 Concept of p. H Hydrolysis of salts
Concept of p. H Hydrolysis of salts
 The plan of the lesson Self-ionization of water Concept of p. H Indicators Hydrolysis of salt Hydrolysis constant Degree of Hydrolysis
The plan of the lesson Self-ionization of water Concept of p. H Indicators Hydrolysis of salt Hydrolysis constant Degree of Hydrolysis
 Self-ionization of water H 2 O + H 2 O ↔ H 3 O+ + OH- For any degree of ionization we will have [H 3 O]+ = [OH-] But at 298 K Kw= [H 3 O+] [OH-] =1. 0 x 10 -14 [H 3 O+]2 =1. 0 x 10 – 14 [H 3 O+] =1. 0 x 10 – 7 mol/l Thus in pure water at 298 K [H 3 O]+ = [OH-] = 1. 0 x 10 – 7 mol/l
Self-ionization of water H 2 O + H 2 O ↔ H 3 O+ + OH- For any degree of ionization we will have [H 3 O]+ = [OH-] But at 298 K Kw= [H 3 O+] [OH-] =1. 0 x 10 -14 [H 3 O+]2 =1. 0 x 10 – 14 [H 3 O+] =1. 0 x 10 – 7 mol/l Thus in pure water at 298 K [H 3 O]+ = [OH-] = 1. 0 x 10 – 7 mol/l
 Concept of p. H The symbol p. H has been taken from danish word ‘potenz the hydrogen ion’which means power of hydrogen ion. The p. H of solution may be defined as negetive logarithm of hydrogen ion concentration. p. H = -log [H+] p. ОH=-log [ОH-] (1) (2)
Concept of p. H The symbol p. H has been taken from danish word ‘potenz the hydrogen ion’which means power of hydrogen ion. The p. H of solution may be defined as negetive logarithm of hydrogen ion concentration. p. H = -log [H+] p. ОH=-log [ОH-] (1) (2)
 Acidic or basic solutions can be distinguished on basis of p. H For neutral solutions [Н+] =10 – 7 or p. H=7 For acidic solutions [H+] >10 – 7 or p. H < 7 For basic solutions [H+] <10 – 7 or p. H > 7
Acidic or basic solutions can be distinguished on basis of p. H For neutral solutions [Н+] =10 – 7 or p. H=7 For acidic solutions [H+] >10 – 7 or p. H < 7 For basic solutions [H+] <10 – 7 or p. H > 7
![Relationship between p. H and p. OH p. H = -log [H+] p. OH Relationship between p. H and p. OH p. H = -log [H+] p. OH](https://present5.com/presentation/-58201108_300341421/image-6.jpg) Relationship between p. H and p. OH p. H = -log [H+] p. OH = -log [OH-] [H+] [OH-] =10 -14 log[H+]+ log[OH-] =log 10 -14 log[H+]+ log[OH-] =14 -log[H+]- log[OH-]=14 p. H + p. OH= 14
Relationship between p. H and p. OH p. H = -log [H+] p. OH = -log [OH-] [H+] [OH-] =10 -14 log[H+]+ log[OH-] =log 10 -14 log[H+]+ log[OH-] =14 -log[H+]- log[OH-]=14 p. H + p. OH= 14
![The Relationship Between p. H, p. OH, [H+] and [OH-] p. H p. OH The Relationship Between p. H, p. OH, [H+] and [OH-] p. H p. OH](https://present5.com/presentation/-58201108_300341421/image-7.jpg) The Relationship Between p. H, p. OH, [H+] and [OH-] p. H p. OH [H+] mol/l [OH-] mol/l 0 14 1. 0 10 -14 2 12 10 -12 4 10 10 -4 10 -10 6 8 10 -6 10 -8 8 6 10 -8 10 -6 10 4 10 -10 10 -4 12 2 10 -12 10 -2 14 0 10 -14 1. 0
The Relationship Between p. H, p. OH, [H+] and [OH-] p. H p. OH [H+] mol/l [OH-] mol/l 0 14 1. 0 10 -14 2 12 10 -12 4 10 10 -4 10 -10 6 8 10 -6 10 -8 8 6 10 -8 10 -6 10 4 10 -10 10 -4 12 2 10 -12 10 -2 14 0 10 -14 1. 0
![The p. H scale goes from 0 to 14 — because [H+][OH-] = The p. H scale goes from 0 to 14 — because [H+][OH-] =](https://present5.com/presentation/-58201108_300341421/image-8.jpg) The p. H scale goes from 0 to 14 — because [H+][OH-] = 10 -14
The p. H scale goes from 0 to 14 — because [H+][OH-] = 10 -14
![[H+] M 100 A strong acid 10 -1 10 -2 10 -3 10 -5 [H+] M 100 A strong acid 10 -1 10 -2 10 -3 10 -5](https://present5.com/presentation/-58201108_300341421/image-9.jpg) [H+] M 100 A strong acid 10 -1 10 -2 10 -3 10 -5 10 -6 10 -7 • [H+] is controlled in all biological organisms, and in virtually all biochemical experiments. 10 -8 10 -9 10 -10 10 -11 10 -13 10 -14 • Most living cells have a very narrow range of tolerance for p. H, i. e. [H+]. • The [H+] concentration will be important (either explicitly or implicitly) for many other topics in biology. 10 -4 10 -12 p. H A strong base • Each p. H unit represents a factor of 14 difference in [H+].
[H+] M 100 A strong acid 10 -1 10 -2 10 -3 10 -5 10 -6 10 -7 • [H+] is controlled in all biological organisms, and in virtually all biochemical experiments. 10 -8 10 -9 10 -10 10 -11 10 -13 10 -14 • Most living cells have a very narrow range of tolerance for p. H, i. e. [H+]. • The [H+] concentration will be important (either explicitly or implicitly) for many other topics in biology. 10 -4 10 -12 p. H A strong base • Each p. H unit represents a factor of 14 difference in [H+].
 INDICATORS Indicators are substances whose solutions change colour due to changes in p. H. These are called acid-base Indicators. Indicator name Litmus Methil Orange Phenolphthalein Colour in Acid Red colourless Colour in Base Blue Yellow Pink Universal indicator Red purple
INDICATORS Indicators are substances whose solutions change colour due to changes in p. H. These are called acid-base Indicators. Indicator name Litmus Methil Orange Phenolphthalein Colour in Acid Red colourless Colour in Base Blue Yellow Pink Universal indicator Red purple
 Universal Indicator To measure p. H, we need an indicator that has a different colour for each value of p. H red 0 ≥ p. H ≥ 3 yellow 3 ≥ p. H ≥ 6 green 7 blue 8 ≥ p. H ≥ 11 purple 11 ≥ p. H ≥ 14 Universal Indicator
Universal Indicator To measure p. H, we need an indicator that has a different colour for each value of p. H red 0 ≥ p. H ≥ 3 yellow 3 ≥ p. H ≥ 6 green 7 blue 8 ≥ p. H ≥ 11 purple 11 ≥ p. H ≥ 14 Universal Indicator
 Hydrolysis OF SALTS Salts are ionic compounds that result from the neutralization reaction of an acid and a base. Usually hydrolysis is a chemical process in which a molecule of water is added to a substance.
Hydrolysis OF SALTS Salts are ionic compounds that result from the neutralization reaction of an acid and a base. Usually hydrolysis is a chemical process in which a molecule of water is added to a substance.
 The ions of salts can have an influence on the p. H of a solution. Ions that come from a strong acid or base do not influence the p. H. WHY? Since strong acids and bases are 100% ionized in water, the ions are unable to reform the molecular acid or the base in water. HCl + H 2 O H 3 O+ + Cl. Na. OH + H 2 O Na+ + OH- + H 2 O “one way arrow” Na. Cl is the salt that comes from a strong acid and a strong base. 13
The ions of salts can have an influence on the p. H of a solution. Ions that come from a strong acid or base do not influence the p. H. WHY? Since strong acids and bases are 100% ionized in water, the ions are unable to reform the molecular acid or the base in water. HCl + H 2 O H 3 O+ + Cl. Na. OH + H 2 O Na+ + OH- + H 2 O “one way arrow” Na. Cl is the salt that comes from a strong acid and a strong base. 13
 What would the p. H of a sodium chloride solution (@ 25 o. C)? p. H = 7 What gives rise to this p. H? Auto hydrolysis of water. H 2 O + H 2 O ↔ H 3 O+ + OH- 14
What would the p. H of a sodium chloride solution (@ 25 o. C)? p. H = 7 What gives rise to this p. H? Auto hydrolysis of water. H 2 O + H 2 O ↔ H 3 O+ + OH- 14
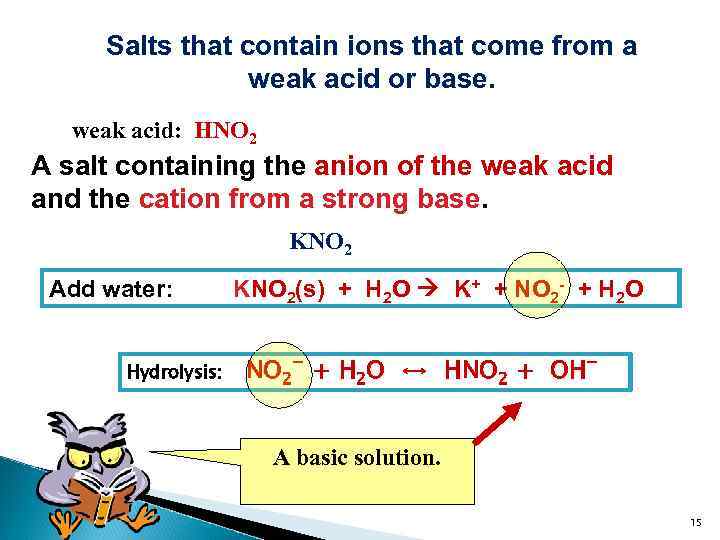 Salts that contain ions that come from a weak acid or base. weak acid: HNO 2 A salt containing the anion of the weak acid and the cation from a strong base. KNO 2 Add water: KNO 2(s) + H 2 O K+ + NO 2 - + H 2 O Hydrolysis: NO 2 - + H 2 O ↔ HNO 2 + OHA basic solution. 15
Salts that contain ions that come from a weak acid or base. weak acid: HNO 2 A salt containing the anion of the weak acid and the cation from a strong base. KNO 2 Add water: KNO 2(s) + H 2 O K+ + NO 2 - + H 2 O Hydrolysis: NO 2 - + H 2 O ↔ HNO 2 + OHA basic solution. 15
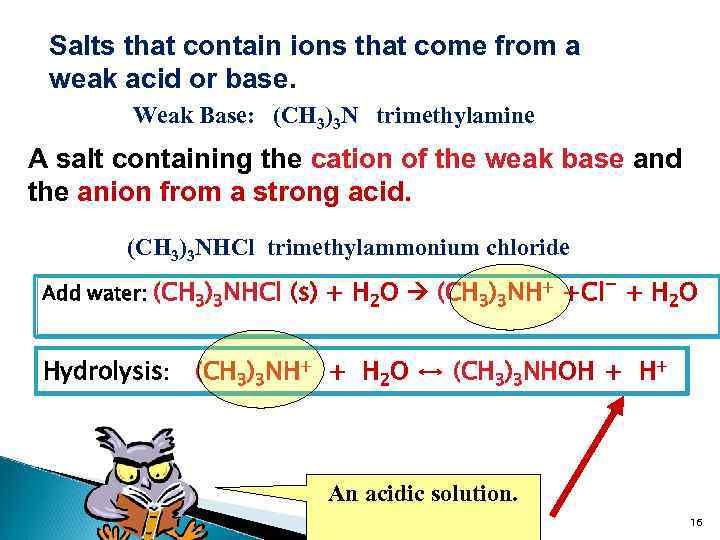 Salts that contain ions that come from a weak acid or base. Weak Base: (CH 3)3 N trimethylamine A salt containing the cation of the weak base and the anion from a strong acid. (CH 3)3 NHCl trimethylammonium chloride Add water: (CH 3)3 NHCl (s) + H 2 O (CH 3)3 NH+ +Cl- + H 2 O Hydrolysis: (CH 3)3 NH+ + H 2 O ↔ (CH 3)3 NHOH + H+ An acidic solution. 16
Salts that contain ions that come from a weak acid or base. Weak Base: (CH 3)3 N trimethylamine A salt containing the cation of the weak base and the anion from a strong acid. (CH 3)3 NHCl trimethylammonium chloride Add water: (CH 3)3 NHCl (s) + H 2 O (CH 3)3 NH+ +Cl- + H 2 O Hydrolysis: (CH 3)3 NH+ + H 2 O ↔ (CH 3)3 NHOH + H+ An acidic solution. 16
 Salts that contain ions that come from a weak acid and weak base. CH 3 COONH 4 ammonium salt A salt containing the cation of the weak base and the anion from a weak acid. Add water: CH 3 COONH 4 + H 2 O CH 3 COOH + NH 4 OH Hydrolysis: CH 3 COO- + NH 4+ + H 2 O CH 3 COOH + NH 4 OH solution will be neutral. 17
Salts that contain ions that come from a weak acid and weak base. CH 3 COONH 4 ammonium salt A salt containing the cation of the weak base and the anion from a weak acid. Add water: CH 3 COONH 4 + H 2 O CH 3 COOH + NH 4 OH Hydrolysis: CH 3 COO- + NH 4+ + H 2 O CH 3 COOH + NH 4 OH solution will be neutral. 17
 What if both ions of a salt come from weak acid and a weak base? Then the Ka and Kb of the acid or base from which the ions come from must be compared. NH 4 CN(aq) Ka(NH 4+) = 5. 6 x 10 -10 Kb(CN-) = 2. 04 x 10 -5 note: Ka was calculated from Kb(NH 3) and Kb from Ka HCN Since Kb(CN-) is greater than Ka(NH 4+), the solution is basic.
What if both ions of a salt come from weak acid and a weak base? Then the Ka and Kb of the acid or base from which the ions come from must be compared. NH 4 CN(aq) Ka(NH 4+) = 5. 6 x 10 -10 Kb(CN-) = 2. 04 x 10 -5 note: Ka was calculated from Kb(NH 3) and Kb from Ka HCN Since Kb(CN-) is greater than Ka(NH 4+), the solution is basic.
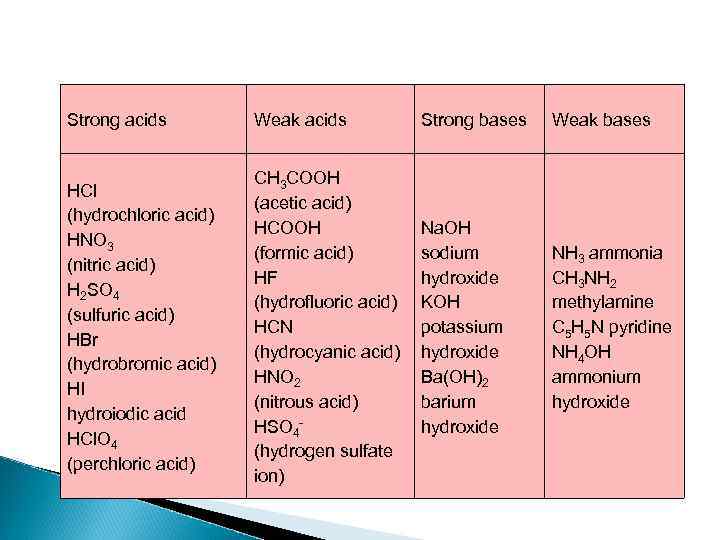 Strong acids Weak acids Strong bases Weak bases HCl (hydrochloric acid) HNO 3 (nitric acid) H 2 SO 4 (sulfuric acid) HBr (hydrobromic acid) HI hydroiodic acid HCl. O 4 (perchloric acid) CH 3 COOH (acetic acid) HCOOH (formic acid) HF (hydrofluoric acid) HCN (hydrocyanic acid) HNO 2 (nitrous acid) HSO 4 - (hydrogen sulfate ion) Na. OH sodium hydroxide KOH potassium hydroxide Ba(OH)2 barium hydroxide NH 3 ammonia CH 3 NH 2 methylamine C 5 H 5 N pyridine NH 4 OH ammonium hydroxide
Strong acids Weak acids Strong bases Weak bases HCl (hydrochloric acid) HNO 3 (nitric acid) H 2 SO 4 (sulfuric acid) HBr (hydrobromic acid) HI hydroiodic acid HCl. O 4 (perchloric acid) CH 3 COOH (acetic acid) HCOOH (formic acid) HF (hydrofluoric acid) HCN (hydrocyanic acid) HNO 2 (nitrous acid) HSO 4 - (hydrogen sulfate ion) Na. OH sodium hydroxide KOH potassium hydroxide Ba(OH)2 barium hydroxide NH 3 ammonia CH 3 NH 2 methylamine C 5 H 5 N pyridine NH 4 OH ammonium hydroxide
 HYDROLYSIS CONSTANT The general equation for the hydrolysis of a salt (BA), Applying the law of chemical equilibrium, we get where K is the equilibrium constant. Since water is present in very large excess in the aqueous solution, its concentration [H 2 O] may be regarded as constant so, where Kw is called the hydrolysis constant.
HYDROLYSIS CONSTANT The general equation for the hydrolysis of a salt (BA), Applying the law of chemical equilibrium, we get where K is the equilibrium constant. Since water is present in very large excess in the aqueous solution, its concentration [H 2 O] may be regarded as constant so, where Kw is called the hydrolysis constant.
 DEGREE OF HYDROLYSIS It is defined as the fraction (or percentage) of the total salt which is hydrolysed at equilibrium. For example, if 90% of a salt solution is hydrolysed, its degree of hydrolysis is 0. 90 or as 90%. It is generally represented by ‘h’.
DEGREE OF HYDROLYSIS It is defined as the fraction (or percentage) of the total salt which is hydrolysed at equilibrium. For example, if 90% of a salt solution is hydrolysed, its degree of hydrolysis is 0. 90 or as 90%. It is generally represented by ‘h’.
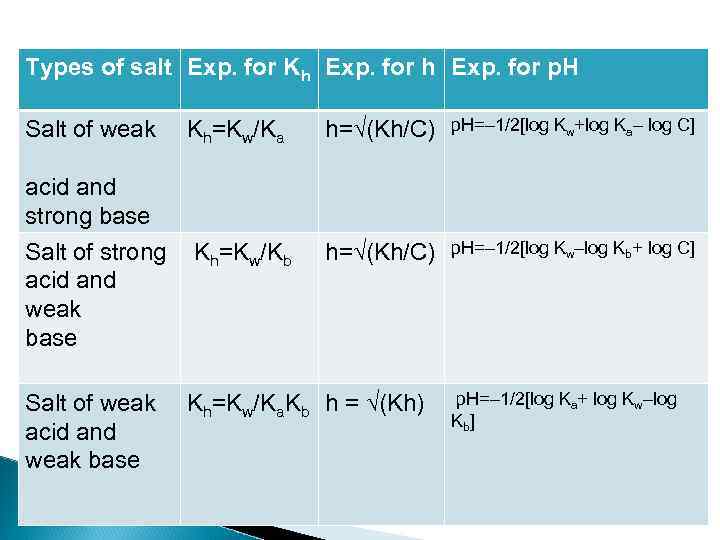 Types of salt Exp. for Kh Exp. for p. H Salt of weak Kh=Kw/Ka acid and strong base h=√(Kh/C) p. H=– 1/2[log Kw+log Ka– log C] Salt of strong Kh=Kw/Kb acid and weak base h=√(Kh/C) p. H=– 1/2[log Kw–log Kb+ log C] Salt of weak acid and weak base Kh=Kw/Ka. Kb h = √(Kh) p. H=– 1/2[log Ka+ log Kw–log Kb]
Types of salt Exp. for Kh Exp. for p. H Salt of weak Kh=Kw/Ka acid and strong base h=√(Kh/C) p. H=– 1/2[log Kw+log Ka– log C] Salt of strong Kh=Kw/Kb acid and weak base h=√(Kh/C) p. H=– 1/2[log Kw–log Kb+ log C] Salt of weak acid and weak base Kh=Kw/Ka. Kb h = √(Kh) p. H=– 1/2[log Ka+ log Kw–log Kb]


