independent_work_2.pptx
- Количество слайдов: 13
 Comparison of Sensitivities and Specificities of Latex Agglutination and an Enzyme-Linked Immunosorbent Assay for Detection of Antibodies to the Human Immunodeficiency Virus in African Sera Review Khaldarova Malika
Comparison of Sensitivities and Specificities of Latex Agglutination and an Enzyme-Linked Immunosorbent Assay for Detection of Antibodies to the Human Immunodeficiency Virus in African Sera Review Khaldarova Malika
 Outline § Introduction § Location and quantity of tested people § Enzyme linked immunosorbent assay(ELISA) § Latex agglutination assay (LA) § Preparation of blood samples § Result § Conclusion
Outline § Introduction § Location and quantity of tested people § Enzyme linked immunosorbent assay(ELISA) § Latex agglutination assay (LA) § Preparation of blood samples § Result § Conclusion
 Introduction • Human immunodeficiency virus(HIV) has become international health problem affecting 5 to 10 million people around the world. • To quantify the populations at risk usually used by enzyme linked immunosorbent assay(ELISA) • However, ELISA is too expensive for Third world countries to implement • Recognizing these problems, biotechnology companies are developing rapid HIV diagnostic tests as latex agglutination assay(LA) which give rapid results, cheap, easy to use and as sensitive and specific as ELISA.
Introduction • Human immunodeficiency virus(HIV) has become international health problem affecting 5 to 10 million people around the world. • To quantify the populations at risk usually used by enzyme linked immunosorbent assay(ELISA) • However, ELISA is too expensive for Third world countries to implement • Recognizing these problems, biotechnology companies are developing rapid HIV diagnostic tests as latex agglutination assay(LA) which give rapid results, cheap, easy to use and as sensitive and specific as ELISA.
 Location and quantity of tested people • Scientists used a latex agglutination(LA) assay in the Mama Yemo Hospital blood bank, Kinshsa, Zaire to evaluate efficacy. • Serum samples was taken from 1, 482 (771 children and 711 adults) patients and tested by LA and ELISA
Location and quantity of tested people • Scientists used a latex agglutination(LA) assay in the Mama Yemo Hospital blood bank, Kinshsa, Zaire to evaluate efficacy. • Serum samples was taken from 1, 482 (771 children and 711 adults) patients and tested by LA and ELISA
 ELISA: procedure • The plasma was stored at 20 F until ELISA and western blood assay could be done • All sera were analyzed for the presence of HIV antibody with the Wellcozyme assay (Wellcome Diagnostics, Dartford, England). • Positive sera were repeatedly reactive in two separate ELISAs, and the results were confirmed by Western blot.
ELISA: procedure • The plasma was stored at 20 F until ELISA and western blood assay could be done • All sera were analyzed for the presence of HIV antibody with the Wellcozyme assay (Wellcome Diagnostics, Dartford, England). • Positive sera were repeatedly reactive in two separate ELISAs, and the results were confirmed by Western blot.
 Latex agglutination assay: procedure • The LA was performed with recombinant gpl 20 antigen • (CBre 3) attached to 0. 5 -p. m polystyrene beads and suspended in 1% bovine serum albumin in phosphatebuffered saline (p. H 7. 6) • The recombinant DNA derived from portions of the HIV envelope gene (env) was expressed in Escherichia coli by using a lambda p. L expression vector. • The polypeptide contains 17 N-terminal amino acids derived from phage gamma and nucleotide linker sequences followed by HIV env amino acids (9). • The expressed polypeptide was purified by standard chromatography procedures and was shown to be free of contamination with E. coli antigens.
Latex agglutination assay: procedure • The LA was performed with recombinant gpl 20 antigen • (CBre 3) attached to 0. 5 -p. m polystyrene beads and suspended in 1% bovine serum albumin in phosphatebuffered saline (p. H 7. 6) • The recombinant DNA derived from portions of the HIV envelope gene (env) was expressed in Escherichia coli by using a lambda p. L expression vector. • The polypeptide contains 17 N-terminal amino acids derived from phage gamma and nucleotide linker sequences followed by HIV env amino acids (9). • The expressed polypeptide was purified by standard chromatography procedures and was shown to be free of contamination with E. coli antigens.
 Latex agglutination assay • The patient sample (25 jsl) (described below) was spread over a 15 -mm circle on a slide • Mixed with 15 tl of 0. 6% latex beads coated with recombinant gp 120 antigen and gently rotated for 5 to 8 min. • A marked flocculent agglutination reaction appeared with HIV-positive sera under highintensity fluorescent light. • HIV-nonreactive sera retained a smooth, creamy homogeneous texture.
Latex agglutination assay • The patient sample (25 jsl) (described below) was spread over a 15 -mm circle on a slide • Mixed with 15 tl of 0. 6% latex beads coated with recombinant gp 120 antigen and gently rotated for 5 to 8 min. • A marked flocculent agglutination reaction appeared with HIV-positive sera under highintensity fluorescent light. • HIV-nonreactive sera retained a smooth, creamy homogeneous texture.
 Preparation of blood samples • Patient samples were prepared in three ways: by the manufacturer's method of using serum diluted 1: 10 with phosphate-buffered saline with 1% bovine albumin, • by using whole blood diluted 1: 10 with phosphate-buffered saline with 1% bovine serum albumin, • by using undiluted whole blood.
Preparation of blood samples • Patient samples were prepared in three ways: by the manufacturer's method of using serum diluted 1: 10 with phosphate-buffered saline with 1% bovine albumin, • by using whole blood diluted 1: 10 with phosphate-buffered saline with 1% bovine serum albumin, • by using undiluted whole blood.
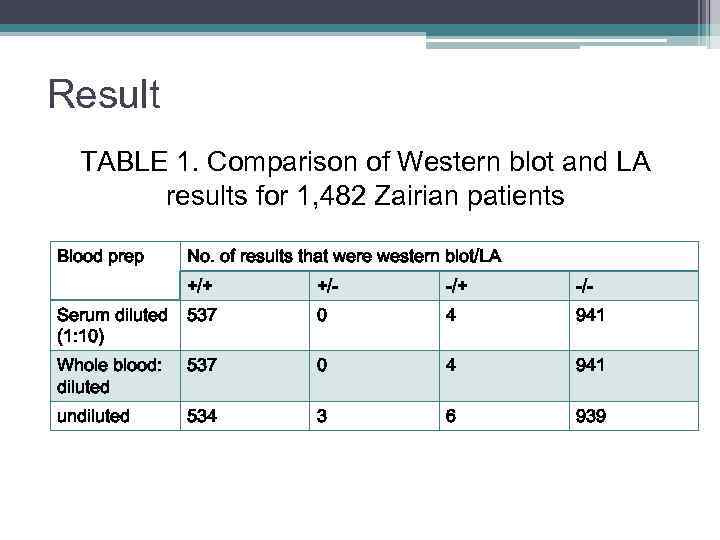 Result TABLE 1. Comparison of Western blot and LA results for 1, 482 Zairian patients Blood prep No. of results that were western blot/LA +/+ +/- -/+ -/- Serum diluted (1: 10) 537 0 4 941 Whole blood: diluted 537 0 4 941 undiluted 534 3 6 939
Result TABLE 1. Comparison of Western blot and LA results for 1, 482 Zairian patients Blood prep No. of results that were western blot/LA +/+ +/- -/+ -/- Serum diluted (1: 10) 537 0 4 941 Whole blood: diluted 537 0 4 941 undiluted 534 3 6 939
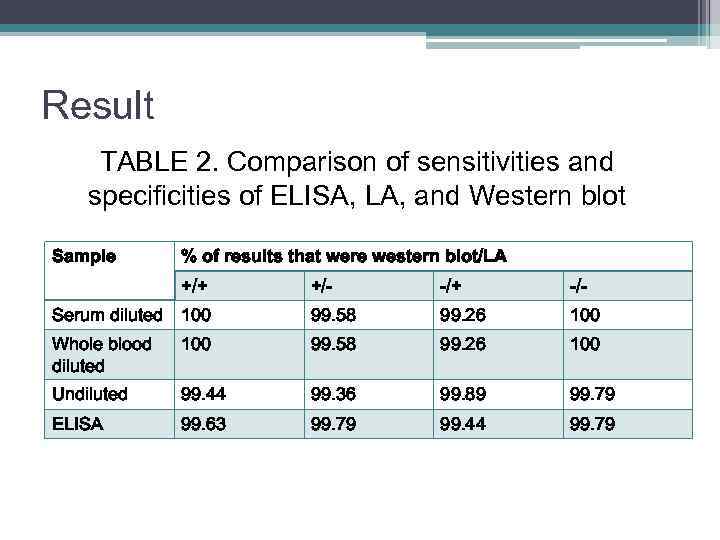 Result TABLE 2. Comparison of sensitivities and specificities of ELISA, LA, and Western blot Sample % of results that were western blot/LA +/+ +/- -/+ -/- Serum diluted 100 99. 58 99. 26 100 Whole blood diluted 100 99. 58 99. 26 100 Undiluted 99. 44 99. 36 99. 89 99. 79 ELISA 99. 63 99. 79 99. 44 99. 79
Result TABLE 2. Comparison of sensitivities and specificities of ELISA, LA, and Western blot Sample % of results that were western blot/LA +/+ +/- -/+ -/- Serum diluted 100 99. 58 99. 26 100 Whole blood diluted 100 99. 58 99. 26 100 Undiluted 99. 44 99. 36 99. 89 99. 79 ELISA 99. 63 99. 79 99. 44 99. 79
 Conclusion • The sensitivities, specificities, and positive and negative predictive values of the latex agglutination assay were compared by using three different blood preparations: 1. diluted sera – the sensitivity was 100% the specificity was 99. 58% the positive and negative predictive values were 99. 26 and 100% 2. diluted or undiluted whole blood -the sensitivity -99. 72% the specificity 99. 47% positive predictive value 99. 07% negative predictive value 99. 89%
Conclusion • The sensitivities, specificities, and positive and negative predictive values of the latex agglutination assay were compared by using three different blood preparations: 1. diluted sera – the sensitivity was 100% the specificity was 99. 58% the positive and negative predictive values were 99. 26 and 100% 2. diluted or undiluted whole blood -the sensitivity -99. 72% the specificity 99. 47% positive predictive value 99. 07% negative predictive value 99. 89%
 References: • HENRY L. FRANCIS, MULANGA KABEYA, ' NSEKE KAFUAMA, CHARLES RIGGINS, ROBERT COLEBUNDERS, ROBERT RYDER, l JAMES CURRAN, 7 LEBUGHE IZALEY, AND THOMAS C. QUINN Project SIDA, ' Blood Bank Center, Marma Yemo Hospital, and Belgian-Zairian Cooperation, Kinshasa, Zaire; National Institnite of Allergy and Infectious Diseases, Bethesda, Maryland 208922; Cambridge Bio. Science Corporation, Worcester, Massachusetts 016054; Centers for Disease Control, Atlanta, Georgia 303337; and Institute of Tropical Medicine, Antwerp, Belgiu. M 5
References: • HENRY L. FRANCIS, MULANGA KABEYA, ' NSEKE KAFUAMA, CHARLES RIGGINS, ROBERT COLEBUNDERS, ROBERT RYDER, l JAMES CURRAN, 7 LEBUGHE IZALEY, AND THOMAS C. QUINN Project SIDA, ' Blood Bank Center, Marma Yemo Hospital, and Belgian-Zairian Cooperation, Kinshasa, Zaire; National Institnite of Allergy and Infectious Diseases, Bethesda, Maryland 208922; Cambridge Bio. Science Corporation, Worcester, Massachusetts 016054; Centers for Disease Control, Atlanta, Georgia 303337; and Institute of Tropical Medicine, Antwerp, Belgiu. M 5
 Conclusion • The latex agglutination assay is a simple, rapid assay for the detection of human immunodeficiency virus that would be useful in Third World countries or other areas where enzyme-linked immunosorbent assays are not available or cannot be used
Conclusion • The latex agglutination assay is a simple, rapid assay for the detection of human immunodeficiency virus that would be useful in Third World countries or other areas where enzyme-linked immunosorbent assays are not available or cannot be used


