7f96837fb487933cdad28906f7fcae96.ppt
- Количество слайдов: 22
 CRT 2010 Washington DC, January 21, 2010 MITRALIGN: Direct Annuloplasty Device Evolution, Technique and Clinical Trial Update Eberhard Grube, MD, FACC, FSCAI St. Elisabeth Hospital, Heart Center Rhein-Ruhr, Essen, Germany Instituto Cardiologico Dante Pazzanese, São Paulo, Brazil
CRT 2010 Washington DC, January 21, 2010 MITRALIGN: Direct Annuloplasty Device Evolution, Technique and Clinical Trial Update Eberhard Grube, MD, FACC, FSCAI St. Elisabeth Hospital, Heart Center Rhein-Ruhr, Essen, Germany Instituto Cardiologico Dante Pazzanese, São Paulo, Brazil
 DISCLOSURES Eberhard Grube, MD Consulting Fees – Abbott Vascular, Boston Scientific Corporation, Cordis, a Johnson & Johnson Company, Medtronic Cardio. Vascular, Inc. Honoraria – Biosensors International , Boston Scientific Corporation, Medtronic Cardio. Vascular, Inc Ownership Interest (Stocks, Stock Options or Other Ownership Interest) – Biosensors International , Medtronic Cardio. Vascular, Inc. I intend to reference unlabeled/ unapproved uses of drugs or devices in my presentation. I intend to reference off-label use of stents and valve prosthesis.
DISCLOSURES Eberhard Grube, MD Consulting Fees – Abbott Vascular, Boston Scientific Corporation, Cordis, a Johnson & Johnson Company, Medtronic Cardio. Vascular, Inc. Honoraria – Biosensors International , Boston Scientific Corporation, Medtronic Cardio. Vascular, Inc Ownership Interest (Stocks, Stock Options or Other Ownership Interest) – Biosensors International , Medtronic Cardio. Vascular, Inc. I intend to reference unlabeled/ unapproved uses of drugs or devices in my presentation. I intend to reference off-label use of stents and valve prosthesis.
 Disclosure Within the past 12 months, the presenter or their spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Physician Name Company/Relationship Eberhard Grube, MD Mitralign: C Key G – Grant and or Research Support E – Equity Interests S - Salary C – Consulting fees, Honoraria R – Royalty Income I – Intellectual Property Rights SB – Speaker’s Bureau O – Ownership OF – Other Financial Benefits‘
Disclosure Within the past 12 months, the presenter or their spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Physician Name Company/Relationship Eberhard Grube, MD Mitralign: C Key G – Grant and or Research Support E – Equity Interests S - Salary C – Consulting fees, Honoraria R – Royalty Income I – Intellectual Property Rights SB – Speaker’s Bureau O – Ownership OF – Other Financial Benefits‘
 MITRALIGN Concept ● Surgical approaches for treatment of FMR are focused on size reduction of the mitral valve annulus. ● A relatively small (1 cm) plication of the posterior annulus can normalize the septo-lateral dimension and reduce MR. ● Mitralign concept emulates suture annuloplasty. - Retrograde LV approach - Surgical pledgets are delivered percutaneously through the posterior annulus - Plicate the annulus
MITRALIGN Concept ● Surgical approaches for treatment of FMR are focused on size reduction of the mitral valve annulus. ● A relatively small (1 cm) plication of the posterior annulus can normalize the septo-lateral dimension and reduce MR. ● Mitralign concept emulates suture annuloplasty. - Retrograde LV approach - Surgical pledgets are delivered percutaneously through the posterior annulus - Plicate the annulus
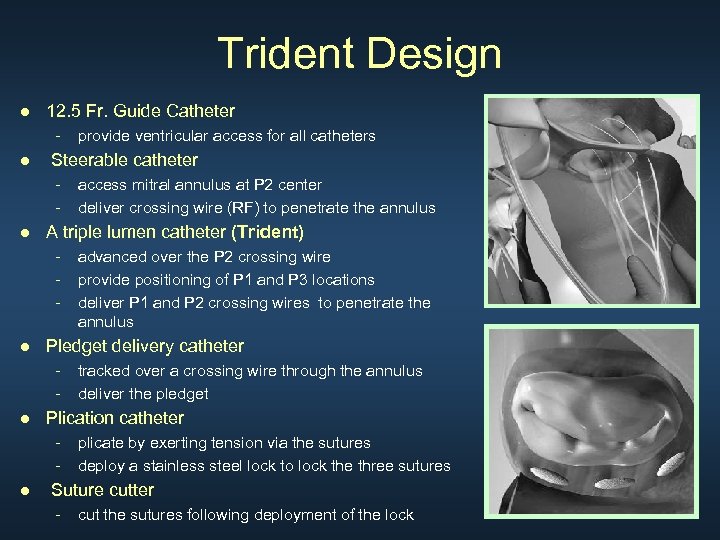 Trident Design ● 12. 5 Fr. Guide Catheter - ● provide ventricular access for all catheters Steerable catheter - access mitral annulus at P 2 center deliver crossing wire (RF) to penetrate the annulus ● A triple lumen catheter (Trident) - advanced over the P 2 crossing wire provide positioning of P 1 and P 3 locations deliver P 1 and P 2 crossing wires to penetrate the annulus ● Pledget delivery catheter - tracked over a crossing wire through the annulus deliver the pledget ● Plication catheter - ● plicate by exerting tension via the sutures deploy a stainless steel lock to lock the three sutures Suture cutter - cut the sutures following deployment of the lock
Trident Design ● 12. 5 Fr. Guide Catheter - ● provide ventricular access for all catheters Steerable catheter - access mitral annulus at P 2 center deliver crossing wire (RF) to penetrate the annulus ● A triple lumen catheter (Trident) - advanced over the P 2 crossing wire provide positioning of P 1 and P 3 locations deliver P 1 and P 2 crossing wires to penetrate the annulus ● Pledget delivery catheter - tracked over a crossing wire through the annulus deliver the pledget ● Plication catheter - ● plicate by exerting tension via the sutures deploy a stainless steel lock to lock the three sutures Suture cutter - cut the sutures following deployment of the lock
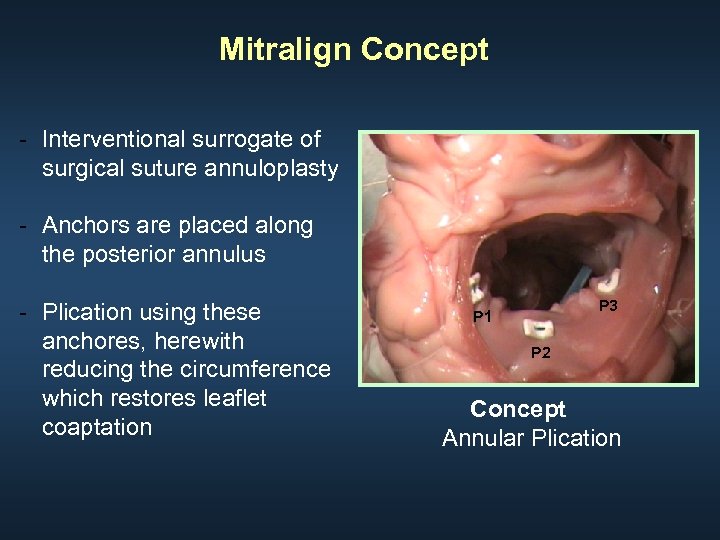 Mitralign Concept - Interventional surrogate of surgical suture annuloplasty - Anchors are placed along the posterior annulus - Plication using these anchores, herewith reducing the circumference which restores leaflet coaptation P 3 P 1 P 2 Concept Annular Plication
Mitralign Concept - Interventional surrogate of surgical suture annuloplasty - Anchors are placed along the posterior annulus - Plication using these anchores, herewith reducing the circumference which restores leaflet coaptation P 3 P 1 P 2 Concept Annular Plication
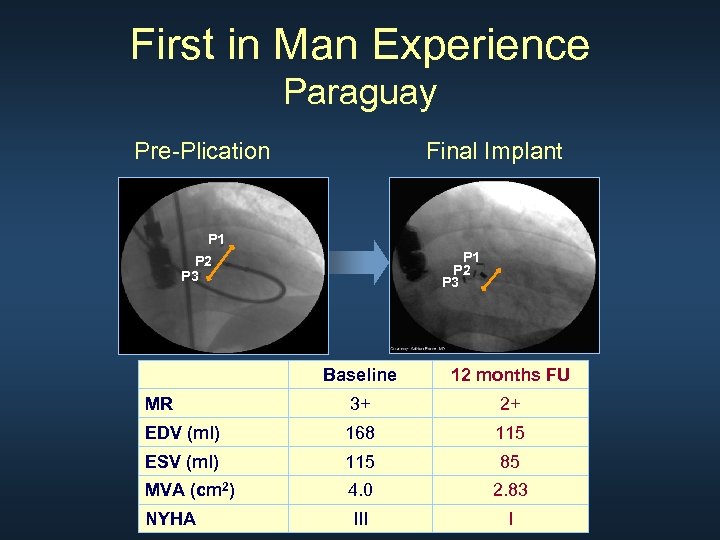 First in Man Experience Paraguay Pre-Plication Final Implant P 1 P 2 P 3 Baseline 12 months FU MR 3+ 2+ EDV (ml) 168 115 ESV (ml) 115 85 MVA (cm 2) 4. 0 2. 83 NYHA III I
First in Man Experience Paraguay Pre-Plication Final Implant P 1 P 2 P 3 Baseline 12 months FU MR 3+ 2+ EDV (ml) 168 115 ESV (ml) 115 85 MVA (cm 2) 4. 0 2. 83 NYHA III I
 Trident Concept First in Man Experience ● Four patients implanted in Paraguay by Dr. A. Ebner • 3 patients with all 3 pledgets • 1 patient with 2 pledges only ● Surgical pledgets can be delivered through the mitral annulus - Mitralign approach can be developed further ● Difficult to manage 3 components (wires, pledgets and sutures) at the same time: - Simpler design is needed
Trident Concept First in Man Experience ● Four patients implanted in Paraguay by Dr. A. Ebner • 3 patients with all 3 pledgets • 1 patient with 2 pledges only ● Surgical pledgets can be delivered through the mitral annulus - Mitralign approach can be developed further ● Difficult to manage 3 components (wires, pledgets and sutures) at the same time: - Simpler design is needed
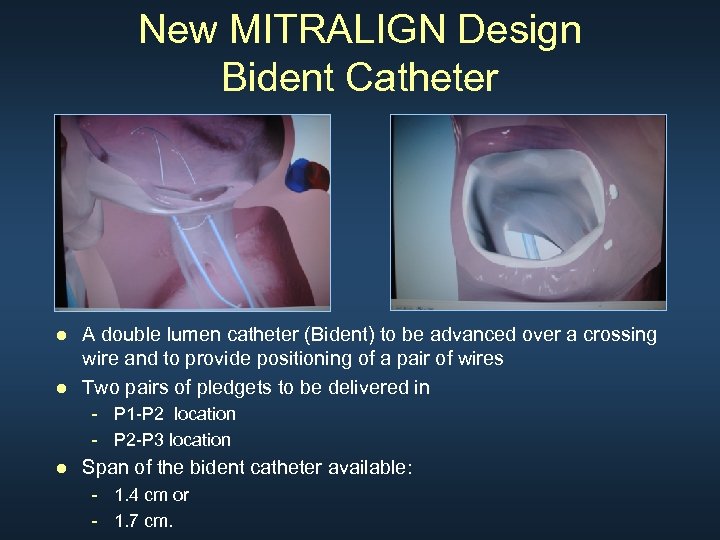 New MITRALIGN Design Bident Catheter ● A double lumen catheter (Bident) to be advanced over a crossing wire and to provide positioning of a pair of wires ● Two pairs of pledgets to be delivered in - P 1 -P 2 location - P 2 -P 3 location ● Span of the bident catheter available: - 1. 4 cm or - 1. 7 cm.
New MITRALIGN Design Bident Catheter ● A double lumen catheter (Bident) to be advanced over a crossing wire and to provide positioning of a pair of wires ● Two pairs of pledgets to be delivered in - P 1 -P 2 location - P 2 -P 3 location ● Span of the bident catheter available: - 1. 4 cm or - 1. 7 cm.
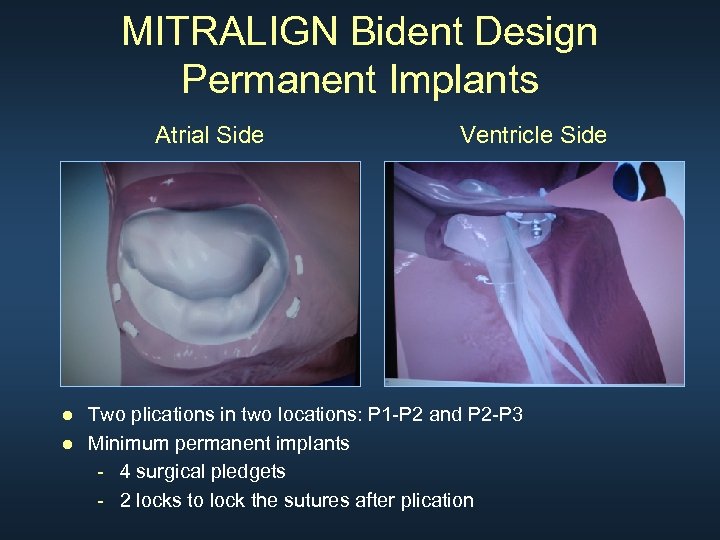 MITRALIGN Bident Design Permanent Implants Atrial Side Ventricle Side ● Two plications in two locations: P 1 -P 2 and P 2 -P 3 ● Minimum permanent implants - 4 surgical pledgets - 2 locks to lock the sutures after plication
MITRALIGN Bident Design Permanent Implants Atrial Side Ventricle Side ● Two plications in two locations: P 1 -P 2 and P 2 -P 3 ● Minimum permanent implants - 4 surgical pledgets - 2 locks to lock the sutures after plication
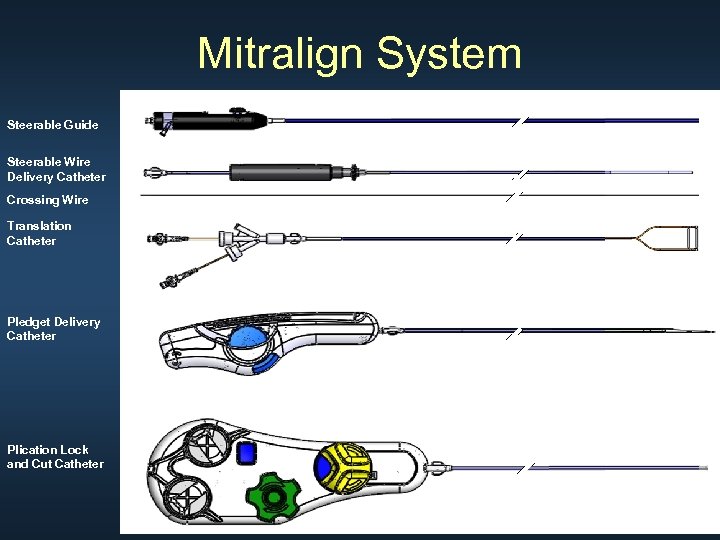 Mitralign System Steerable Guide Steerable Wire Delivery Catheter Crossing Wire Translation Catheter Pledget Delivery Catheter Plication Lock and Cut Catheter
Mitralign System Steerable Guide Steerable Wire Delivery Catheter Crossing Wire Translation Catheter Pledget Delivery Catheter Plication Lock and Cut Catheter
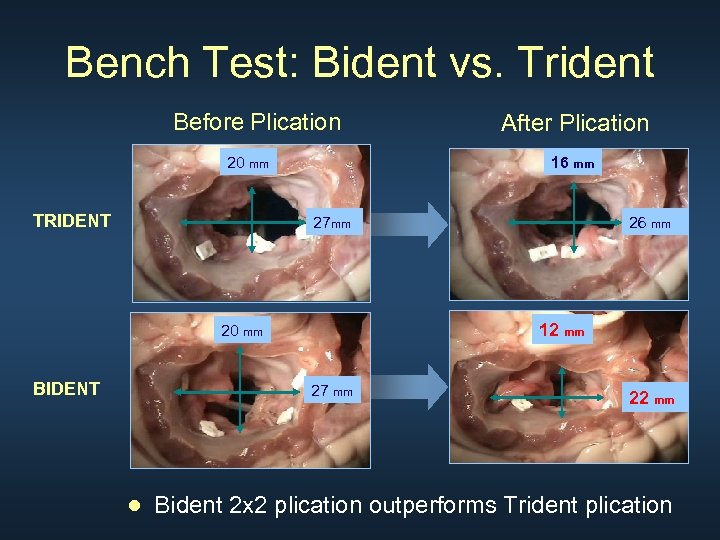 Bench Test: Bident vs. Trident Before Plication 20 mm TRIDENT 16 mm 27 mm 26 mm 12 mm 20 mm BIDENT After Plication 27 mm 22 mm ● Bident 2 x 2 plication outperforms Trident plication
Bench Test: Bident vs. Trident Before Plication 20 mm TRIDENT 16 mm 27 mm 26 mm 12 mm 20 mm BIDENT After Plication 27 mm 22 mm ● Bident 2 x 2 plication outperforms Trident plication
 Pre-Clinical Study: Bident ● MITRALIGN Bident Design simplifies the procedure.
Pre-Clinical Study: Bident ● MITRALIGN Bident Design simplifies the procedure.
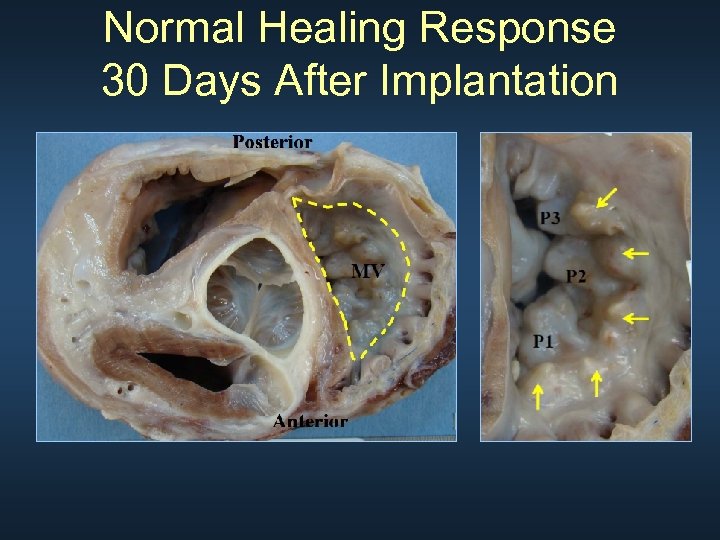 Normal Healing Response 30 Days After Implantation
Normal Healing Response 30 Days After Implantation
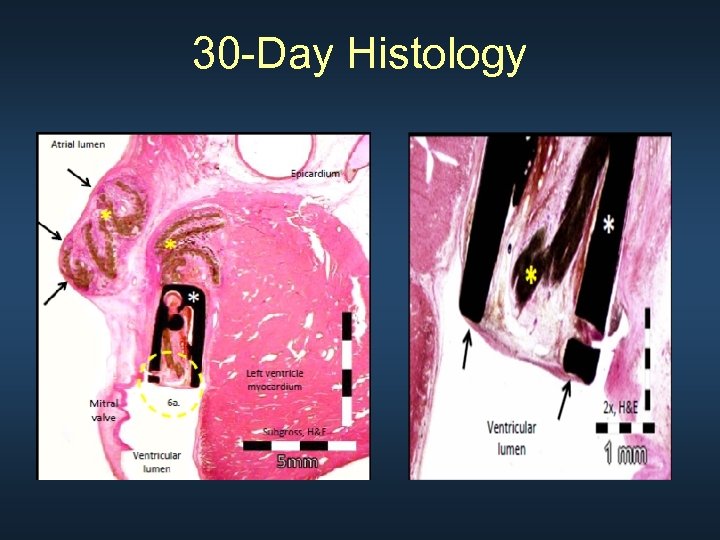 30 -Day Histology
30 -Day Histology
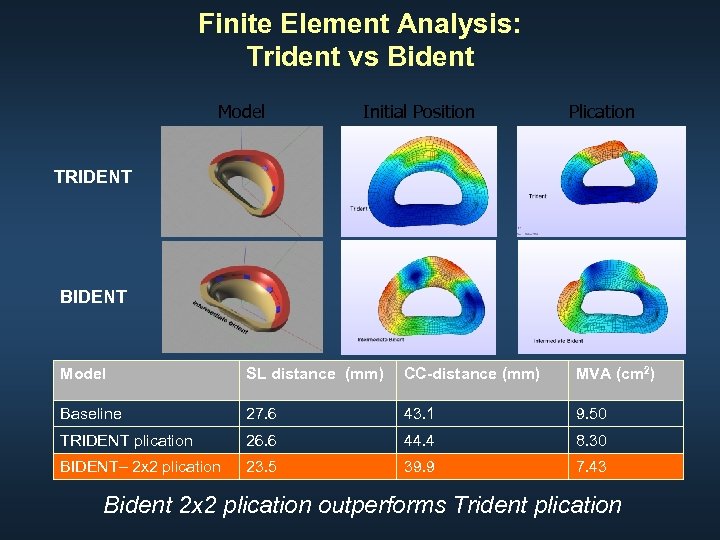 Finite Element Analysis: Trident vs Bident Model Initial Position Plication TRIDENT BIDENT Model SL distance (mm) CC-distance (mm) MVA (cm 2) Baseline 27. 6 43. 1 9. 50 TRIDENT plication 26. 6 44. 4 8. 30 BIDENT– 2 x 2 plication 23. 5 39. 9 7. 43 Bident 2 x 2 plication outperforms Trident plication
Finite Element Analysis: Trident vs Bident Model Initial Position Plication TRIDENT BIDENT Model SL distance (mm) CC-distance (mm) MVA (cm 2) Baseline 27. 6 43. 1 9. 50 TRIDENT plication 26. 6 44. 4 8. 30 BIDENT– 2 x 2 plication 23. 5 39. 9 7. 43 Bident 2 x 2 plication outperforms Trident plication
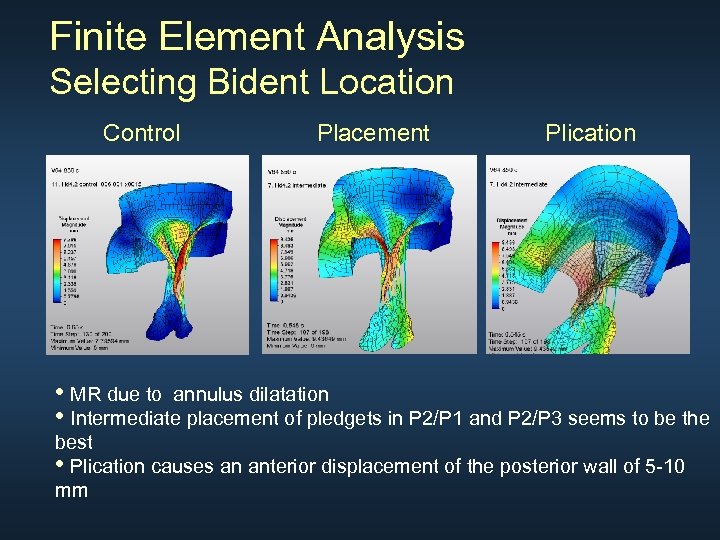 Finite Element Analysis Selecting Bident Location Control Placement Plication • MR due to annulus dilatation • Intermediate placement of pledgets in P 2/P 1 and P 2/P 3 seems to be the best • Plication causes an anterior displacement of the posterior wall of 5 -10 mm
Finite Element Analysis Selecting Bident Location Control Placement Plication • MR due to annulus dilatation • Intermediate placement of pledgets in P 2/P 1 and P 2/P 3 seems to be the best • Plication causes an anterior displacement of the posterior wall of 5 -10 mm
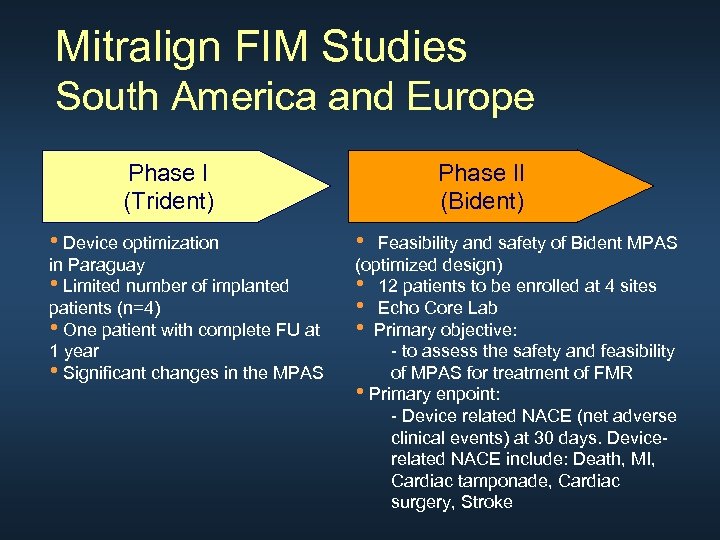 Mitralign FIM Studies South America and Europe Phase I (Trident) • Device optimization in Paraguay • Limited number of implanted patients (n=4) • One patient with complete FU at 1 year • Significant changes in the MPAS Phase II (Bident) • Feasibility and safety of Bident MPAS (optimized design) • 12 patients to be enrolled at 4 sites • Echo Core Lab • Primary objective: - to assess the safety and feasibility of MPAS for treatment of FMR • Primary enpoint: - Device related NACE (net adverse clinical events) at 30 days. Devicerelated NACE include: Death, MI, Cardiac tamponade, Cardiac surgery, Stroke
Mitralign FIM Studies South America and Europe Phase I (Trident) • Device optimization in Paraguay • Limited number of implanted patients (n=4) • One patient with complete FU at 1 year • Significant changes in the MPAS Phase II (Bident) • Feasibility and safety of Bident MPAS (optimized design) • 12 patients to be enrolled at 4 sites • Echo Core Lab • Primary objective: - to assess the safety and feasibility of MPAS for treatment of FMR • Primary enpoint: - Device related NACE (net adverse clinical events) at 30 days. Devicerelated NACE include: Death, MI, Cardiac tamponade, Cardiac surgery, Stroke
 Bident FIM Study Clinical Sites ● Study PI: - Eberhard Grube, MD ● Study sites and Site PIs - St. Elisabeth Krankenhaus, Heart Center Rhein-Ruhr • Dr. E. Grube - Helios Heart Center, Siegburg, Germany: • Dr. L. Buellesfeld - Asklepios Klinik St. Georg, Hamburg, Germany: • Dr. K. H. Kuck - Dante Pazzanesse Cardiology Center, Sao Paulo, Brazil: • Drs. A. Abizaid, C. Esteves, E. Grube - Klinikum-Oldenburg, Germany: • Dr. A. Elsaesser ● Echo Core Lab - Med. Star, Washington , DC, USA • Dr. Neil Weissman
Bident FIM Study Clinical Sites ● Study PI: - Eberhard Grube, MD ● Study sites and Site PIs - St. Elisabeth Krankenhaus, Heart Center Rhein-Ruhr • Dr. E. Grube - Helios Heart Center, Siegburg, Germany: • Dr. L. Buellesfeld - Asklepios Klinik St. Georg, Hamburg, Germany: • Dr. K. H. Kuck - Dante Pazzanesse Cardiology Center, Sao Paulo, Brazil: • Drs. A. Abizaid, C. Esteves, E. Grube - Klinikum-Oldenburg, Germany: • Dr. A. Elsaesser ● Echo Core Lab - Med. Star, Washington , DC, USA • Dr. Neil Weissman
 Inclusion Criteria ● Patient is ≥ 18 years old ● Functional Mitral Regurgitation ≥ 2+ ● NYHA Class: II-IV ● EF% ≥ 30% ● Mitral plane to apex dimension ≥ 5 cm ● Structurally normal mitral valve ● No significant calcification ● No thrombus in LV or LA
Inclusion Criteria ● Patient is ≥ 18 years old ● Functional Mitral Regurgitation ≥ 2+ ● NYHA Class: II-IV ● EF% ≥ 30% ● Mitral plane to apex dimension ≥ 5 cm ● Structurally normal mitral valve ● No significant calcification ● No thrombus in LV or LA
 Conclusions ● Mitralign Percutaneous Annuloplasty System is emerging technology for direct annuloplasty. ● First experience with the Trident System was promising but change in design concept was needed. - To simplify the procedure - To possibly improve outcomes ● Safety and feasibility of the new Bident System will be studied in the new FIM study in Germany and Brazil.
Conclusions ● Mitralign Percutaneous Annuloplasty System is emerging technology for direct annuloplasty. ● First experience with the Trident System was promising but change in design concept was needed. - To simplify the procedure - To possibly improve outcomes ● Safety and feasibility of the new Bident System will be studied in the new FIM study in Germany and Brazil.
 Thank you for your attention !
Thank you for your attention !


