e2b52a04e5b10d33bf7db5f27ae9200a.ppt
- Количество слайдов: 29

Coupling of carbon and nitrogen cycles through humic redox reactions in an alpine stream Diane Mc. Knight, Matt Miller, Rose Cory and Mark Williams Depart. Civil, Environmental & Architectural Engineering, University of Colorado

NWTLTER: C & N transport and reactivity in Green Lakes Valley Response of pristine, cold regions to climate change and N enrichment
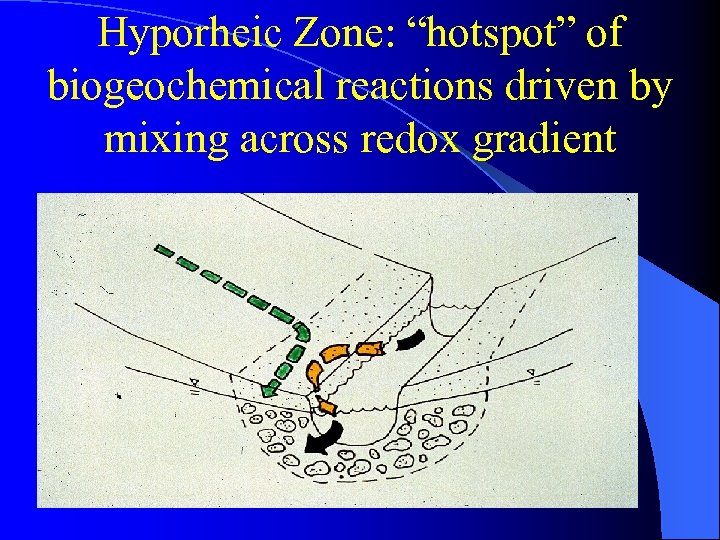
Hyporheic Zone: “hotspot” of biogeochemical reactions driven by mixing across redox gradient
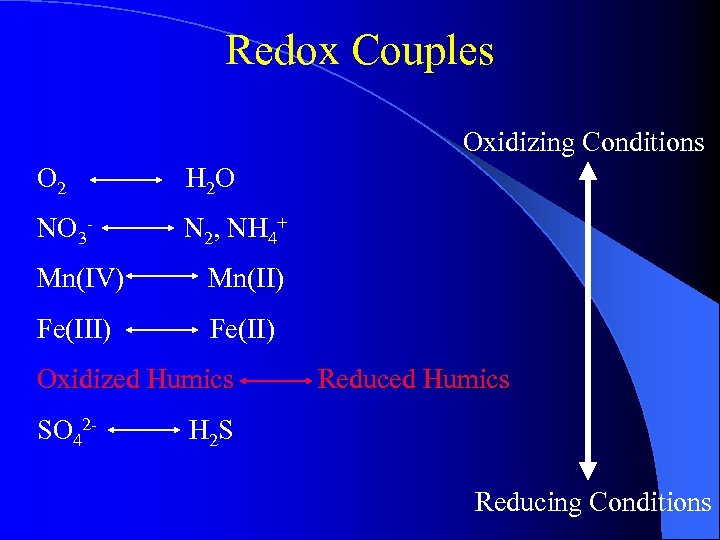
Redox Couples Oxidizing Conditions O 2 H 2 O NO 3 - N 2, NH 4+ Mn(IV) Mn(II) Fe(II) Oxidized Humics SO 42 - Reduced Humics H 2 S Reducing Conditions
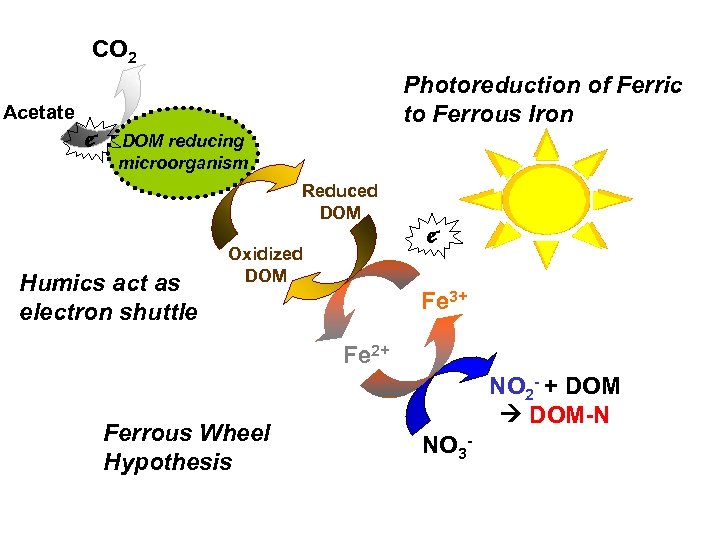
CO 2 Photoreduction of Ferric to Ferrous Iron Acetate e- DOM reducing microorganism Reduced DOM Humics act as electron shuttle Oxidized DOM e. Fe 3+ Fe 2+ Ferrous Wheel Hypothesis NO 2 - + DOM-N NO 3 -
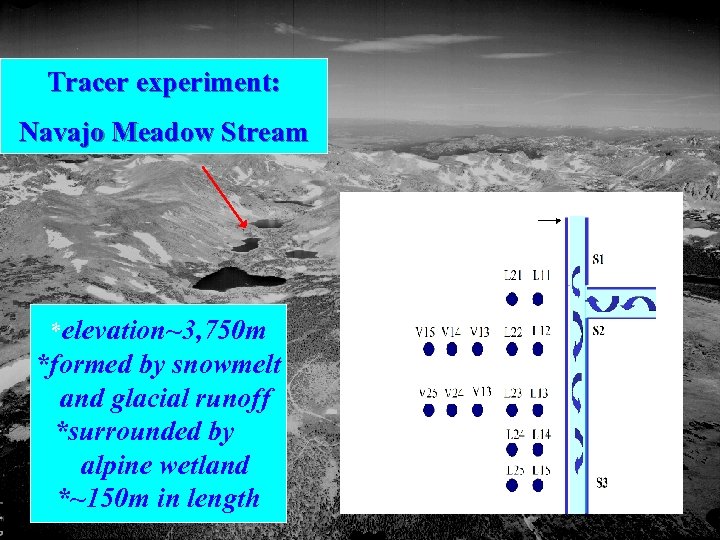
Tracer experiment: Navajo Meadow Stream *elevation~3, 750 m *formed by snowmelt and glacial runoff *surrounded by alpine wetland *~150 m in length

Approach: Tracer injection experiment and modeling with OTIS Main Channel: Lateral inflow Advection Dispersion Storage Zone: Transient storage s Transient storage
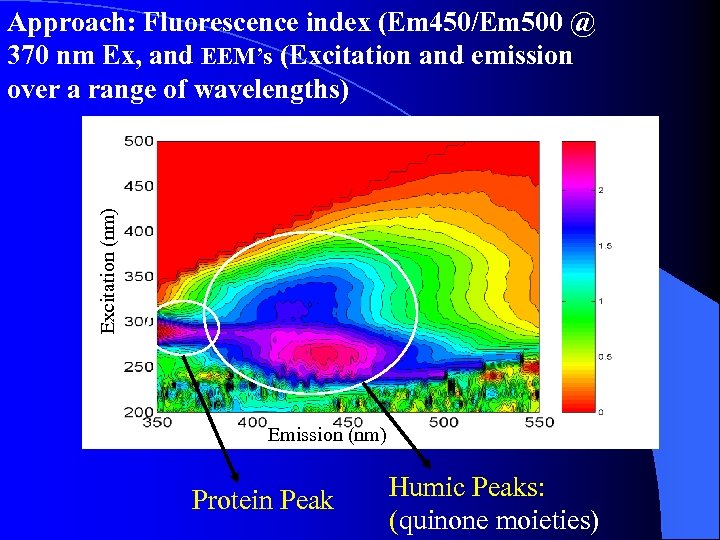
Excitation (nm) Approach: Fluorescence index (Em 450/Em 500 @ 370 nm Ex, and EEM’s (Excitation and emission over a range of wavelengths) Emission (nm) Protein Peak Humic Peaks: (quinone moieties)
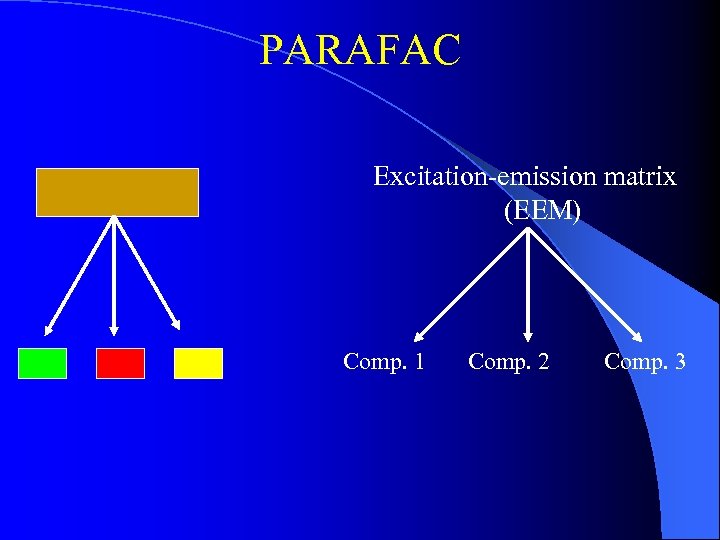
PARAFAC Excitation-emission matrix (EEM) Comp. 1 Comp. 2 Comp. 3
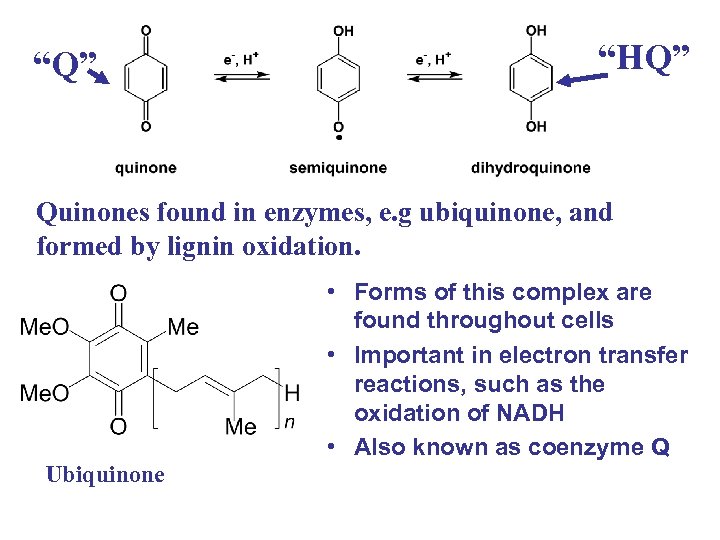
“Q” “HQ” Quinones found in enzymes, e. g ubiquinone, and formed by lignin oxidation. Ubiquinone • Forms of this complex are found throughout cells • Important in electron transfer reactions, such as the oxidation of NADH • Also known as coenzyme Q
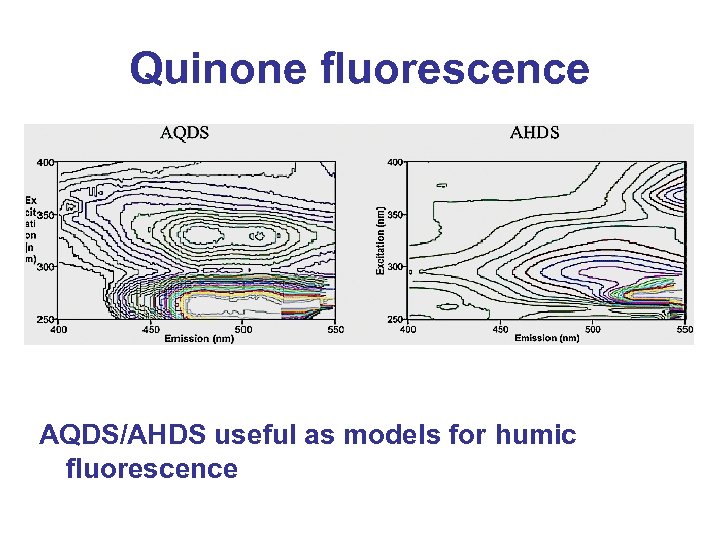
Quinone fluorescence AQDS/AHDS useful as models for humic fluorescence
![Stream Br- Addition, July 10 Background [Br-] = 0 mg/L Reach 1 Reach 2 Stream Br- Addition, July 10 Background [Br-] = 0 mg/L Reach 1 Reach 2](https://present5.com/presentation/e2b52a04e5b10d33bf7db5f27ae9200a/image-12.jpg)
Stream Br- Addition, July 10 Background [Br-] = 0 mg/L Reach 1 Reach 2 Reach 3
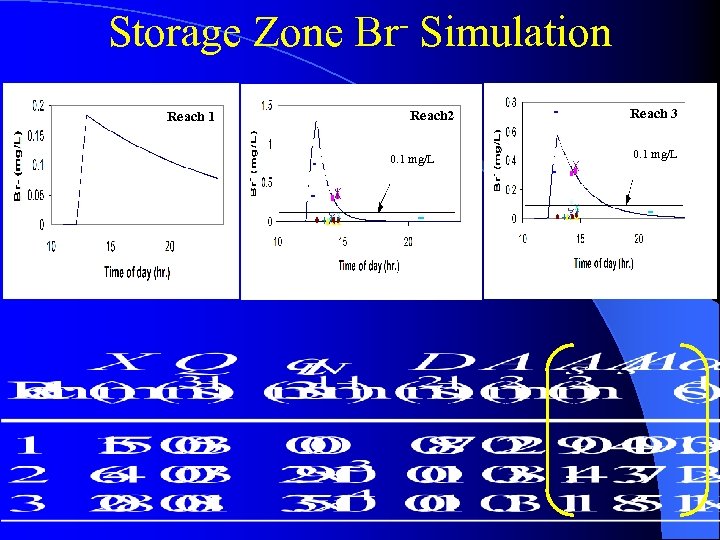
Storage Zone Br- Simulation Reach 1 Reach 2 0. 1 mg/L Reach 3 0. 1 mg/L
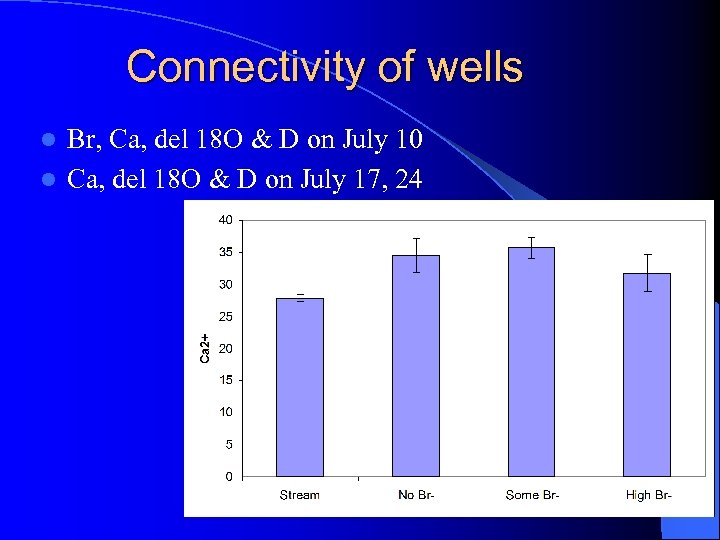
Connectivity of wells Br, Ca, del 18 O & D on July 10 l Ca, del 18 O & D on July 17, 24 l
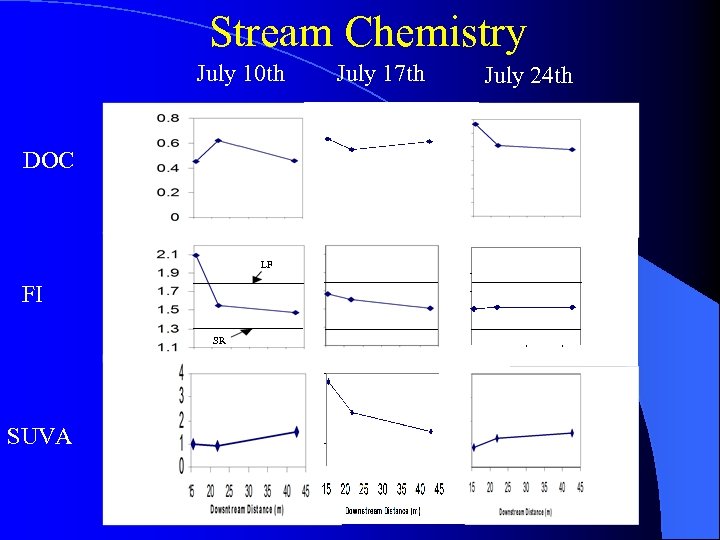
Stream Chemistry July 10 th DOC LF FI SR SUVA July 17 th July 24 th
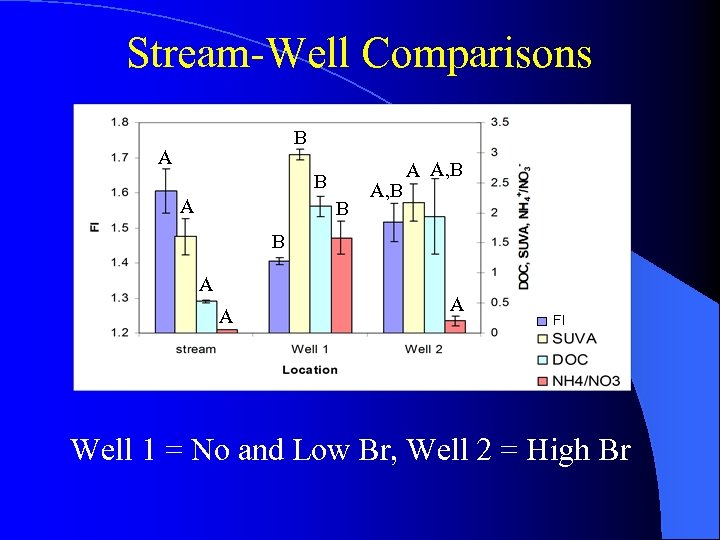
Stream-Well Comparisons B A A B A, B A A, B B A A A FI Well 1 = No and Low Br, Well 2 = High Br
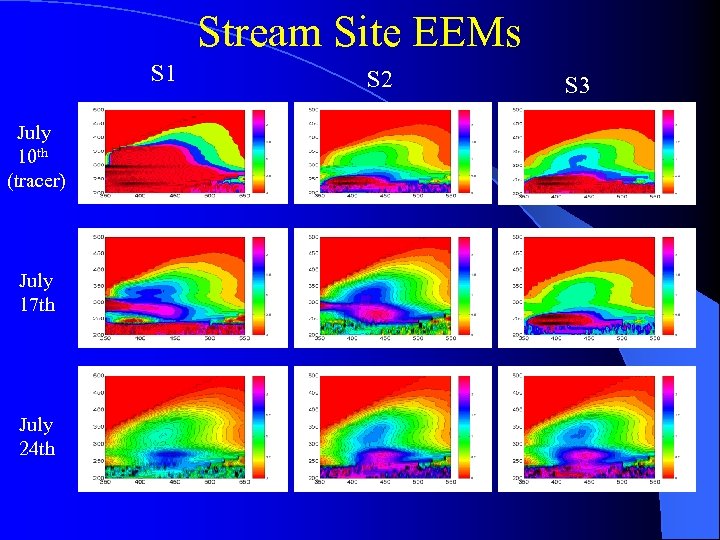
Stream Site EEMs S 1 July 10 th (tracer) July 17 th July 24 th S 2 S 3
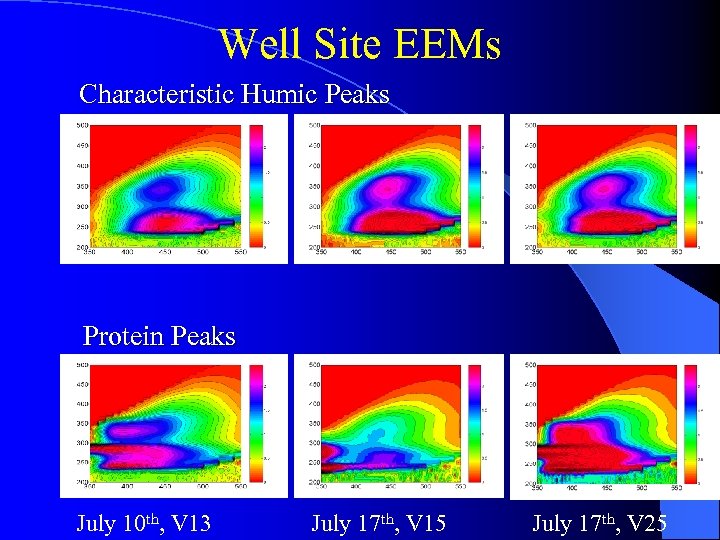
Well Site EEMs Characteristic Humic Peaks Protein Peaks July 10 th, V 13 July 17 th, V 15 July 17 th, V 25
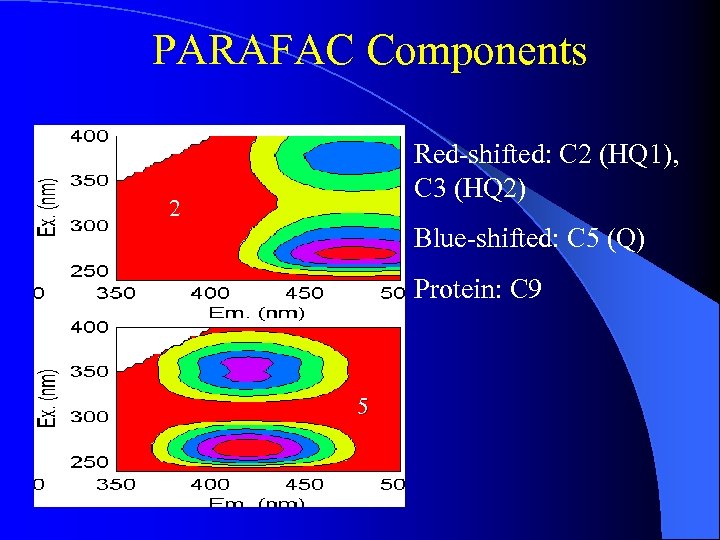
PARAFAC Components Red-shifted: C 2 (HQ 1), C 3 (HQ 2) 2 Blue-shifted: C 5 (Q) Protein: C 9 5
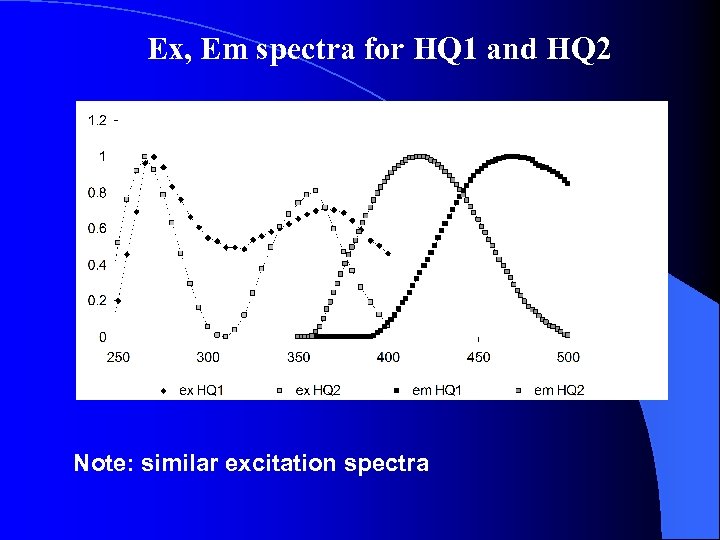
Ex, Em spectra for HQ 1 and HQ 2 Note: similar excitation spectra
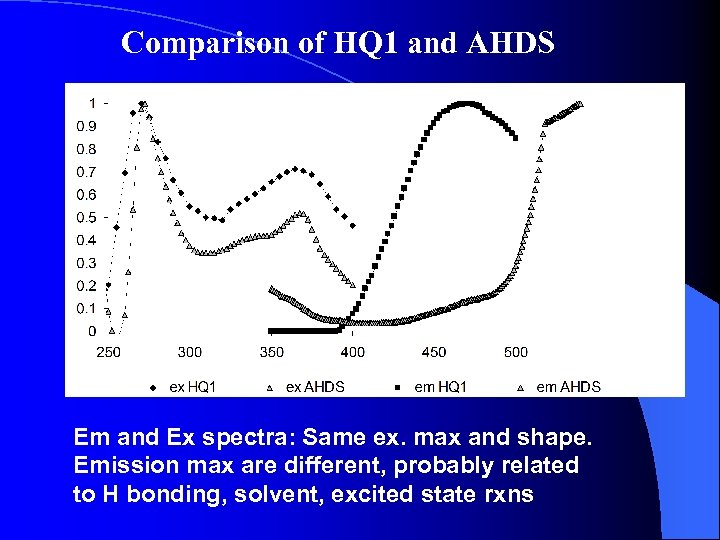
Comparison of HQ 1 and AHDS Em and Ex spectra: Same ex. max and shape. Emission max are different, probably related to H bonding, solvent, excited state rxns
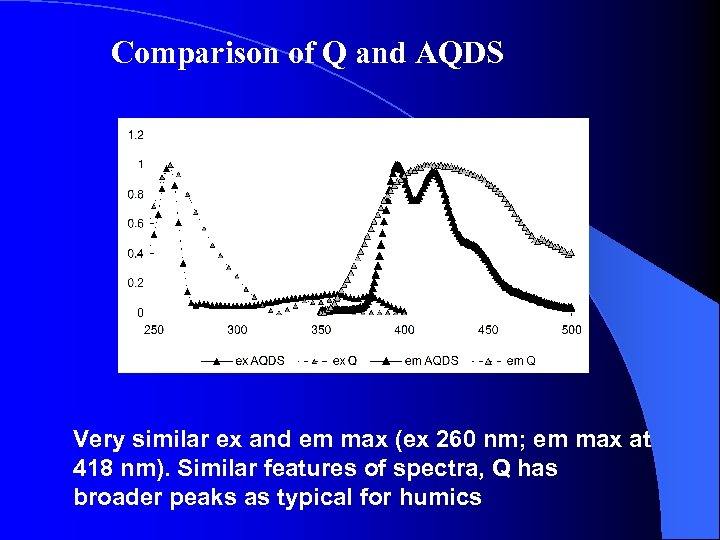
Comparison of Q and AQDS Very similar ex and em max (ex 260 nm; em max at 418 nm). Similar features of spectra, Q has broader peaks as typical for humics
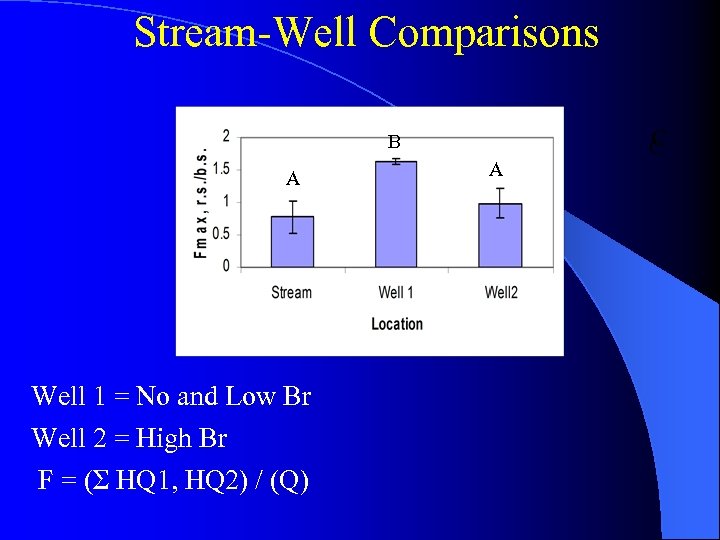
Stream-Well Comparisons B A Well 1 = No and Low Br Well 2 = High Br F = (Σ HQ 1, HQ 2) / (Q) A C C
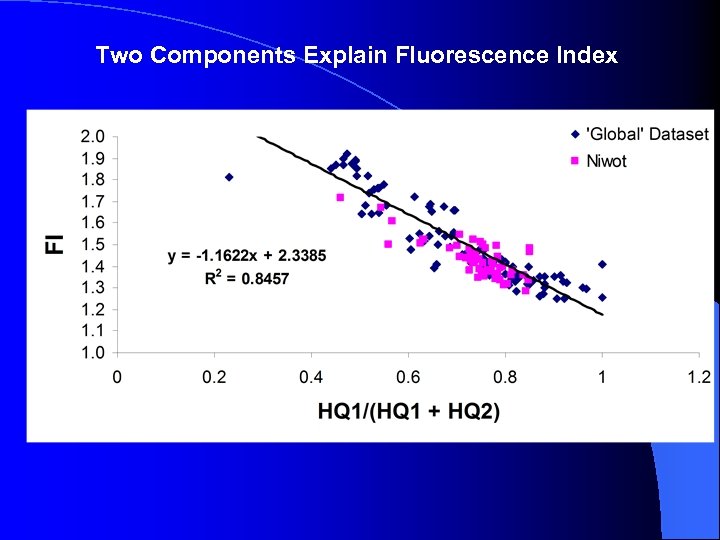
Two Components Explain Fluorescence Index
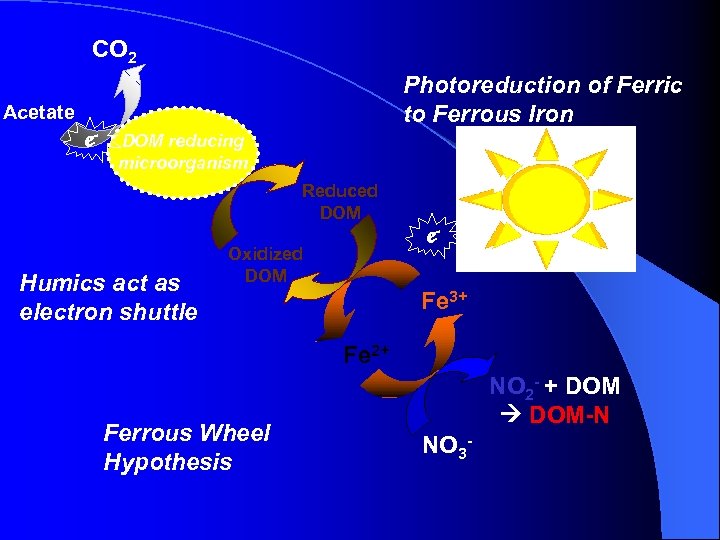
CO 2 Photoreduction of Ferric to Ferrous Iron Acetate e- DOM reducing microorganism Reduced DOM Humics act as electron shuttle Oxidized DOM e. Fe 3+ Fe 2+ Ferrous Wheel Hypothesis NO 2 - + DOM-N NO 3 -
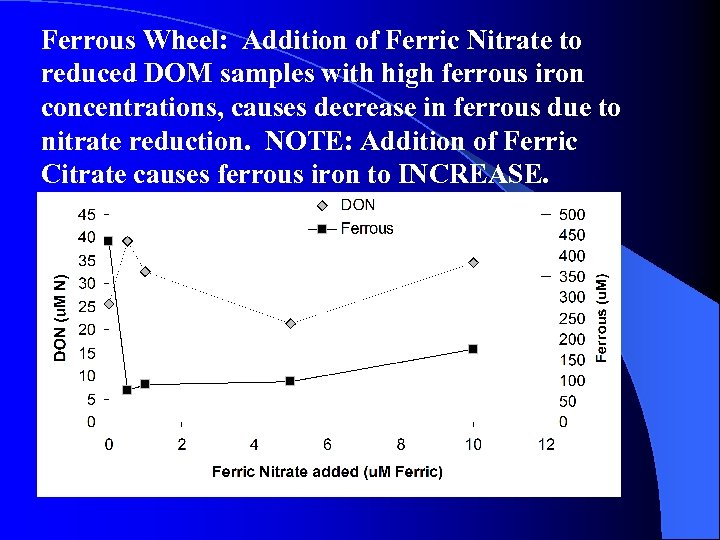
Ferrous Wheel: Addition of Ferric Nitrate to reduced DOM samples with high ferrous iron concentrations, causes decrease in ferrous due to nitrate reduction. NOTE: Addition of Ferric Citrate causes ferrous iron to INCREASE.

Hyporheic zone interactions, e. g. humic redox!!, hotspot of C & N interactions, influencing N transport in alpine systems. Fluorescence index = HQ 1/HQ 2 FI increases with microbial sources (primary and secondary) Nitrogen and Carbon cycling coupled by biotic and chemical processes

Questions?
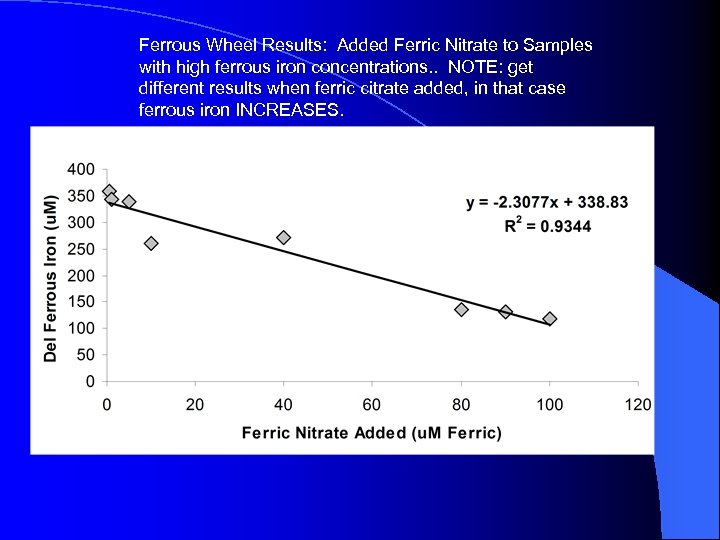
Ferrous Wheel Results: Added Ferric Nitrate to Samples with high ferrous iron concentrations. . NOTE: get different results when ferric citrate added, in that case ferrous iron INCREASES.
e2b52a04e5b10d33bf7db5f27ae9200a.ppt