e5fa8db3b8ed6223aa02ec0caf8dbbe0.ppt
- Количество слайдов: 22
 Cost-effectiveness and QALY Assessment Ilana F. Gareen, Ph. D. Brown University Center for Statistical Sciences igareen@stat. brown. edu ACRIN Cardiovascular Committee
Cost-effectiveness and QALY Assessment Ilana F. Gareen, Ph. D. Brown University Center for Statistical Sciences igareen@stat. brown. edu ACRIN Cardiovascular Committee
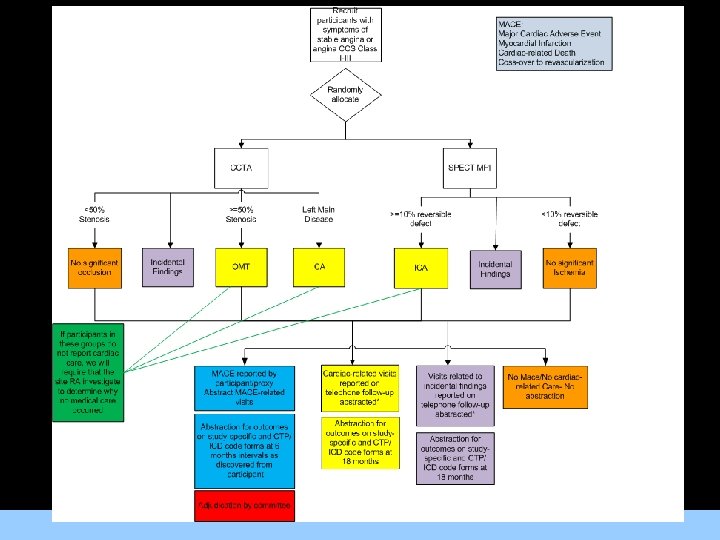
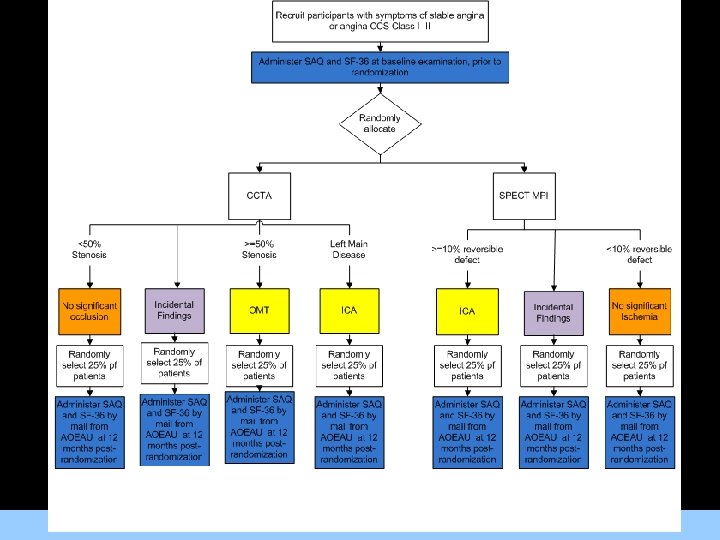
 RESCUE AIMS Primary Aim Compare outcomes of participants with symptoms of stable angina undergoing CCTA as initial method of CAD diagnosis (Group A) to SPECT MPI (Group B) as a guide to OMT. Secondary Aims 1. To evaluate the ability of available prognostic indices to predict revascularization or MACE using CCTA information and to develop new indices using the RESCUE trial data. 2. To determine the cost, effectiveness, and incremental cost-effectiveness of CCTA versus SPECT MPI in the evaluation of participants with symptoms of stable angina. 3. To compare angina symptoms and self-reported health status of participants with symptoms of stable angina undergoing CCTA as initial method of CAD diagnosis (Group A) to SPECT MPI (Group B) as a guide to OMT. ACRIN Cardiovascular Committee
RESCUE AIMS Primary Aim Compare outcomes of participants with symptoms of stable angina undergoing CCTA as initial method of CAD diagnosis (Group A) to SPECT MPI (Group B) as a guide to OMT. Secondary Aims 1. To evaluate the ability of available prognostic indices to predict revascularization or MACE using CCTA information and to develop new indices using the RESCUE trial data. 2. To determine the cost, effectiveness, and incremental cost-effectiveness of CCTA versus SPECT MPI in the evaluation of participants with symptoms of stable angina. 3. To compare angina symptoms and self-reported health status of participants with symptoms of stable angina undergoing CCTA as initial method of CAD diagnosis (Group A) to SPECT MPI (Group B) as a guide to OMT. ACRIN Cardiovascular Committee
 For Images/Graphics
For Images/Graphics
 For Images/Graphics
For Images/Graphics
 For Images/Graphics
For Images/Graphics
 For Images/Graphics
For Images/Graphics
 For Images/Graphics
For Images/Graphics
 For Images/Graphics
For Images/Graphics
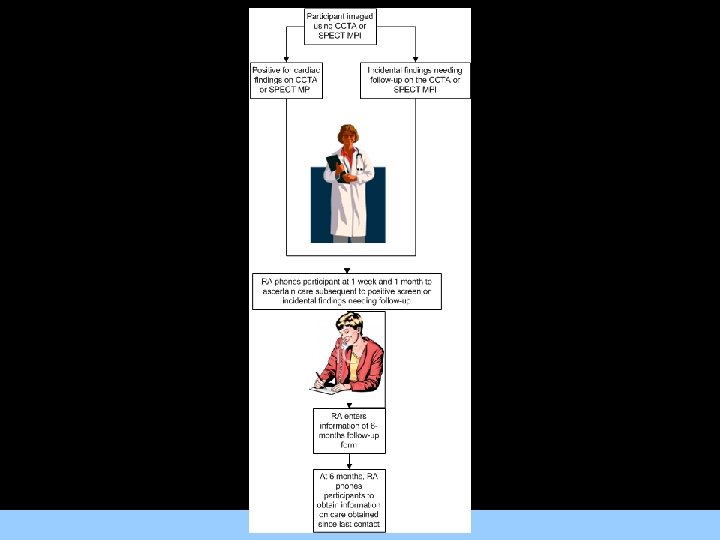
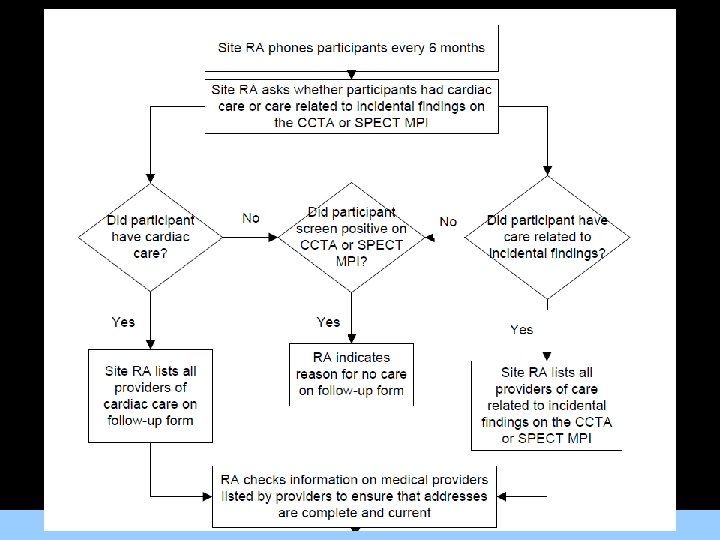
 Triggering Medical Record Abstraction • Medical chart abstraction will be triggered by participant responses during telephone follow up For Images/Graphics and by initial CCTA and SPECT MPI results.
Triggering Medical Record Abstraction • Medical chart abstraction will be triggered by participant responses during telephone follow up For Images/Graphics and by initial CCTA and SPECT MPI results.
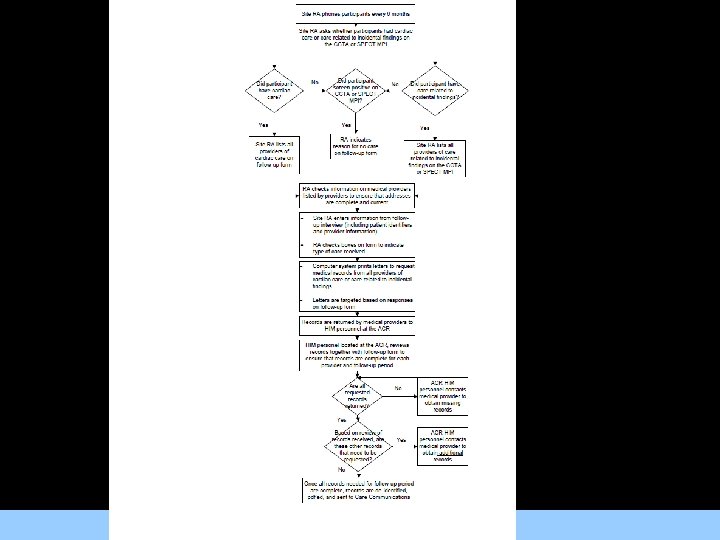
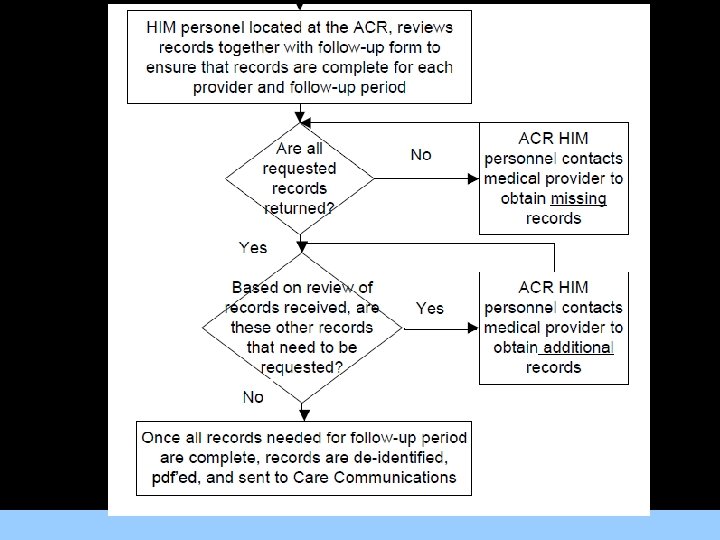
 Triggering Medical Record Abstraction • All records relating to the following will be abstracted: For Images/Graphics – MACE/revascularization events; – Medical care for cardiac issues; – Medical care for OMT; – Medical care related to incidental findings on the CCTA or SPECT MPI examinations.
Triggering Medical Record Abstraction • All records relating to the following will be abstracted: For Images/Graphics – MACE/revascularization events; – Medical care for cardiac issues; – Medical care for OMT; – Medical care related to incidental findings on the CCTA or SPECT MPI examinations.
 Centralized Process • HIM staff at the ACR will collect, prepare records for shipment to the centralized abstraction facility • Medical records abstraction and coding will be For a. Images/Graphics performed by single central medical record abstraction company. • Centralizing the abstraction process with a single group of abstractors simplifies quality control, ensuring consistent and reliable coding of medical conditions and care.
Centralized Process • HIM staff at the ACR will collect, prepare records for shipment to the centralized abstraction facility • Medical records abstraction and coding will be For a. Images/Graphics performed by single central medical record abstraction company. • Centralizing the abstraction process with a single group of abstractors simplifies quality control, ensuring consistent and reliable coding of medical conditions and care.
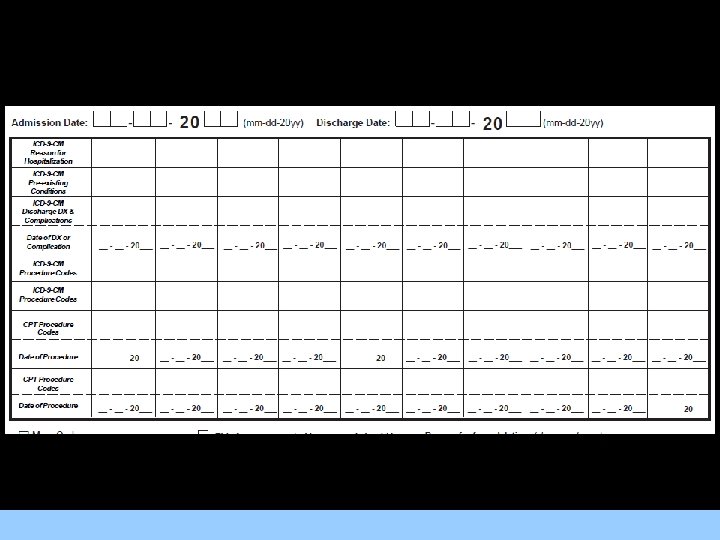
 Collection of Abstraction Data • All abstraction will be done directly onto forms on laptop computers. These computer forms incorporate range checks and International For Images/Graphics Classification of Disease (ICD) and Current Procedural Terminology (CPT) look-ups which ensure that data are as error-free as possible. • The data on the laptops are routinely backed up and uploaded to the central computer at the conclusion of the abstraction assignment at each site.
Collection of Abstraction Data • All abstraction will be done directly onto forms on laptop computers. These computer forms incorporate range checks and International For Images/Graphics Classification of Disease (ICD) and Current Procedural Terminology (CPT) look-ups which ensure that data are as error-free as possible. • The data on the laptops are routinely backed up and uploaded to the central computer at the conclusion of the abstraction assignment at each site.
 For Images/Graphics
For Images/Graphics
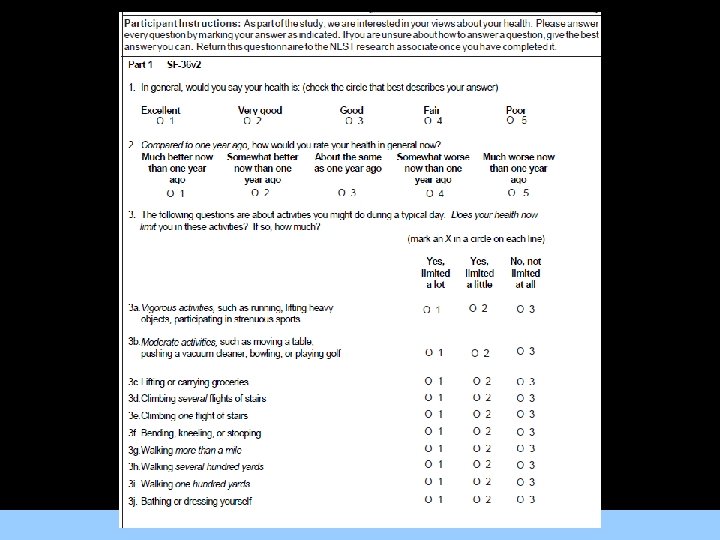
 Patient Reported Outcomes • The importance of patient self-reported outcomes is widely recognized in the literature and by the US Food and Drug Administration. For Images/Graphics – Global measures of health, such as the 36 -Item Short Form Survey Instrument, version 2 (SF-36 v 2), are useful to compare participant health status across studies of different health conditions as well as to derive health utilities for use in costeffectiveness analyses. – Disease-specific measures of participant health status, such as the Seattle Angina Questionnaire (SAQ), are useful to collect detailed information regarding the specific disease outcome of interest.
Patient Reported Outcomes • The importance of patient self-reported outcomes is widely recognized in the literature and by the US Food and Drug Administration. For Images/Graphics – Global measures of health, such as the 36 -Item Short Form Survey Instrument, version 2 (SF-36 v 2), are useful to compare participant health status across studies of different health conditions as well as to derive health utilities for use in costeffectiveness analyses. – Disease-specific measures of participant health status, such as the Seattle Angina Questionnaire (SAQ), are useful to collect detailed information regarding the specific disease outcome of interest.
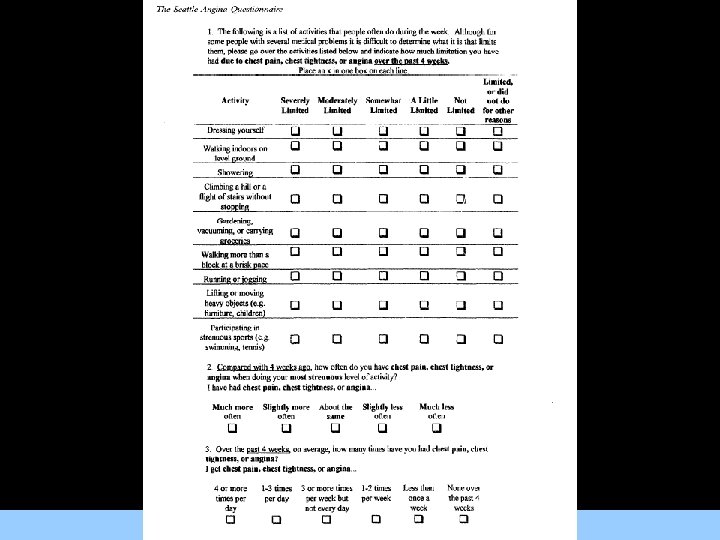
 Quality of Life Measurement Tools • Both the SF-36 and the SAQ have been used to assess health-related Qo. L in patients with angina and have been demonstrated to be internally consistent with good testretest reliability. For Images/Graphics • Selected subscales from both tools have been correlated with patient angina severity measured using the CCS Classification • Both of these tools have been used to measure participant health status in a prior large clinical trial of PCI and OMT interventions for patients with angina.
Quality of Life Measurement Tools • Both the SF-36 and the SAQ have been used to assess health-related Qo. L in patients with angina and have been demonstrated to be internally consistent with good testretest reliability. For Images/Graphics • Selected subscales from both tools have been correlated with patient angina severity measured using the CCS Classification • Both of these tools have been used to measure participant health status in a prior large clinical trial of PCI and OMT interventions for patients with angina.
 For Images/Graphics
For Images/Graphics
 For Images/Graphics
For Images/Graphics
 For Images/Graphics
For Images/Graphics
 Administration of Qo. L Tools: • Administered at the sites at the Baseline examination prior to randomization. For Images/Graphics • Administered by the ACRIN Outcomes and Economics Unit (AOEU) located within the ACRIN Biostatistics Center at the Brown University Center for Statistical Sciences at 12 months post-randomization.
Administration of Qo. L Tools: • Administered at the sites at the Baseline examination prior to randomization. For Images/Graphics • Administered by the ACRIN Outcomes and Economics Unit (AOEU) located within the ACRIN Biostatistics Center at the Brown University Center for Statistical Sciences at 12 months post-randomization.
 Ensuring Completion of Qo. L Tools: • The site will fax Participant Contact Information Forms to the AOEU following the baseline visit. For Images/Graphics • AEOU personnel will mail copies of Qo. L tools to participants along with pre-addressed, stamped envelopes for return mailing to the AOEU. Participants will be provided with a toll-free number (which is answered by the AOEU RA) should they require assistance with reading questionnaires.
Ensuring Completion of Qo. L Tools: • The site will fax Participant Contact Information Forms to the AOEU following the baseline visit. For Images/Graphics • AEOU personnel will mail copies of Qo. L tools to participants along with pre-addressed, stamped envelopes for return mailing to the AOEU. Participants will be provided with a toll-free number (which is answered by the AOEU RA) should they require assistance with reading questionnaires.
 Ensuring Completion of Qo. L Tools: • If the questionnaires are not received at the AOEU within 10 working days of the date of the mailing, an AOEU RA will telephone the participant – To determine whether the questionnaires were received – If the participant has the questionnaires, the participant will be asked to complete and return them. – If the participant has not received the questionnaires, the AOEU RA will confirm the delivery address and send the tools to the participant. For Images/Graphics
Ensuring Completion of Qo. L Tools: • If the questionnaires are not received at the AOEU within 10 working days of the date of the mailing, an AOEU RA will telephone the participant – To determine whether the questionnaires were received – If the participant has the questionnaires, the participant will be asked to complete and return them. – If the participant has not received the questionnaires, the AOEU RA will confirm the delivery address and send the tools to the participant. For Images/Graphics
 Ensuring Completion of Qo. L Tools: • If questionnaires are not returned within 20 working days thereafter, the AOEU RA will attempt to complete the questionnaires in a telephone interview. – Telephone interviews will be conducted only as a final measure to avoid any biases introduced by differences in the method of administration of the questionnaires, and the mode of administration of all such questionnaires will be documented in the trial database. – The AOEU RA will not attempt to interpret a question; training will be provided to help ensure that the AOEU RAs facilitate completion of the Qo. L questionnaires in a standardized fashion. For Images/Graphics
Ensuring Completion of Qo. L Tools: • If questionnaires are not returned within 20 working days thereafter, the AOEU RA will attempt to complete the questionnaires in a telephone interview. – Telephone interviews will be conducted only as a final measure to avoid any biases introduced by differences in the method of administration of the questionnaires, and the mode of administration of all such questionnaires will be documented in the trial database. – The AOEU RA will not attempt to interpret a question; training will be provided to help ensure that the AOEU RAs facilitate completion of the Qo. L questionnaires in a standardized fashion. For Images/Graphics


