (CFSAN) Kara Morgan (OC) Don Burr (CFSAN) Hitelia Castellanos (ORA) Tara Doran (ORA) Carie Jasperse (OCC) Andrew Becher (OCC) Cathy Mc. Dermott (OFVM) Sherri Mc. Garry (OFVM) • • • Tim Mc. Grath (ORA) Jenny Murphy (CVM) Eric Nelson (CVM) Deep Patel (OC) Sherry Purvis-Wynn (ORA) Shannon Rutherford (CVM) Pete Salsbury (CFSAN) Marianna Solomotis (CFSAN) Lawrence Sproul (ORA) Nadine Steinberg (CVM) Iris Valentin-Bon (OC) 3 Julianna Wittig (CVM)
 Information Technology (IT) WG Charge Defining and understanding the requirements for developing an integrated electronic information management backbone, and undertaking technical projects to create an interoperable and integrated national food safety system. WG Co-Chair: Tiffani Onifade (FL) WG Co-Chair: Don Jaccard (FDA) Current Projects • Developing a data dictionary to standardize terminology and facilitate the capture & exchange of common data elements • Establishing a plan to increase the number of agencies capturing baseline set of common data elements for a subset of programs/activities 4
Information Technology (IT) WG Charge Defining and understanding the requirements for developing an integrated electronic information management backbone, and undertaking technical projects to create an interoperable and integrated national food safety system. WG Co-Chair: Tiffani Onifade (FL) WG Co-Chair: Don Jaccard (FDA) Current Projects • Developing a data dictionary to standardize terminology and facilitate the capture & exchange of common data elements • Establishing a plan to increase the number of agencies capturing baseline set of common data elements for a subset of programs/activities 4
 Information Technology WG (FDA Reps) • Don Jaccard (OFVM) • Barbara Cassens (ORA) • Lauren Gore (ORA) 5
Information Technology WG (FDA Reps) • Don Jaccard (OFVM) • Barbara Cassens (ORA) • Lauren Gore (ORA) 5
 Local Engagement WG Charge Current Projects To incorporate the Food Safety • Modernization Act (FSMA) deliverables that include “local” agencies with PFP projects & provide specific recommendations for engagement of local agencies within the • vision for an IFSS. WG Co-Chair: Pat Maloney (MA) WG Co-Chair: Liz O’Malley (FDA) WG Co-Chair: Davene Sarrocco-Smith (OH) Members to review the Food Safety Modernization Act (FSMA) & how it applies to working with local agencies. Review FSMA provisions and PFP projects to determine where local engagement is appropriate and best way to engage. 6
Local Engagement WG Charge Current Projects To incorporate the Food Safety • Modernization Act (FSMA) deliverables that include “local” agencies with PFP projects & provide specific recommendations for engagement of local agencies within the • vision for an IFSS. WG Co-Chair: Pat Maloney (MA) WG Co-Chair: Liz O’Malley (FDA) WG Co-Chair: Davene Sarrocco-Smith (OH) Members to review the Food Safety Modernization Act (FSMA) & how it applies to working with local agencies. Review FSMA provisions and PFP projects to determine where local engagement is appropriate and best way to engage. 6
 Local Engagement WG (FDA Reps) • • • Jeff Farrar (OFVM) Laurie Farmer (ORA) Lauren Gore (ORA) Liz O’Malley (ORA) Pete Salsbury (CFSAN) 7
Local Engagement WG (FDA Reps) • • • Jeff Farrar (OFVM) Laurie Farmer (ORA) Lauren Gore (ORA) Liz O’Malley (ORA) Pete Salsbury (CFSAN) 7
 Response & Recall WG Charge • Develop response guides and records for event responses. • • • WG Co-Chair: Travis Goodman (FDA) WG Co-Chair: Ben Miller (MN) Drafted an Index of investigation, response & recovery guidance documents Drafted a Guide for States on Records Collection for More Successful Tracebacks Completed White paper on traceback investigations Drafted a PFP Quick Start Response Guide Current Projects • • Complete the items above Identify 2 -3 new Response & Recall Projects 8
Response & Recall WG Charge • Develop response guides and records for event responses. • • • WG Co-Chair: Travis Goodman (FDA) WG Co-Chair: Ben Miller (MN) Drafted an Index of investigation, response & recovery guidance documents Drafted a Guide for States on Records Collection for More Successful Tracebacks Completed White paper on traceback investigations Drafted a PFP Quick Start Response Guide Current Projects • • Complete the items above Identify 2 -3 new Response & Recall Projects 8
 Response & Recall WG (FDA Reps) • Jeff Farrar (OFVM) • Travis Goodman (ORA) • Roberta Hammond (CORE) • Pamela Le. Blanc (CORE) • Wanda Lenger (ORA) • Priscilla Neves (ORA) • Mahala Shillingford (OFVM) • Cecilia Wolyniak (ORA) • Lauren Yeung (ORA) 9
Response & Recall WG (FDA Reps) • Jeff Farrar (OFVM) • Travis Goodman (ORA) • Roberta Hammond (CORE) • Pamela Le. Blanc (CORE) • Wanda Lenger (ORA) • Priscilla Neves (ORA) • Mahala Shillingford (OFVM) • Cecilia Wolyniak (ORA) • Lauren Yeung (ORA) 9
 National Feed Assignment WG Charge Current Projects Develop methods of sharing feed collection samples between federal & state agencies. • Domestic Import Sampling Assignment • Develop a database for sharing information relative to feed samples • Complete White Paper on Retail Food Salvage for Animal Feed WG Co-Chair: Danielson (TN) WG Co-Chair: Darlene Krieger (FDA) 10
National Feed Assignment WG Charge Current Projects Develop methods of sharing feed collection samples between federal & state agencies. • Domestic Import Sampling Assignment • Develop a database for sharing information relative to feed samples • Complete White Paper on Retail Food Salvage for Animal Feed WG Co-Chair: Danielson (TN) WG Co-Chair: Darlene Krieger (FDA) 10
 National Feed Assignment WG (FDA Reps) • • • Mary Bartholomew (CVM) Darlene Krieger (ORA) Sherri Mc. Garry (OFVM) Kara Morgan (OC) Jenny Murphy (CVM) • • Eric Nelson (CVM) Deep Patel (OC) Iris Valentin-Bon (ORA) Julianna Wittig (CVM) 11
National Feed Assignment WG (FDA Reps) • • • Mary Bartholomew (CVM) Darlene Krieger (ORA) Sherri Mc. Garry (OFVM) Kara Morgan (OC) Jenny Murphy (CVM) • • Eric Nelson (CVM) Deep Patel (OC) Iris Valentin-Bon (ORA) Julianna Wittig (CVM) 11
 PETNet Expansion WG Charge Current Project Provide mechanism for information sharing and monitoring between federal & state agencies for pet-food related incidents so that these agencies can determine quickly best response & action. • Review current Pet Event Tracking Network (PETNet) system for improvement needs. • Expand PETNet system to include food producing animals: – Develop appropriate questions for reporting livestock feed concerns – Develop the livestock portal – Determine how/if the livestock and PETNet portals will interact WG Co-Chair: Liz Higgins (NM) WG Co-Chair: Jenny Murphy (FDA) 12
PETNet Expansion WG Charge Current Project Provide mechanism for information sharing and monitoring between federal & state agencies for pet-food related incidents so that these agencies can determine quickly best response & action. • Review current Pet Event Tracking Network (PETNet) system for improvement needs. • Expand PETNet system to include food producing animals: – Develop appropriate questions for reporting livestock feed concerns – Develop the livestock portal – Determine how/if the livestock and PETNet portals will interact WG Co-Chair: Liz Higgins (NM) WG Co-Chair: Jenny Murphy (FDA) 12
 PETNet Expansion WG (FDA Reps) • • • Krisztina Atkinson (CVM) Jean Bowman (CVM) Alberto Chiesa (CVM) Zoe Gill (CVM) April Hodges (CVM) Caleb Michaud (ORA) • • Jenny Murphy (CVM) Jackie Queen (CVM) Marianne Ross (CVM) Renee Shibukawa-Kent (CVM) • Lauren Yeung (CVM) 13
PETNet Expansion WG (FDA Reps) • • • Krisztina Atkinson (CVM) Jean Bowman (CVM) Alberto Chiesa (CVM) Zoe Gill (CVM) April Hodges (CVM) Caleb Michaud (ORA) • • Jenny Murphy (CVM) Jackie Queen (CVM) Marianne Ross (CVM) Renee Shibukawa-Kent (CVM) • Lauren Yeung (CVM) 13
 Training & Certification WG Charge Current Projects To provide support for visionary development, best practices, appropriate content and recommendations toward development of an integrated food safety training & certification system. • Linking FSMA Sections to the IFPTI Curriculum Framework • Develop a process that would identify and prioritize training and certification needs • Develop a process to identify and qualify existing analyses, bodies of knowledge, or other underlying frameworks for job related curriculum that fit the IFSS curriculum framework. WG Co-Chair: Allan Bateson (FDA) WG Co-Chair: Steve James (FDA) WG Co-Chair: Keith Johnson (ND) WG Co-Chair: Dave Read (MN) 14
Training & Certification WG Charge Current Projects To provide support for visionary development, best practices, appropriate content and recommendations toward development of an integrated food safety training & certification system. • Linking FSMA Sections to the IFPTI Curriculum Framework • Develop a process that would identify and prioritize training and certification needs • Develop a process to identify and qualify existing analyses, bodies of knowledge, or other underlying frameworks for job related curriculum that fit the IFSS curriculum framework. WG Co-Chair: Allan Bateson (FDA) WG Co-Chair: Steve James (FDA) WG Co-Chair: Keith Johnson (ND) WG Co-Chair: Dave Read (MN) 14
 Training & Certification WG (FDA Reps) • • • Allan Bateson (ORA) Atin Datta (CFSAN) Jim Fear (ORA) Steve James (ORA) Karyn Johnstone (CVM) • • • Thomas Hughes (ORA) Cindy Leonard (ORA) Kara Lynch (ORA) John Marcello (ORA) Pete Salsbury (CFSAN) 15
Training & Certification WG (FDA Reps) • • • Allan Bateson (ORA) Atin Datta (CFSAN) Jim Fear (ORA) Steve James (ORA) Karyn Johnstone (CVM) • • • Thomas Hughes (ORA) Cindy Leonard (ORA) Kara Lynch (ORA) John Marcello (ORA) Pete Salsbury (CFSAN) 15
 Take Home Messages • The PFP plays a collaborative role in building an IFSS. • We need to identify the most significant IFSS gaps and “fund the gaps”. • PFP & FSMA have the same goal; to build an IFSS. • FSMA work has been aligned/merged with the dynamic PFP Workgroups. • Integration will not happen overnight, but working in partnership, we are making strides towards having an IFSS become a reality. 16
Take Home Messages • The PFP plays a collaborative role in building an IFSS. • We need to identify the most significant IFSS gaps and “fund the gaps”. • PFP & FSMA have the same goal; to build an IFSS. • FSMA work has been aligned/merged with the dynamic PFP Workgroups. • Integration will not happen overnight, but working in partnership, we are making strides towards having an IFSS become a reality. 16
 Relevant PFP Accomplishments & Outputs from Related Efforts to Support the Building an IFSS 17
Relevant PFP Accomplishments & Outputs from Related Efforts to Support the Building an IFSS 17
 FDA/ORA/Office of Partnerships Central hub for: • Information sharing • Communication with partnering agencies & trade organizations • Execution of partnerships, grants, & cooperative agreements • Links to public documents & information • Program Summaries • Contact Information 18
FDA/ORA/Office of Partnerships Central hub for: • Information sharing • Communication with partnering agencies & trade organizations • Execution of partnerships, grants, & cooperative agreements • Links to public documents & information • Program Summaries • Contact Information 18
 Utilization of State Programs as Partners FY 12 Inspection Contract Programs Program # of Awards Food* 46 Feed 35 Egg 8 Tissue Residue 21 MQSA** 44 Medical Device 1 Milk Residue 1 Totals $15 M *43 states, 1 territory **Mammography Quality Standards Act ***Analysis of nearly 4 million milk residue samples 19
Utilization of State Programs as Partners FY 12 Inspection Contract Programs Program # of Awards Food* 46 Feed 35 Egg 8 Tissue Residue 21 MQSA** 44 Medical Device 1 Milk Residue 1 Totals $15 M *43 states, 1 territory **Mammography Quality Standards Act ***Analysis of nearly 4 million milk residue samples 19
 Expanding Support Opportunities FY 12 Grants & Cooperative Agreements Programs # of Awards Food Protection Task Forces 23 Food Emergency Response Network (FERN) 34 Ruminant Feed Ban Support (BSE) 11 Rapid Response Teams 19 Innovative Food Defense 3 Small Conference Grants 9 FSMA Emergency Response & Risk Based Inspections 36 Manufactured Food Regulatory Program Standards 26 ISO 17025: 2005 Laboratory Accreditation 31 Voluntary Nat’l Retail Food Regulatory Program Standards 38 Integrated Laboratory System 1 Alliances 2 Total Funding $44. 3 M 20
Expanding Support Opportunities FY 12 Grants & Cooperative Agreements Programs # of Awards Food Protection Task Forces 23 Food Emergency Response Network (FERN) 34 Ruminant Feed Ban Support (BSE) 11 Rapid Response Teams 19 Innovative Food Defense 3 Small Conference Grants 9 FSMA Emergency Response & Risk Based Inspections 36 Manufactured Food Regulatory Program Standards 26 ISO 17025: 2005 Laboratory Accreditation 31 Voluntary Nat’l Retail Food Regulatory Program Standards 38 Integrated Laboratory System 1 Alliances 2 Total Funding $44. 3 M 20
 National Program Standards: Manufactured Food Regulatory Program Standards (MFRPS) • Uniform foundation for the design and management of state programs responsible for regulating food manufacturers. • Institute a comprehensive quality assurance & standardization program. • FDA conducts Program Assessment Validation Audits (PAVAs) at 18 and 36 months; & comprehensive audit at 60 months. 21
National Program Standards: Manufactured Food Regulatory Program Standards (MFRPS) • Uniform foundation for the design and management of state programs responsible for regulating food manufacturers. • Institute a comprehensive quality assurance & standardization program. • FDA conducts Program Assessment Validation Audits (PAVAs) at 18 and 36 months; & comprehensive audit at 60 months. 21
 Building a Stronger MFRPS Community • 41 programs in 40 States – Increased from 28 programs in 2010 to 41 programs in 2013 • Food. SHIELD: Workgroup & Quarterly Webinar • MFRP Alliance – 1 st Annual MFRPS Training Course conducted March, 2012, – 2 nd Annual MFRPS Training conducted March 19 -21, 2013. – Strengthen: Community, Collaboration, Resources, & Program Advancement. 22
Building a Stronger MFRPS Community • 41 programs in 40 States – Increased from 28 programs in 2010 to 41 programs in 2013 • Food. SHIELD: Workgroup & Quarterly Webinar • MFRP Alliance – 1 st Annual MFRPS Training Course conducted March, 2012, – 2 nd Annual MFRPS Training conducted March 19 -21, 2013. – Strengthen: Community, Collaboration, Resources, & Program Advancement. 22
 National Program Standards: Voluntary National Retail Foods Regulatory Program Standards (VNRFRPS) • Standards provide: – A guide to design and management of a retail food program. – A program foundation. – A tool to evaluate the effectiveness of food safety interventions. • 553 regulatory jurisdictions enrolled (as of April 2013) • Additional information available at http: //www. fda. gov/Food. Safety/Retail. Food. Protection/Pr ogram. Standards/default. htm. 23
National Program Standards: Voluntary National Retail Foods Regulatory Program Standards (VNRFRPS) • Standards provide: – A guide to design and management of a retail food program. – A program foundation. – A tool to evaluate the effectiveness of food safety interventions. • 553 regulatory jurisdictions enrolled (as of April 2013) • Additional information available at http: //www. fda. gov/Food. Safety/Retail. Food. Protection/Pr ogram. Standards/default. htm. 23
 Rapid Response Teams (RRT) • Multi-year Cooperative Agreement – Awards range from $300, 000 - $500, 000 – Awards typically 3 years in length – 9 began in 2008/2009 – 10 added in 2012 • Why RRTs? – White House & Congressional * Did You Know 9 RRTs are in Public Health Agencies 10 RRTs are in Departments of Agriculture interest in improving response and food safety. • Purpose: – Improve response by unifying & coordinating partners – Develop effective models for response that can be replicated. 24
Rapid Response Teams (RRT) • Multi-year Cooperative Agreement – Awards range from $300, 000 - $500, 000 – Awards typically 3 years in length – 9 began in 2008/2009 – 10 added in 2012 • Why RRTs? – White House & Congressional * Did You Know 9 RRTs are in Public Health Agencies 10 RRTs are in Departments of Agriculture interest in improving response and food safety. • Purpose: – Improve response by unifying & coordinating partners – Develop effective models for response that can be replicated. 24
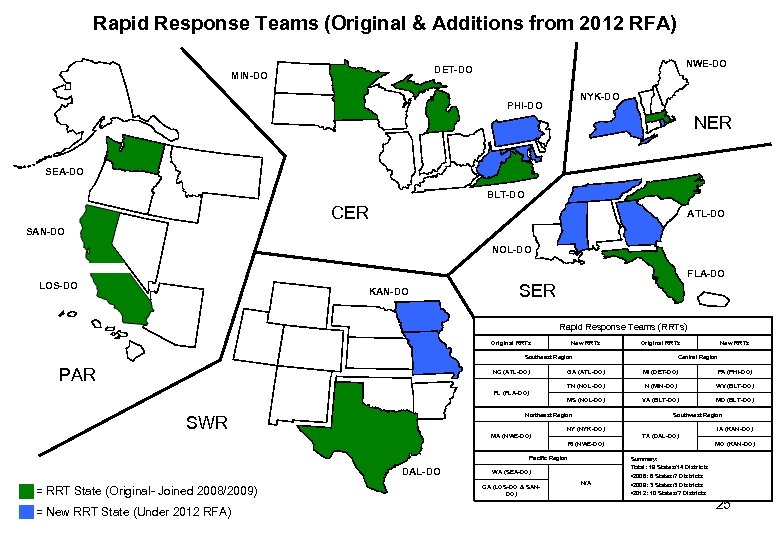 Rapid Response Teams (Original & Additions from 2012 RFA) NWE-DO DET-DO MIN-DO NYK-DO PHI-DO NER SEA-DO BLT-DO CER ATL-DO SAN-DO NOL-DO FLA-DO LOS-DO KAN-DO SER Rapid Response Teams (RRTs) Original RRTs New RRTs Original RRTs Southeast Region PAR NC (ATL-DO) New RRTs Central Region GA (ATL-DO) MI (DET-DO) PA (PHI-DO) TN (NOL-DO) N (MIN-DO) WV (BLT-DO) MS (NOL-DO) VA (BLT-DO) MD (BLT-DO) FL (FLA-DO) Northeast Region SWR Southwest Region NY (NYK-DO) MA (NWE-DO) IA (KAN-DO) TX (DAL-DO) RI (NWE-DO) Pacific Region DAL-DO = RRT State (Original- Joined 2008/2009) = New RRT State (Under 2012 RFA) WA (SEA-DO) CA (LOS-DO & SANDO) N/A MO (KAN-DO) Summary: Total: 19 States/14 Districts • 2008: 6 States/7 Districts • 2009: 3 States/3 Districts • 2012: 10 States/7 Districts 25
Rapid Response Teams (Original & Additions from 2012 RFA) NWE-DO DET-DO MIN-DO NYK-DO PHI-DO NER SEA-DO BLT-DO CER ATL-DO SAN-DO NOL-DO FLA-DO LOS-DO KAN-DO SER Rapid Response Teams (RRTs) Original RRTs New RRTs Original RRTs Southeast Region PAR NC (ATL-DO) New RRTs Central Region GA (ATL-DO) MI (DET-DO) PA (PHI-DO) TN (NOL-DO) N (MIN-DO) WV (BLT-DO) MS (NOL-DO) VA (BLT-DO) MD (BLT-DO) FL (FLA-DO) Northeast Region SWR Southwest Region NY (NYK-DO) MA (NWE-DO) IA (KAN-DO) TX (DAL-DO) RI (NWE-DO) Pacific Region DAL-DO = RRT State (Original- Joined 2008/2009) = New RRT State (Under 2012 RFA) WA (SEA-DO) CA (LOS-DO & SANDO) N/A MO (KAN-DO) Summary: Total: 19 States/14 Districts • 2008: 6 States/7 Districts • 2009: 3 States/3 Districts • 2012: 10 States/7 Districts 25
 Bringing Together the Regulatory Community • Operationalizing mutual needs to make the regulatory community “Partners” programs stronger and more efficient with minimal duplication of efforts. • Utilization of the Joint Work Planning document from the PFP work group. • Developing mechanisms to engage other stakeholders in the process by: – Bringing their ideas to the table for future work; – Inclusion of overall continuous improvement toward a fully integrated food safety system. 26
Bringing Together the Regulatory Community • Operationalizing mutual needs to make the regulatory community “Partners” programs stronger and more efficient with minimal duplication of efforts. • Utilization of the Joint Work Planning document from the PFP work group. • Developing mechanisms to engage other stakeholders in the process by: – Bringing their ideas to the table for future work; – Inclusion of overall continuous improvement toward a fully integrated food safety system. 26
 Accomplishments of 2010 -2012 PFP WGs • Compliance/enforcement coordination pilot. • Developed local work planning process to reduce redundant inspections/leverage resources. • Developed Business Process Evaluation Tool to evaluate IT gaps/needs relative to Inspection Systems. • Developed the “National Standards for Food/Feed Testing Laboratories”. • Developed recommendation for a process to review/update MFRPS. 27
Accomplishments of 2010 -2012 PFP WGs • Compliance/enforcement coordination pilot. • Developed local work planning process to reduce redundant inspections/leverage resources. • Developed Business Process Evaluation Tool to evaluate IT gaps/needs relative to Inspection Systems. • Developed the “National Standards for Food/Feed Testing Laboratories”. • Developed recommendation for a process to review/update MFRPS. 27
 Thank You for Your Attention We Welcome Your Questions, Comments & Suggestions 28
Thank You for Your Attention We Welcome Your Questions, Comments & Suggestions 28


