0fd0b6ffdd7ac382e234f50a7992c825.ppt
- Количество слайдов: 34

CRO and Job Opportunities 동아대 문화인류학과 전공특화 프로그램 Dec 10, 2010 Aviva Park(Hye Sook Park) Confidential
The Dreamers l 자료Superstark_MV_Web. wmv Confidential 2

Agenda ØBrief of Clinical Trial ØBusiness Prospect ØCompany Introduction ØJob and JD ØMajor Distribution of Parexel Korea Employee Confidential 3

Brief of Clinical Trial Confidential 4 4
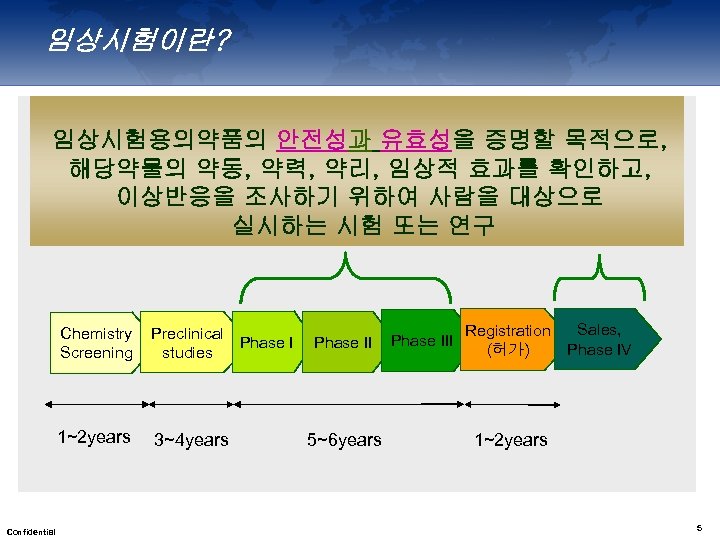
임상시험이란? 임상시험용의약품의 안전성과 유효성을 증명할 목적으로, 해당약물의 약동, 약력, 약리, 임상적 효과를 확인하고, 이상반응을 조사하기 위하여 사람을 대상으로 실시하는 시험 또는 연구 Chemistry Screening 1~2 years Confidential Preclinical Phase I studies 3~4 years Phase II 5~6 years Phase III Registration (허가) Sales, Phase IV 1~2 years 5
![임상시험관리기준 KGCP [식품의약품안전청고시 제 2009 -122호, 2009. 08. 24] 임상시험의 계획 시행 실시 모니터링 임상시험관리기준 KGCP [식품의약품안전청고시 제 2009 -122호, 2009. 08. 24] 임상시험의 계획 시행 실시 모니터링](https://present5.com/presentation/0fd0b6ffdd7ac382e234f50a7992c825/image-6.jpg)
임상시험관리기준 KGCP [식품의약품안전청고시 제 2009 -122호, 2009. 08. 24] 임상시험의 계획 시행 실시 모니터링 점검 자료의 기록 및 분석 임상시험결과보고서 작성 등에 관한 기준을 정함 정확하고 신뢰성 있는 결과의 획득 피험자 비밀 보장 피험자 권익 보호 Confidential 6

ICH-GCP ICH Guideline for Good Clinical Practice l ICH 란? ü International Conference on Harmonization of Technical Requirement for Registration of Pharmaceuticals for Human Use ü 3 regions: European Union, United States, Japan ü 표준화된 약물 등록의 요건을 합의하기 위해 구성 l ICH-GCP ü KGCP (임상시험관리기준)과 거의 동일한 내용 ü Should be followed when generating clinical trial data that are intended to be submitted to regulatory authority ü May be applied to other clinical investigations that may have impact on the safety and well-being of human subjects Confidential 7

CFR Code of Federal Regulation (미국 연방규정집) l 미국 연방등록사무소(OFR)에서 발행하며, 50개 분야 (Title) 로 나누어져 있 는 미국의 대표적인 규정집 l Title of 21 CFR : Food and Drugs - Chapter 1 : Food and Drug administration, Department of Health and Human Services ü ü ü Confidential 21 CFR Part 11 – Electronic Records and Electronic Signature 21 CFR Part 50 – Protection of Human Subjects 21 CFR Part 54 – Financial Disclosure by Clinical Investigators 21 CFR Part 56 – Institutional Review Boards 21 CFR Part 312 – Investigational New Drug Application 21 CFR Part 314 – Application for FDA approval to Market a New Drug 8
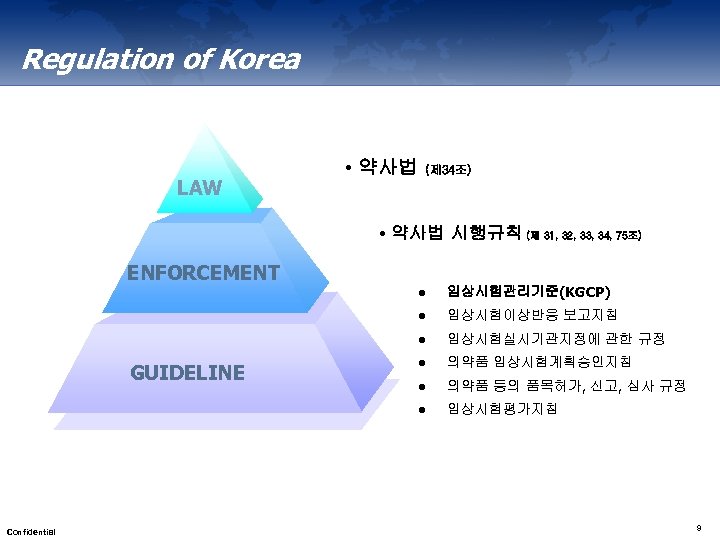
Regulation of Korea LAW • 약사법 (제 34조) • 약사법 시행규칙 (제 31, 32, 33, 34, 75조) ENFORCEMENT l l 임상시험실시기관지정에 관한 규정 l 의약품 임상시험계획승인지침 l 의약품 등의 품목허가, 신고, 심사 규정 l Confidential 임상시험이상반응 보고지침 l GUIDELINE 임상시험관리기준(KGCP) 임상시험평가지침 9

Business Prospect Confidential 10 10

Business Growth and Prospect 1980 1990 건강보험 시행, 국민소득증가 1987, 물질 특허제, 연구개발 투자 증대 2000 의약분업, 처방증가 약가인하 압력, 노령화 가속 2010 Confidential 11
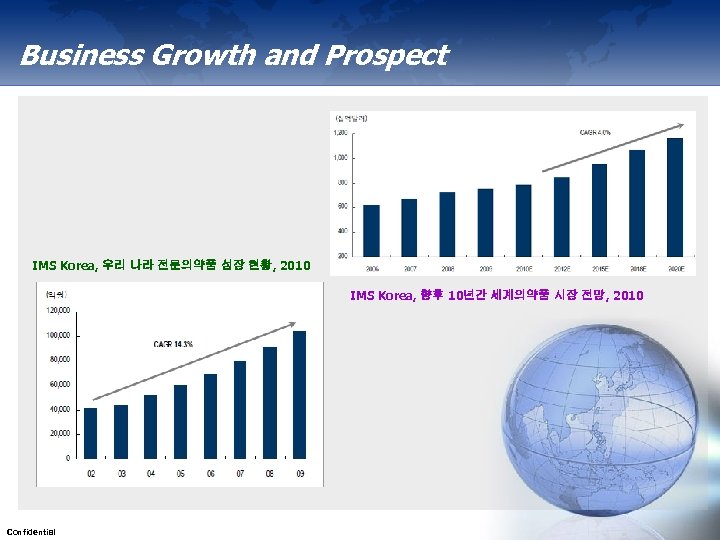
Business Growth and Prospect IMS Korea, 우리 나라 전문의약품 성장 현황, 2010 IMS Korea, 향후 10년간 세계의약품 시장 전망, 2010 Confidential 12
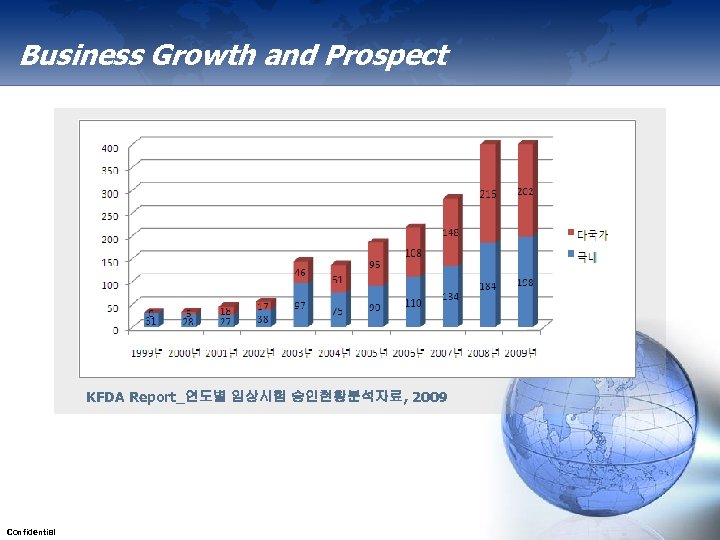
Business Growth and Prospect KFDA Report_연도별 임상시험 승인현황분석자료, 2009 Confidential 13

Competitive Pressures for CROs Small CROs serve niche requirements; large CROs, the global trial portfolio needs. How can the mid-size CRO differentiate? Publish date: Nov 1, 2010 By: Lisa Henderson Source: Applied Clinical Trials Trends over the past five years have indicated the changing relationships between the pharmaceutical sponsors and the CRO. Historically, the CROs have always indicated they wanted to use their brains, and be a more strategic partner to pharma. But the complaint was that they were the arms and legs, just doing what the pharma said. These "mindless" activities translated into a high number of transactional relationships that are still prevalent in the industry. Increasingly, the transactional relationship is giving way to the strategic relationship. The strategic relationships allow pharma to take advantage of the brains and mine the capabilities of a limited number of CROs, to hopefully bring efficiencies to the process. This trend has been borne out by both industry surveys and recent headlines. In the June 2009 issue of Applied Clinical Trials, an article based on the results of a global CRO usage survey indicated plans by large and mid-size sponsors to use fewer than five CROs in that year. The authors wrote that significant differences in relationship structure are determined by the sponsor's size. Large and mid-size went more strategic, while the small pharma chose transactional. The authors based this on the smaller project size where task-oriented outsourcing best met their needs, as well as a belief that they couldn't compete with the larger sponsors financially or for attention. Most recently in the headlines are deals that clearly reflect this trend toward fewer outsourcing partners. For example, reports that Glaxo. Smith. Kline was entering into strategic relationships with Parexel and PPD, following its mid-year announcement to whittle its current roster of 30 CROs it was working with down to a few. Confidential 14

Competitive Pressures for CROs Small CROs serve niche requirements; large CROs, the global trial portfolio needs. How can the mid-size CRO differentiate? Publish date: Nov 1, 2010 By: Lisa Henderson Source: Applied Clinical Trials On its side, Parexel has announced similar deals with Eli Lilly and Bristol-Myers Squibb. Covance too entered into a strategic agreement with Sanofi for lab-related business. An article in the Wall Street Journal, posted after the GSK-Parexel-PPD announcement, forecasted that these large CRO-pharma deals will squeeze the mid-size CRO. Research and analysts interviewed for the article noted that mid-size CROs suffer from being too small to be considered a broad global vendor, and too big to be a niche player. These industry observers noted that the best thing to do was to get big quick via M&A or capital infusion, or find a way to differentiate in a specialty that allows them to stand out. Large CROs have 50 percent of the global market and revenues above $500 million. These CROs include (in no particular order) Parexel, PPD, ICON, Quintiles, and Covance. Mid-size CROs feature revenues below $500 million, with Kendle coming close at $475 million for 2008 revenues. PRA, Pharma. Net, INC, RPS, and Medpace also fall in the $100 million to $500 million range. In the coming months, we will feature news and articles on these and other competitive pressures faced by all sized CROs. In the meantime, this issue features an article based on the results of our Salary Survey. These results show that the competitive pressures faced by the CROs and pharma in regard to M&As and global trials, trickle down in a big way on individual workload. Confidential 15

Company Introduction Confidential 16 16

45 50 PAREXEL HAS SUPPORTED NEARLY ALL OF THE TOP FIFTY DRUGS ON THE MARKET Confidential 17

PAREXEL International Vision Strives to be the premier provider to the bio/pharmaceutical and medical device industries for development and commercialization of new medical therapies worldwide. Mission Combine the strength of our expertise, experience and innovation to advance the worldwide success of the bio/pharmaceutical and medical device industries in preventing and curing disease. Think Globally, Act Locally Confidential 18

Milestones: + 25 Years of Biopharmaceutical Industry Progress 2009 Product Line Strategy Award, Frost & Sullivan Awards 2009 Best Performing CRO, Bio. Singapore Awards 2008 Most Innovative Patient Recruitment Strategy, GCPj Awards 2008 Best Contract Research Organization, Script Awards 2007 Asia Pacific Healthcare Best Practice Award, Frost & Sullivan Awards 2007 APEX Full Acquisition by PAREXEL 2003 Alliance between PAREXEL & APEX 1999 Foundation of APEX In Taiwan 1995 PAREXEL expands operation in Japan 1982 Foundation of PAREXEL Confidential 19
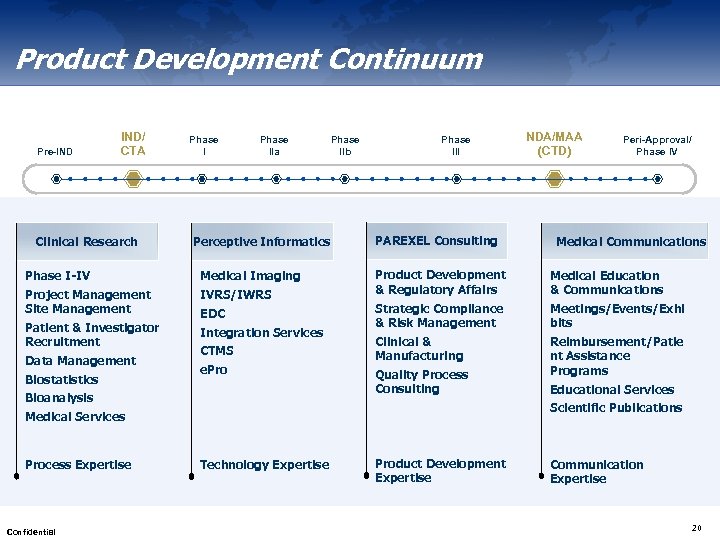
Product Development Continuum Pre-IND IND/ CTA Clinical Research Phase IIa Perceptive Informatics Phase I-IV Medical Imaging Project Management Site Management IVRS/IWRS Patient & Investigator Recruitment Integration Services Data Management Biostatistics EDC CTMS e. Pro Bioanalysis Phase IIb Phase III PAREXEL Consulting Confidential Peri-Approval/ Phase IV Medical Communications Product Development & Regulatory Affairs Medical Education & Communications Strategic Compliance & Risk Management Meetings/Events/Exhi bits Clinical & Manufacturing Reimbursement/Patie nt Assistance Programs Quality Process Consulting Educational Services Scientific Publications Medical Services Process Expertise NDA/MAA (CTD) Technology Expertise Product Development Expertise Communication Expertise 20
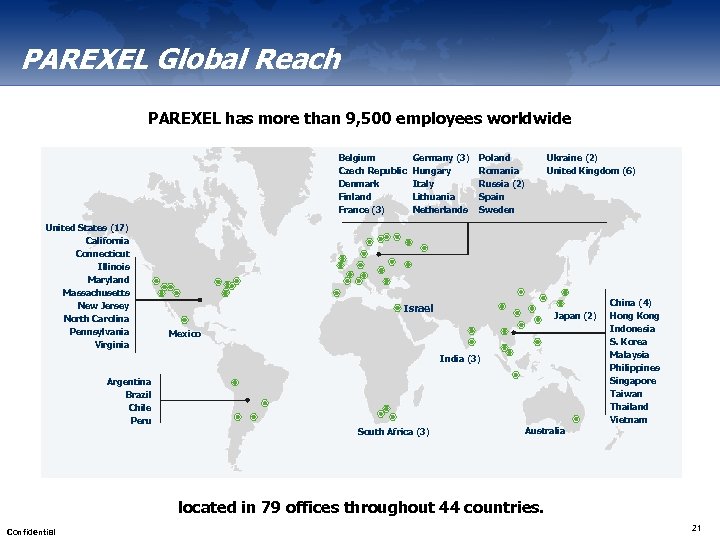
PAREXEL Global Reach PAREXEL has more than 9, 500 employees worldwide Belgium Czech Republic Denmark Finland France (3) United States (17) California Connecticut Illinois Maryland Massachusetts New Jersey North Carolina Pennsylvania Virginia Germany (3) Hungary Italy Lithuania Netherlands Poland Romania Russia (2) Spain Sweden Israel Ukraine (2) United Kingdom (6) Japan (2) Mexico India (3) Argentina Brazil Chile Peru South Africa (3) China (4) Hong Kong Indonesia S. Korea Malaysia Philippines Singapore Taiwan Thailand Vietnam Australia located in 79 offices throughout 44 countries. Confidential 21

Job Opportunities Confidential 22 22

CRA Clinical Research Associate l Essential Function § To perform all clinical monitoring / site management activities ü ü ü ü l the ownership of investigator sites the selection of investigator/sites collating Regulatory Documentation initiating and monitoring of sites performing site documentation verification data collection drug accountability in accordance with ICH GCP guidelines Education Educated to degree level (biological science, pharmacy or other health related discipline preferred) or equivalent nursing qualification/experience. l Language Skills Competent in written and oral English. l Minimum Work Experience Substantial monitoring experience in clinical research. Confidential 23
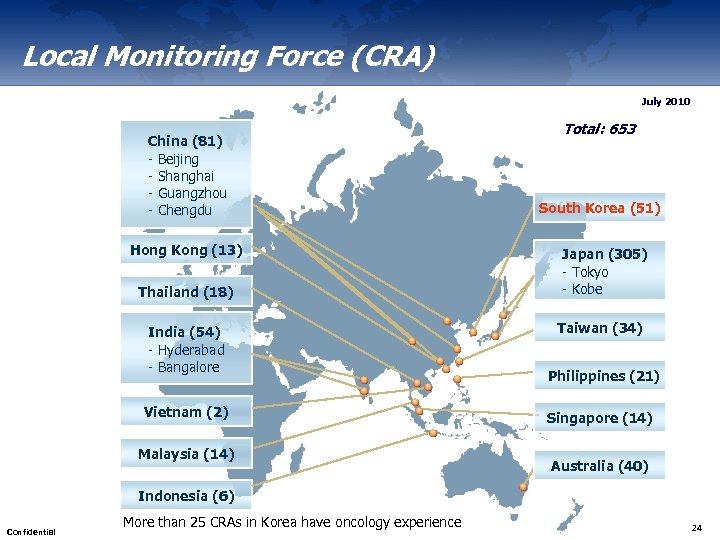
Local Monitoring Force (CRA) July 2010 China (81) - Beijing - Shanghai - Guangzhou - Chengdu Hong Kong (13) Thailand (18) India (54) - Hyderabad - Bangalore Vietnam (2) Malaysia (14) Total: 653 South Korea (51) Japan (305) - Tokyo - Kobe Taiwan (34) Philippines (21) Singapore (14) Australia (40) Indonesia (6) Confidential More than 25 CRAs in Korea have oncology experience 24

COL/PL Clinical Operational Leader/Project Leader l Essential Function § Coordinating the functional team members and their activities across all geographies § liaising with project leadership and the sponsor to ensure that the Global Research § l Operations deliverables (timeline, quality, productivity) are met Overall accountability for the execution of the clinical operations strategy on the project Education Educated to degree level (biological science, pharmacy or other health related discipline preferred) or equivalent nursing qualification/experience l Language Skills Competent in written and oral English • Minimum Work Experience Substantial experience in clinical research including relevant experience as a team Leader in Clinical functions or proven experience in coordinating clinical trials. Relevant Work experience in clinical research Experience in Coordination of Clinical Trials – Preferably relevant working experience as an Associate Clinical Operations Leader or proven experience in a similar role Individuals should have a strong understanding of the cross functional activities Confidential 25
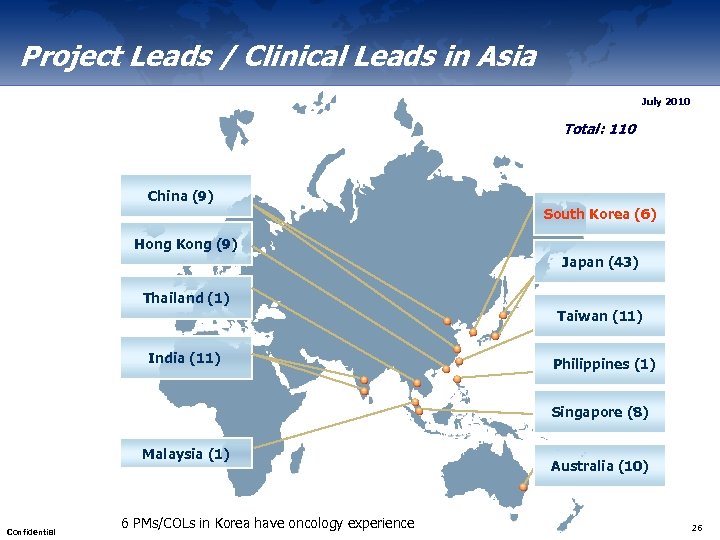
Project Leads / Clinical Leads in Asia July 2010 Total: 110 China (9) South Korea (6) Hong Kong (9) Japan (43) Thailand (1) Taiwan (11) India (11) Philippines (1) Singapore (8) Malaysia (1) Confidential 6 PMs/COLs in Korea have oncology experience Australia (10) 26

CTS/CMA CTS(Clinical Trial Specialist) l Essential Function Conduct and facilitate specific start-up activities such as § Site identification § Feasibilities § SRP collection § Review and approval § ICF customization and approval § EC and RA submissions § Preparation and negotiation of Clinical Site Agreements (CSAs) at a site level CMA(Clinical Monitoring Associate) l Essential Function § to perform remote clinical monitoring tasks and remote visits for designated projects in accordance with relevant SOPs, study specific procedures and regulations. § to include managing/coordinating/supporting clinical monitoring activities remotely ü ü ü Confidential Issue resolution Patient enrollment management Handling protocol- and/or ICF- and/or contract amendments Drug / supply management Ensuring timely data entry at a site level 27
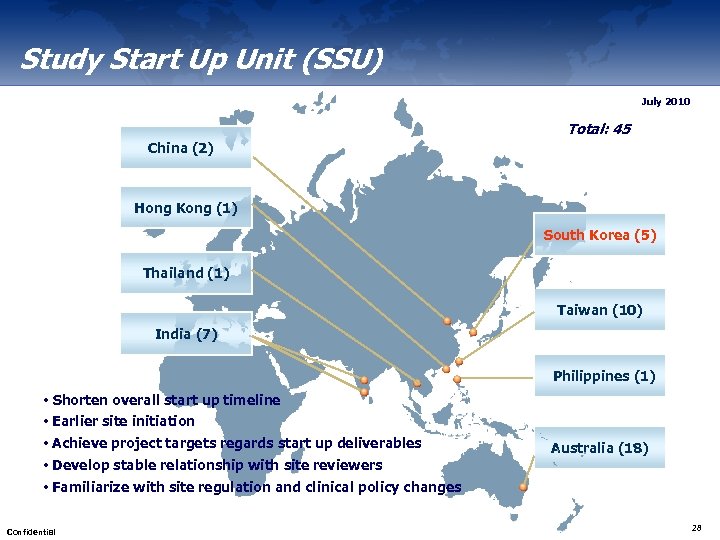
Study Start Up Unit (SSU) July 2010 Total: 45 China (2) Hong Kong (1) South Korea (5) Thailand (1) Taiwan (10) India (7) Philippines (1) • Shorten overall start up timeline • Earlier site initiation • Achieve project targets regards start up deliverables Australia (18) • Develop stable relationship with site reviewers • Familiarize with site regulation and clinical policy changes Confidential 28

Other Role Ø Quality Management Ø Consultant Ø Data Specialist Ø … Confidential 29

Major Distribution of Parexel Korea Employee Confidential 30 30
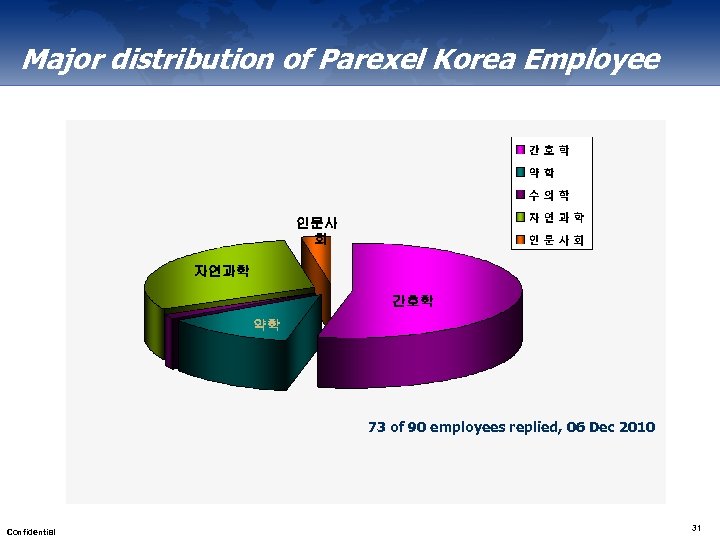
Major distribution of Parexel Korea Employee 인문사 회 자연과학 간호학 약학 73 of 90 employees replied, 06 Dec 2010 Confidential 31

Open Discussions & Questions? 32

Thank You 33
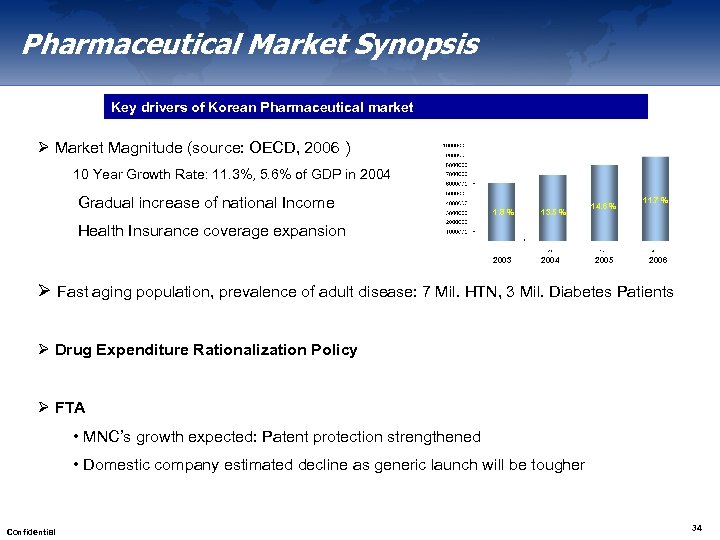
Pharmaceutical Market Synopsis Key drivers of Korean Pharmaceutical market Ø Market Magnitude (source: OECD, 2006 ) 10 Year Growth Rate: 11. 3%, 5. 6% of GDP in 2004 Gradual increase of national Income 1. 8 % 13. 5 % 2003 2004 14. 6 % 11. 7 % Health Insurance coverage expansion 2005 2006 Ø Fast aging population, prevalence of adult disease: 7 Mil. HTN, 3 Mil. Diabetes Patients Ø Drug Expenditure Rationalization Policy Ø FTA • MNC’s growth expected: Patent protection strengthened • Domestic company estimated decline as generic launch will be tougher Confidential 34
0fd0b6ffdd7ac382e234f50a7992c825.ppt