d359d7e91fb14df8184648b65b5f9df6.ppt
- Количество слайдов: 26

CONFIDENTIAL "International congresses, including booth materials and panels, controls related to non-CME medical presentations, and interactions between commercial associates and health care professionals before/after CME events: Which guidelines apply? " Dr. José F. Zamarriego Izquierdo Paris, May 29 th 2008 Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE OF PRACTICE g CONFIDENTIAL INTRODUCTION 1991: Adoption of the Spanish Code on the basis of EFPIA Code (adapted in 1992). 2002: Approved a much more stringent and precise new version of the Code. 2004: New version with additional measures to reinforce this self-regulation system. 2005: New adaptation, incorporating elements from EFPIA Code new version (Nov 04). 2008: Code New Version & New Code: Interaction with Patient Organizations (July 08). g DOCUMENTS (www. farmaindustria. es) 4 Spanish Code Of Practice 4 Rules of Procedure for the Control Bodies 4 Implementation Guide 4 Queries (Questions And Answers) on the Interpretation of the Code a. The Code covers all promotional actions addressed at Spanish healthcare professionals wherever they may take place (i. e. Spain, USA, South Africa, China. . ). a. Applicable to members of Farmaindustria and to the pharmaceutical companies adhered to the Code. Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE OF PRACTICE g CONFIDENTIAL GOALS GUARANTEE Information available and distributed to healthcare professional able to prescribe and dispense medicines, being accurate, balanced, fair, objective and complete. a That such promotion is done respecting the most stringent ethical principles of professionalism and responsibility. a ESTABLISH a. Establish a framework for the conduct of promotional activities by the industry, offering guidance and facilitating pharmaceutical companies of applicable rules. Spanish Code of Practice for the Promotion of Medicines compliance by

THE SPANISH CODE OF PRACTICE CONFIDENTIAL CODE COVERAGE 4 FARMAINDUSTRIA MEMBERS (ASSOCIATES) - NATIONAL COMPANIES: 98 (Large 10, Medium 6, Small 82) - INTERNATIONAL COMPANIES: 118 (Large 27, Medium 27, Small 64) 4 ADHERE COMPANIES (National & International) - Companies that voluntarily submit their promotion activities to the Code - Companies that are EFPIA members Approximately 250 Pharmaceutical Companies representing the 98% total sales of prescription medicines in Spanish Code of Practice for the Promotion of Medicines
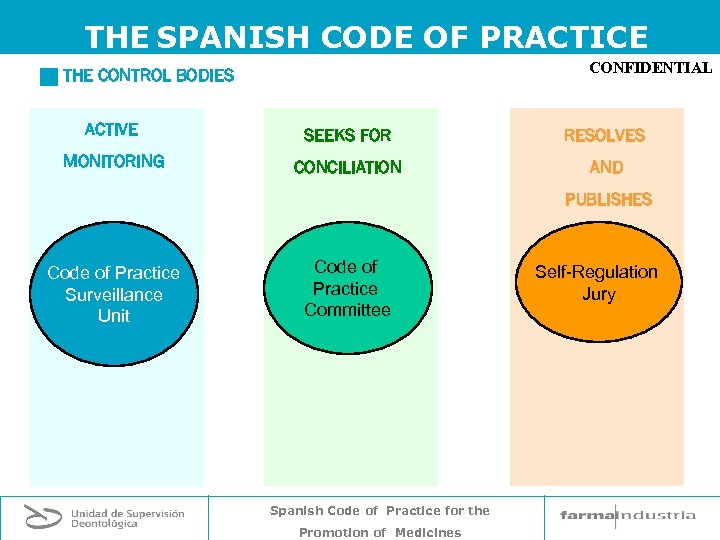
THE SPANISH CODE OF PRACTICE CONFIDENTIAL g THE CONTROL BODIES ACTIVE SEEKS FOR RESOLVES MONITORING CONCILIATION AND PUBLISHES Code of Practice Surveillance Unit Code of Practice Committee Spanish Code of Practice for the Promotion of Medicines Self-Regulation Jury
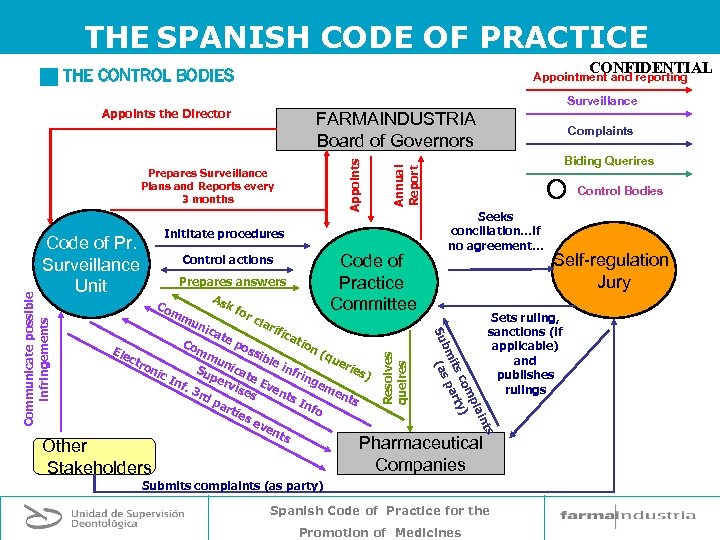
THE SPANISH CODE OF PRACTICE CONFIDENTIAL g THE CONTROL BODIES Appointment and reporting Surveillance Appoints the Director Prepares Surveillance Plans and Reports every 3 months Inititate procedures Code of Practice Committee Control Bodies Seeks conciliation…if no agreement. . . Pharmaceutical Companies Other Stakeholders Submits complaints (as party) Spanish Code of Practice for the Promotion of Medicines Self-regulation Jury Sets ruling, sanctions (if applicable) and publishes rulings nts lai mp co y) its art bm as p ( Prepares answers As kf Co or mm cla un rif ica te tio Co po n( ssi mm Ele qu ble ctr un eri on inf ica es Su ic rin te ) pe In ge Ev rvi f. 3 me ses en rd nts ts pa In fo rti es ev en ts Resolves queires Control actions Biding Querires Su Communicate possible infringements Code of Pr. Surveillance Unit Complaints Annual Report Appoints FARMAINDUSTRIA Board of Governors

THE SPANISH CODE OF PRACTICE CONFIDENTIAL g g INTRODUCTION, PURPOSE AND SCOPE OF THE CODE ARTICLES 1 MARKETING AUTHORIZATION 2 INFORMATION TO BE MADE AVAILABLE 3 INFORMATION AND ITS SUBSTANTIATION 4 ACCEPTABILITY OF PROMOTION 5 TRANSPARENCY OF PROMOTION 6 USE OF REFERENCE QUOTATIONS 7 DISTRIBUTION OF PROMOTIONAL MATERIAL 8 PROMOTION VIA INTERNET 9 SCIENTIFC SERVICE AND REVIEW OF PROMOTIONAL MATERIAL 10 GIFTS AND INDUCEMENTS 11 HOSPITALITY AND MEETINGS Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE OF PRACTICE CONFIDENTIAL g ARTICLES 12 PHARMACEUTICAL COMPANY STAFF 13 SAMPLES 14 STUDIES 15 RULES OF APPLICATION OF THE CODE 16 REQUEST FOR QUERIES 17 CONTROL OF THE BODIE COMPLIANCE 18 INFRINGEMENTS AND SANCTIONS 19 IMPLEMENTATION GUIDES AND COLLABORATION AGREEMENTS 20 DISCLOSURE AND COMPLILATION OF DECISIONS 21 DATE OF ENTRY INTO FORCE OF THE CODE Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE OF PRACTICE CONFIDENTIAL 11. HOSPITALITY AND MEETINGS CONGRESSES AND SCIENTIFIC EVENTS FUNDED BY THE INDUSTRY 4 The behaviour of the pharmaceutical companies must be guided by this principles: 4 Scientific goals must represent the main focus in the organization of such meetings 88. 4 Hospitality should be reasonable at all times, and must always be secondary to the main purpose of the meeting A useful criterion Would the company like that all details of the organization of the event to be widely disseminated? Spanish Code of Practice for the Promotion of Medicines
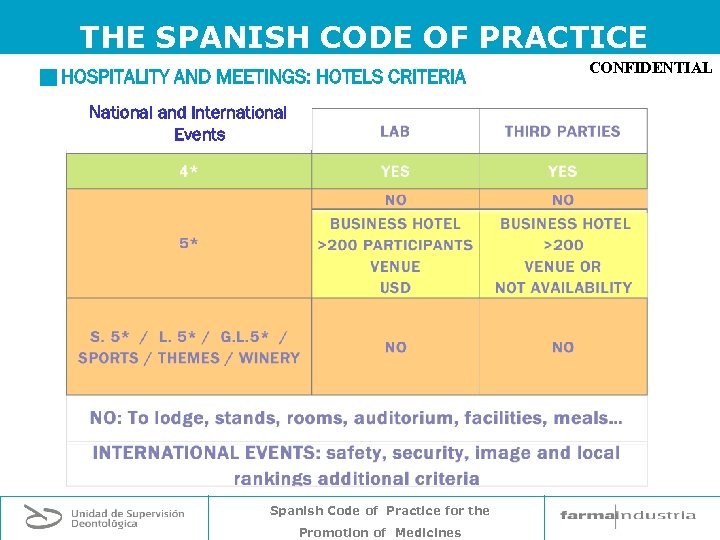
THE SPANISH CODE OF PRACTICE g HOSPITALITY AND MEETINGS: HOTELS CRITERIA National and International Events Spanish Code of Practice for the Promotion of Medicines CONFIDENTIAL

THE SPANISH CODE OF PRACTICE CONFIDENTIAL GENERAL STANDARDS APPLICABLE IN SPAIN: EVENTS & HOSPITALITY Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE OF PRACTICE CONFIDENTIAL g EVENTS ORGANIZED BY THIRD PARTIES Located on Farmaindustria intranet there is a useful tool, available to all its members, through which the national and international events organized by third parties that have been reviewed by the Unit can be consulted. 4 System available to companies Compliance Officers only. Spanish Code of Practice for the Promotion of Medicines
THE SPANISH CODE OF PRACTICE CONFIDENTIAL g EVENTS ORGANIZED BY THIRD PARTIES Spanish Code of Practice for the Promotion of Medicines
THE SPANISH CODE OF PRACTICE CONFIDENTIAL g NON PERMITTED PARTICIPATION Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE OF PRACTICE CONFIDENTIAL g POSSIBLE PARTICIPATION WITH COMMENTS Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE OF PRACTICE CONFIDENTIAL g POSSIBLE PARTICIPATION Spanish Code of Practice for the Promotion of Medicines
THE SPANISH CODE OF PRACTICE CONFIDENTIAL g ON STUDY BY THE SURVEILLANCE UNIT Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE OF PRACTICE CONFIDENTIAL g EVENTS ORGANIZED BY THIRD PARTIES Positive effects: When a national event is marked as non permitted: - Laboratories contact the organisers to let them know that there are some items that do not comply with the Code. - Organisers contact the Surveillance Unit for advise, make the correct changes for an approval qualification and send a signed letter certifying it. - Surveillance Unit changes the non permitted mark for the possible participation with comments marked and send an email to the laboratories to let them know the new conditions. Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE OF PRACTICE CONFIDENTIAL g CASE STUDY Procedure followed once an event which might; (i) cause an infringement of the Code of Practice or (ii) represents a material risk for pharmaceutical industry image; is identified: 4 Contact with the organizers (Scientific Society or Technical Secretary), 4 If necessary give a brief explanation of our self-regulation system (Article 11) and the role of the Surveillance Unit, 4 Indicate aspects related with the meeting that do not allow pharmaceutical industry collaboration. Explain why, 4 Propose solutions, alternatives, recommendations, modifications, 4 Inform companies about the result of this process and qualify the event. Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE OF PRACTICE CONFIDENTIAL g CASE STUDY GOLF TOURNAMENT DURING THE MEETING Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE OF PRACTICE CONFIDENTIAL g CASE STUDY Hereby confirm that the golf tournament has been cancelled and that no golf tournament will take place. The President of the Organization Committee. Spanish Code of Practice for the Promotion of Medicines
THE SPANISH CODE OF PRACTICE CONFIDENTIAL g CASE STUDY Spanish Code of Practice for the Promotion of Medicines
THE SPANISH CODE OF PRACTICE g CODE COMPLIANCE NETWORK (CCN) Spanish Code of Practice for the Promotion of Medicines CONFIDENTIAL
THE SPANISH CODE OF PRACTICE g CODE COMPLIANCE NETWORK (CCN) Spanish Code of Practice for the Promotion of Medicines CONFIDENTIAL

CONCLUSIONS CONFIDENTIAL KEEPING AND REINFORCING THE CRITICAL ASPECTS, THE SELF-REGULATION SYSTEM IS A STRENGTH OF THE PHARMACEUTICAL INDUSTRY TO POSITIVELY IMPROVE ITS IMAGE BEFORE THE WHOLE SOCIETY (PUBLIC ADMINISTRATION, HEALTHCARE PROFESSIONALS AND PATIENTS) Spanish Code of Practice for the Promotion of Medicines

THE SPANISH CODE SURVILLANCE UNIT Velázquez 109 E - 28006 Madrid - Spain Tel. +34 91 7452050 Fax +34 91 7450408 usd@codigo. farmaindustria. es www. farmaindustria. es
d359d7e91fb14df8184648b65b5f9df6.ppt