da1ae5754a3612c9791131f42860bc0d.ppt
- Количество слайдов: 45
 CONFIDENTIAL Update on International Medical Affairs Alexander Petersen, Mc. Kinsey Stanley Bukofzer, Abbott Laboratories March 30, 2005, 4: 45 -5: 30 p. m. This report is solely for the use of client personnel. No part of it may be circulated, quoted, or reproduced for distribution outside the client organization without prior written approval from Mc. Kinsey & Company. This material was used by Mc. Kinsey & Company during an oral presentation; it is not a complete record of the discussion.
CONFIDENTIAL Update on International Medical Affairs Alexander Petersen, Mc. Kinsey Stanley Bukofzer, Abbott Laboratories March 30, 2005, 4: 45 -5: 30 p. m. This report is solely for the use of client personnel. No part of it may be circulated, quoted, or reproduced for distribution outside the client organization without prior written approval from Mc. Kinsey & Company. This material was used by Mc. Kinsey & Company during an oral presentation; it is not a complete record of the discussion.
 NJE-192515. 016 -20050310 -sugn. HR 1 AGENDA 4: 45 A map of international medical affairs Alexander Petersen, Mc. Kinsey & Company 5: 00 Managing the complexity – One perspective Stanley Bukofzer, Abbott Laboratories 5: 15 Questions from the audience 1
NJE-192515. 016 -20050310 -sugn. HR 1 AGENDA 4: 45 A map of international medical affairs Alexander Petersen, Mc. Kinsey & Company 5: 00 Managing the complexity – One perspective Stanley Bukofzer, Abbott Laboratories 5: 15 Questions from the audience 1
 NJE-192515. 016 -20050310 -sugn. HR 1 INTRODUCTION • Medical affairs is a complex area to manage internationally • Local-level officials, doctors, and industry figures have their own views of compliance and proper medical affairs conduct, resulting in a tremendous diversity of regulations, guidance, and norms across different national contexts • At the same time, there are regional forces that are increasingly influential on local-level opinion and policy • Managing medical affairs without taking into account local and regional variation can create compliance and business risk • Five specific areas—communications with physicians, meetings and conventions, clinical trials, medical education, and direct-to -consumer information—provide a vivid demonstration of the diversity of international affairs 2
NJE-192515. 016 -20050310 -sugn. HR 1 INTRODUCTION • Medical affairs is a complex area to manage internationally • Local-level officials, doctors, and industry figures have their own views of compliance and proper medical affairs conduct, resulting in a tremendous diversity of regulations, guidance, and norms across different national contexts • At the same time, there are regional forces that are increasingly influential on local-level opinion and policy • Managing medical affairs without taking into account local and regional variation can create compliance and business risk • Five specific areas—communications with physicians, meetings and conventions, clinical trials, medical education, and direct-to -consumer information—provide a vivid demonstration of the diversity of international affairs 2
 NJE-192515. 016 -20050310 -sugn. HR 1 WHAT INDUSTRY IS SAYING – EXAMPLES When the government implemented the CTD in Belgium, they went too far. For several months, academic trials were halted. – Belgian Medical Director I was hired as the head of medical affairs; now, I spend most of my time being the head of risk management – Italian Medical Director With countries constantly changing their rules, it is getting harder and harder for headquarters to set meaningful global standards - Head of medical affairs, global pharmaceutical company The Medical Visits Charter would have been much worse if the industry had not proposed its own reforms – French Medical Director 3
NJE-192515. 016 -20050310 -sugn. HR 1 WHAT INDUSTRY IS SAYING – EXAMPLES When the government implemented the CTD in Belgium, they went too far. For several months, academic trials were halted. – Belgian Medical Director I was hired as the head of medical affairs; now, I spend most of my time being the head of risk management – Italian Medical Director With countries constantly changing their rules, it is getting harder and harder for headquarters to set meaningful global standards - Head of medical affairs, global pharmaceutical company The Medical Visits Charter would have been much worse if the industry had not proposed its own reforms – French Medical Director 3
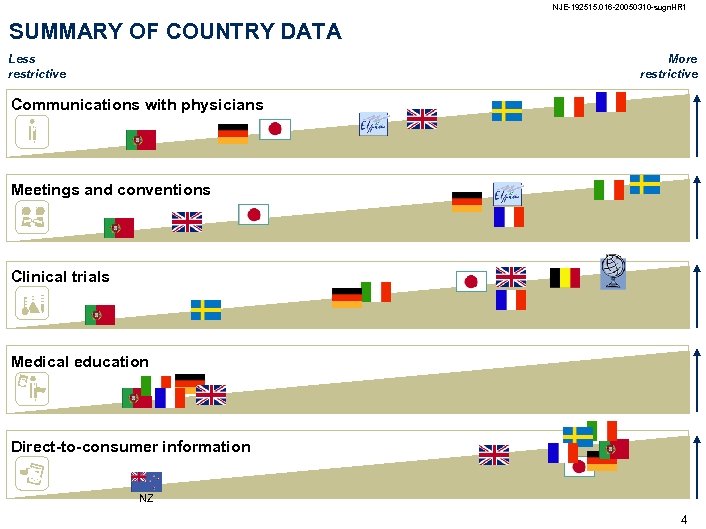 NJE-192515. 016 -20050310 -sugn. HR 1 SUMMARY OF COUNTRY DATA Less restrictive More restrictive Communications with physicians Meetings and conventions Clinical trials Medical education Direct-to-consumer information NZ 4
NJE-192515. 016 -20050310 -sugn. HR 1 SUMMARY OF COUNTRY DATA Less restrictive More restrictive Communications with physicians Meetings and conventions Clinical trials Medical education Direct-to-consumer information NZ 4
 NJE-192515. 016 -20050310 -sugn. HR 1 COMMUNICATIONS WITH PHYSICIANS • Frequency of office visits • Other channels of communication (phone, fax, email) • Limitations on leaving samples Italy Sweden Japan Germany France Portugal EFPIA Increasing restrictions UK 5
NJE-192515. 016 -20050310 -sugn. HR 1 COMMUNICATIONS WITH PHYSICIANS • Frequency of office visits • Other channels of communication (phone, fax, email) • Limitations on leaving samples Italy Sweden Japan Germany France Portugal EFPIA Increasing restrictions UK 5
 NJE-192515. 016 -20050310 -sugn. HR 1 COMMUNICATIONS WITH PHYSICIANS • Frequency of office visits • Other channels of communication (phone, fax, email) • Limitations on leaving samplesis even illegal to track doctor “It Italy Sweden UK Japan Germany France Portugal EFPIA Increasing restrictions “EFPIA expects us to ask for prior permission of a doctor to send him/her a fax, e-fax, email, automated calls or text messages” prescribing information” “The rules aren’t quite clear, but it would be risky to make more than 5 visits/year to a doctor” 6
NJE-192515. 016 -20050310 -sugn. HR 1 COMMUNICATIONS WITH PHYSICIANS • Frequency of office visits • Other channels of communication (phone, fax, email) • Limitations on leaving samplesis even illegal to track doctor “It Italy Sweden UK Japan Germany France Portugal EFPIA Increasing restrictions “EFPIA expects us to ask for prior permission of a doctor to send him/her a fax, e-fax, email, automated calls or text messages” prescribing information” “The rules aren’t quite clear, but it would be risky to make more than 5 visits/year to a doctor” 6
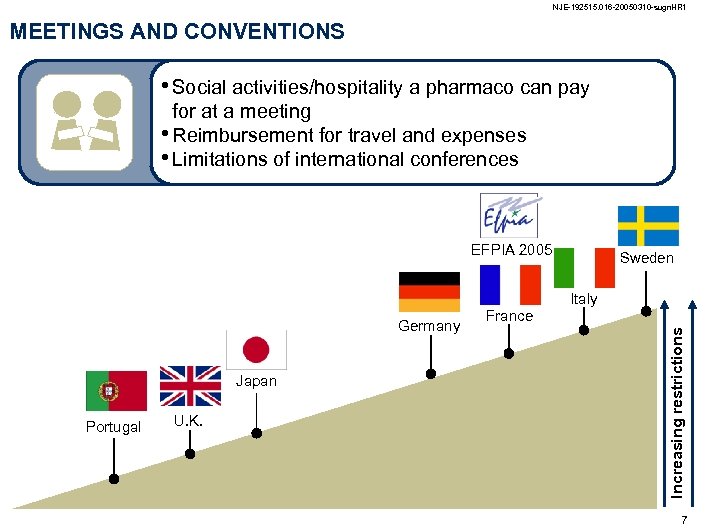 NJE-192515. 016 -20050310 -sugn. HR 1 MEETINGS AND CONVENTIONS • Social activities/hospitality a pharmaco can pay for at a meeting • Reimbursement for travel and expenses • Limitations of international conferences EFPIA 2005 Sweden Italy Japan Portugal U. K. France Increasing restrictions Germany 7
NJE-192515. 016 -20050310 -sugn. HR 1 MEETINGS AND CONVENTIONS • Social activities/hospitality a pharmaco can pay for at a meeting • Reimbursement for travel and expenses • Limitations of international conferences EFPIA 2005 Sweden Italy Japan Portugal U. K. France Increasing restrictions Germany 7
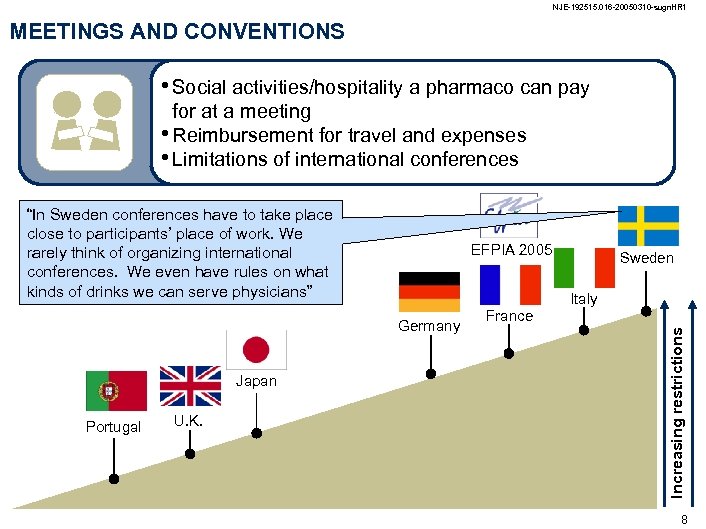 NJE-192515. 016 -20050310 -sugn. HR 1 MEETINGS AND CONVENTIONS • Social activities/hospitality a pharmaco can pay for at a meeting • Reimbursement for travel and expenses • Limitations of international conferences “In Sweden conferences have to take place close to participants’ place of work. We rarely think of organizing international conferences. We even have rules on what kinds of drinks we can serve physicians” EFPIA 2005 Italy Portugal U. K. France Increasing restrictions Germany Japan Sweden 8
NJE-192515. 016 -20050310 -sugn. HR 1 MEETINGS AND CONVENTIONS • Social activities/hospitality a pharmaco can pay for at a meeting • Reimbursement for travel and expenses • Limitations of international conferences “In Sweden conferences have to take place close to participants’ place of work. We rarely think of organizing international conferences. We even have rules on what kinds of drinks we can serve physicians” EFPIA 2005 Italy Portugal U. K. France Increasing restrictions Germany Japan Sweden 8
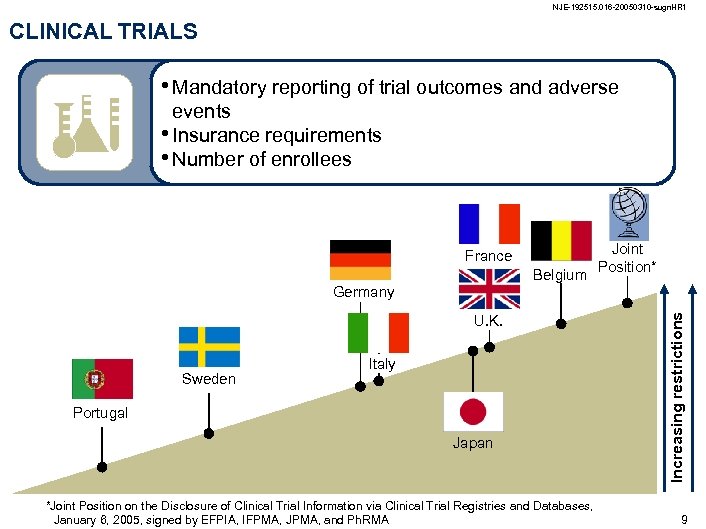 NJE-192515. 016 -20050310 -sugn. HR 1 CLINICAL TRIALS • Mandatory reporting of trial outcomes and adverse events • Insurance requirements • Number of enrollees France Joint Belgium Position* U. K. Sweden Italy Portugal Japan *Joint Position on the Disclosure of Clinical Trial Information via Clinical Trial Registries and Databases, January 6, 2005, signed by EFPIA, IFPMA, JPMA, and Ph. RMA Increasing restrictions Germany 9
NJE-192515. 016 -20050310 -sugn. HR 1 CLINICAL TRIALS • Mandatory reporting of trial outcomes and adverse events • Insurance requirements • Number of enrollees France Joint Belgium Position* U. K. Sweden Italy Portugal Japan *Joint Position on the Disclosure of Clinical Trial Information via Clinical Trial Registries and Databases, January 6, 2005, signed by EFPIA, IFPMA, JPMA, and Ph. RMA Increasing restrictions Germany 9
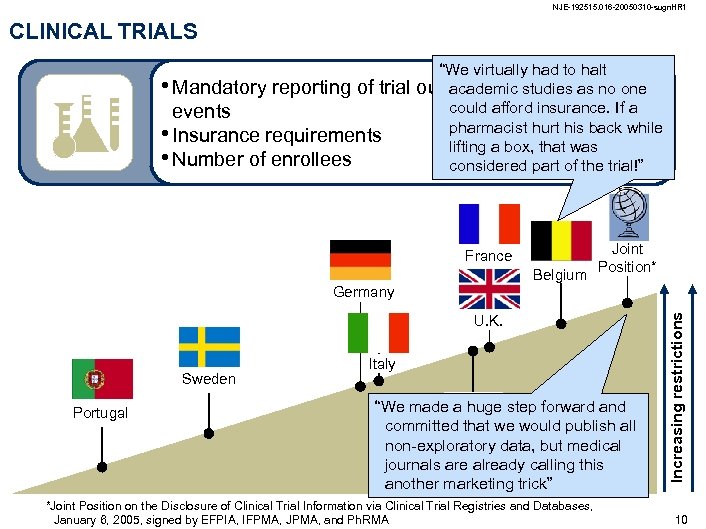 NJE-192515. 016 -20050310 -sugn. HR 1 CLINICAL TRIALS • Mandatory reporting of trial events • Insurance requirements • Number of enrollees “We virtually had to halt outcomes and adverse one academic studies as no could afford insurance. If a pharmacist hurt his back while lifting a box, that was considered part of the trial!” France Joint Belgium Position* U. K. Sweden Portugal Italy “We made a huge step forward and committed that we would publish all Japan non-exploratory data, but medical journals are already calling this another marketing trick” *Joint Position on the Disclosure of Clinical Trial Information via Clinical Trial Registries and Databases, January 6, 2005, signed by EFPIA, IFPMA, JPMA, and Ph. RMA Increasing restrictions Germany 10
NJE-192515. 016 -20050310 -sugn. HR 1 CLINICAL TRIALS • Mandatory reporting of trial events • Insurance requirements • Number of enrollees “We virtually had to halt outcomes and adverse one academic studies as no could afford insurance. If a pharmacist hurt his back while lifting a box, that was considered part of the trial!” France Joint Belgium Position* U. K. Sweden Portugal Italy “We made a huge step forward and committed that we would publish all Japan non-exploratory data, but medical journals are already calling this another marketing trick” *Joint Position on the Disclosure of Clinical Trial Information via Clinical Trial Registries and Databases, January 6, 2005, signed by EFPIA, IFPMA, JPMA, and Ph. RMA Increasing restrictions Germany 10
 NJE-192515. 016 -20050310 -sugn. HR 1 MEDICAL EDUCATION • Limits on content • Financial limits of sponsorship • Independence of pharmaco from those who receive Italy Germany Portugal U. K. France Increasing restrictions funds (speakers, CME) 11
NJE-192515. 016 -20050310 -sugn. HR 1 MEDICAL EDUCATION • Limits on content • Financial limits of sponsorship • Independence of pharmaco from those who receive Italy Germany Portugal U. K. France Increasing restrictions funds (speakers, CME) 11
 NJE-192515. 016 -20050310 -sugn. HR 1 MEDICAL EDUCATION • Limits on content • Financial limits of sponsorship • Independence of pharmaco from those who receive Italy Germany Portugal U. K. France “In France, continuing medical education is not yet on the authorities’ radar” Increasing restrictions funds (speakers, CME) 12
NJE-192515. 016 -20050310 -sugn. HR 1 MEDICAL EDUCATION • Limits on content • Financial limits of sponsorship • Independence of pharmaco from those who receive Italy Germany Portugal U. K. France “In France, continuing medical education is not yet on the authorities’ radar” Increasing restrictions funds (speakers, CME) 12
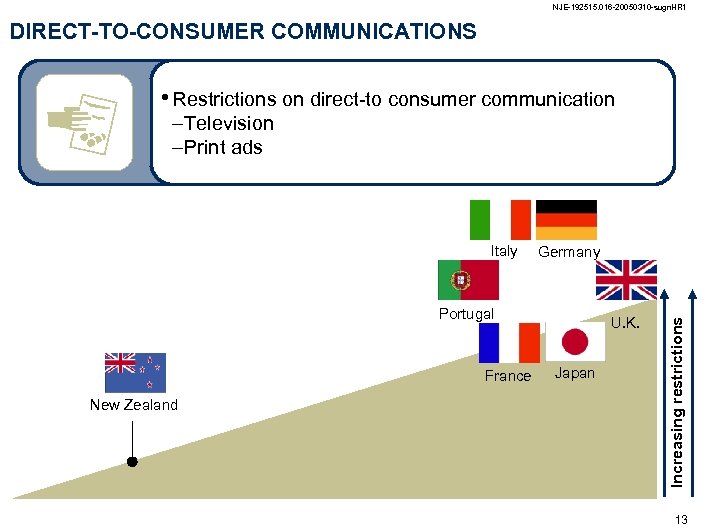 NJE-192515. 016 -20050310 -sugn. HR 1 DIRECT-TO-CONSUMER COMMUNICATIONS • Restrictions on direct-to consumer communication –Television –Print ads Germany Portugal France New Zealand U. K. Japan Increasing restrictions Italy 13
NJE-192515. 016 -20050310 -sugn. HR 1 DIRECT-TO-CONSUMER COMMUNICATIONS • Restrictions on direct-to consumer communication –Television –Print ads Germany Portugal France New Zealand U. K. Japan Increasing restrictions Italy 13
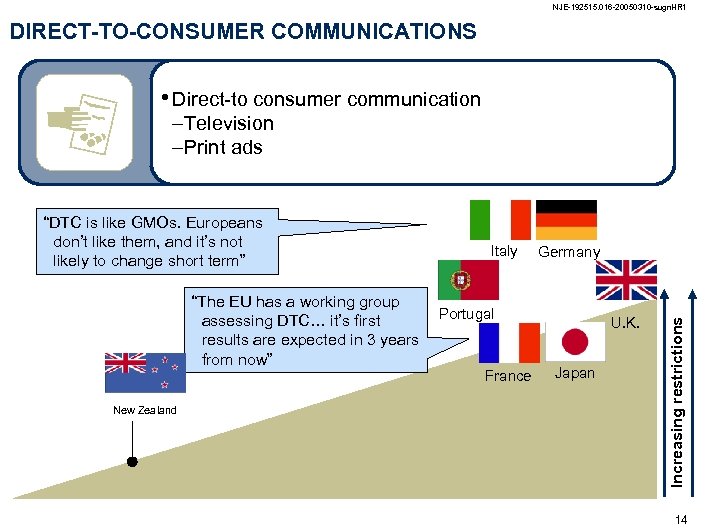 NJE-192515. 016 -20050310 -sugn. HR 1 DIRECT-TO-CONSUMER COMMUNICATIONS • Direct-to consumer communication –Television –Print ads “The EU has a working group assessing DTC… it’s first results are expected in 3 years from now” New Zealand Italy Germany Portugal France U. K. Japan Increasing restrictions “DTC is like GMOs. Europeans don’t like them, and it’s not likely to change short term” 14
NJE-192515. 016 -20050310 -sugn. HR 1 DIRECT-TO-CONSUMER COMMUNICATIONS • Direct-to consumer communication –Television –Print ads “The EU has a working group assessing DTC… it’s first results are expected in 3 years from now” New Zealand Italy Germany Portugal France U. K. Japan Increasing restrictions “DTC is like GMOs. Europeans don’t like them, and it’s not likely to change short term” 14
 NJE-192515. 016 -20050310 -sugn. HR 1 MANAGEMENT CHALLENGES DRIVEN BY LOCAL DIVERSITY • Do I need to rethink my model of communicating medical information to customers? • How do I stay abreast of the new developments, especially in making sure country operations are always compliant? • Should I attempt to have a single set of comprehensive corporate-wide guidelines around medical affairs? • Without a corporate-wide standard, how do I explain my compliance posture to my customers? • How do I coordinate trial strategy in a world of more transparency? 15
NJE-192515. 016 -20050310 -sugn. HR 1 MANAGEMENT CHALLENGES DRIVEN BY LOCAL DIVERSITY • Do I need to rethink my model of communicating medical information to customers? • How do I stay abreast of the new developments, especially in making sure country operations are always compliant? • Should I attempt to have a single set of comprehensive corporate-wide guidelines around medical affairs? • Without a corporate-wide standard, how do I explain my compliance posture to my customers? • How do I coordinate trial strategy in a world of more transparency? 15
 CONFIDENTIAL Managing the Complexity – One Perspective Stan Bukofzer Divisional Vice President and Head of Global Medical Affairs
CONFIDENTIAL Managing the Complexity – One Perspective Stan Bukofzer Divisional Vice President and Head of Global Medical Affairs
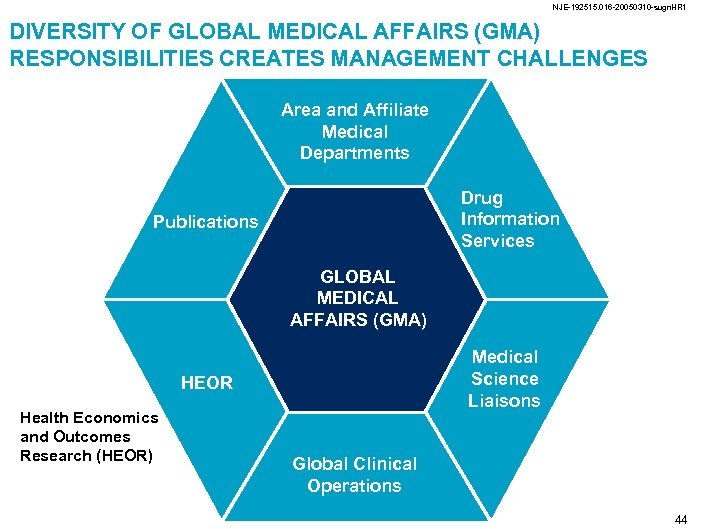
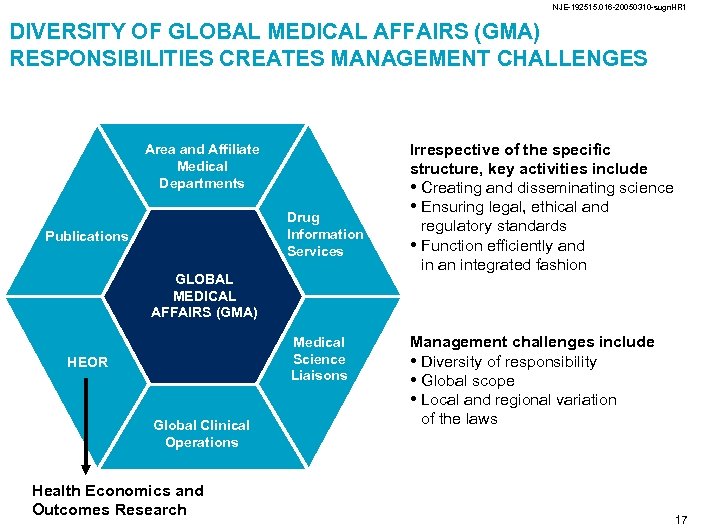 NJE-192515. 016 -20050310 -sugn. HR 1 DIVERSITY OF GLOBAL MEDICAL AFFAIRS (GMA) RESPONSIBILITIES CREATES MANAGEMENT CHALLENGES Area and Affiliate Medical Departments Drug Information Services Publications GLOBAL MEDICAL AFFAIRS (GMA) Medical Science Liaisons HEOR Global Clinical Operations Health Economics and Outcomes Research Irrespective of the specific structure, key activities include • Creating and disseminating science • Ensuring legal, ethical and regulatory standards • Function efficiently and in an integrated fashion Management challenges include • Diversity of responsibility • Global scope • Local and regional variation of the laws 17
NJE-192515. 016 -20050310 -sugn. HR 1 DIVERSITY OF GLOBAL MEDICAL AFFAIRS (GMA) RESPONSIBILITIES CREATES MANAGEMENT CHALLENGES Area and Affiliate Medical Departments Drug Information Services Publications GLOBAL MEDICAL AFFAIRS (GMA) Medical Science Liaisons HEOR Global Clinical Operations Health Economics and Outcomes Research Irrespective of the specific structure, key activities include • Creating and disseminating science • Ensuring legal, ethical and regulatory standards • Function efficiently and in an integrated fashion Management challenges include • Diversity of responsibility • Global scope • Local and regional variation of the laws 17
 NJE-192515. 016 -20050310 -sugn. HR 1 ABBOTT’S GUIDING PRINCIPLES TO MANAGE GMA Structural separation of GMA from commercial • GMA reporting to R&D Centralized decision making within GMA • Simple and clear processes Cross functional communication teams that include regional and local structures • Specific Communication Channels Application of global clinical SOPs • Clear Understanding of Legal and Regulatory Requirements as well as ethical and best practices Standardized clinical training world-wide 18
NJE-192515. 016 -20050310 -sugn. HR 1 ABBOTT’S GUIDING PRINCIPLES TO MANAGE GMA Structural separation of GMA from commercial • GMA reporting to R&D Centralized decision making within GMA • Simple and clear processes Cross functional communication teams that include regional and local structures • Specific Communication Channels Application of global clinical SOPs • Clear Understanding of Legal and Regulatory Requirements as well as ethical and best practices Standardized clinical training world-wide 18
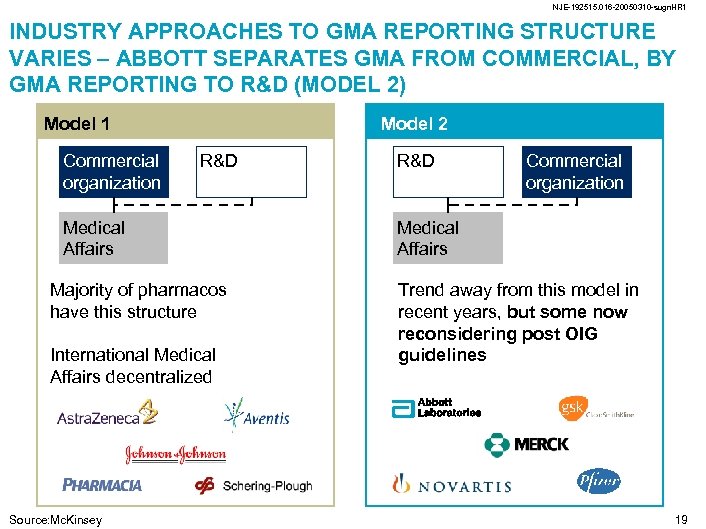 NJE-192515. 016 -20050310 -sugn. HR 1 INDUSTRY APPROACHES TO GMA REPORTING STRUCTURE VARIES – ABBOTT SEPARATES GMA FROM COMMERCIAL, BY GMA REPORTING TO R&D (MODEL 2) Model 1 Commercial organization Model 2 R&D Medical Affairs Majority of pharmacos have this structure International Medical Affairs decentralized Source: Mc. Kinsey R&D Commercial organization Medical Affairs Trend away from this model in recent years, but some now reconsidering post OIG guidelines 19
NJE-192515. 016 -20050310 -sugn. HR 1 INDUSTRY APPROACHES TO GMA REPORTING STRUCTURE VARIES – ABBOTT SEPARATES GMA FROM COMMERCIAL, BY GMA REPORTING TO R&D (MODEL 2) Model 1 Commercial organization Model 2 R&D Medical Affairs Majority of pharmacos have this structure International Medical Affairs decentralized Source: Mc. Kinsey R&D Commercial organization Medical Affairs Trend away from this model in recent years, but some now reconsidering post OIG guidelines 19
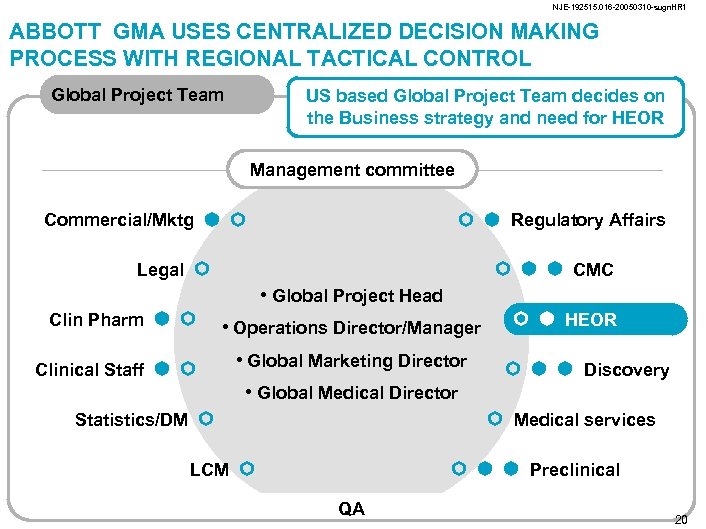 NJE-192515. 016 -20050310 -sugn. HR 1 ABBOTT GMA USES CENTRALIZED DECISION MAKING PROCESS WITH REGIONAL TACTICAL CONTROL Global Project Team US based Global Project Team decides on the Business strategy and need for HEOR Management committee Commercial/Mktg Regulatory Affairs Legal CMC • Global Project Head Clin Pharm Clinical Staff • Operations Director/Manager • Global Marketing Director • Global Medical Director Statistics/DM HEOR Discovery Medical services LCM Preclinical QA 20
NJE-192515. 016 -20050310 -sugn. HR 1 ABBOTT GMA USES CENTRALIZED DECISION MAKING PROCESS WITH REGIONAL TACTICAL CONTROL Global Project Team US based Global Project Team decides on the Business strategy and need for HEOR Management committee Commercial/Mktg Regulatory Affairs Legal CMC • Global Project Head Clin Pharm Clinical Staff • Operations Director/Manager • Global Marketing Director • Global Medical Director Statistics/DM HEOR Discovery Medical services LCM Preclinical QA 20
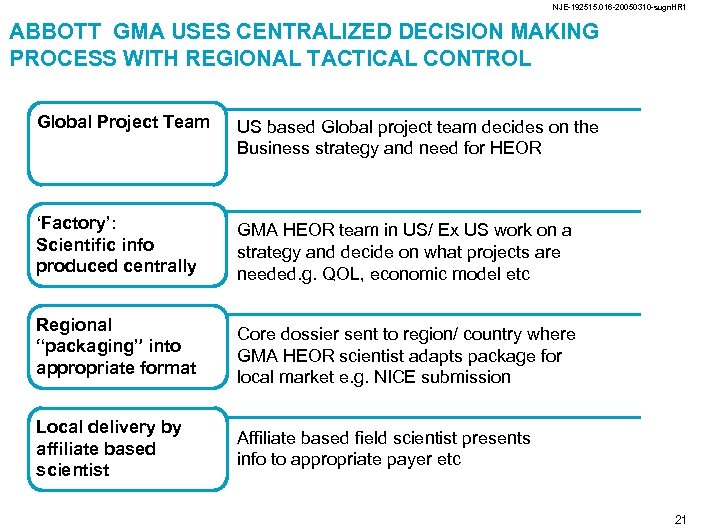 NJE-192515. 016 -20050310 -sugn. HR 1 ABBOTT GMA USES CENTRALIZED DECISION MAKING PROCESS WITH REGIONAL TACTICAL CONTROL Global Project Team US based Global project team decides on the Business strategy and need for HEOR ‘Factory’: Scientific info produced centrally GMA HEOR team in US/ Ex US work on a strategy and decide on what projects are needed. g. QOL, economic model etc Regional “packaging” into appropriate format Local delivery by affiliate based scientist Core dossier sent to region/ country where GMA HEOR scientist adapts package for local market e. g. NICE submission Affiliate based field scientist presents info to appropriate payer etc 21
NJE-192515. 016 -20050310 -sugn. HR 1 ABBOTT GMA USES CENTRALIZED DECISION MAKING PROCESS WITH REGIONAL TACTICAL CONTROL Global Project Team US based Global project team decides on the Business strategy and need for HEOR ‘Factory’: Scientific info produced centrally GMA HEOR team in US/ Ex US work on a strategy and decide on what projects are needed. g. QOL, economic model etc Regional “packaging” into appropriate format Local delivery by affiliate based scientist Core dossier sent to region/ country where GMA HEOR scientist adapts package for local market e. g. NICE submission Affiliate based field scientist presents info to appropriate payer etc 21
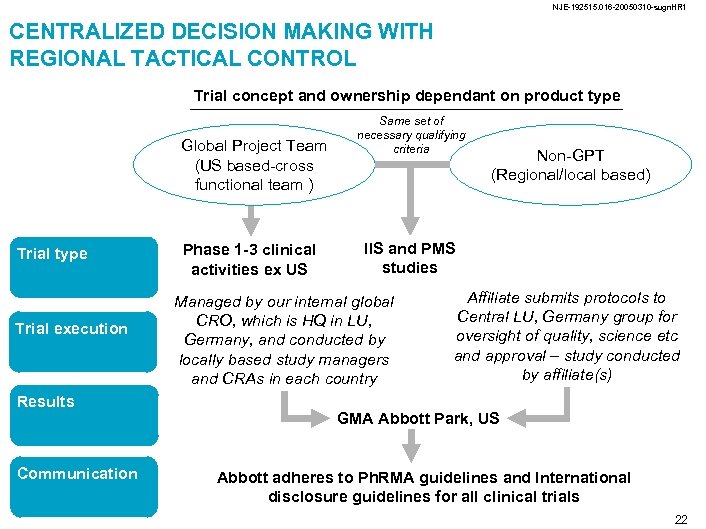 NJE-192515. 016 -20050310 -sugn. HR 1 CENTRALIZED DECISION MAKING WITH REGIONAL TACTICAL CONTROL Trial concept and ownership dependant on product type Global Project Team (US based-cross functional team ) Trial type Trial execution Results Communication Phase 1 -3 clinical activities ex US Same set of necessary qualifying criteria Non-GPT (Regional/local based) IIS and PMS studies Managed by our internal global CRO, which is HQ in LU, Germany, and conducted by locally based study managers and CRAs in each country Affiliate submits protocols to Central LU, Germany group for oversight of quality, science etc and approval – study conducted by affiliate(s) GMA Abbott Park, US Abbott adheres to Ph. RMA guidelines and International disclosure guidelines for all clinical trials 22
NJE-192515. 016 -20050310 -sugn. HR 1 CENTRALIZED DECISION MAKING WITH REGIONAL TACTICAL CONTROL Trial concept and ownership dependant on product type Global Project Team (US based-cross functional team ) Trial type Trial execution Results Communication Phase 1 -3 clinical activities ex US Same set of necessary qualifying criteria Non-GPT (Regional/local based) IIS and PMS studies Managed by our internal global CRO, which is HQ in LU, Germany, and conducted by locally based study managers and CRAs in each country Affiliate submits protocols to Central LU, Germany group for oversight of quality, science etc and approval – study conducted by affiliate(s) GMA Abbott Park, US Abbott adheres to Ph. RMA guidelines and International disclosure guidelines for all clinical trials 22
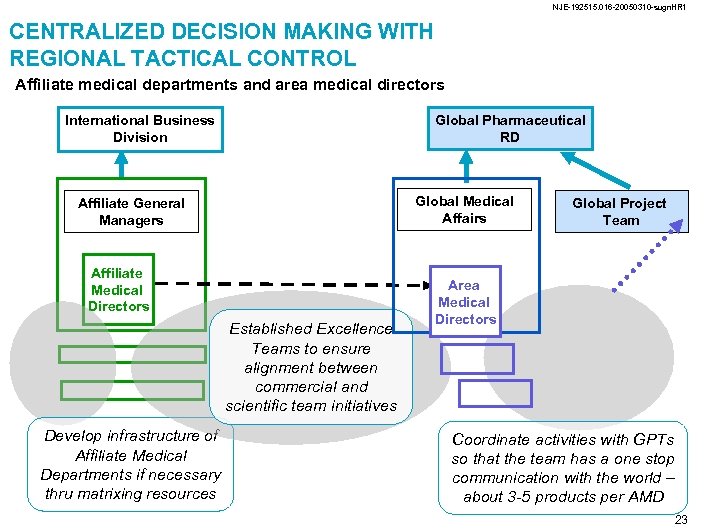 NJE-192515. 016 -20050310 -sugn. HR 1 CENTRALIZED DECISION MAKING WITH REGIONAL TACTICAL CONTROL Affiliate medical departments and area medical directors International Business Division Global Pharmaceutical RD Global Medical Affairs Affiliate General Managers Affiliate Medical Directors Established Excellence Teams to ensure alignment between commercial and scientific team initiatives Develop infrastructure of Affiliate Medical Departments if necessary thru matrixing resources Global Project Team Area Medical Directors Coordinate activities with GPTs so that the team has a one stop communication with the world – about 3 -5 products per AMD 23
NJE-192515. 016 -20050310 -sugn. HR 1 CENTRALIZED DECISION MAKING WITH REGIONAL TACTICAL CONTROL Affiliate medical departments and area medical directors International Business Division Global Pharmaceutical RD Global Medical Affairs Affiliate General Managers Affiliate Medical Directors Established Excellence Teams to ensure alignment between commercial and scientific team initiatives Develop infrastructure of Affiliate Medical Departments if necessary thru matrixing resources Global Project Team Area Medical Directors Coordinate activities with GPTs so that the team has a one stop communication with the world – about 3 -5 products per AMD 23
 NJE-192515. 016 -20050310 -sugn. HR 1 GLOBAL MEDICAL AFFAIRS HAS DEVELOPED A STRONG SET OF GLOBAL SOPs BASED ON A CLEAR UNDERSTANDING OF LEGAL REGULATORY REQUIREMENTS Try to keep abreast of rapidly changing environment • Trends in legislation at state and federal/country level • Address new requirements in their formative stages • Participate in industry organizations • Bench mark and apply Best Practices Standard global SOPs generally use US/ EU laws and regulations as a base – where affiliate more stringent/ different local laws exist, an additional local SOP will be issued Where applicable ICH practice guidance are reflected – many not yet actually adopted, but best practice dictates use 24
NJE-192515. 016 -20050310 -sugn. HR 1 GLOBAL MEDICAL AFFAIRS HAS DEVELOPED A STRONG SET OF GLOBAL SOPs BASED ON A CLEAR UNDERSTANDING OF LEGAL REGULATORY REQUIREMENTS Try to keep abreast of rapidly changing environment • Trends in legislation at state and federal/country level • Address new requirements in their formative stages • Participate in industry organizations • Bench mark and apply Best Practices Standard global SOPs generally use US/ EU laws and regulations as a base – where affiliate more stringent/ different local laws exist, an additional local SOP will be issued Where applicable ICH practice guidance are reflected – many not yet actually adopted, but best practice dictates use 24
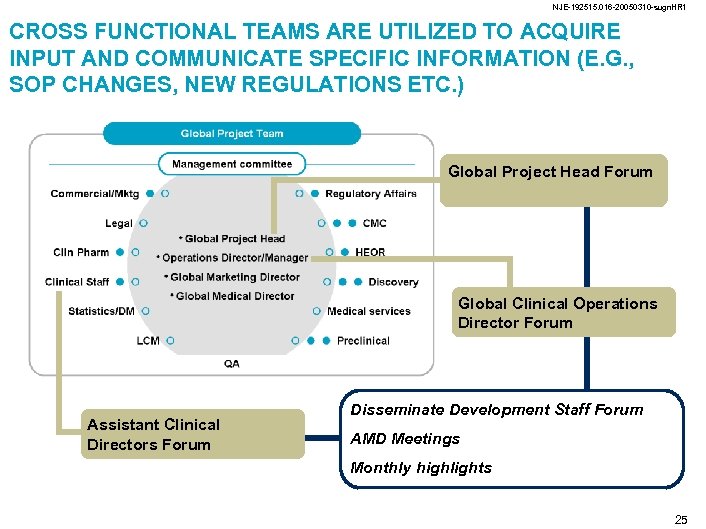 NJE-192515. 016 -20050310 -sugn. HR 1 CROSS FUNCTIONAL TEAMS ARE UTILIZED TO ACQUIRE INPUT AND COMMUNICATE SPECIFIC INFORMATION (E. G. , SOP CHANGES, NEW REGULATIONS ETC. ) Global Project Head Forum Global Clinical Operations Director Forum Assistant Clinical Directors Forum Disseminate Development Staff Forum AMD Meetings Monthly highlights 25
NJE-192515. 016 -20050310 -sugn. HR 1 CROSS FUNCTIONAL TEAMS ARE UTILIZED TO ACQUIRE INPUT AND COMMUNICATE SPECIFIC INFORMATION (E. G. , SOP CHANGES, NEW REGULATIONS ETC. ) Global Project Head Forum Global Clinical Operations Director Forum Assistant Clinical Directors Forum Disseminate Development Staff Forum AMD Meetings Monthly highlights 25
 NJE-192515. 016 -20050310 -sugn. HR 1 STANDARDIZED TOOLS FOR CLINICAL TRAINING WORLDWIDE (e. g. TRAINING MATERIALS AVAILABLE THROUGH WEB BASED PORTAL) Web Site • Central Repository for Training allowing Global Access to CBTs and Training links • Single site ensures Consistency/Training • Requires minimal internal IT support • Is compatible with Abbott’s other training systems • Mandatory legal and ethical training of relevant roles 26
NJE-192515. 016 -20050310 -sugn. HR 1 STANDARDIZED TOOLS FOR CLINICAL TRAINING WORLDWIDE (e. g. TRAINING MATERIALS AVAILABLE THROUGH WEB BASED PORTAL) Web Site • Central Repository for Training allowing Global Access to CBTs and Training links • Single site ensures Consistency/Training • Requires minimal internal IT support • Is compatible with Abbott’s other training systems • Mandatory legal and ethical training of relevant roles 26
 Appendix
Appendix
 NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: USA VS. EUROPE Key facts Communications with Physicians Meetings and Conventions Clinical Trials Medical Education Direct to Consumer Information Vs. Europe • • • Samples and visits are generally left up to the company’s discretion MSL’s pretty prevalent for KOL communications • • FMV for speakers No special limitations on international meetings • Some countries discourage international meetings (though none ban it) • IIS (non-sponsored) trials AE reports are discretionary • IIS AE reporting is mandatory • • • Pharmaco cannot provide the content Cannot hire presenters w/in the company Must mention all therapies for a condition • Very little guidance at this point • DTC advertising allowed, but with content control from DDMAC • DTC advertising being considered in EU review that will produce report three years from now, but observers think cost control, doctors’ resistance will never let it happen • Varies by country, but generally more restrictive than USA MSL is still a growing role in Europe. Growth won’t be even, because some countries are still less restrictive (e. g. Portugal, Germany) so reps are still an effective information channel 28
NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: USA VS. EUROPE Key facts Communications with Physicians Meetings and Conventions Clinical Trials Medical Education Direct to Consumer Information Vs. Europe • • • Samples and visits are generally left up to the company’s discretion MSL’s pretty prevalent for KOL communications • • FMV for speakers No special limitations on international meetings • Some countries discourage international meetings (though none ban it) • IIS (non-sponsored) trials AE reports are discretionary • IIS AE reporting is mandatory • • • Pharmaco cannot provide the content Cannot hire presenters w/in the company Must mention all therapies for a condition • Very little guidance at this point • DTC advertising allowed, but with content control from DDMAC • DTC advertising being considered in EU review that will produce report three years from now, but observers think cost control, doctors’ resistance will never let it happen • Varies by country, but generally more restrictive than USA MSL is still a growing role in Europe. Growth won’t be even, because some countries are still less restrictive (e. g. Portugal, Germany) so reps are still an effective information channel 28
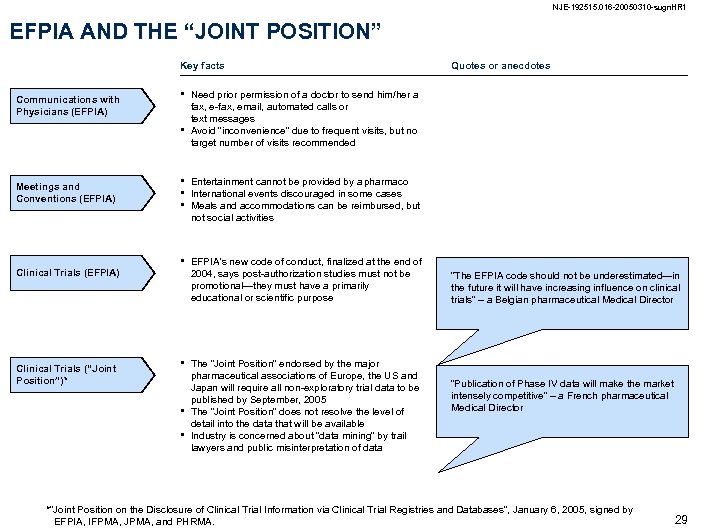 NJE-192515. 016 -20050310 -sugn. HR 1 EFPIA AND THE “JOINT POSITION” Key facts Communications with Physicians (EFPIA) • • Meetings and Conventions (EFPIA) Clinical Trials (“Joint Position”)* Quotes or anecdotes Need prior permission of a doctor to send him/her a fax, e-fax, email, automated calls or text messages Avoid “inconvenience” due to frequent visits, but no target number of visits recommended • • • Entertainment cannot be provided by a pharmaco International events discouraged in some cases Meals and accommodations can be reimbursed, but not social activities • EFPIA’s new code of conduct, finalized at the end of 2004, says post-authorization studies must not be promotional—they must have a primarily educational or scientific purpose • • • The “Joint Position” endorsed by the major pharmaceutical associations of Europe, the US and Japan will require all non-exploratory trial data to be published by September, 2005 The “Joint Position” does not resolve the level of detail into the data that will be available Industry is concerned about “data mining” by trail lawyers and public misinterpretation of data “The EFPIA code should not be underestimated—in the future it will have increasing influence on clinical trials” – a Belgian pharmaceutical Medical Director “Publication of Phase IV data will make the market intensely competitive” – a French pharmaceutical Medical Director *“Joint Position on the Disclosure of Clinical Trial Information via Clinical Trial Registries and Databases”, January 6, 2005, signed by EFPIA, IFPMA, JPMA, and PHRMA. 29
NJE-192515. 016 -20050310 -sugn. HR 1 EFPIA AND THE “JOINT POSITION” Key facts Communications with Physicians (EFPIA) • • Meetings and Conventions (EFPIA) Clinical Trials (“Joint Position”)* Quotes or anecdotes Need prior permission of a doctor to send him/her a fax, e-fax, email, automated calls or text messages Avoid “inconvenience” due to frequent visits, but no target number of visits recommended • • • Entertainment cannot be provided by a pharmaco International events discouraged in some cases Meals and accommodations can be reimbursed, but not social activities • EFPIA’s new code of conduct, finalized at the end of 2004, says post-authorization studies must not be promotional—they must have a primarily educational or scientific purpose • • • The “Joint Position” endorsed by the major pharmaceutical associations of Europe, the US and Japan will require all non-exploratory trial data to be published by September, 2005 The “Joint Position” does not resolve the level of detail into the data that will be available Industry is concerned about “data mining” by trail lawyers and public misinterpretation of data “The EFPIA code should not be underestimated—in the future it will have increasing influence on clinical trials” – a Belgian pharmaceutical Medical Director “Publication of Phase IV data will make the market intensely competitive” – a French pharmaceutical Medical Director *“Joint Position on the Disclosure of Clinical Trial Information via Clinical Trial Registries and Databases”, January 6, 2005, signed by EFPIA, IFPMA, JPMA, and PHRMA. 29
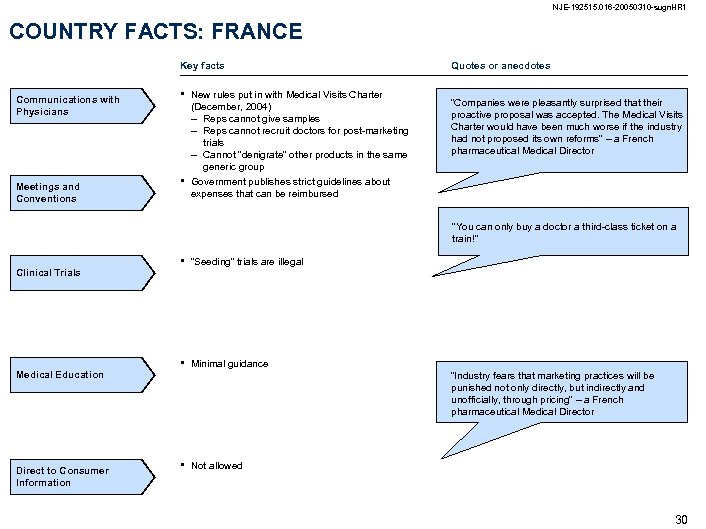 NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: FRANCE Key facts Communications with Physicians Meetings and Conventions • • New rules put in with Medical Visits Charter (December, 2004) – Reps cannot give samples – Reps cannot recruit doctors for post-marketing trials – Cannot “denigrate” other products in the same generic group Government publishes strict guidelines about expenses that can be reimbursed Quotes or anecdotes “Companies were pleasantly surprised that their proactive proposal was accepted. The Medical Visits Charter would have been much worse if the industry had not proposed its own reforms” – a French pharmaceutical Medical Director “You can only buy a doctor a third-class ticket on a train!” Clinical Trials Medical Education Direct to Consumer Information • “Seeding” trials are illegal • Minimal guidance “Industry fears that marketing practices will be punished not only directly, but indirectly and unofficially, through pricing” – a French pharmaceutical Medical Director • Not allowed 30
NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: FRANCE Key facts Communications with Physicians Meetings and Conventions • • New rules put in with Medical Visits Charter (December, 2004) – Reps cannot give samples – Reps cannot recruit doctors for post-marketing trials – Cannot “denigrate” other products in the same generic group Government publishes strict guidelines about expenses that can be reimbursed Quotes or anecdotes “Companies were pleasantly surprised that their proactive proposal was accepted. The Medical Visits Charter would have been much worse if the industry had not proposed its own reforms” – a French pharmaceutical Medical Director “You can only buy a doctor a third-class ticket on a train!” Clinical Trials Medical Education Direct to Consumer Information • “Seeding” trials are illegal • Minimal guidance “Industry fears that marketing practices will be punished not only directly, but indirectly and unofficially, through pricing” – a French pharmaceutical Medical Director • Not allowed 30
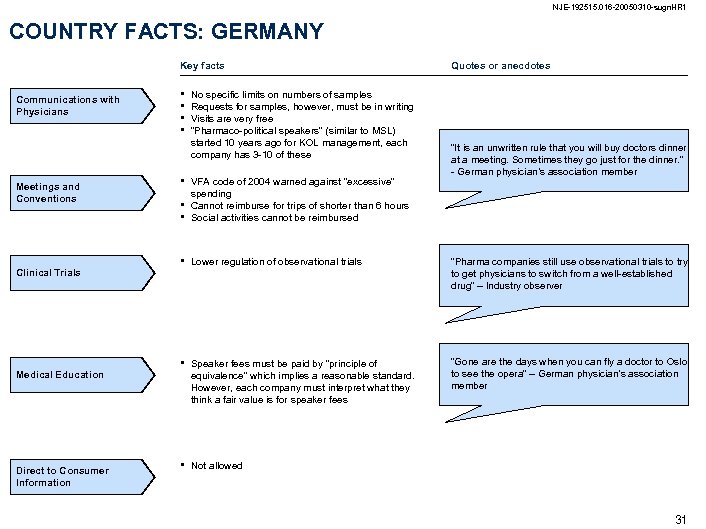 NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: GERMANY Key facts Communications with Physicians Meetings and Conventions Clinical Trials Medical Education Direct to Consumer Information • • • No specific limits on numbers of samples Requests for samples, however, must be in writing Visits are very free “Pharmaco-political speakers” (similar to MSL) started 10 years ago for KOL management, each company has 3 -10 of these Quotes or anecdotes “It is an unwritten rule that you will buy doctors dinner at a meeting. Sometimes they go just for the dinner. ” - German physician’s association member • • VFA code of 2004 warned against “excessive” spending Cannot reimburse for trips of shorter than 6 hours Social activities cannot be reimbursed • Lower regulation of observational trials “Pharma companies still use observational trials to try to get physicians to switch from a well-established drug” – Industry observer • Speaker fees must be paid by “principle of equivalence” which implies a reasonable standard. However, each company must interpret what they think a fair value is for speaker fees “Gone are the days when you can fly a doctor to Oslo to see the opera” – German physician’s association member • Not allowed 31
NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: GERMANY Key facts Communications with Physicians Meetings and Conventions Clinical Trials Medical Education Direct to Consumer Information • • • No specific limits on numbers of samples Requests for samples, however, must be in writing Visits are very free “Pharmaco-political speakers” (similar to MSL) started 10 years ago for KOL management, each company has 3 -10 of these Quotes or anecdotes “It is an unwritten rule that you will buy doctors dinner at a meeting. Sometimes they go just for the dinner. ” - German physician’s association member • • VFA code of 2004 warned against “excessive” spending Cannot reimburse for trips of shorter than 6 hours Social activities cannot be reimbursed • Lower regulation of observational trials “Pharma companies still use observational trials to try to get physicians to switch from a well-established drug” – Industry observer • Speaker fees must be paid by “principle of equivalence” which implies a reasonable standard. However, each company must interpret what they think a fair value is for speaker fees “Gone are the days when you can fly a doctor to Oslo to see the opera” – German physician’s association member • Not allowed 31
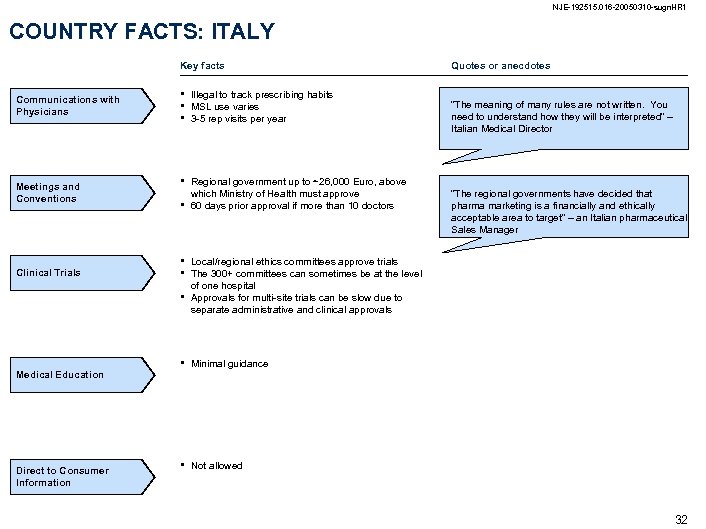 NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: ITALY Key facts Communications with Physicians Meetings and Conventions Clinical Trials • • • Illegal to track prescribing habits MSL use varies 3 -5 rep visits per year • Regional government up to ~26, 000 Euro, above which Ministry of Health must approve 60 days prior approval if more than 10 doctors • • Medical Education Direct to Consumer Information Quotes or anecdotes “The meaning of many rules are not written. You need to understand how they will be interpreted” – Italian Medical Director “The regional governments have decided that pharma marketing is a financially and ethically acceptable area to target” – an Italian pharmaceutical Sales Manager Local/regional ethics committees approve trials The 300+ committees can sometimes be at the level of one hospital Approvals for multi-site trials can be slow due to separate administrative and clinical approvals • Minimal guidance • Not allowed 32
NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: ITALY Key facts Communications with Physicians Meetings and Conventions Clinical Trials • • • Illegal to track prescribing habits MSL use varies 3 -5 rep visits per year • Regional government up to ~26, 000 Euro, above which Ministry of Health must approve 60 days prior approval if more than 10 doctors • • Medical Education Direct to Consumer Information Quotes or anecdotes “The meaning of many rules are not written. You need to understand how they will be interpreted” – Italian Medical Director “The regional governments have decided that pharma marketing is a financially and ethically acceptable area to target” – an Italian pharmaceutical Sales Manager Local/regional ethics committees approve trials The 300+ committees can sometimes be at the level of one hospital Approvals for multi-site trials can be slow due to separate administrative and clinical approvals • Minimal guidance • Not allowed 32
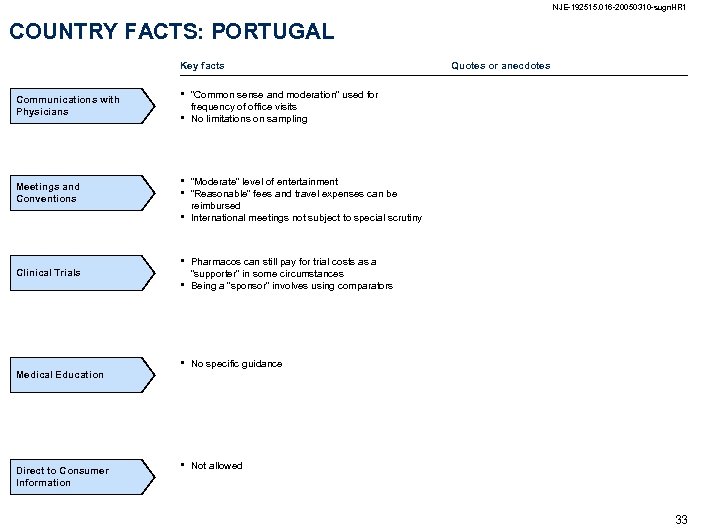 NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: PORTUGAL Key facts Communications with Physicians Meetings and Conventions • • • Clinical Trials Medical Education Direct to Consumer Information • Quotes or anecdotes “Common sense and moderation” used for frequency of office visits No limitations on sampling “Moderate” level of entertainment “Reasonable” fees and travel expenses can be reimbursed International meetings not subject to special scrutiny • Pharmacos can still pay for trial costs as a “supporter” in some circumstances Being a “sponsor” involves using comparators • No specific guidance • Not allowed 33
NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: PORTUGAL Key facts Communications with Physicians Meetings and Conventions • • • Clinical Trials Medical Education Direct to Consumer Information • Quotes or anecdotes “Common sense and moderation” used for frequency of office visits No limitations on sampling “Moderate” level of entertainment “Reasonable” fees and travel expenses can be reimbursed International meetings not subject to special scrutiny • Pharmacos can still pay for trial costs as a “supporter” in some circumstances Being a “sponsor” involves using comparators • No specific guidance • Not allowed 33
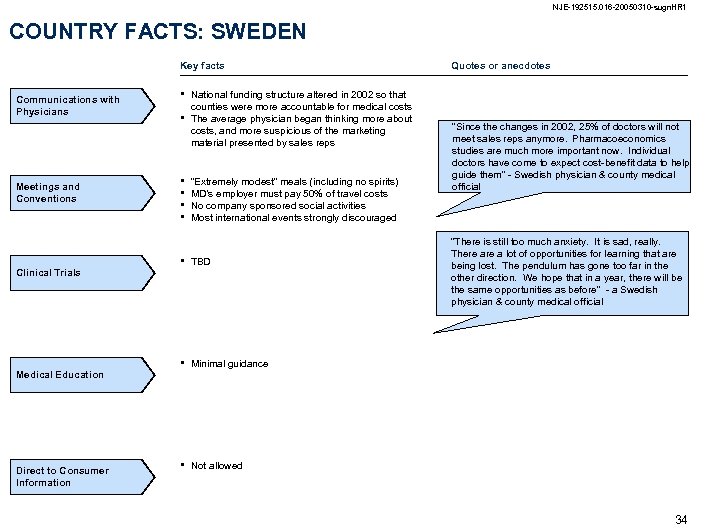 NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: SWEDEN Key facts Communications with Physicians Meetings and Conventions Clinical Trials Medical Education Direct to Consumer Information • • National funding structure altered in 2002 so that counties were more accountable for medical costs The average physician began thinking more about costs, and more suspicious of the marketing material presented by sales reps • • “Extremely modest” meals (including no spirits) MD’s employer must pay 50% of travel costs No company sponsored social activities Most international events strongly discouraged • TBD • “Since the changes in 2002, 25% of doctors will not meet sales reps anymore. Pharmacoeconomics studies are much more important now. Individual doctors have come to expect cost-benefit data to help guide them” - Swedish physician & county medical official “There is still too much anxiety. It is sad, really. There a lot of opportunities for learning that are being lost. The pendulum has gone too far in the other direction. We hope that in a year, there will be the same opportunities as before” - a Swedish physician & county medical official Minimal guidance • Quotes or anecdotes Not allowed 34
NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: SWEDEN Key facts Communications with Physicians Meetings and Conventions Clinical Trials Medical Education Direct to Consumer Information • • National funding structure altered in 2002 so that counties were more accountable for medical costs The average physician began thinking more about costs, and more suspicious of the marketing material presented by sales reps • • “Extremely modest” meals (including no spirits) MD’s employer must pay 50% of travel costs No company sponsored social activities Most international events strongly discouraged • TBD • “Since the changes in 2002, 25% of doctors will not meet sales reps anymore. Pharmacoeconomics studies are much more important now. Individual doctors have come to expect cost-benefit data to help guide them” - Swedish physician & county medical official “There is still too much anxiety. It is sad, really. There a lot of opportunities for learning that are being lost. The pendulum has gone too far in the other direction. We hope that in a year, there will be the same opportunities as before” - a Swedish physician & county medical official Minimal guidance • Quotes or anecdotes Not allowed 34
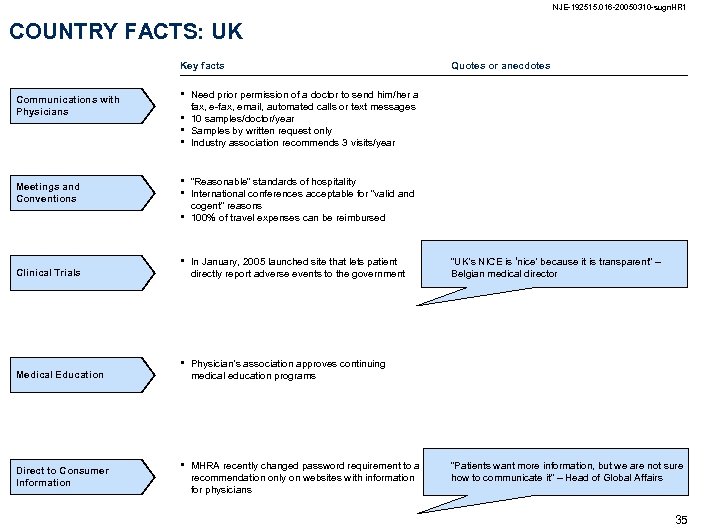 NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: UK Key facts Communications with Physicians Meetings and Conventions • • Clinical Trials Medical Education Direct to Consumer Information Quotes or anecdotes Need prior permission of a doctor to send him/her a fax, e-fax, email, automated calls or text messages 10 samples/doctor/year Samples by written request only Industry association recommends 3 visits/year “Reasonable” standards of hospitality International conferences acceptable for “valid and cogent” reasons 100% of travel expenses can be reimbursed • In January, 2005 launched site that lets patient directly report adverse events to the government • Physician’s association approves continuing medical education programs • MHRA recently changed password requirement to a recommendation only on websites with information for physicians “UK’s NICE is ‘nice’ because it is transparent” – Belgian medical director “Patients want more information, but we are not sure how to communicate it” – Head of Global Affairs 35
NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: UK Key facts Communications with Physicians Meetings and Conventions • • Clinical Trials Medical Education Direct to Consumer Information Quotes or anecdotes Need prior permission of a doctor to send him/her a fax, e-fax, email, automated calls or text messages 10 samples/doctor/year Samples by written request only Industry association recommends 3 visits/year “Reasonable” standards of hospitality International conferences acceptable for “valid and cogent” reasons 100% of travel expenses can be reimbursed • In January, 2005 launched site that lets patient directly report adverse events to the government • Physician’s association approves continuing medical education programs • MHRA recently changed password requirement to a recommendation only on websites with information for physicians “UK’s NICE is ‘nice’ because it is transparent” – Belgian medical director “Patients want more information, but we are not sure how to communicate it” – Head of Global Affairs 35
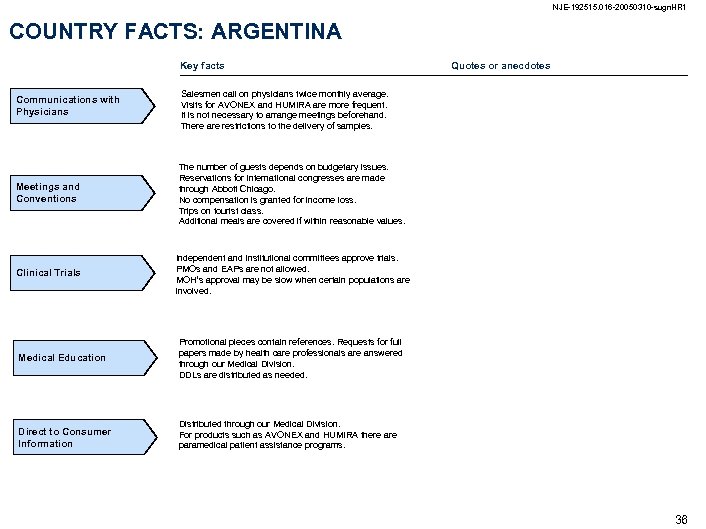 NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: ARGENTINA Key facts Communications with Physicians Salesmen call on physicians twice monthly average. Visits for AVONEX and HUMIRA are more frequent. It is not necessary to arrange meetings beforehand. There are restrictions to the delivery of samples. Meetings and Conventions The number of guests depends on budgetary issues. Reservations for international congresses are made through Abbott Chicago. No compensation is granted for income loss. Trips on tourist class. Additional meals are covered if within reasonable values. Clinical Trials Quotes or anecdotes Independent and institutional committees approve trials. PMOs and EAPs are not allowed. MOH’s approval may be slow when certain populations are involved. Medical Education Promotional pieces contain references. Requests for full papers made by health care professionals are answered through our Medical Division. DDLs are distributed as needed. Direct to Consumer Information Distributed through our Medical Division. For products such as AVONEX and HUMIRA there are paramedical patient assistance programs. 36
NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: ARGENTINA Key facts Communications with Physicians Salesmen call on physicians twice monthly average. Visits for AVONEX and HUMIRA are more frequent. It is not necessary to arrange meetings beforehand. There are restrictions to the delivery of samples. Meetings and Conventions The number of guests depends on budgetary issues. Reservations for international congresses are made through Abbott Chicago. No compensation is granted for income loss. Trips on tourist class. Additional meals are covered if within reasonable values. Clinical Trials Quotes or anecdotes Independent and institutional committees approve trials. PMOs and EAPs are not allowed. MOH’s approval may be slow when certain populations are involved. Medical Education Promotional pieces contain references. Requests for full papers made by health care professionals are answered through our Medical Division. DDLs are distributed as needed. Direct to Consumer Information Distributed through our Medical Division. For products such as AVONEX and HUMIRA there are paramedical patient assistance programs. 36
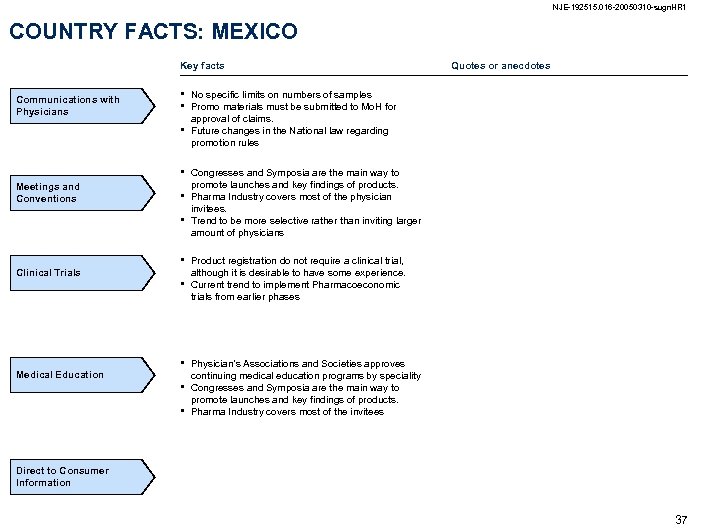 NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: MEXICO Key facts Communications with Physicians • • Meetings and Conventions • • • Clinical Trials Medical Education • • Quotes or anecdotes No specific limits on numbers of samples Promo materials must be submitted to Mo. H for approval of claims. Future changes in the National law regarding promotion rules Congresses and Symposia are the main way to promote launches and key findings of products. Pharma Industry covers most of the physician invitees. Trend to be more selective rather than inviting larger amount of physicians Product registration do not require a clinical trial, although it is desirable to have some experience. Current trend to implement Pharmacoeconomic trials from earlier phases Physician’s Associations and Societies approves continuing medical education programs by speciality Congresses and Symposia are the main way to promote launches and key findings of products. Pharma Industry covers most of the invitees Direct to Consumer Information 37
NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: MEXICO Key facts Communications with Physicians • • Meetings and Conventions • • • Clinical Trials Medical Education • • Quotes or anecdotes No specific limits on numbers of samples Promo materials must be submitted to Mo. H for approval of claims. Future changes in the National law regarding promotion rules Congresses and Symposia are the main way to promote launches and key findings of products. Pharma Industry covers most of the physician invitees. Trend to be more selective rather than inviting larger amount of physicians Product registration do not require a clinical trial, although it is desirable to have some experience. Current trend to implement Pharmacoeconomic trials from earlier phases Physician’s Associations and Societies approves continuing medical education programs by speciality Congresses and Symposia are the main way to promote launches and key findings of products. Pharma Industry covers most of the invitees Direct to Consumer Information 37
 NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: BRAZIL Key facts Communications with Physicians Quotes or anecdotes • • Regulatory restrictions for promotional claims; Claims against competitors allowed since based on published data; No limits for visits, no prior permission request; Physicians can be invited to participate in PMOS; Samples allowed, no amount limits. • TBD Regulatory restrictions about promotional contents and sponsorship; 100% of travel expenses can be reimbursed, never including entertainment; Promotion restricted to healthcare professionals; students not allowed at exhibition area. • TBD • • Continuous improvements on regulations; Specific rules for Early Access Programs; PMOS allowed; Sponsorship means any kind of support; • TBD • CME organized by pharmacos, medical societies or upon agreement between both; Kind of activity not frequently done by local pharmacos; • TBD Regulatory restrictions for DTC regarding contents and targets; Just OTCs are allowed to be promoted to consumers; prescribed drugs never. • TBD • • • Meetings and Conventions • • • Clinical Trials Medical Education Direct to Consumer Information • • • 38
NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: BRAZIL Key facts Communications with Physicians Quotes or anecdotes • • Regulatory restrictions for promotional claims; Claims against competitors allowed since based on published data; No limits for visits, no prior permission request; Physicians can be invited to participate in PMOS; Samples allowed, no amount limits. • TBD Regulatory restrictions about promotional contents and sponsorship; 100% of travel expenses can be reimbursed, never including entertainment; Promotion restricted to healthcare professionals; students not allowed at exhibition area. • TBD • • Continuous improvements on regulations; Specific rules for Early Access Programs; PMOS allowed; Sponsorship means any kind of support; • TBD • CME organized by pharmacos, medical societies or upon agreement between both; Kind of activity not frequently done by local pharmacos; • TBD Regulatory restrictions for DTC regarding contents and targets; Just OTCs are allowed to be promoted to consumers; prescribed drugs never. • TBD • • • Meetings and Conventions • • • Clinical Trials Medical Education Direct to Consumer Information • • • 38
 NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: JAPAN Key facts Communications with Physicians • • Meetings and Conventions • • Clinical Trials Medical Education Direct to Consumer Information Regulated by the rule from Japan Pharmaceutical Manufacturers Association. For example, invitation to dinner is not allowed in case of physicians of national/public hospitals. - Frequency of office visits: Frequent visit is required. On the other hand, physicains working for National Univ. /Hospital need to get special approval from their hospitals when honorium is paid by company. - Other channels of communication (phone, fax, email) Telephone, e-mail are less welcome comparing to western countries but face-to-face. Government published guidelines about reasonable expenses for honorarium, travel and food, etc. in case of physicians of national/public hospitals. Those physicians need prior permission by the institutes when attending the meetings. Social activities/hospitality a pharmaco can pay for at a meeting Highly stricted for physicians who are goverment employees. Reimbursement for travel and expenses Same as above for the physicians who are goverment employees. Under the contract, reimbursement and travel are often included in the study fee and no direct reimbursement to physicians. For ad-hoc meetings, not many restrictions for private university/hospital physicians but for goverment employee physicians need written permission from their hospitals Limitations of international conferences It is difficult to invite goverment employee physicians to a company sponsored conferences if of which purpose is to advertise either specific compnay or product. There should be always publicity • • • Cannot support investigator-initiated study for non-registration purpose without IND Mandatory data reporting Yes Adverse events Yes Mandatory reporting of unfavorable outcomes Yes Insurance requirements No # enrollees Not many because no insentive to physicians (money is to be paid to hospitals not to individual physicians) • … • • Individual Product DTC is illegal Password protection for physician info on websites Only required to answer Yes or No to a question "Are you medical people ? " No password protection. Promotion permitted on the physician/professional part of the site Only product information (package insert and interview form) Other criteria that define the difference between consumer ? information? and ? advertising? No advertisement with product on newspaper, TV, magazine, and radio etc. • • 39
NJE-192515. 016 -20050310 -sugn. HR 1 COUNTRY FACTS: JAPAN Key facts Communications with Physicians • • Meetings and Conventions • • Clinical Trials Medical Education Direct to Consumer Information Regulated by the rule from Japan Pharmaceutical Manufacturers Association. For example, invitation to dinner is not allowed in case of physicians of national/public hospitals. - Frequency of office visits: Frequent visit is required. On the other hand, physicains working for National Univ. /Hospital need to get special approval from their hospitals when honorium is paid by company. - Other channels of communication (phone, fax, email) Telephone, e-mail are less welcome comparing to western countries but face-to-face. Government published guidelines about reasonable expenses for honorarium, travel and food, etc. in case of physicians of national/public hospitals. Those physicians need prior permission by the institutes when attending the meetings. Social activities/hospitality a pharmaco can pay for at a meeting Highly stricted for physicians who are goverment employees. Reimbursement for travel and expenses Same as above for the physicians who are goverment employees. Under the contract, reimbursement and travel are often included in the study fee and no direct reimbursement to physicians. For ad-hoc meetings, not many restrictions for private university/hospital physicians but for goverment employee physicians need written permission from their hospitals Limitations of international conferences It is difficult to invite goverment employee physicians to a company sponsored conferences if of which purpose is to advertise either specific compnay or product. There should be always publicity • • • Cannot support investigator-initiated study for non-registration purpose without IND Mandatory data reporting Yes Adverse events Yes Mandatory reporting of unfavorable outcomes Yes Insurance requirements No # enrollees Not many because no insentive to physicians (money is to be paid to hospitals not to individual physicians) • … • • Individual Product DTC is illegal Password protection for physician info on websites Only required to answer Yes or No to a question "Are you medical people ? " No password protection. Promotion permitted on the physician/professional part of the site Only product information (package insert and interview form) Other criteria that define the difference between consumer ? information? and ? advertising? No advertisement with product on newspaper, TV, magazine, and radio etc. • • 39
 NJE-192515. 016 -20050310 -sugn. HR 1 ITALY’S PROSECUTION OF GSK IN 2003 -2004 REVEALS THE COMPLIANCE RISK INHERENT IN MARKETING & MED AFFAIRS “The Guardia di Finanza (Gi. F) have alleged that the Italian subsidiary of GSK, based in Verona, had spent 228 million Euros ($278 million) between 1999 and 2002 on cash, gifts and bribes to doctors and other medical professionals to encourage them to prescribe GSK's drug products. ” “The most serious allegations are that 60 Italian oncologists received payments for every patient treated with GSK's small cell lung and ovarian cancer treatment Hycamtin (topotecan). ” “The police said GSK employees disguised incentives and gifts under names such as ‘medical phase IV, ‘ ‘field selling’ and ‘other promotional expenses. ’ Alleged gifts ranged from foreign holidays to cameras and computer equipment, as well as straight cash payments” Italy’s regulations pre-date 2003 -04 and are among the most strict in Europe • It is illegal to collect prescribing information on specific doctors • Sales reps (“information agents”) must report to R & D, not Marketing • Information agents can only go to a doctor’s office by prior appointment These regulations were not always enforced. The GSK case may indicate a future trend of tougher enforcement. The stakes in this case were high: management was left vulnerable to criminal charges, including 73 directors and the managing director of GSK’s Italian subsidiary Source: “ 300 Glaxo. Smith. Kline employees named in illegal incentives probe in Italy. ” Pharma Marketletter, June 7, 2004 40
NJE-192515. 016 -20050310 -sugn. HR 1 ITALY’S PROSECUTION OF GSK IN 2003 -2004 REVEALS THE COMPLIANCE RISK INHERENT IN MARKETING & MED AFFAIRS “The Guardia di Finanza (Gi. F) have alleged that the Italian subsidiary of GSK, based in Verona, had spent 228 million Euros ($278 million) between 1999 and 2002 on cash, gifts and bribes to doctors and other medical professionals to encourage them to prescribe GSK's drug products. ” “The most serious allegations are that 60 Italian oncologists received payments for every patient treated with GSK's small cell lung and ovarian cancer treatment Hycamtin (topotecan). ” “The police said GSK employees disguised incentives and gifts under names such as ‘medical phase IV, ‘ ‘field selling’ and ‘other promotional expenses. ’ Alleged gifts ranged from foreign holidays to cameras and computer equipment, as well as straight cash payments” Italy’s regulations pre-date 2003 -04 and are among the most strict in Europe • It is illegal to collect prescribing information on specific doctors • Sales reps (“information agents”) must report to R & D, not Marketing • Information agents can only go to a doctor’s office by prior appointment These regulations were not always enforced. The GSK case may indicate a future trend of tougher enforcement. The stakes in this case were high: management was left vulnerable to criminal charges, including 73 directors and the managing director of GSK’s Italian subsidiary Source: “ 300 Glaxo. Smith. Kline employees named in illegal incentives probe in Italy. ” Pharma Marketletter, June 7, 2004 40
 NJE-192515. 016 -20050310 -sugn. HR 1 SWEDEN RECENTLY ENACTED REGULATIONS ON DOCTOR MEETINGS STRICTER THAN MOST EUROPEAN COUNTRIES* • Host companies can reimburse doctors no more than 50% of their costs for attending a conference • Travel should only be in economy class, unless there is a “negligible” price difference for business class • No social or recreational activities may be provided by the host company. Meals at conferences should be “extremely modest. ” • “Alcoholic drinks in the form of wine and beer may only be offered in limited quantities and only with food. No spirits may be offered. ” • Conferences should take place “at participants’ place of work or in the same town or city or as near as possible, ” deviating only when “pedagogical, practical, economic or other similar reasons so dictate” • Conferences outside Sweden “are only permitted… if the majority of participant professionals are not from Sweden and equivalent sessions cannot be held within the country. ”** Similar limitations apply to site visits to international studies *“Agreement On Forms Of Cooperation Between Pharmaceutical Companies And Medical Professionals In The Public Healthcare Sector” signed by the Landstingsförbundet (the Federation of Swedish County Councils) and Läkemedelsindustriföreningen (the Swedish Association of the Pharmaceutical Industry) in June, 2004 ** An exception is allowed for one region, southern Denmark 41
NJE-192515. 016 -20050310 -sugn. HR 1 SWEDEN RECENTLY ENACTED REGULATIONS ON DOCTOR MEETINGS STRICTER THAN MOST EUROPEAN COUNTRIES* • Host companies can reimburse doctors no more than 50% of their costs for attending a conference • Travel should only be in economy class, unless there is a “negligible” price difference for business class • No social or recreational activities may be provided by the host company. Meals at conferences should be “extremely modest. ” • “Alcoholic drinks in the form of wine and beer may only be offered in limited quantities and only with food. No spirits may be offered. ” • Conferences should take place “at participants’ place of work or in the same town or city or as near as possible, ” deviating only when “pedagogical, practical, economic or other similar reasons so dictate” • Conferences outside Sweden “are only permitted… if the majority of participant professionals are not from Sweden and equivalent sessions cannot be held within the country. ”** Similar limitations apply to site visits to international studies *“Agreement On Forms Of Cooperation Between Pharmaceutical Companies And Medical Professionals In The Public Healthcare Sector” signed by the Landstingsförbundet (the Federation of Swedish County Councils) and Läkemedelsindustriföreningen (the Swedish Association of the Pharmaceutical Industry) in June, 2004 ** An exception is allowed for one region, southern Denmark 41
 NJE-192515. 016 -20050310 -sugn. HR 1 EXTRA 42
NJE-192515. 016 -20050310 -sugn. HR 1 EXTRA 42
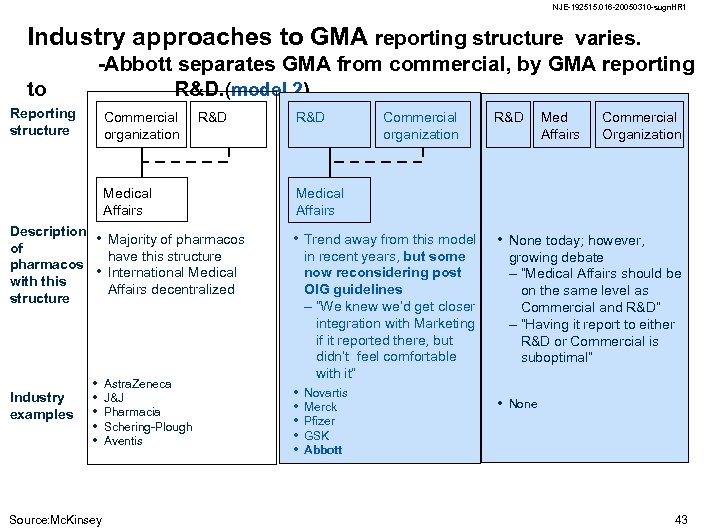 NJE-192515. 016 -20050310 -sugn. HR 1 Industry approaches to GMA reporting structure varies. to -Abbott separates GMA from commercial, by GMA reporting R&D. (model 2) Reporting structure Commercial organization R&D Medical Affairs Description of pharmacos with this structure Industry examples • Majority of pharmacos • • • Source: Mc. Kinsey R&D Med Affairs Commercial Organization Medical Affairs • Trend away from this model have this structure International Medical Affairs decentralized Astra. Zeneca J&J Pharmacia Schering-Plough Aventis Commercial organization in recent years, but some now reconsidering post OIG guidelines – “We knew we’d get closer integration with Marketing if it reported there, but didn’t feel comfortable with it” • • • Novartis Merck Pfizer GSK Abbott • None today; however, growing debate – “Medical Affairs should be on the same level as Commercial and R&D” – “Having it report to either R&D or Commercial is suboptimal” • None 43
NJE-192515. 016 -20050310 -sugn. HR 1 Industry approaches to GMA reporting structure varies. to -Abbott separates GMA from commercial, by GMA reporting R&D. (model 2) Reporting structure Commercial organization R&D Medical Affairs Description of pharmacos with this structure Industry examples • Majority of pharmacos • • • Source: Mc. Kinsey R&D Med Affairs Commercial Organization Medical Affairs • Trend away from this model have this structure International Medical Affairs decentralized Astra. Zeneca J&J Pharmacia Schering-Plough Aventis Commercial organization in recent years, but some now reconsidering post OIG guidelines – “We knew we’d get closer integration with Marketing if it reported there, but didn’t feel comfortable with it” • • • Novartis Merck Pfizer GSK Abbott • None today; however, growing debate – “Medical Affairs should be on the same level as Commercial and R&D” – “Having it report to either R&D or Commercial is suboptimal” • None 43
 NJE-192515. 016 -20050310 -sugn. HR 1 DIVERSITY OF GLOBAL MEDICAL AFFAIRS (GMA) RESPONSIBILITIES CREATES MANAGEMENT CHALLENGES Area and Affiliate Medical Departments Drug Information Services Publications GLOBAL MEDICAL AFFAIRS (GMA) Medical Science Liaisons HEOR Health Economics and Outcomes Research (HEOR) Global Clinical Operations 44
NJE-192515. 016 -20050310 -sugn. HR 1 DIVERSITY OF GLOBAL MEDICAL AFFAIRS (GMA) RESPONSIBILITIES CREATES MANAGEMENT CHALLENGES Area and Affiliate Medical Departments Drug Information Services Publications GLOBAL MEDICAL AFFAIRS (GMA) Medical Science Liaisons HEOR Health Economics and Outcomes Research (HEOR) Global Clinical Operations 44


