a3e393fc239b7a1cb2b75f4dbe46d828.ppt
- Количество слайдов: 65
 Colorectal Cancer: New Approaches Daniel G. Haller, MD Professor of Medicine Abramson Cancer Center at the University of Pennsylvania Philadelphia, PA USA St. Louis, MO 10/1/04
Colorectal Cancer: New Approaches Daniel G. Haller, MD Professor of Medicine Abramson Cancer Center at the University of Pennsylvania Philadelphia, PA USA St. Louis, MO 10/1/04
 Incidence of colorectal cancer in the U. S. and Western Europe 2003 (n~300, 000) Stage I 24% Stage IV 22% Initially eligible for systemic therapy n~60, 000 Stage II 26% Stage III 29% Eligible for adjuvant chemotherapy n~160, 000 (55%)
Incidence of colorectal cancer in the U. S. and Western Europe 2003 (n~300, 000) Stage I 24% Stage IV 22% Initially eligible for systemic therapy n~60, 000 Stage II 26% Stage III 29% Eligible for adjuvant chemotherapy n~160, 000 (55%)
 Environmental Associates for Colorectal Cancer Dietary Factors: Dietary Fiber Dietary Fat Red Meat Alcohol Folate Methionine Calcium and vitamin D Non-Dietary Factors: Body Mass Index Physical Activity Smoking Aspirin Endoscopy Use
Environmental Associates for Colorectal Cancer Dietary Factors: Dietary Fiber Dietary Fat Red Meat Alcohol Folate Methionine Calcium and vitamin D Non-Dietary Factors: Body Mass Index Physical Activity Smoking Aspirin Endoscopy Use
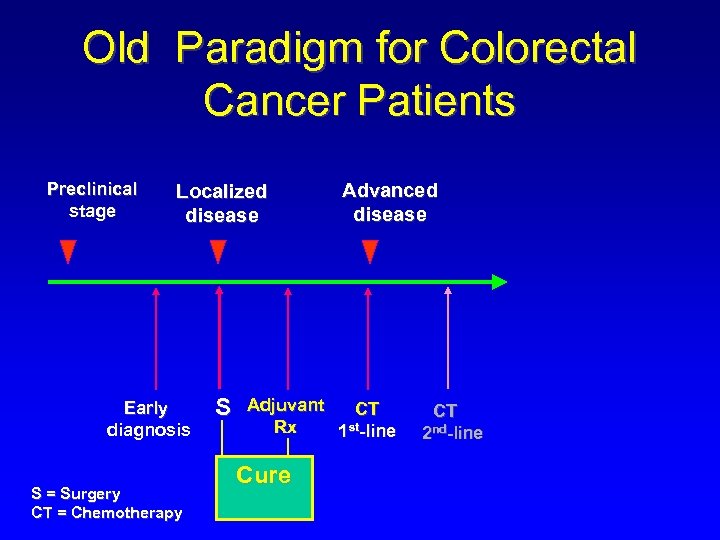 Old Paradigm for Colorectal Cancer Patients Preclinical stage Localized disease Early diagnosis S = Surgery CT = Chemotherapy S Advanced disease Adjuvant CT Rx 1 st-line Cure CT 2 nd-line
Old Paradigm for Colorectal Cancer Patients Preclinical stage Localized disease Early diagnosis S = Surgery CT = Chemotherapy S Advanced disease Adjuvant CT Rx 1 st-line Cure CT 2 nd-line
 Key Therapeutic Agents in CRC: Historical Perspective ~1960: 5 -FU is the cornerstone of first-line therapy in MCRC ~1985: Addition of LV (biochemical modulator) to 5 -FU bolus regimens 1998: Irinotecan as single agent approved as second-line in CRC 2000: Irinotecan approved as first-line in CRC in bolus regimen (IFL) 2001: Capecitabine approved as first-line in CRC 2002: Oxaliplatin approved as second-line agent 2004: Oxaliplatin approved as first-line agent 2004: Introduction of biologics – bevacizumab – cetuximab 5 -FU = 5 -fluorouracil; MCRC = metastatic CRC; LV = leucovorin; IFL = irinotecan/5 -FU/LV;
Key Therapeutic Agents in CRC: Historical Perspective ~1960: 5 -FU is the cornerstone of first-line therapy in MCRC ~1985: Addition of LV (biochemical modulator) to 5 -FU bolus regimens 1998: Irinotecan as single agent approved as second-line in CRC 2000: Irinotecan approved as first-line in CRC in bolus regimen (IFL) 2001: Capecitabine approved as first-line in CRC 2002: Oxaliplatin approved as second-line agent 2004: Oxaliplatin approved as first-line agent 2004: Introduction of biologics – bevacizumab – cetuximab 5 -FU = 5 -fluorouracil; MCRC = metastatic CRC; LV = leucovorin; IFL = irinotecan/5 -FU/LV;
 5 -Fluorouracil; 5 -FU
5 -Fluorouracil; 5 -FU
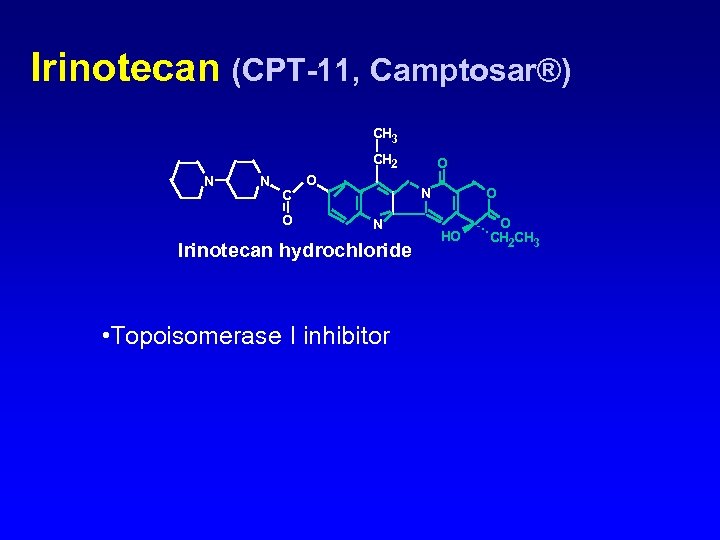 Irinotecan (CPT-11, Camptosar®) CH 3 CH 2 N N C O O O N N Irinotecan hydrochloride • Topoisomerase I inhibitor O HO O CH 2 CH 3
Irinotecan (CPT-11, Camptosar®) CH 3 CH 2 N N C O O O N N Irinotecan hydrochloride • Topoisomerase I inhibitor O HO O CH 2 CH 3
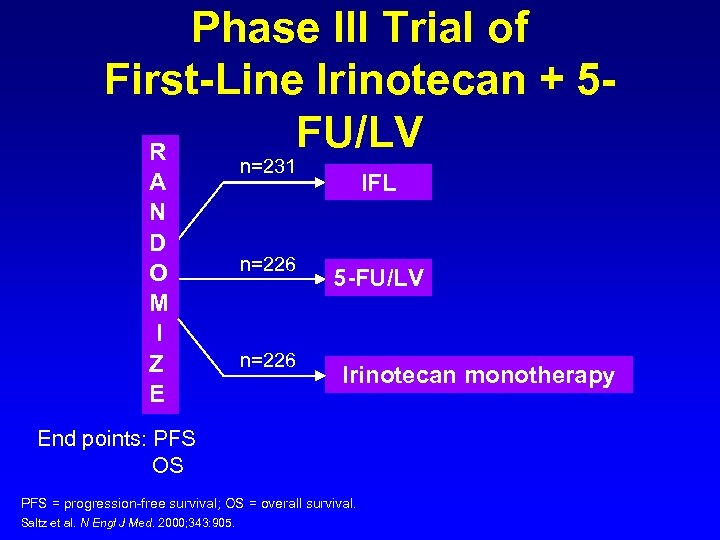 Phase III Trial of First-Line Irinotecan + 5 FU/LV R A N D O M I Z E n=231 n=226 IFL 5 -FU/LV Irinotecan monotherapy End points: PFS OS PFS = progression-free survival; OS = overall survival. Saltz et al. N Engl J Med. 2000; 343: 905.
Phase III Trial of First-Line Irinotecan + 5 FU/LV R A N D O M I Z E n=231 n=226 IFL 5 -FU/LV Irinotecan monotherapy End points: PFS OS PFS = progression-free survival; OS = overall survival. Saltz et al. N Engl J Med. 2000; 343: 905.
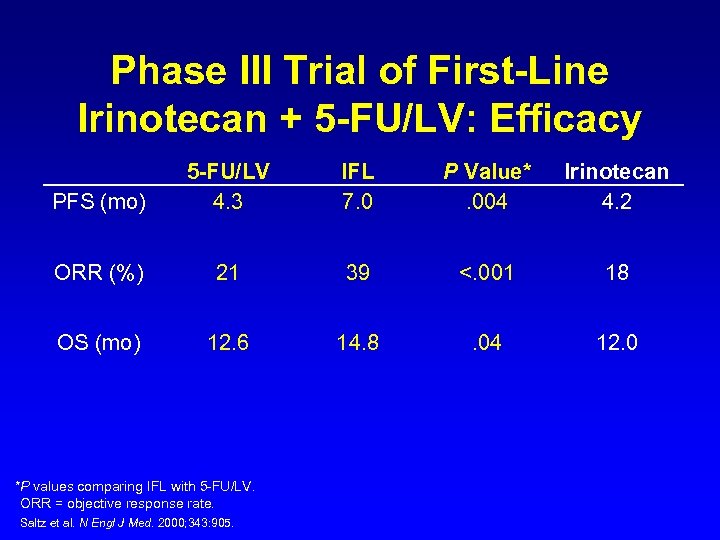 Phase III Trial of First-Line Irinotecan + 5 -FU/LV: Efficacy PFS (mo) 5 -FU/LV 4. 3 IFL 7. 0 P Value*. 004 Irinotecan 4. 2 ORR (%) 21 39 <. 001 18 OS (mo) 12. 6 14. 8 . 04 12. 0 *P values comparing IFL with 5 -FU/LV. ORR = objective response rate. Saltz et al. N Engl J Med. 2000; 343: 905.
Phase III Trial of First-Line Irinotecan + 5 -FU/LV: Efficacy PFS (mo) 5 -FU/LV 4. 3 IFL 7. 0 P Value*. 004 Irinotecan 4. 2 ORR (%) 21 39 <. 001 18 OS (mo) 12. 6 14. 8 . 04 12. 0 *P values comparing IFL with 5 -FU/LV. ORR = objective response rate. Saltz et al. N Engl J Med. 2000; 343: 905.
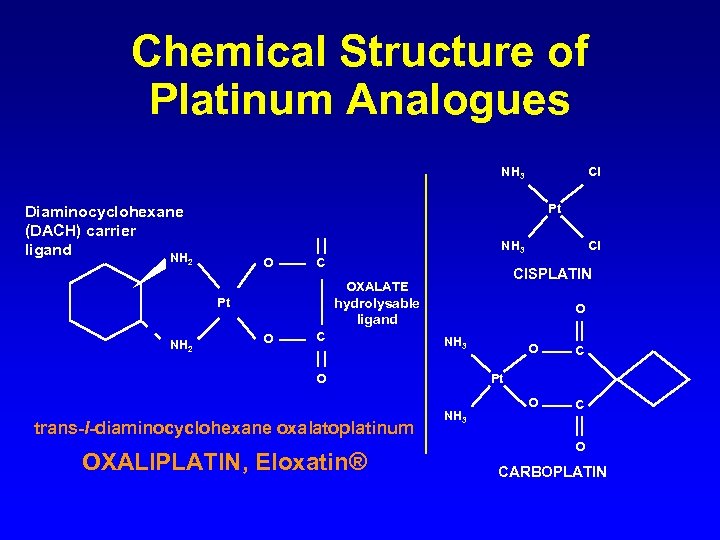 Chemical Structure of Platinum Analogues NH 3 Pt Diaminocyclohexane (DACH) carrier ligand NH NH 3 O 2 C Cl CISPLATIN OXALATE hydrolysable ligand Pt NH 2 Cl O C O NH 3 O trans-l-diaminocyclohexane oxalatoplatinum OXALIPLATIN, Eloxatin® O C Pt NH 3 O CARBOPLATIN
Chemical Structure of Platinum Analogues NH 3 Pt Diaminocyclohexane (DACH) carrier ligand NH NH 3 O 2 C Cl CISPLATIN OXALATE hydrolysable ligand Pt NH 2 Cl O C O NH 3 O trans-l-diaminocyclohexane oxalatoplatinum OXALIPLATIN, Eloxatin® O C Pt NH 3 O CARBOPLATIN
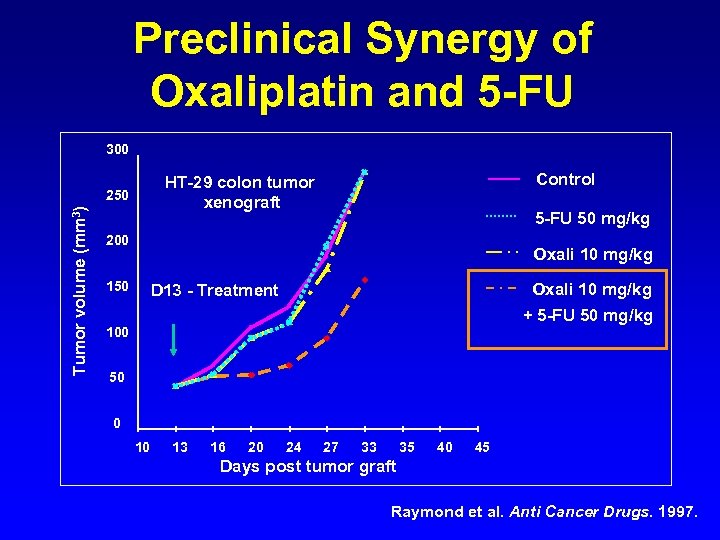 Preclinical Synergy of Oxaliplatin and 5 -FU 300 250 Tumor volume (mm 3) Control HT-29 colon tumor xenograft 5 -FU 50 mg/kg 200 Oxali 10 mg/kg 150 Oxali 10 mg/kg D 13 - Treatment + 5 -FU 50 mg/kg 100 50 0 10 13 16 20 24 27 33 35 40 45 Days post tumor graft Raymond et al. Anti Cancer Drugs. 1997.
Preclinical Synergy of Oxaliplatin and 5 -FU 300 250 Tumor volume (mm 3) Control HT-29 colon tumor xenograft 5 -FU 50 mg/kg 200 Oxali 10 mg/kg 150 Oxali 10 mg/kg D 13 - Treatment + 5 -FU 50 mg/kg 100 50 0 10 13 16 20 24 27 33 35 40 45 Days post tumor graft Raymond et al. Anti Cancer Drugs. 1997.
 Infusional 5 -FU/LV Regimens Oxaliplatin FOLFOX 4 85 400 600 x Irinotecan 400 600 LV 5 FU 2 q 2 w “Douillard” 180 400 FOLFOX 6 100 2400 x s. LV 5 FU 2 q 2 w 400 m. FOLFOX 6 85 m. FOLFOX 7 130 (85) 2400 x s. LV 5 FU 2 q 2 w 2400 x vs. LV 5 FU 2 q 2 w FOLFIRI 180
Infusional 5 -FU/LV Regimens Oxaliplatin FOLFOX 4 85 400 600 x Irinotecan 400 600 LV 5 FU 2 q 2 w “Douillard” 180 400 FOLFOX 6 100 2400 x s. LV 5 FU 2 q 2 w 400 m. FOLFOX 6 85 m. FOLFOX 7 130 (85) 2400 x s. LV 5 FU 2 q 2 w 2400 x vs. LV 5 FU 2 q 2 w FOLFIRI 180
 Oxaliplatin Second-Line Registrational Trial IFL Failures R A N D O M I Z E n=272 n=270 n=274 Infusional LV 5 FU 2 FOLFOX 4 Oxaliplatin Primary end point: OS Secondary end points: TTP, ORR, safety Rothenberg et al. JCO 6/03.
Oxaliplatin Second-Line Registrational Trial IFL Failures R A N D O M I Z E n=272 n=270 n=274 Infusional LV 5 FU 2 FOLFOX 4 Oxaliplatin Primary end point: OS Secondary end points: TTP, ORR, safety Rothenberg et al. JCO 6/03.
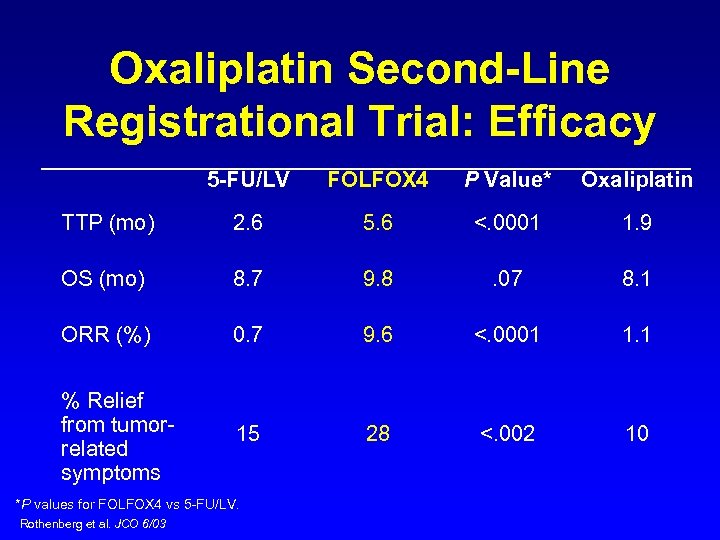 Oxaliplatin Second-Line Registrational Trial: Efficacy 5 -FU/LV FOLFOX 4 P Value* Oxaliplatin TTP (mo) 2. 6 5. 6 <. 0001 1. 9 OS (mo) 8. 7 9. 8 . 07 8. 1 ORR (%) 0. 7 9. 6 <. 0001 1. 1 % Relief from tumorrelated symptoms 15 28 <. 002 10 *P values for FOLFOX 4 vs 5 -FU/LV. Rothenberg et al. JCO 6/03
Oxaliplatin Second-Line Registrational Trial: Efficacy 5 -FU/LV FOLFOX 4 P Value* Oxaliplatin TTP (mo) 2. 6 5. 6 <. 0001 1. 9 OS (mo) 8. 7 9. 8 . 07 8. 1 ORR (%) 0. 7 9. 6 <. 0001 1. 1 % Relief from tumorrelated symptoms 15 28 <. 002 10 *P values for FOLFOX 4 vs 5 -FU/LV. Rothenberg et al. JCO 6/03
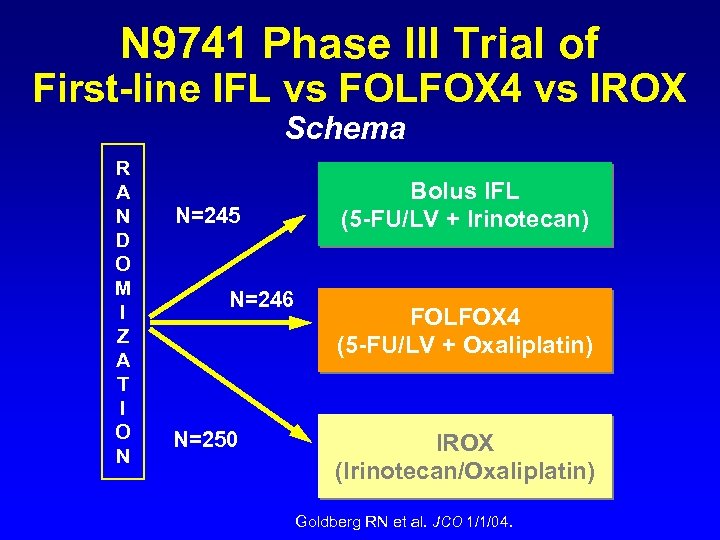 N 9741 Phase III Trial of First-line IFL vs FOLFOX 4 vs IROX Schema R A N D O M I Z A T I O N N=245 N=246 N=250 Bolus IFL (5 -FU/LV + Irinotecan) FOLFOX 4 (5 -FU/LV + Oxaliplatin) IROX (Irinotecan/Oxaliplatin) Goldberg RN et al. JCO 1/1/04.
N 9741 Phase III Trial of First-line IFL vs FOLFOX 4 vs IROX Schema R A N D O M I Z A T I O N N=245 N=246 N=250 Bolus IFL (5 -FU/LV + Irinotecan) FOLFOX 4 (5 -FU/LV + Oxaliplatin) IROX (Irinotecan/Oxaliplatin) Goldberg RN et al. JCO 1/1/04.
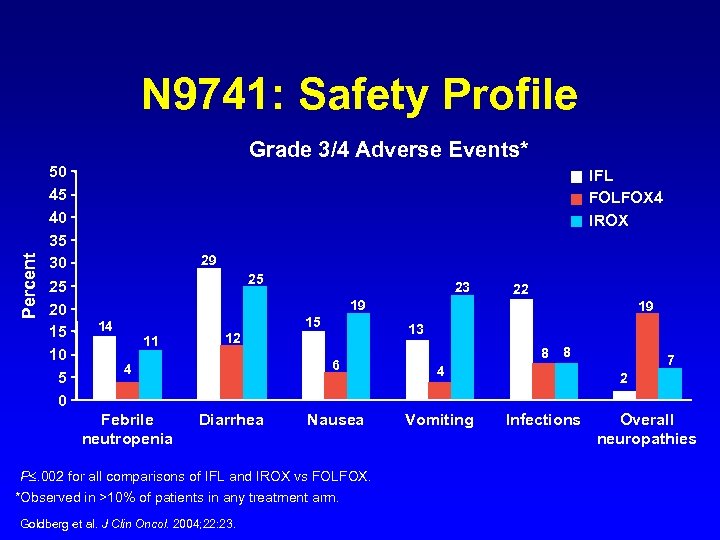 N 9741: Safety Profile Percent Grade 3/4 Adverse Events* 50 45 40 35 30 25 20 15 10 5 0 IFL FOLFOX 4 IROX 29 25 23 19 15 14 11 Febrile neutropenia 19 13 12 8 8 6 4 Diarrhea 4 Nausea Vomiting P. 002 for all comparisons of IFL and IROX vs FOLFOX. *Observed in >10% of patients in any treatment arm. Goldberg et al. J Clin Oncol. 2004; 22: 23. 22 7 2 Infections Overall neuropathies
N 9741: Safety Profile Percent Grade 3/4 Adverse Events* 50 45 40 35 30 25 20 15 10 5 0 IFL FOLFOX 4 IROX 29 25 23 19 15 14 11 Febrile neutropenia 19 13 12 8 8 6 4 Diarrhea 4 Nausea Vomiting P. 002 for all comparisons of IFL and IROX vs FOLFOX. *Observed in >10% of patients in any treatment arm. Goldberg et al. J Clin Oncol. 2004; 22: 23. 22 7 2 Infections Overall neuropathies
 N 9741 Phase III Trial of First-line IFL vs FOLFOX Results IFL FOLFOX p value 31% 45% 0. 002 Median Time to Progression (mo) 6. 9 8. 7 0. 0014 Overall Survival (mo) 14. 8 19. 5 0. 0001 RR FOLFOX: Second-line approval in US 8/02; First line approval 2/04 Goldberg RN et al. JCO 1/1/04
N 9741 Phase III Trial of First-line IFL vs FOLFOX Results IFL FOLFOX p value 31% 45% 0. 002 Median Time to Progression (mo) 6. 9 8. 7 0. 0014 Overall Survival (mo) 14. 8 19. 5 0. 0001 RR FOLFOX: Second-line approval in US 8/02; First line approval 2/04 Goldberg RN et al. JCO 1/1/04
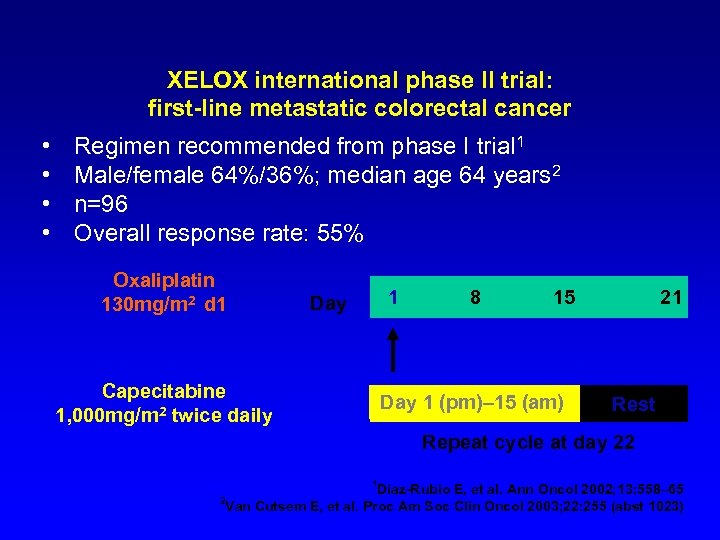 XELOX international phase II trial: first-line metastatic colorectal cancer • • Regimen recommended from phase I trial 1 Male/female 64%/36%; median age 64 years 2 n=96 Overall response rate: 55% Oxaliplatin 130 mg/m 2 d 1 1 Day Capecitabine 1, 000 mg/m 2 twice daily 8 15 Day 1 (pm)– 15 (am) 21 Rest Repeat cycle at day 22 1 Díaz-Rubio E, et al. Ann Oncol 2002; 13: 558– 65 Van Cutsem E, et al. Proc Am Soc Clin Oncol 2003; 22: 255 (abst 1023) 2
XELOX international phase II trial: first-line metastatic colorectal cancer • • Regimen recommended from phase I trial 1 Male/female 64%/36%; median age 64 years 2 n=96 Overall response rate: 55% Oxaliplatin 130 mg/m 2 d 1 1 Day Capecitabine 1, 000 mg/m 2 twice daily 8 15 Day 1 (pm)– 15 (am) 21 Rest Repeat cycle at day 22 1 Díaz-Rubio E, et al. Ann Oncol 2002; 13: 558– 65 Van Cutsem E, et al. Proc Am Soc Clin Oncol 2003; 22: 255 (abst 1023) 2
 Is There an Optimal Sequence in Therapy of Metastatic CRC? • 5 -FU, oxaliplatin, and irinotecan all have activity in first- and second-line settings • No compelling data exist to choose one sequence unless there is comorbid illness
Is There an Optimal Sequence in Therapy of Metastatic CRC? • 5 -FU, oxaliplatin, and irinotecan all have activity in first- and second-line settings • No compelling data exist to choose one sequence unless there is comorbid illness
 The Evolution of Chemotherapy for CRC
The Evolution of Chemotherapy for CRC
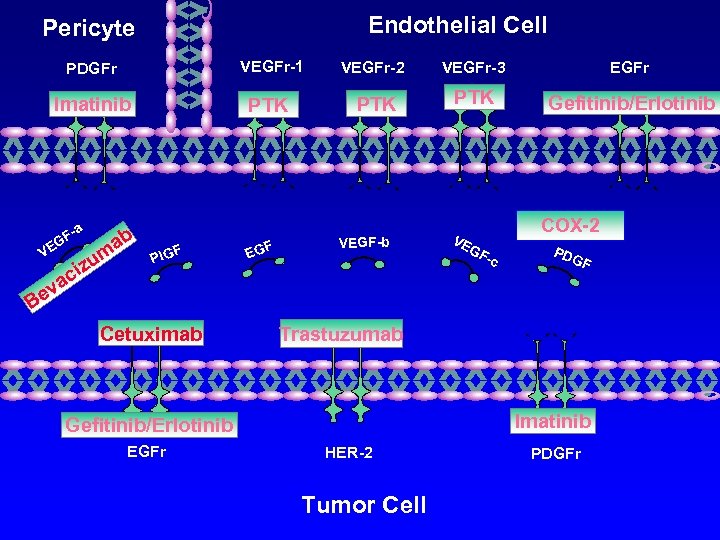 Endothelial Cell Pericyte PDGFr VEGFr-1 VEGFr-2 VEGFr-3 EGFr Imatinib PTK PTK Gefitinib/Erlotinib -a F EG ab m zu ci V F Pl. G F EG VEGF-b COX-2 VE GF -c PD GF a ev B Cetuximab Trastuzumab Imatinib Gefitinib/Erlotinib EGFr HER-2 Tumor Cell PDGFr
Endothelial Cell Pericyte PDGFr VEGFr-1 VEGFr-2 VEGFr-3 EGFr Imatinib PTK PTK Gefitinib/Erlotinib -a F EG ab m zu ci V F Pl. G F EG VEGF-b COX-2 VE GF -c PD GF a ev B Cetuximab Trastuzumab Imatinib Gefitinib/Erlotinib EGFr HER-2 Tumor Cell PDGFr
 Bevacizumab (Avastin™) • A recombinant humanized anti-VEGF MAb – human Ig. G 1 framework with antigen-binding regions from a murine MAb • Binds all forms of VEGF and prevents receptor binding • Effectively depletes circulating VEGF • Targets angiogenic components of tumor, stroma, and endothelial cells – Effect on drug delivery • Terminal half-life, 17 -21 days
Bevacizumab (Avastin™) • A recombinant humanized anti-VEGF MAb – human Ig. G 1 framework with antigen-binding regions from a murine MAb • Binds all forms of VEGF and prevents receptor binding • Effectively depletes circulating VEGF • Targets angiogenic components of tumor, stroma, and endothelial cells – Effect on drug delivery • Terminal half-life, 17 -21 days
 Phase III Study in Metastatic CRC: 5 -FU/LV/CPT-11 +/- bevacizumab • Previously untreated metastatic colorectal cancer • n=900 Previously untreated metastatic CRC Randomization 5 -FU/LV/CPT-11 (Saltz regimen) 5 -FU/LV/CPT-11/ bevacizumab Primary endpoint: survival 5 -FU/LV/ bevacizumab
Phase III Study in Metastatic CRC: 5 -FU/LV/CPT-11 +/- bevacizumab • Previously untreated metastatic colorectal cancer • n=900 Previously untreated metastatic CRC Randomization 5 -FU/LV/CPT-11 (Saltz regimen) 5 -FU/LV/CPT-11/ bevacizumab Primary endpoint: survival 5 -FU/LV/ bevacizumab
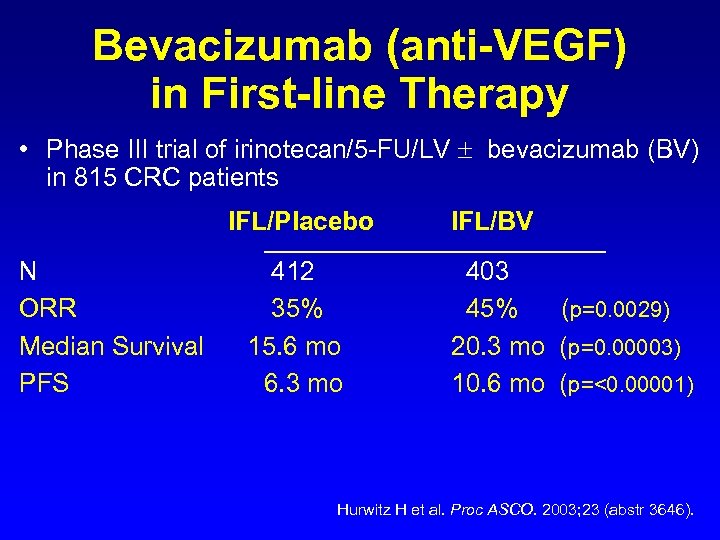 Bevacizumab (anti-VEGF) in First-line Therapy • Phase III trial of irinotecan/5 -FU/LV bevacizumab (BV) in 815 CRC patients IFL/Placebo N ORR Median Survival PFS 412 35% 15. 6 mo 6. 3 mo IFL/BV 403 45% (p=0. 0029) 20. 3 mo (p=0. 00003) 10. 6 mo (p=<0. 00001) Hurwitz H et al. Proc ASCO. 2003; 23 (abstr 3646).
Bevacizumab (anti-VEGF) in First-line Therapy • Phase III trial of irinotecan/5 -FU/LV bevacizumab (BV) in 815 CRC patients IFL/Placebo N ORR Median Survival PFS 412 35% 15. 6 mo 6. 3 mo IFL/BV 403 45% (p=0. 0029) 20. 3 mo (p=0. 00003) 10. 6 mo (p=<0. 00001) Hurwitz H et al. Proc ASCO. 2003; 23 (abstr 3646).
 Bevacizumab • Approved by FDA 2. 26. 04 • 1 st line therapy with any IV 5 -FU-based chemotherapy • 5 mg/kg Q 2 W dose • Safety – Hypertension – Perforation – Cardiovascular events
Bevacizumab • Approved by FDA 2. 26. 04 • 1 st line therapy with any IV 5 -FU-based chemotherapy • 5 mg/kg Q 2 W dose • Safety – Hypertension – Perforation – Cardiovascular events
 Cetuximab (C 225, Erbitux™) • Chimeric monoclonal antibody to EGFR – ABX-EGF: fully humanized • Inhibits EGFR function and downstream signal transduction pathways, promoting apoptosis • Synergistic with chemotherapy and radiation O‘Dwyer PJ, Benson AB III. Semin Oncol. 2002; 29(suppl 14): 10.
Cetuximab (C 225, Erbitux™) • Chimeric monoclonal antibody to EGFR – ABX-EGF: fully humanized • Inhibits EGFR function and downstream signal transduction pathways, promoting apoptosis • Synergistic with chemotherapy and radiation O‘Dwyer PJ, Benson AB III. Semin Oncol. 2002; 29(suppl 14): 10.
 “BOND” Trial • Randomized Phase II trial in CPT 11 -refractory CRC • Cetuximab+CPT-11 versus Cetuximab alone • 2: 1 randomization, 300 pts • 1 o endpoint: response rate
“BOND” Trial • Randomized Phase II trial in CPT 11 -refractory CRC • Cetuximab+CPT-11 versus Cetuximab alone • 2: 1 randomization, 300 pts • 1 o endpoint: response rate
 Cetuximab in Irinotecan. Refractory EGFR+ Patients Cetuximab + (n=111) Irinotecan (n=218) PR 11% 23% TTP 1. 5 mo 4. 1 mo Overall Survival 6. 9 mo 8. 6 mo Cunningham D et al. NEJM 7/04 Van Laethem JL et al. Proc ASCO. 2003; 23 (abstr 1058).
Cetuximab in Irinotecan. Refractory EGFR+ Patients Cetuximab + (n=111) Irinotecan (n=218) PR 11% 23% TTP 1. 5 mo 4. 1 mo Overall Survival 6. 9 mo 8. 6 mo Cunningham D et al. NEJM 7/04 Van Laethem JL et al. Proc ASCO. 2003; 23 (abstr 1058).
 Cetuximab Approval • 2/04 • For CPT-11 failures, in combination with CPT-11 • Monotherapy, for CPT-11 “intolerant” patients • EGFR status known ? ? ? • Role in 1 st line therapy?
Cetuximab Approval • 2/04 • For CPT-11 failures, in combination with CPT-11 • Monotherapy, for CPT-11 “intolerant” patients • EGFR status known ? ? ? • Role in 1 st line therapy?
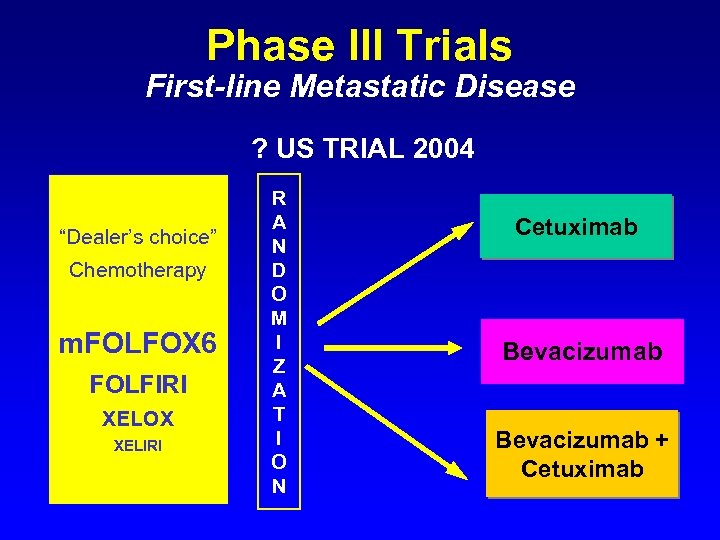 Phase III Trials First-line Metastatic Disease ? US TRIAL 2004 “Dealer’s choice” Chemotherapy m. FOLFOX 6 FOLFIRI XELOX XELIRI R A N D O M I Z A T I O N Cetuximab Bevacizumab + Cetuximab
Phase III Trials First-line Metastatic Disease ? US TRIAL 2004 “Dealer’s choice” Chemotherapy m. FOLFOX 6 FOLFIRI XELOX XELIRI R A N D O M I Z A T I O N Cetuximab Bevacizumab + Cetuximab
 The future of treatment of colon cancer • Cost: Shrag, NEJM, 7/2004 – Average patient with 1 st line FOLFOX+ bevacizumab (8 months) followed by CPT 11 + cetuximab (4 months)= $161, 000 USD
The future of treatment of colon cancer • Cost: Shrag, NEJM, 7/2004 – Average patient with 1 st line FOLFOX+ bevacizumab (8 months) followed by CPT 11 + cetuximab (4 months)= $161, 000 USD
 Adjuvant Therapy of Colon Cancer 1990 5 -FU/lev better than surgery alone 1994 5 -FU/LV better than surgery alone 1998 5 -FU/LV better than 5 -FU/lev 1998 6 months = 12 months 1998 Levamisole unnecessary 1998 HDLV = LDLV 1998 Weekly = monthly 2002 LV 5 FU 2 = monthly bolus
Adjuvant Therapy of Colon Cancer 1990 5 -FU/lev better than surgery alone 1994 5 -FU/LV better than surgery alone 1998 5 -FU/LV better than 5 -FU/lev 1998 6 months = 12 months 1998 Levamisole unnecessary 1998 HDLV = LDLV 1998 Weekly = monthly 2002 LV 5 FU 2 = monthly bolus
 Adjuvant Therapy of Colon Cancer 1990 5 -FU/lev better than surgery alone 1994 5 -FU/LV better than surgery alone 1998 5 -FU/LV better than 5 -FU/lev 1998 6 months = 12 months 1998 Levamisole unnecessary 1998 HDLV = LDLV 1998 Weekly = monthly 2002 LV 5 FU 2 = monthly bolus
Adjuvant Therapy of Colon Cancer 1990 5 -FU/lev better than surgery alone 1994 5 -FU/LV better than surgery alone 1998 5 -FU/LV better than 5 -FU/lev 1998 6 months = 12 months 1998 Levamisole unnecessary 1998 HDLV = LDLV 1998 Weekly = monthly 2002 LV 5 FU 2 = monthly bolus
 CRC: AJCC 6 th Edition Staging Guidelines • The AJCC 6 th edition staging manual refined stages II and III of the TNM system: – Smooth nodules in fat are considered LNs – Stage II divided into IIA (T 3) and IIB (T 4) – Stage III divided into • IIIA (T 1 -2 N 1 M 0) • IIIB (T 3 -4 N 1 M 0) • IIIC (TAny. N 2 M 0) – N 2 denotes metastases to 4 or more regional lymph nodes American Joint Committee on Cancer (AJCC) Cancer Staging Manual, 6 th Edition (2002).
CRC: AJCC 6 th Edition Staging Guidelines • The AJCC 6 th edition staging manual refined stages II and III of the TNM system: – Smooth nodules in fat are considered LNs – Stage II divided into IIA (T 3) and IIB (T 4) – Stage III divided into • IIIA (T 1 -2 N 1 M 0) • IIIB (T 3 -4 N 1 M 0) • IIIC (TAny. N 2 M 0) – N 2 denotes metastases to 4 or more regional lymph nodes American Joint Committee on Cancer (AJCC) Cancer Staging Manual, 6 th Edition (2002).
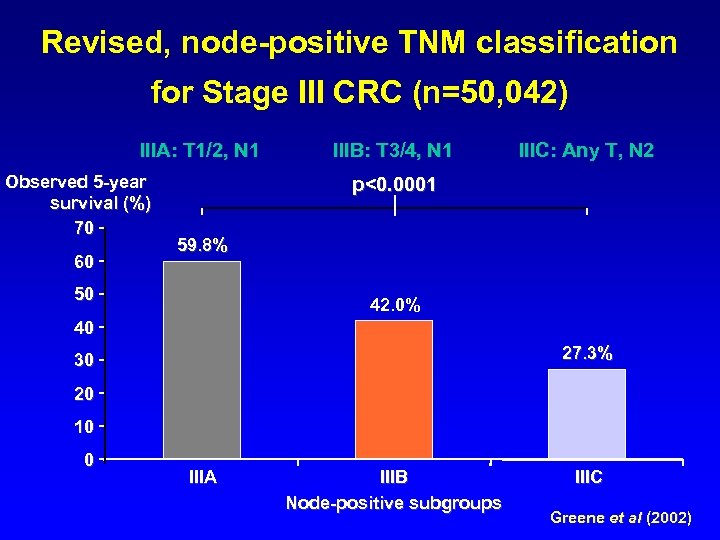 Revised, node-positive TNM classification for Stage III CRC (n=50, 042) IIIA: T 1/2, N 1 Observed 5 -year survival (%) 70 60 IIIB: T 3/4, N 1 IIIC: Any T, N 2 p<0. 0001 59. 8% 50 42. 0% 40 27. 3% 30 20 10 0 IIIA IIIB Node-positive subgroups IIIC Greene et al (2002)
Revised, node-positive TNM classification for Stage III CRC (n=50, 042) IIIA: T 1/2, N 1 Observed 5 -year survival (%) 70 60 IIIB: T 3/4, N 1 IIIC: Any T, N 2 p<0. 0001 59. 8% 50 42. 0% 40 27. 3% 30 20 10 0 IIIA IIIB Node-positive subgroups IIIC Greene et al (2002)
 Model-Derived Estimates of 5 year DFS (%) with Surgery plus Adjuvant Therapy
Model-Derived Estimates of 5 year DFS (%) with Surgery plus Adjuvant Therapy
 Adjuvant Therapy for Stage II Colon Cancer , ? CPT-11 Oxalii FU/LV Surgery
Adjuvant Therapy for Stage II Colon Cancer , ? CPT-11 Oxalii FU/LV Surgery
 Adjuvant Therapy for Stage II Colon Cancer: Cancer Care Ontario Metaanalysis • Data: 62 randomized trials; 11 metaanalysis – Primary analysis based on subset of 3586 pts in whom deaths in pts with stage II disease were provided – OR: 0. 82 (95% CI 0. 67 -1. 01; p=0. 07) • ASCO Guidelines : 8/15/04 JCO
Adjuvant Therapy for Stage II Colon Cancer: Cancer Care Ontario Metaanalysis • Data: 62 randomized trials; 11 metaanalysis – Primary analysis based on subset of 3586 pts in whom deaths in pts with stage II disease were provided – OR: 0. 82 (95% CI 0. 67 -1. 01; p=0. 07) • ASCO Guidelines : 8/15/04 JCO
 Adjuvant Therapy for Stage III Colon Cancer ? ? ? CPT-11 OXALI FU/LV Surgery
Adjuvant Therapy for Stage III Colon Cancer ? ? ? CPT-11 OXALI FU/LV Surgery
 Extending Benefit in Stage II/III colon cancer • Do combination therapies offer advantages over 5 -FU alone? – oxaliplatin-based regimens: MOSAIC, NSABP C 0 -7 – irinotecan-based regimens: CALGB, PETACC-3/V-307, FFCD/FNCLCC • Can convenience of administration be improved? – replace 5 -FU with capecitabine • Can we further improve results? The role of biologics: – anti-EGFR (cetuximab) – anti-VEGF (bevacizumab)
Extending Benefit in Stage II/III colon cancer • Do combination therapies offer advantages over 5 -FU alone? – oxaliplatin-based regimens: MOSAIC, NSABP C 0 -7 – irinotecan-based regimens: CALGB, PETACC-3/V-307, FFCD/FNCLCC • Can convenience of administration be improved? – replace 5 -FU with capecitabine • Can we further improve results? The role of biologics: – anti-EGFR (cetuximab) – anti-VEGF (bevacizumab)
 MOSAIC: Treatment arms FOLFOX 4: LV 5 FU 2 + Oxaliplatin 85 mg/m² R LV 5 FU 2 Endpoints n n Primary: – 3 -yr Disease Free Survival (DFS) Secondary: – Safety (including long-term) – Overall Survival (OS)
MOSAIC: Treatment arms FOLFOX 4: LV 5 FU 2 + Oxaliplatin 85 mg/m² R LV 5 FU 2 Endpoints n n Primary: – 3 -yr Disease Free Survival (DFS) Secondary: – Safety (including long-term) – Overall Survival (OS)
 DFS by treatment arm (ITT) Probability FOLFOX 4 (n=1123) LV 5 FU 2 (n=1123) Hazard ratio: 0. 77 [0. 65 – 0. 92] 3 -year 77. 8% 72. 9% p < 0. 01 DFS (months) 23% risk reduction in the FOLFOX 4 arm
DFS by treatment arm (ITT) Probability FOLFOX 4 (n=1123) LV 5 FU 2 (n=1123) Hazard ratio: 0. 77 [0. 65 – 0. 92] 3 -year 77. 8% 72. 9% p < 0. 01 DFS (months) 23% risk reduction in the FOLFOX 4 arm
 Probability Disease-Free Survival Stage III patients FOLFOX 4 (n=672) LV 5 FU 2 (n=675) Hazard ratio: 0. 76 [0. 62 -0. 92] DFS (months) 24% risk reduction for stage III patients in the FOLFOX 4 arm 3 -year 71. 8% 65. 5%
Probability Disease-Free Survival Stage III patients FOLFOX 4 (n=672) LV 5 FU 2 (n=675) Hazard ratio: 0. 76 [0. 62 -0. 92] DFS (months) 24% risk reduction for stage III patients in the FOLFOX 4 arm 3 -year 71. 8% 65. 5%
 3 Year DFS vs 5 Year OS: Approved by ODAC 5 yr OS= 0. 0002+0. 998*3 yr DFS
3 Year DFS vs 5 Year OS: Approved by ODAC 5 yr OS= 0. 0002+0. 998*3 yr DFS
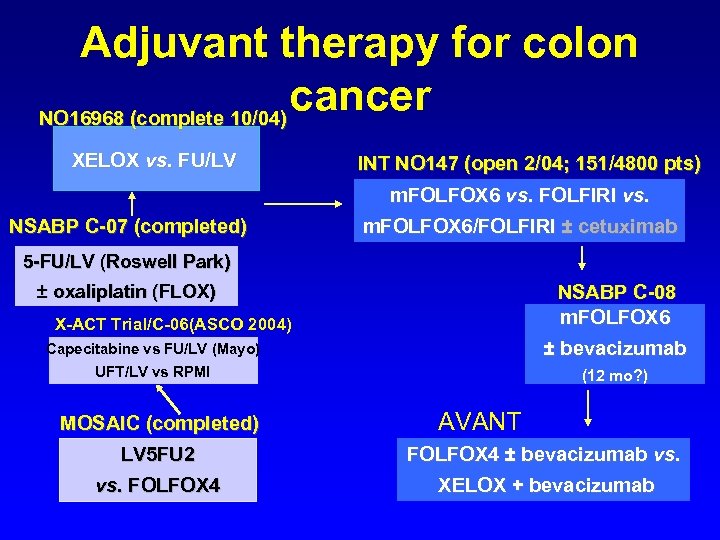 Adjuvant therapy for colon cancer NO 16968 (complete 10/04) XELOX vs. FU/LV INT NO 147 (open 2/04; 151/4800 pts) m. FOLFOX 6 vs. FOLFIRI vs. NSABP C-07 (completed) m. FOLFOX 6/FOLFIRI ± cetuximab 5 -FU/LV (Roswell Park) NSABP C-08 m. FOLFOX 6 ± oxaliplatin (FLOX) X-ACT Trial/C-06(ASCO 2004) Capecitabine vs FU/LV (Mayo) ± bevacizumab UFT/LV vs RPMI (12 mo? ) MOSAIC (completed) AVANT LV 5 FU 2 FOLFOX 4 ± bevacizumab vs. FOLFOX 4 XELOX + bevacizumab
Adjuvant therapy for colon cancer NO 16968 (complete 10/04) XELOX vs. FU/LV INT NO 147 (open 2/04; 151/4800 pts) m. FOLFOX 6 vs. FOLFIRI vs. NSABP C-07 (completed) m. FOLFOX 6/FOLFIRI ± cetuximab 5 -FU/LV (Roswell Park) NSABP C-08 m. FOLFOX 6 ± oxaliplatin (FLOX) X-ACT Trial/C-06(ASCO 2004) Capecitabine vs FU/LV (Mayo) ± bevacizumab UFT/LV vs RPMI (12 mo? ) MOSAIC (completed) AVANT LV 5 FU 2 FOLFOX 4 ± bevacizumab vs. FOLFOX 4 XELOX + bevacizumab
 Surveillance • Chau, et. al. ECCO 2003 • 550 pts with st II and III CRC treated with bolus or PVI; median f/U • CEA each visit, CT C/A/P at 12 and 24 months; non-randomized • 154 pts detected by – Sx (65), CEA (31), CT (49) • OS better in pts who had CT-detected tumor • 33 pts proceeded to potentially curative surgery
Surveillance • Chau, et. al. ECCO 2003 • 550 pts with st II and III CRC treated with bolus or PVI; median f/U • CEA each visit, CT C/A/P at 12 and 24 months; non-randomized • 154 pts detected by – Sx (65), CEA (31), CT (49) • OS better in pts who had CT-detected tumor • 33 pts proceeded to potentially curative surgery
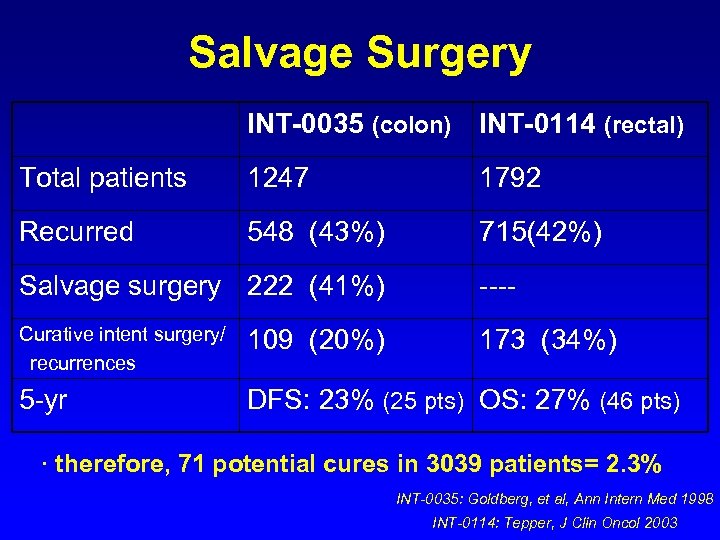 Salvage Surgery INT-0035 (colon) INT-0114 (rectal) Total patients 1247 1792 Recurred 548 (43%) 715(42%) Salvage surgery 222 (41%) ---- Curative intent surgery/ recurrences 109 (20%) 173 (34%) 5 -yr DFS: 23% (25 pts) OS: 27% (46 pts) · therefore, 71 potential cures in 3039 patients= 2. 3% INT-0035: Goldberg, et al, Ann Intern Med 1998 INT-0114: Tepper, J Clin Oncol 2003
Salvage Surgery INT-0035 (colon) INT-0114 (rectal) Total patients 1247 1792 Recurred 548 (43%) 715(42%) Salvage surgery 222 (41%) ---- Curative intent surgery/ recurrences 109 (20%) 173 (34%) 5 -yr DFS: 23% (25 pts) OS: 27% (46 pts) · therefore, 71 potential cures in 3039 patients= 2. 3% INT-0035: Goldberg, et al, Ann Intern Med 1998 INT-0114: Tepper, J Clin Oncol 2003
 Liver metastases from colorectal cancer n. Liver is the most common site of metastases from CRC n - 50 to 75% of patients with advanced CRC will develop liver metastases (1) n - 15 to 25% of patients have liver metastases at presentation (1, 2) n - 20 to 35% of patients will have metastatic disease confined to the liver (3) n. Improving the outlook of advanced colorectal cancer necessitates better management of liver metastases • 1 - N. Kemeny, F. Fata, J. Hepatobiliary Pancreas Surg. , 1999; 6: 39 -49 2 - JK. Seifert, J. R. Coll. Surg. Edinb. , 1998; 43: 141 -54 3 - MM. Borner, Ann. Oncol. , 1999; 10, 6: 623 -26
Liver metastases from colorectal cancer n. Liver is the most common site of metastases from CRC n - 50 to 75% of patients with advanced CRC will develop liver metastases (1) n - 15 to 25% of patients have liver metastases at presentation (1, 2) n - 20 to 35% of patients will have metastatic disease confined to the liver (3) n. Improving the outlook of advanced colorectal cancer necessitates better management of liver metastases • 1 - N. Kemeny, F. Fata, J. Hepatobiliary Pancreas Surg. , 1999; 6: 39 -49 2 - JK. Seifert, J. R. Coll. Surg. Edinb. , 1998; 43: 141 -54 3 - MM. Borner, Ann. Oncol. , 1999; 10, 6: 623 -26
 Advances in surgery of liver metastases Scheele, 1995 (1) Period 1960 -1979 1980 -1992 52 382 114 11. 5% 3. 4% 1. 8% 1980 -1986 1987 -1992 78 130 Operative mortality 5. 1% 0. 8% Operative morbidity 53% 24% 450 ml 150 ml N. of patients Operative mortality Doci, 1995 (2) Period N. of patients Transfusions 1 - J. Scheele, et al. , World J. Surg. , 1995; 19: 59 -71 2 - R. Doci, et al. , Br. J. Surgery, 1995; 82: 377 -81 1989 -1992
Advances in surgery of liver metastases Scheele, 1995 (1) Period 1960 -1979 1980 -1992 52 382 114 11. 5% 3. 4% 1. 8% 1980 -1986 1987 -1992 78 130 Operative mortality 5. 1% 0. 8% Operative morbidity 53% 24% 450 ml 150 ml N. of patients Operative mortality Doci, 1995 (2) Period N. of patients Transfusions 1 - J. Scheele, et al. , World J. Surg. , 1995; 19: 59 -71 2 - R. Doci, et al. , Br. J. Surgery, 1995; 82: 377 -81 1989 -1992
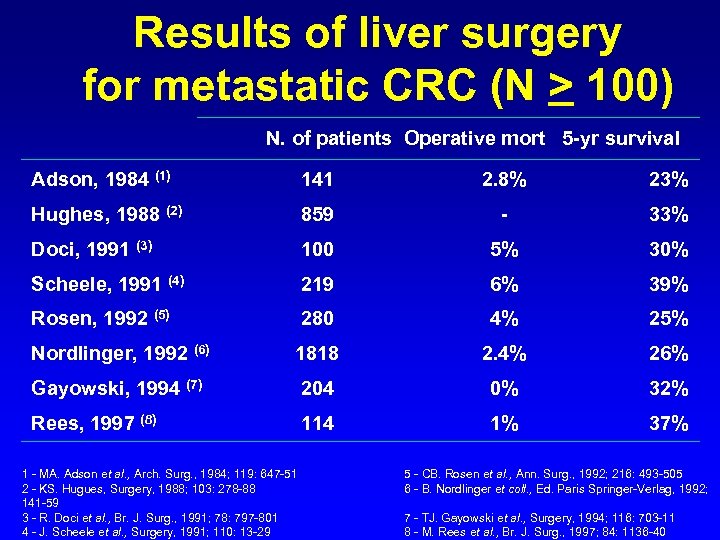 Results of liver surgery for metastatic CRC (N > 100) N. of patients Operative mort 5 -yr survival Adson, 1984 (1) 141 2. 8% 23% Hughes, 1988 (2) 859 - 33% Doci, 1991 (3) 100 5% 30% Scheele, 1991 (4) 219 6% 39% Rosen, 1992 (5) 280 4% 25% Nordlinger, 1992 (6) 1818 2. 4% 26% Gayowski, 1994 (7) 204 0% 32% Rees, 1997 (8) 114 1% 37% 1 - MA. Adson et al. , Arch. Surg. , 1984; 119: 647 -51 2 - KS. Hugues, Surgery, 1988; 103: 278 -88 141 -59 3 - R. Doci et al. , Br. J. Surg. , 1991; 78: 797 -801 4 - J. Scheele et al. , Surgery, 1991; 110: 13 -29 5 - CB. Rosen et al. , Ann. Surg. , 1992; 216: 493 -505 6 - B. Nordlinger et coll. , Ed. Paris Springer-Verlag, 1992; 7 - TJ. Gayowski et al. , Surgery, 1994; 116: 703 -11 8 - M. Rees et al. , Br. J. Surg. , 1997; 84: 1136 -40
Results of liver surgery for metastatic CRC (N > 100) N. of patients Operative mort 5 -yr survival Adson, 1984 (1) 141 2. 8% 23% Hughes, 1988 (2) 859 - 33% Doci, 1991 (3) 100 5% 30% Scheele, 1991 (4) 219 6% 39% Rosen, 1992 (5) 280 4% 25% Nordlinger, 1992 (6) 1818 2. 4% 26% Gayowski, 1994 (7) 204 0% 32% Rees, 1997 (8) 114 1% 37% 1 - MA. Adson et al. , Arch. Surg. , 1984; 119: 647 -51 2 - KS. Hugues, Surgery, 1988; 103: 278 -88 141 -59 3 - R. Doci et al. , Br. J. Surg. , 1991; 78: 797 -801 4 - J. Scheele et al. , Surgery, 1991; 110: 13 -29 5 - CB. Rosen et al. , Ann. Surg. , 1992; 216: 493 -505 6 - B. Nordlinger et coll. , Ed. Paris Springer-Verlag, 1992; 7 - TJ. Gayowski et al. , Surgery, 1994; 116: 703 -11 8 - M. Rees et al. , Br. J. Surg. , 1997; 84: 1136 -40
 Ongoing Trials in Patients With Liver Metastases • EORTC Study 40983 – FOLFOX + surgery vs surgery alone – Primary endpoint: 3 -yr RFS; also resectability – 350 patients, completed accrual (started 10/01) • CLOCC Trial – FOLFOX +/- RFA for unresectable disease • NCCTG/NSABP – Is HAI necessary with optimal SYS therapy? – Adjuvant XELOX ± HAI FUd. R
Ongoing Trials in Patients With Liver Metastases • EORTC Study 40983 – FOLFOX + surgery vs surgery alone – Primary endpoint: 3 -yr RFS; also resectability – 350 patients, completed accrual (started 10/01) • CLOCC Trial – FOLFOX +/- RFA for unresectable disease • NCCTG/NSABP – Is HAI necessary with optimal SYS therapy? – Adjuvant XELOX ± HAI FUd. R
 Rectal Cancer Portion of Rectum Upper 1/3 Middle 1/3 Lower 1/3 Left upper valve of Houston Cm. from anal verge Right middle valve of Houston 15 Peritoneum 11 Ampulla of Rectum Left lower valve of Houston 7 2 Anal verge Cohen AM, et al. Cancer: Principles & Practice of Oncology. 5 th ed. 1997, p. 1197
Rectal Cancer Portion of Rectum Upper 1/3 Middle 1/3 Lower 1/3 Left upper valve of Houston Cm. from anal verge Right middle valve of Houston 15 Peritoneum 11 Ampulla of Rectum Left lower valve of Houston 7 2 Anal verge Cohen AM, et al. Cancer: Principles & Practice of Oncology. 5 th ed. 1997, p. 1197
 Rectal Cancer • OBJECTIVES OF TREATMENT – Cure – Quality of Life • sphincter preservation vs colostomy (distal lesions) • anorectal function – acute and late toxicity of treatment
Rectal Cancer • OBJECTIVES OF TREATMENT – Cure – Quality of Life • sphincter preservation vs colostomy (distal lesions) • anorectal function – acute and late toxicity of treatment
 Management of Early Rectal Cancer • SURGERY (TME) • RADIATION – reduce locoregional failure – reduce distant failure (± chemotherapy) • CHEMOTHERAPY – reduce distant failure – reduce locoregional failure (± radiation)
Management of Early Rectal Cancer • SURGERY (TME) • RADIATION – reduce locoregional failure – reduce distant failure (± chemotherapy) • CHEMOTHERAPY – reduce distant failure – reduce locoregional failure (± radiation)
 Local failure: 1980 s– 2000 s 35 Local failure (%) 30 25 20 15 10 5 0 Surgery only Surgery RT Surgery CTRT TME + RT/CTRT
Local failure: 1980 s– 2000 s 35 Local failure (%) 30 25 20 15 10 5 0 Surgery only Surgery RT Surgery CTRT TME + RT/CTRT
 Distant metastases: 1980 s – 2000 s Distant metastases (%) 40 35 30 25 20 15 10 5 0 Surgery only Surgery RT Surgery CTRT TME +RT/CTRT
Distant metastases: 1980 s – 2000 s Distant metastases (%) 40 35 30 25 20 15 10 5 0 Surgery only Surgery RT Surgery CTRT TME +RT/CTRT
 Adjuvant Therapy of Rectal Cancer • Postoperative – pathologic staging • EUS, MRI – immediate surgery • patient bias • physician bias • referral patterns – Positive adjuvant trials in US in postoperative setting • Preoperative – improved resectability – sphincter preservation – reduced damage to normal tissue – Only positive trial from Swedish study (2500 c. Gy/5 fx)
Adjuvant Therapy of Rectal Cancer • Postoperative – pathologic staging • EUS, MRI – immediate surgery • patient bias • physician bias • referral patterns – Positive adjuvant trials in US in postoperative setting • Preoperative – improved resectability – sphincter preservation – reduced damage to normal tissue – Only positive trial from Swedish study (2500 c. Gy/5 fx)
 Pre-operative adjuvant therapy in rectal cancer: recent advances • Emphasis on curative resection in addition to sphincter preservation – – pre-operative staging (CRM unsafe) pre-operative tumor down-staging surgical technique (TME) accurate pathological staging (R 0) CRM = circumferential resection margin; TME = total mesorectal excision
Pre-operative adjuvant therapy in rectal cancer: recent advances • Emphasis on curative resection in addition to sphincter preservation – – pre-operative staging (CRM unsafe) pre-operative tumor down-staging surgical technique (TME) accurate pathological staging (R 0) CRM = circumferential resection margin; TME = total mesorectal excision
 Preoperative vs Postoperative Chemoradiation • INT-0147 – terminated prematurely secondary to low accrual • NSABP R-03 – terminated prematurely secondary to low accrual • German Trial – completed accrual!
Preoperative vs Postoperative Chemoradiation • INT-0147 – terminated prematurely secondary to low accrual • NSABP R-03 – terminated prematurely secondary to low accrual • German Trial – completed accrual!
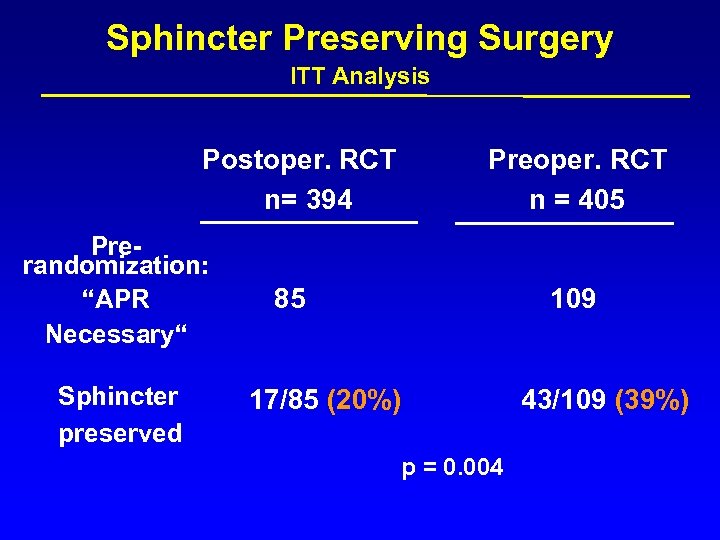 Sphincter Preserving Surgery ITT Analysis Postoper. RCT n= 394 Prerandomization: “APR Necessary“ Sphincter preserved Preoper. RCT n = 405 85 109 17/85 (20%) p = 0. 004 43/109 (39%)
Sphincter Preserving Surgery ITT Analysis Postoper. RCT n= 394 Prerandomization: “APR Necessary“ Sphincter preserved Preoper. RCT n = 405 85 109 17/85 (20%) p = 0. 004 43/109 (39%)
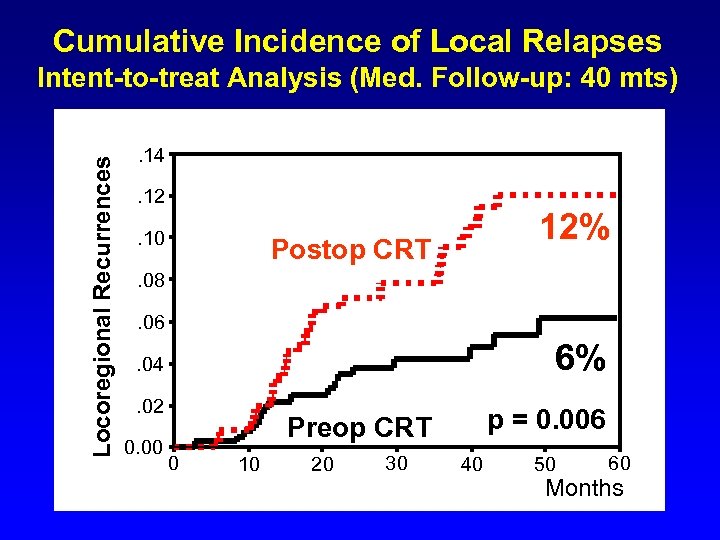 Cumulative Incidence of Local Relapses Locoregional Recurrences Intent-to-treat Analysis (Med. Follow-up: 40 mts). 14. 12. 10 12% Postop CRT . 08. 06 6% . 04. 02 0. 00 p = 0. 006 Preop CRT 0 10 20 30 40 50 60 Months
Cumulative Incidence of Local Relapses Locoregional Recurrences Intent-to-treat Analysis (Med. Follow-up: 40 mts). 14. 12. 10 12% Postop CRT . 08. 06 6% . 04. 02 0. 00 p = 0. 006 Preop CRT 0 10 20 30 40 50 60 Months
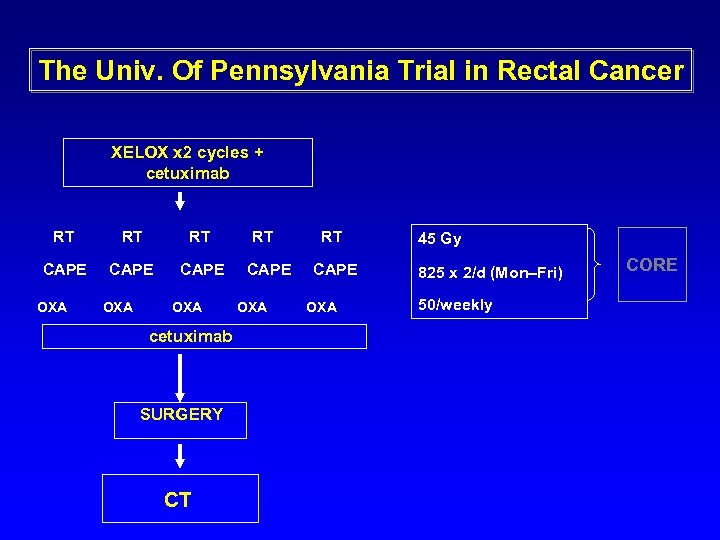 The Univ. Of Pennsylvania Trial in Rectal Cancer XELOX x 2 cycles + cetuximab RT RT RT CAPE OXA OXA cetuximab SURGERY CT RT CAPE OXA 45 Gy 825 x 2/d (Mon–Fri) 50/weekly CORE
The Univ. Of Pennsylvania Trial in Rectal Cancer XELOX x 2 cycles + cetuximab RT RT RT CAPE OXA OXA cetuximab SURGERY CT RT CAPE OXA 45 Gy 825 x 2/d (Mon–Fri) 50/weekly CORE
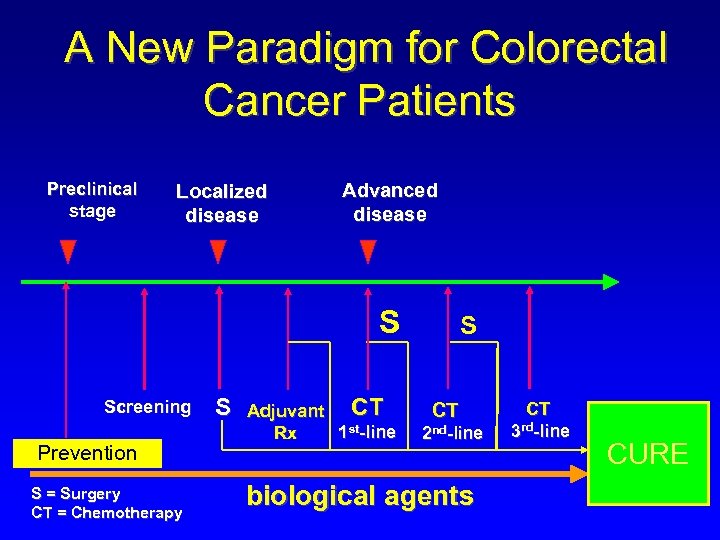 A New Paradigm for Colorectal Cancer Patients Preclinical stage Localized disease Advanced disease S Screening Prevention S = Surgery CT = Chemotherapy S CT Adjuvant 1 st-line Rx S CT 2 nd-line biological agents CT 3 rd-line CURE
A New Paradigm for Colorectal Cancer Patients Preclinical stage Localized disease Advanced disease S Screening Prevention S = Surgery CT = Chemotherapy S CT Adjuvant 1 st-line Rx S CT 2 nd-line biological agents CT 3 rd-line CURE
 The future of treatment of colon cancer • • • Improve efficacy Improve convenience Reduce duration Reduce toxicity Individualize treatment –Patient selection (anatomic staging, prognostic markers) –Drug selection (proteinomics, pharmacogenomics, SNPs) • • Cost/benefit Confusion…
The future of treatment of colon cancer • • • Improve efficacy Improve convenience Reduce duration Reduce toxicity Individualize treatment –Patient selection (anatomic staging, prognostic markers) –Drug selection (proteinomics, pharmacogenomics, SNPs) • • Cost/benefit Confusion…
 Therapy for Colorectal Cancer “There are only two tragedies in life: one is not getting what one wants, and the other is getting it. ” Oscar Wilde
Therapy for Colorectal Cancer “There are only two tragedies in life: one is not getting what one wants, and the other is getting it. ” Oscar Wilde


