c80781d722e98fe4fe45e3e7436aed13.ppt
- Количество слайдов: 40

CRT 2010 Washington, USA, Feb 21 -23, 2010 Update on Atrial Appendage Closure Technology Horst Sievert, Nina Wunderlich Cardio. Vascular Center Frankfurt, Germany

DISCLOSURES Horst Sievert, MD Consulting Fees – Access. Closure, Inc. , AGA Medical Corporation, Angiomed, Ardian, Inc. , Arstasis, Inc. , Avinger, Bridge. Point Medical, Cardio. Kinetix, Inc. , Cardio. MEMS Inc. , Coherex Medical, Inc. , CSI Medical, Endo. Cross, Boston Scientific Corporation, ev 3, Inc. , Flow. Cardia, Inc. , W. L. Gore & Associates, Inc. , Abbott Vascular, Lumen Biomedical, Kensey Nash Corporation, Kyoto Medical, NDC, NMT Medical, Inc. , OAS, Occlutech, Osprey Medical, Inc. , Ovalis, Inc. , Pathway Medical Technologies, Inc. , Pfm Medical, Inc. , Pendra. Care, Percardia Inc. , Remon Medical Technologies, ROX Medical, Sadra Medical, Sorin Group, Spectranetics, Square One Medical, Viacor, Inc. , St. Jude Medical, Medinol Ltd. , Lutonix, Inc. Grants/Contracted Research – Abbott Vascular, Access. Closure, Inc. , AGA Medical Corporation, Angiomed, Boston Scientific Corporation, Cardio. Kinetix, Inc. , Co. Aptus Medical Corporation, Cordis, a Johnson & Johnson company, CSI Medical, Edwards Lifesciences LLC, ev 3, Inc. , W. L. Gore & Associates, Inc. , Kensey Nash Corporation , Mind Guard, NDC, Neovasc, NMT Medical, Inc. , Percardia Inc. , Sorin Group, St. Jude Medical, Terumo Medical Corporation, Top. Spin Medical Ltd. , St. Jude Medical, Lumen Biomedical Ownership Interest (Stocks, Stock Options or Other Ownership Interest) – Cardio-Kinetics Inc. , Access. Closure, Inc. , Co. Aptus Medical Corporation, Lumen Biomedical, Cierra I intend to reference unlabeled/ unapproved uses of drugs or devices in my presentation. I intend to reference PFO, ASD devices, carotid stents, and EPD.
Why to close the left atrial appendage? • To prevent thrombus formation in atrial fibrillation • LAA closure had been a logical but unproven concept until last year

How to occlude the LAA ?

PLAATO™ Device e. PTFE membrane LA anchors LAA Nitinol Cage

Watchman Device • • • Nitinol frame PET membrane row of fixation barbs around the mid perimeter • 21, 24, 27, 30, 33 mm CE mark
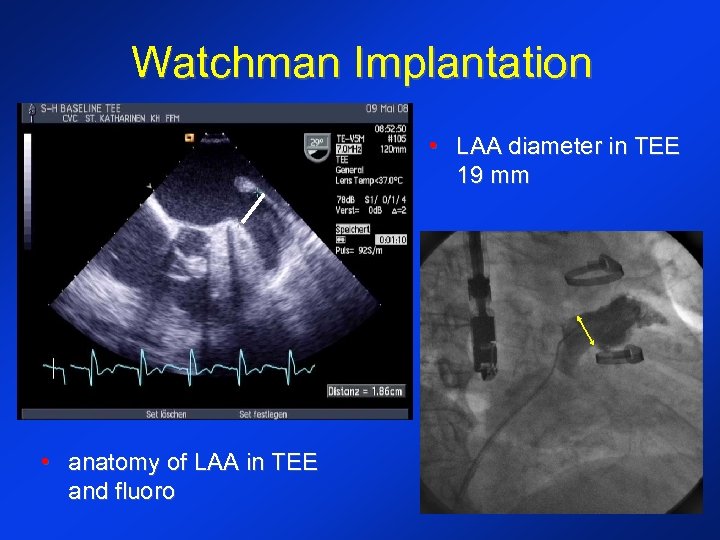
Watchman Implantation • LAA diameter in TEE 19 mm • anatomy of LAA in TEE and fluoro
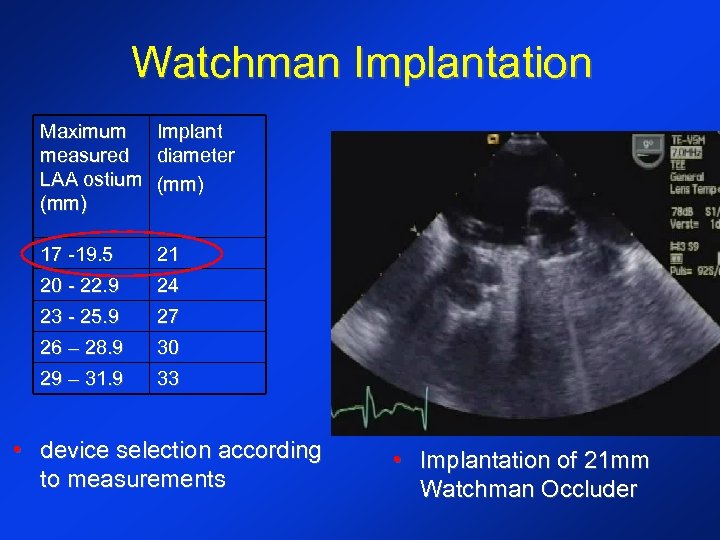
Watchman Implantation Maximum Implant measured diameter LAA ostium (mm) 17 -19. 5 21 20 - 22. 9 24 23 - 25. 9 27 26 – 28. 9 30 29 – 31. 9 33 • device selection according to measurements • Implantation of 21 mm Watchman Occluder
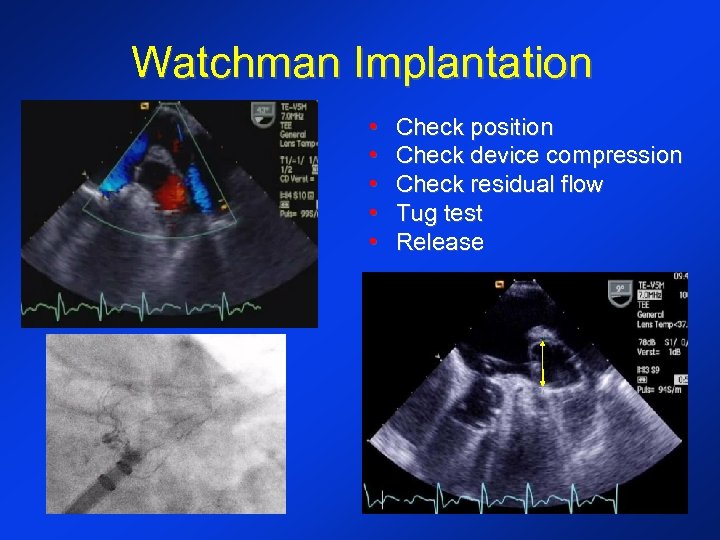
Watchman Implantation • • • Check position Check device compression Check residual flow Tug test Release



Protect AF (System for Embolic PROTECTion in Patients with Atrial Fibrillation) • • • Multicenter Prospective randomized WATCHMAN vs coumadin 2: 1 Non-inferiority trial 800 pts (enrollment closed June 2008) > 900 patient-years

In- & Exclusion Major inclusion criteria Major exclusion criteria • Non valvular AF with Chads 2 score ≥ 1 • No contraindications to coumadin • No co-morbidities mandating chronic warfarin use other than AF • LAA thrombus • Large PFO with significant atrial septal aneurysm • Mobile aortic atheroma • Symptomatic carotid artery disease

PROTECT AF Trial Endpoints • Primary Efficacy Endpoint • All stroke • Cardiovascular and unexplained death • Systemic embolization • Primary Safety Endpoint • Device embolization requiring retrieval • Pericardial effusion requiring intervention • Cranial bleeds and gastrointestinal bleeds • Any bleed that requires ≥ 2 u. PRBC
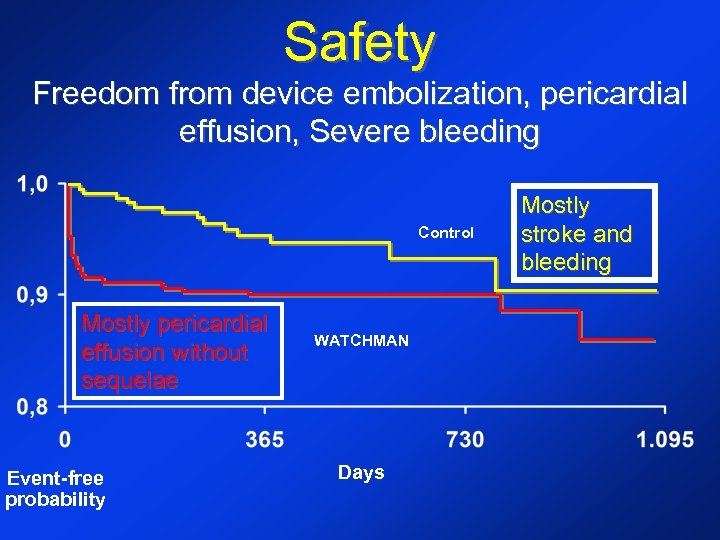
Safety Freedom from device embolization, pericardial effusion, Severe bleeding Control Mostly pericardial effusion without sequelae Event-free probability WATCHMAN Days Mostly stroke and bleeding
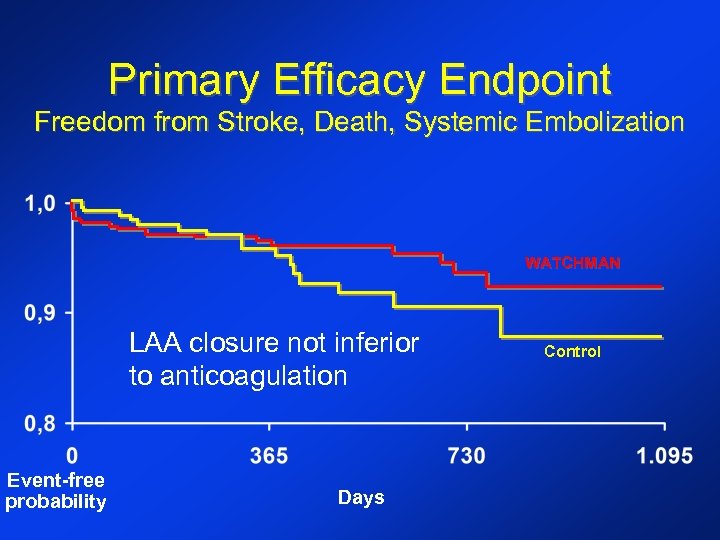
Primary Efficacy Endpoint Freedom from Stroke, Death, Systemic Embolization WATCHMAN LAA closure not inferior to anticoagulation Event-free probability Days Control

Other significant findings • LAA occlusion is • Noninferior for all strokes - 26% lower rate in device group • Superior for hemorrhagic stroke - 91% lower in device group • Noninferior for mortality - 39% lower rate in device group • Most events in the device group were periprocedural pericardial effusions which did not occur later on

PROTECT AF • We now have the "Proof of concept": - Left atrial appendage closure prevents stroke - It is as effective as anticoagulation • As expected there are more early safety events after LAA closure due to pericardial effusions • Under anticoagulation therapy there are more late safety events due to stroke and bleeding
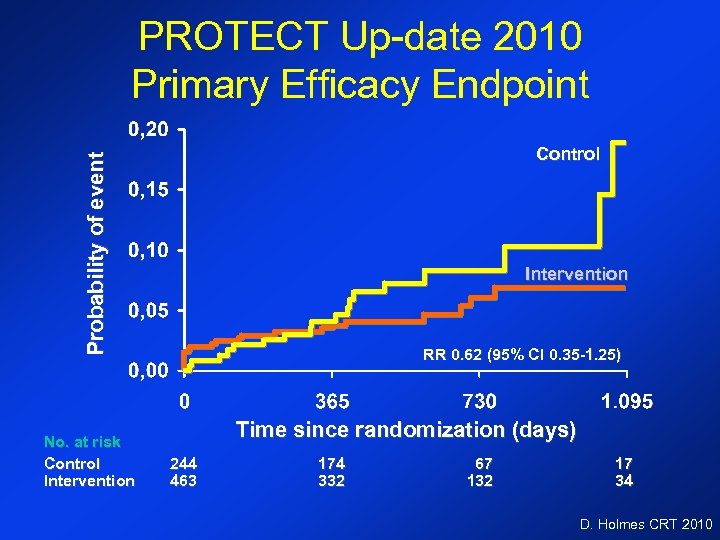
PROTECT Up-date 2010 Primary Efficacy Endpoint Probability of event Control No. at risk Control Intervention RR 0. 62 (95% CI 0. 35 -1. 25) Time since randomization (days) 244 463 174 332 67 132 17 34 D. Holmes CRT 2010
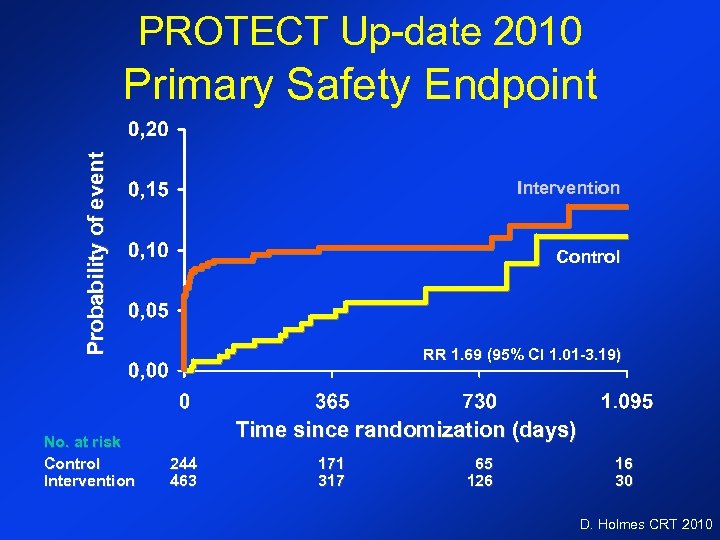
PROTECT Up-date 2010 Probability of event Primary Safety Endpoint No. at risk Control Intervention Control RR 1. 69 (95% CI 1. 01 -3. 19) Time since randomization (days) 244 463 171 317 65 126 16 30 D. Holmes CRT 2010

Amplatzer Cardiac Plug • Distal plug and proximal disk connected by a flexible central waist • Hooks • Plug size range: 16 - 30 mm • Disk size range: 20 - 36 mm • Delivery system: 9 F - 13 F
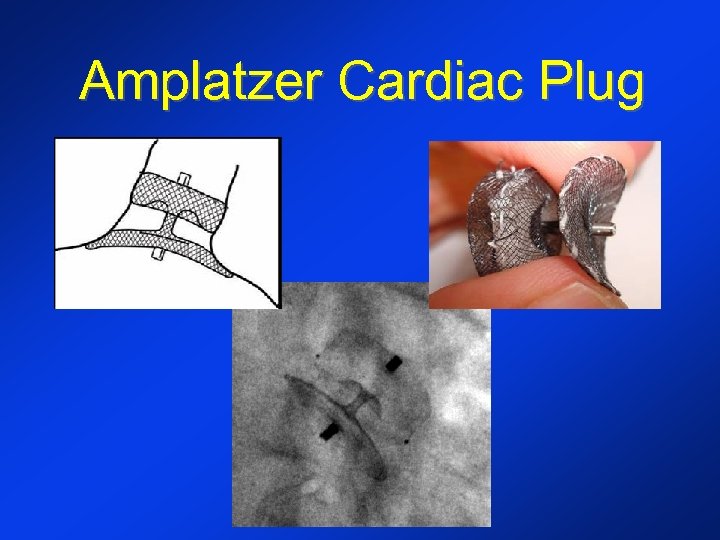
Amplatzer Cardiac Plug
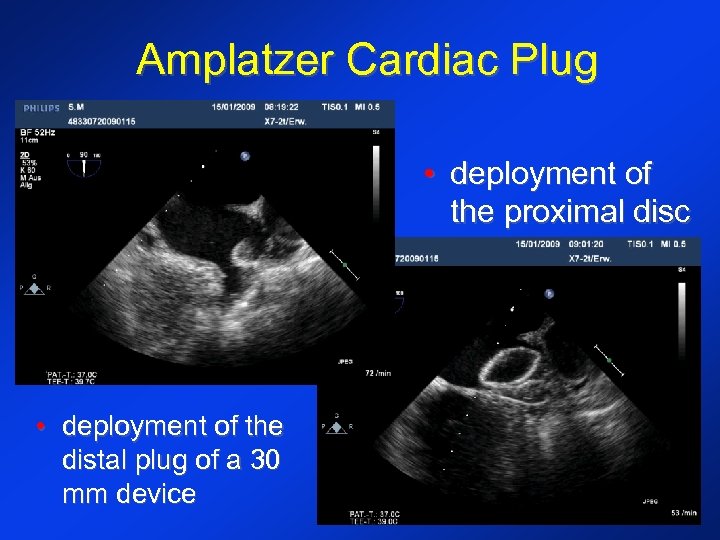
Amplatzer Cardiac Plug • deployment of the proximal disc • deployment of the distal plug of a 30 mm device
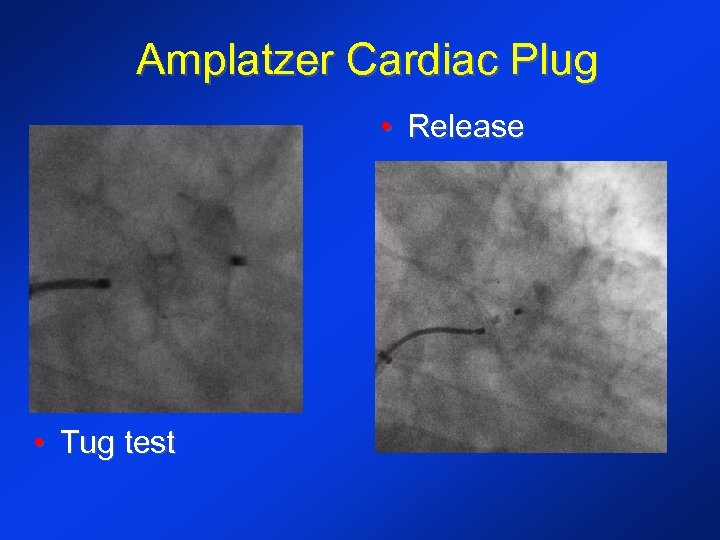
Amplatzer Cardiac Plug • Release • Tug test
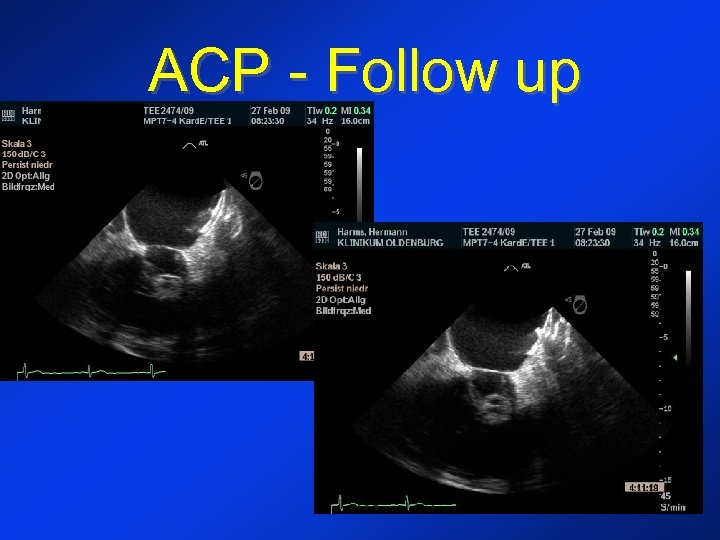
ACP - Follow up

Amplatzer Cardiac Plug Frankfurt Experience • • • N = 41, 39 % female 34 % paroxysmal, 66 % permanent AF Concomitant diseases – Prior stroke/TIA – Heart failure – Diabetes – Hypertension – Coronary artery disease 39 % 20 % 22 % 90 % 34 %

Acute Results • LAA occlusion attempted • Device retrieved • before release and replaced by another device 6 • no further attempt 2 • Device implanted • Device size 16 - 30 mm (22 ± 4) • Mean procedure time 74 min • Mean Flouroscopy time 13 min 41 • Residual flow (intraprocedural) 3 / 38 / 41

Procedural Complications • Death • Tamponade • Pericardial effusion pericardiocenthesis • • 0 0 3 0% 0% 8% 0 1 0% 3% 2 5% 1 Groin complications Device embolization no sequelae • Air embolism with transient ST- depression, no sequelae – Will be prevented by a device design modification
New Approaches • Epicardial • Endocardial

Epicardial • Epitek • Atri. Cure • Sentre. Heart • Others • Medtronic
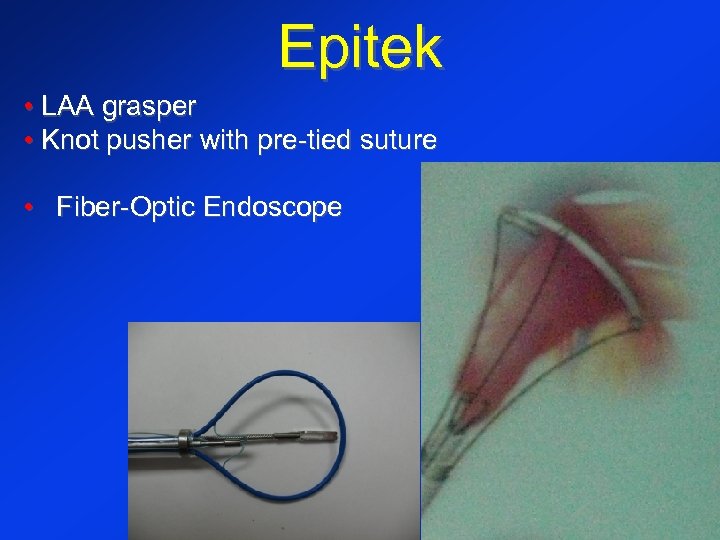
Epitek • LAA grasper • Knot pusher with pre-tied suture • Fiber-Optic Endoscope
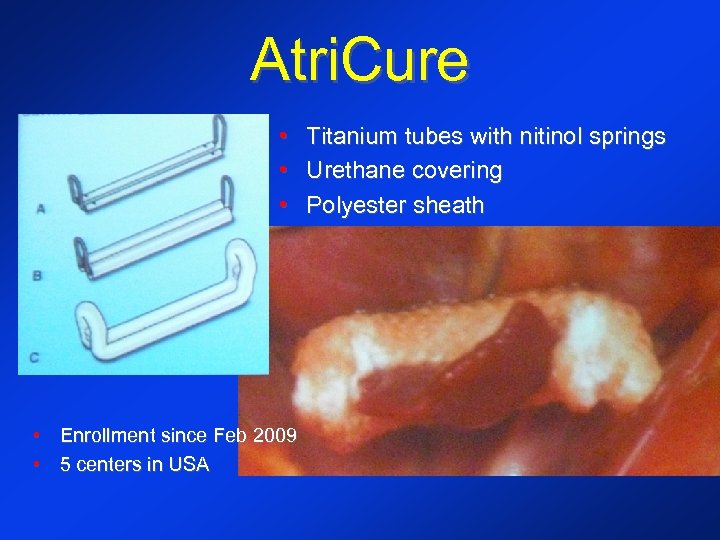
Atri. Cure • • Enrollment since Feb 2009 • 5 centers in USA Titanium tubes with nitinol springs Urethane covering Polyester sheath

Others • Sentre. Heart • Aegis Medical Working on a device… …but… …currently no details available

Endocardial • Atritech – 4 th generation • Occlutech • Others • AGA • Coherex • Gore • Custom Medical Devices
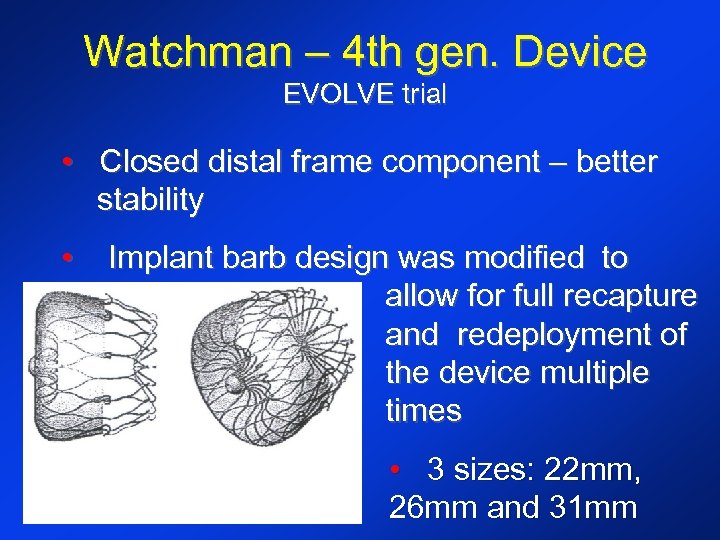
Watchman – 4 th gen. Device EVOLVE trial • Closed distal frame component – better stability • Implant barb design was modified to allow for full recapture and redeployment of the device multiple times • 3 sizes: 22 mm, 26 mm and 31 mm

Occlutech • • Self-expanding flexible nitinol meshwork Patch to close the LAA The new Occlutech Connector Clinical trial 2010?

Coherex Medical Clinical trial 2010

Custom medical Devices • Balloon inflatable deployment method • Wireless design • Allows a single patch to take the size and shape of any LAA • Soft device • Cannot cause perforations • Bioabsorbable material • Prevents chronic erosions • CE Mark

Transcatheter Patch • • • Animal work since 2002 Human implantations since 2005 International registry for occlusion of the left atrial appendage in high risk patients with atrial fibrillation à 15 human implantations

Take Home Messages • Atrial fibrillation is a frequent cause of stroke • Thrombi originate in the left atrial appendage • Catheter closure of the atrial appendage is feasible and relatively safe • The randomized trial with the Watchman device showed that the procedure is safe and effective in stroke reduction and not inferior compared to anticoagulation • Currently two devices are available for LAA closure • Many others are currently under development
c80781d722e98fe4fe45e3e7436aed13.ppt