d4fe2a0f1530c5f2c822db93675d1069.ppt
- Количество слайдов: 43
 Colon Cancer Chemoprevention UWCCC Chemoprevention Program And Mayo Clinic Chemoprevention Program Spirit of Eagles September 6, 2007 Howard Bailey, MD and Paul Limburg, MD, MPH
Colon Cancer Chemoprevention UWCCC Chemoprevention Program And Mayo Clinic Chemoprevention Program Spirit of Eagles September 6, 2007 Howard Bailey, MD and Paul Limburg, MD, MPH
 Colon Cancer Chemoprevention § Need/Rationale § 150, 000 expected to be diagnosed with colorectal cancer in 2007 § >50, 000 expected to die from colorectal cancer in 2007
Colon Cancer Chemoprevention § Need/Rationale § 150, 000 expected to be diagnosed with colorectal cancer in 2007 § >50, 000 expected to die from colorectal cancer in 2007
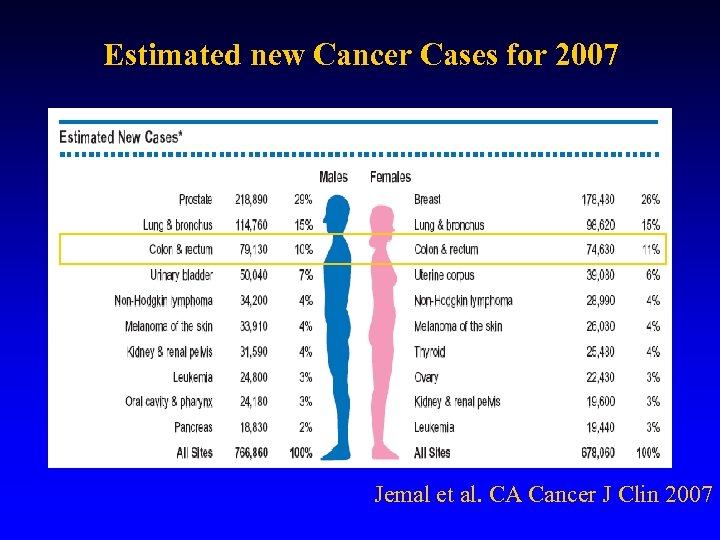 Estimated new Cancer Cases for 2007 Jemal et al. CA Cancer J Clin 2007
Estimated new Cancer Cases for 2007 Jemal et al. CA Cancer J Clin 2007
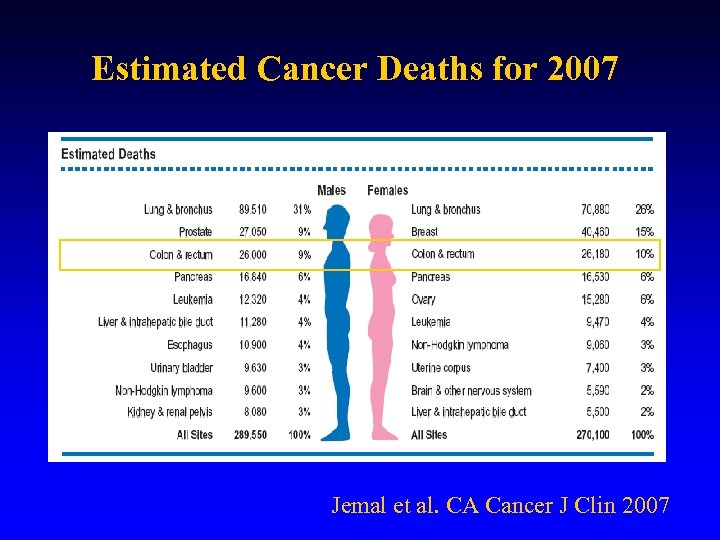 Estimated Cancer Deaths for 2007 Jemal et al. CA Cancer J Clin 2007
Estimated Cancer Deaths for 2007 Jemal et al. CA Cancer J Clin 2007
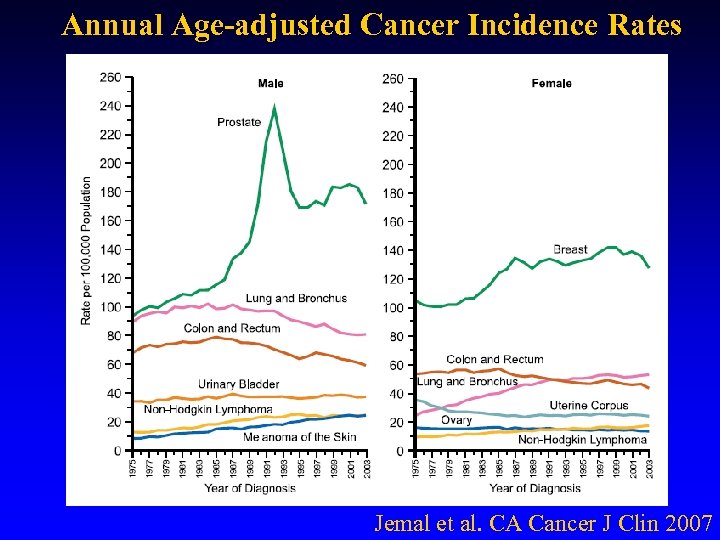 Annual Age-adjusted Cancer Incidence Rates Jemal et al. CA Cancer J Clin 2007
Annual Age-adjusted Cancer Incidence Rates Jemal et al. CA Cancer J Clin 2007
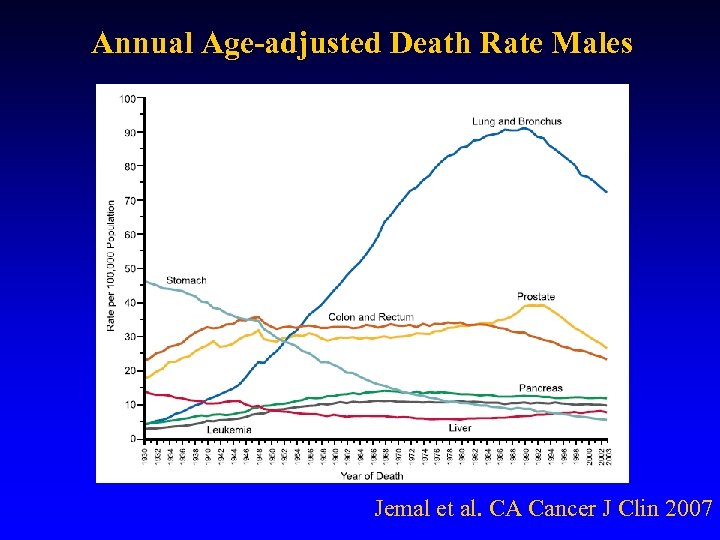 Annual Age-adjusted Death Rate Males Jemal et al. CA Cancer J Clin 2007
Annual Age-adjusted Death Rate Males Jemal et al. CA Cancer J Clin 2007
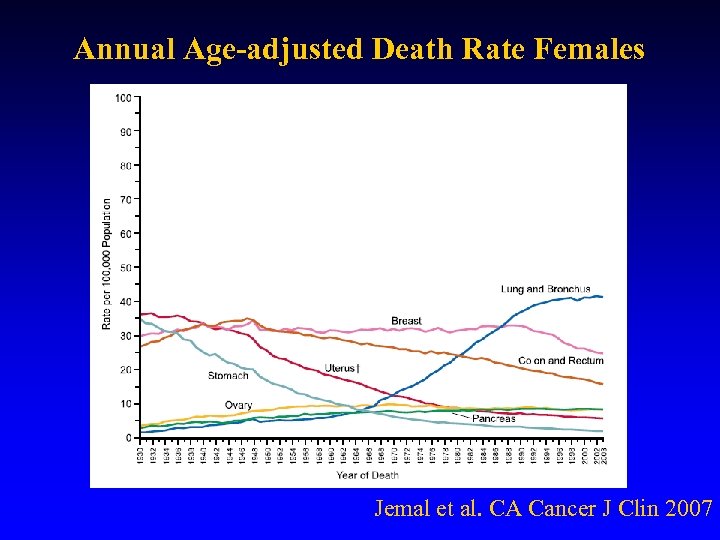 Annual Age-adjusted Death Rate Females Jemal et al. CA Cancer J Clin 2007
Annual Age-adjusted Death Rate Females Jemal et al. CA Cancer J Clin 2007
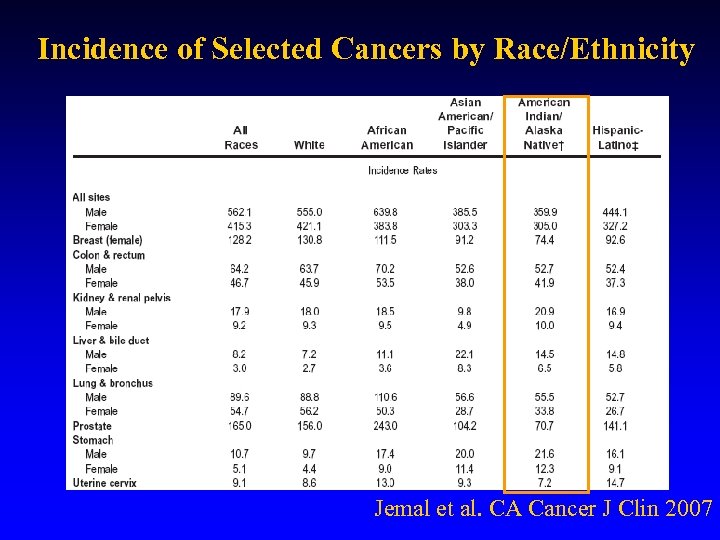 Incidence of Selected Cancers by Race/Ethnicity Jemal et al. CA Cancer J Clin 2007
Incidence of Selected Cancers by Race/Ethnicity Jemal et al. CA Cancer J Clin 2007
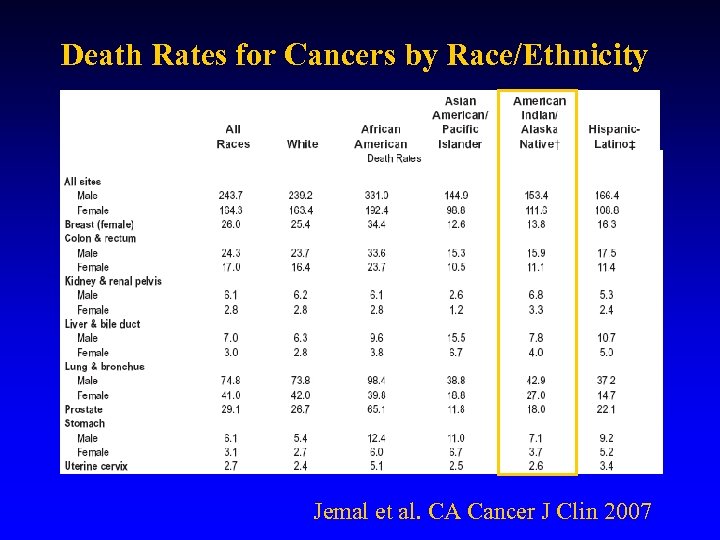 Death Rates for Cancers by Race/Ethnicity Jemal et al. CA Cancer J Clin 2007
Death Rates for Cancers by Race/Ethnicity Jemal et al. CA Cancer J Clin 2007
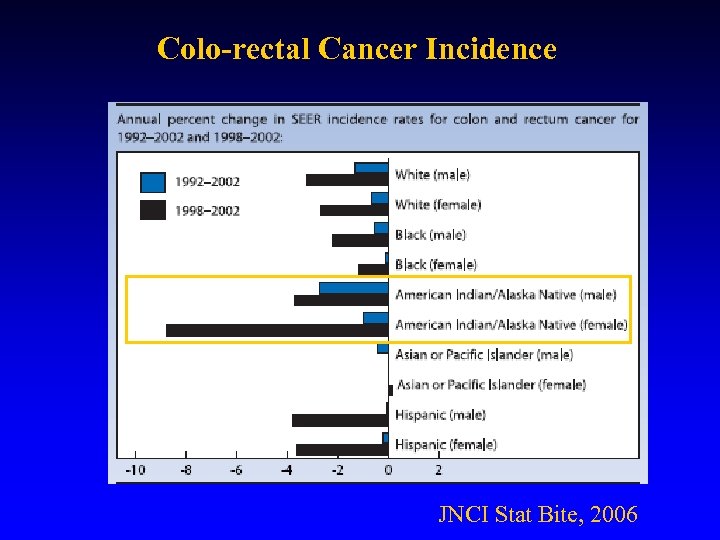 Colo-rectal Cancer Incidence JNCI Stat Bite, 2006
Colo-rectal Cancer Incidence JNCI Stat Bite, 2006
 Colon Cancer • Above data implies while colorectal cancer remains a significant health issue, it appears to be lessening including for American Indians/Alaska Natives. • Cancer mortality, in general, is less in American Indians/Alaska Natives than general population 165 compared to 201 per 100, 000; however mortality rates for Alaska Natives and Northern Plains Tribes is higher, especially for colorectal, lung, liver, stomach, gall bladder and kidney cancers.
Colon Cancer • Above data implies while colorectal cancer remains a significant health issue, it appears to be lessening including for American Indians/Alaska Natives. • Cancer mortality, in general, is less in American Indians/Alaska Natives than general population 165 compared to 201 per 100, 000; however mortality rates for Alaska Natives and Northern Plains Tribes is higher, especially for colorectal, lung, liver, stomach, gall bladder and kidney cancers.
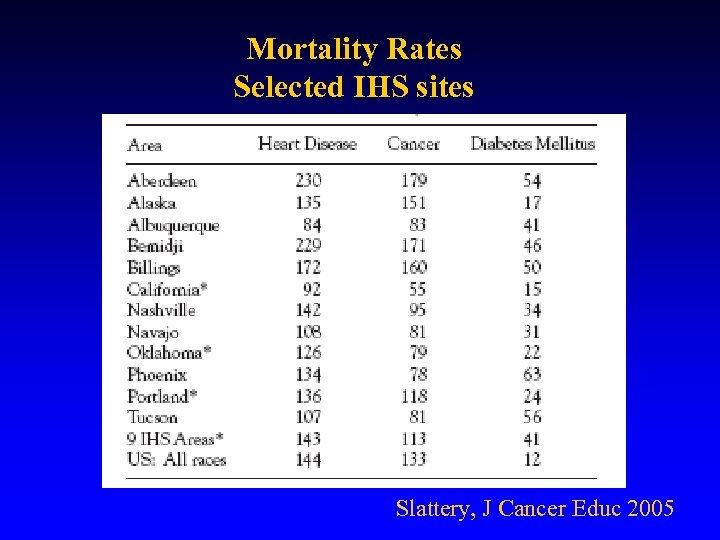 Mortality Rates Selected IHS sites Slattery, J Cancer Educ 2005
Mortality Rates Selected IHS sites Slattery, J Cancer Educ 2005
 Colon Cancer in Indian Country • Harwell et al. (Am J Prev Med 30: 493, 2006) – Age adjusted 6 -yr cancer incidence rates in Montana, 1991 -96 and 1997 -2002 – All cancers incidence rates higher in American Indians than whites – Colon cancer incidence twice as high in American Indian males as white males
Colon Cancer in Indian Country • Harwell et al. (Am J Prev Med 30: 493, 2006) – Age adjusted 6 -yr cancer incidence rates in Montana, 1991 -96 and 1997 -2002 – All cancers incidence rates higher in American Indians than whites – Colon cancer incidence twice as high in American Indian males as white males
 Colon Cancer Prevention • For the US population it remains important despite decreasing incidence and mortality • This appears especially true for Northern Plains Tribes and Alaska Natives • Many ways to accomplish this through lifestyle, screening, etc. . • Will focus on the potential of chemoprevention of colon cancer
Colon Cancer Prevention • For the US population it remains important despite decreasing incidence and mortality • This appears especially true for Northern Plains Tribes and Alaska Natives • Many ways to accomplish this through lifestyle, screening, etc. . • Will focus on the potential of chemoprevention of colon cancer
 Chemoprevention of Cancer § The study of carcinogenesis has led to the current dogma that human carcinogenesis is a multi-year process § Example – normal colonic mucosa to hyperplastic polyps to adenomatous polyps to carcinoma may take 10 -30 years § Thus providing an opportunity to intervene prior to accumulated mutations or phenotypic changes
Chemoprevention of Cancer § The study of carcinogenesis has led to the current dogma that human carcinogenesis is a multi-year process § Example – normal colonic mucosa to hyperplastic polyps to adenomatous polyps to carcinoma may take 10 -30 years § Thus providing an opportunity to intervene prior to accumulated mutations or phenotypic changes
 Candidate Agents • COX-2 inhibitors, selective or nonselective • Diet and Nutraceuticals • Antioxidants/Vitamins • Statins • Difluoromethylornithine (DFMO) • Others
Candidate Agents • COX-2 inhibitors, selective or nonselective • Diet and Nutraceuticals • Antioxidants/Vitamins • Statins • Difluoromethylornithine (DFMO) • Others
 Selective COX-2 Inhibitors • Celecoxib: FDA approved for adenomatous polyp prevention for individuals with Familial Adenomatous Polyposis – These data and retrospective data have led to extensive study of COX-2 inhibitors for sporadic adenomas as well
Selective COX-2 Inhibitors • Celecoxib: FDA approved for adenomatous polyp prevention for individuals with Familial Adenomatous Polyposis – These data and retrospective data have led to extensive study of COX-2 inhibitors for sporadic adenomas as well
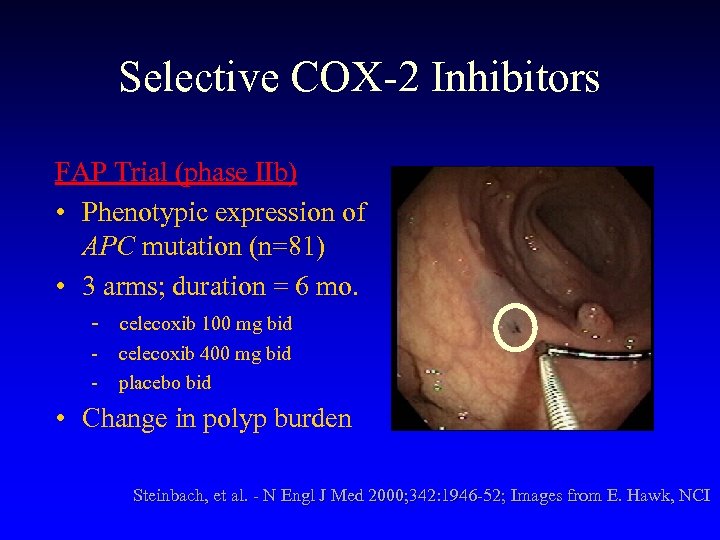 Selective COX-2 Inhibitors FAP Trial (phase IIb) • Phenotypic expression of APC mutation (n=81) • 3 arms; duration = 6 mo. - celecoxib 100 mg bid - celecoxib 400 mg bid placebo bid • Change in polyp burden Steinbach, et al. - N Engl J Med 2000; 342: 1946 -52; Images from E. Hawk, NCI
Selective COX-2 Inhibitors FAP Trial (phase IIb) • Phenotypic expression of APC mutation (n=81) • 3 arms; duration = 6 mo. - celecoxib 100 mg bid - celecoxib 400 mg bid placebo bid • Change in polyp burden Steinbach, et al. - N Engl J Med 2000; 342: 1946 -52; Images from E. Hawk, NCI
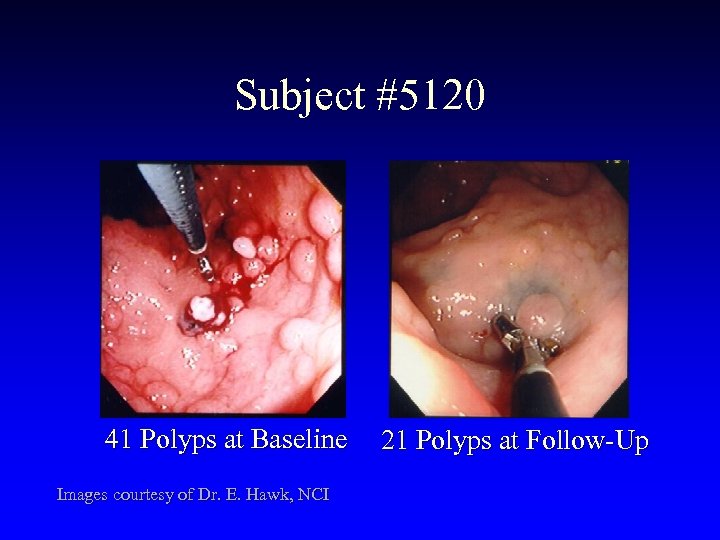 Subject #5120 41 Polyps at Baseline Images courtesy of Dr. E. Hawk, NCI 21 Polyps at Follow-Up
Subject #5120 41 Polyps at Baseline Images courtesy of Dr. E. Hawk, NCI 21 Polyps at Follow-Up
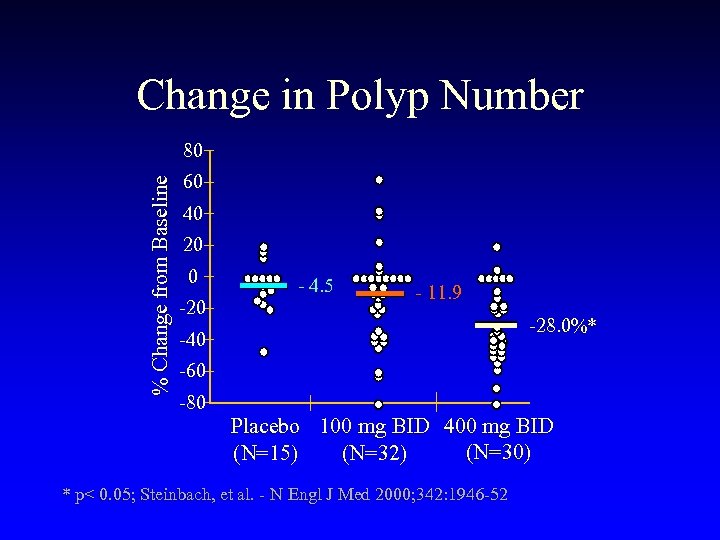 Change in Polyp Number % Change from Baseline 80 60 40 20 0 -20 - 4. 5 - 11. 9 -28. 0%* -40 -60 -80 Placebo 100 mg BID 400 mg BID (N=30) (N=32) (N=15) * p< 0. 05; Steinbach, et al. - N Engl J Med 2000; 342: 1946 -52
Change in Polyp Number % Change from Baseline 80 60 40 20 0 -20 - 4. 5 - 11. 9 -28. 0%* -40 -60 -80 Placebo 100 mg BID 400 mg BID (N=30) (N=32) (N=15) * p< 0. 05; Steinbach, et al. - N Engl J Med 2000; 342: 1946 -52
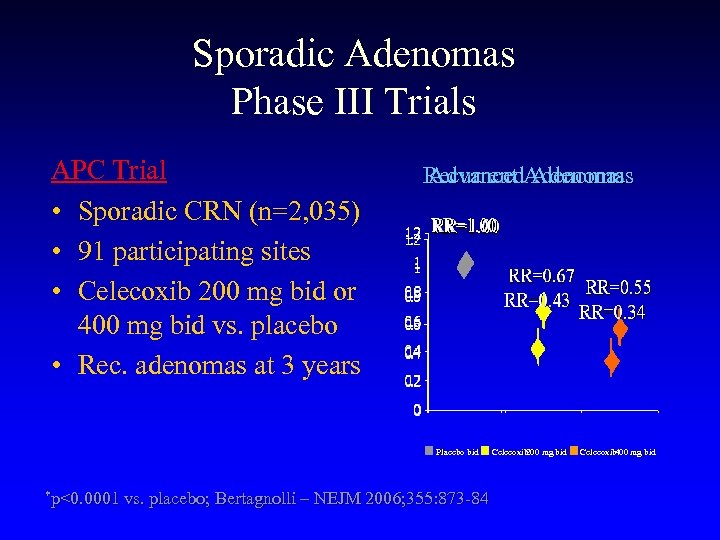 Sporadic Adenomas Phase III Trials APC Trial • Sporadic CRN (n=2, 035) • 91 participating sites • Celecoxib 200 mg bid or 400 mg bid vs. placebo • Rec. adenomas at 3 years Advanced Adenomas Recurrent Adenomas Placebo bid *p<0. 0001 vs. placebo; Bertagnolli – NEJM 2006; 355: 873 -84 Celecoxib mg bid 200 Celecoxib 400 mg bid
Sporadic Adenomas Phase III Trials APC Trial • Sporadic CRN (n=2, 035) • 91 participating sites • Celecoxib 200 mg bid or 400 mg bid vs. placebo • Rec. adenomas at 3 years Advanced Adenomas Recurrent Adenomas Placebo bid *p<0. 0001 vs. placebo; Bertagnolli – NEJM 2006; 355: 873 -84 Celecoxib mg bid 200 Celecoxib 400 mg bid
 Phase III Trials Pre. SAP Trial (n=1, 561) • 107 participating sites • Celecoxib 400 mg qd vs. placebo • Rec. adenomas at 3 years RR=0. 64 (0. 56 -0. 75); any RR=0. 49 (0. 33 -0. 73); adv. APPROVe Trial (n=2, 587) • 108 participating sites • Rofecoxib 25 mg qd vs. placebo • Rec. adenomas at 3 years RR=0. 76 (0. 69 -0. 83); any RR=0. 70 (0. 57 -0. 73); N Engl J Med 2006; 355: 885 -95 and Gastroenterol 2006; epub adv.
Phase III Trials Pre. SAP Trial (n=1, 561) • 107 participating sites • Celecoxib 400 mg qd vs. placebo • Rec. adenomas at 3 years RR=0. 64 (0. 56 -0. 75); any RR=0. 49 (0. 33 -0. 73); adv. APPROVe Trial (n=2, 587) • 108 participating sites • Rofecoxib 25 mg qd vs. placebo • Rec. adenomas at 3 years RR=0. 76 (0. 69 -0. 83); any RR=0. 70 (0. 57 -0. 73); N Engl J Med 2006; 355: 885 -95 and Gastroenterol 2006; epub adv.
 Cardiovascular Toxicity Celecoxib • APC Trial • N=2, 035 subjects • Follow-up = 2. 8 -3. 1 years • CV deaths (%): Rofecoxib • APPROVe Trial • N=2, 586 subjects • Follow-up = 3, 327 pt-years • CV Adverse events (%): –Placebo (1%); RR=1. 0 – 200 mg BID (2. 3%) ; RR=2. 3 – 400 mg BID (3. 4%) ; RR=3. 4 www. theoaklandpress. com 12/18/04 www. washingtonpost. com; ; 4/7/05 and 1092 -102 N Engl J Med. 2005; 352: 1071 -80 –Placebo (2%); RR=1. 0 – 25 mg QD (3. 6%); RR=1. 9
Cardiovascular Toxicity Celecoxib • APC Trial • N=2, 035 subjects • Follow-up = 2. 8 -3. 1 years • CV deaths (%): Rofecoxib • APPROVe Trial • N=2, 586 subjects • Follow-up = 3, 327 pt-years • CV Adverse events (%): –Placebo (1%); RR=1. 0 – 200 mg BID (2. 3%) ; RR=2. 3 – 400 mg BID (3. 4%) ; RR=3. 4 www. theoaklandpress. com 12/18/04 www. washingtonpost. com; ; 4/7/05 and 1092 -102 N Engl J Med. 2005; 352: 1071 -80 –Placebo (2%); RR=1. 0 – 25 mg QD (3. 6%); RR=1. 9
 Celecoxib and Colon Cancer Prevention • Psaty and Potter (NEJM 355: 950, 2006) – Reviewed APC and Pre. SAP trials and concluded the following – Celecoxib decreases adenoma formation – Celecoxib increases the risk of cardiovascular adverse events – The potential increase in CV event/mortality outweighs the projected decrease in colon cancer incidence
Celecoxib and Colon Cancer Prevention • Psaty and Potter (NEJM 355: 950, 2006) – Reviewed APC and Pre. SAP trials and concluded the following – Celecoxib decreases adenoma formation – Celecoxib increases the risk of cardiovascular adverse events – The potential increase in CV event/mortality outweighs the projected decrease in colon cancer incidence
 Non-selective COX-2 inhibitors • Retrospective data supporting potential preventive effects • Cardiovascular issues appear less, but remain • Aspirin § Sandler et al. NEJM 348: 883, 2003 § 635 subjects with previous colo-rectal CA randomized to ASA 325 mg/d or placebo § 1 or more adenomas in 27% of placebo vs 17% ASA
Non-selective COX-2 inhibitors • Retrospective data supporting potential preventive effects • Cardiovascular issues appear less, but remain • Aspirin § Sandler et al. NEJM 348: 883, 2003 § 635 subjects with previous colo-rectal CA randomized to ASA 325 mg/d or placebo § 1 or more adenomas in 27% of placebo vs 17% ASA
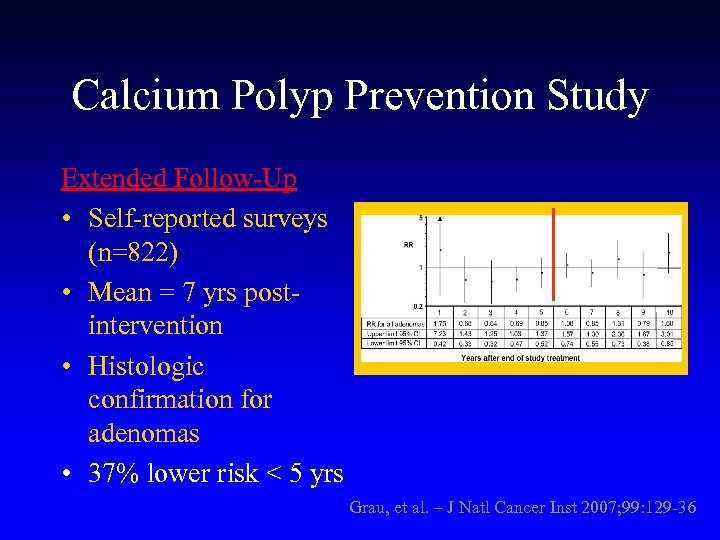 Calcium Polyp Prevention Study Extended Follow-Up • Self-reported surveys (n=822) • Mean = 7 yrs postintervention • Histologic confirmation for adenomas • 37% lower risk < 5 yrs Grau, et al. – J Natl Cancer Inst 2007; 99: 129 -36
Calcium Polyp Prevention Study Extended Follow-Up • Self-reported surveys (n=822) • Mean = 7 yrs postintervention • Histologic confirmation for adenomas • 37% lower risk < 5 yrs Grau, et al. – J Natl Cancer Inst 2007; 99: 129 -36
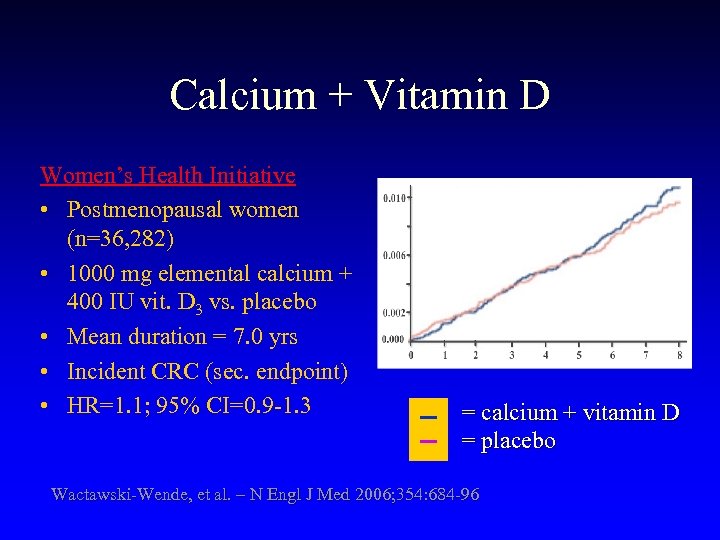 Calcium + Vitamin D Women’s Health Initiative • Postmenopausal women (n=36, 282) • 1000 mg elemental calcium + 400 IU vit. D 3 vs. placebo • Mean duration = 7. 0 yrs • Incident CRC (sec. endpoint) • HR=1. 1; 95% CI=0. 9 -1. 3 = calcium + vitamin D = placebo Wactawski-Wende, et al. – N Engl J Med 2006; 354: 684 -96
Calcium + Vitamin D Women’s Health Initiative • Postmenopausal women (n=36, 282) • 1000 mg elemental calcium + 400 IU vit. D 3 vs. placebo • Mean duration = 7. 0 yrs • Incident CRC (sec. endpoint) • HR=1. 1; 95% CI=0. 9 -1. 3 = calcium + vitamin D = placebo Wactawski-Wende, et al. – N Engl J Med 2006; 354: 684 -96
 Nutraceutical Trials Agent(s) N Endpoint Risk Estimate Fiber, Fat 201 Rec. adenoma 1. 2 (0. 6 -2. 2) Fiber, Fat, BC* 424 Rec. adenoma 1. 2 (0. 8 -2. 0) Fiber 1, 429 Rec. adenoma 0. 9 (0. 7 -1. 1) Fiber, Fat, F&V† 2, 079 Rec. adenoma 1. 0 (0. 9 -1. 1) Vitamins C, E 200 Rec. adenoma 0. 9 (0. 5 -1. 5) Vitamins C, E 864 Rec. adenoma 1. 1 (0. 9 -1. 3) Vitamin E 29, 133 Inc. cancer 0. 8 (0. 6 -1. 1) Selenium 598 Prev. adenoma 0. 7 (0. 4 -1. 1) Antioxidants + Calcium 93 Rec. adenoma 0. 3 (0. 1 -0. 8) Calcium 930 Rec. adenoma 0. 8 (0. 7 -1. 0) *Beta carotene; †Fruits & vegetables
Nutraceutical Trials Agent(s) N Endpoint Risk Estimate Fiber, Fat 201 Rec. adenoma 1. 2 (0. 6 -2. 2) Fiber, Fat, BC* 424 Rec. adenoma 1. 2 (0. 8 -2. 0) Fiber 1, 429 Rec. adenoma 0. 9 (0. 7 -1. 1) Fiber, Fat, F&V† 2, 079 Rec. adenoma 1. 0 (0. 9 -1. 1) Vitamins C, E 200 Rec. adenoma 0. 9 (0. 5 -1. 5) Vitamins C, E 864 Rec. adenoma 1. 1 (0. 9 -1. 3) Vitamin E 29, 133 Inc. cancer 0. 8 (0. 6 -1. 1) Selenium 598 Prev. adenoma 0. 7 (0. 4 -1. 1) Antioxidants + Calcium 93 Rec. adenoma 0. 3 (0. 1 -0. 8) Calcium 930 Rec. adenoma 0. 8 (0. 7 -1. 0) *Beta carotene; †Fruits & vegetables
 Meta-analysis of Antioxidants for GI Cancer Prevention Bjelakovic et al. Lancet 364: 1219, 2004
Meta-analysis of Antioxidants for GI Cancer Prevention Bjelakovic et al. Lancet 364: 1219, 2004
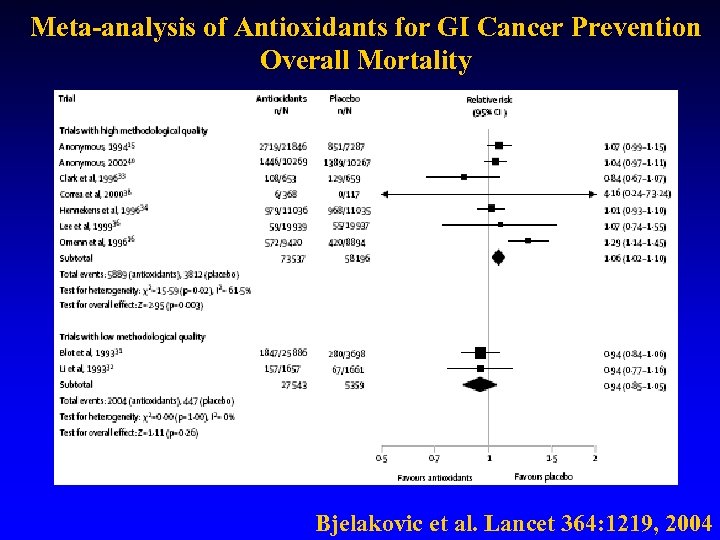 Meta-analysis of Antioxidants for GI Cancer Prevention Overall Mortality Bjelakovic et al. Lancet 364: 1219, 2004
Meta-analysis of Antioxidants for GI Cancer Prevention Overall Mortality Bjelakovic et al. Lancet 364: 1219, 2004
 Statins and Colon Cancer • Lipid lowering agents have been associated with decreased incidence of various cancers • Dale et al (JAMA 295: 74, 2006) performed metaanalysis of randomized through 2005 and observed no effect on cancer incidence or death rate • Poynter et al. (NEJM 352: 2184, 2005) case control study in Israel of approx 2000 colon cancer patients vs 2000 controls; statin use of >5 yrs was associated with a 50% risk reduction
Statins and Colon Cancer • Lipid lowering agents have been associated with decreased incidence of various cancers • Dale et al (JAMA 295: 74, 2006) performed metaanalysis of randomized through 2005 and observed no effect on cancer incidence or death rate • Poynter et al. (NEJM 352: 2184, 2005) case control study in Israel of approx 2000 colon cancer patients vs 2000 controls; statin use of >5 yrs was associated with a 50% risk reduction
 Difluoromethylornithine (DFMO) • DFMO is a specific polyamine inhibitor approved for treatment of African Sleeping sickness • Phase 2 and 3 studies in other tissue sites have observed safety and tolerability; longer duration studies are pending including in preventing colonic polyps
Difluoromethylornithine (DFMO) • DFMO is a specific polyamine inhibitor approved for treatment of African Sleeping sickness • Phase 2 and 3 studies in other tissue sites have observed safety and tolerability; longer duration studies are pending including in preventing colonic polyps
 Chemoprevention of Colon Cancer • Promising results with COX-2 inhibitors have been severely compromised by other organ toxicity • Dietary measures or nutrient-based supplements have mainly been negative, potential exceptions include calcium ± vitamin D and selenium • We continue to explore other options for the chemoprevention of colon cancer
Chemoprevention of Colon Cancer • Promising results with COX-2 inhibitors have been severely compromised by other organ toxicity • Dietary measures or nutrient-based supplements have mainly been negative, potential exceptions include calcium ± vitamin D and selenium • We continue to explore other options for the chemoprevention of colon cancer
 Chemoprevention of Colon Cancer • Future directions – Earlier markers of risk • Protein signatures • Aberrant crypt formation within colon • Genomic signatures – Less invasive measures of ongoing chemopreventive effectiveness • Finding the equivalent of checking your cholesterol level for risk of heart disease
Chemoprevention of Colon Cancer • Future directions – Earlier markers of risk • Protein signatures • Aberrant crypt formation within colon • Genomic signatures – Less invasive measures of ongoing chemopreventive effectiveness • Finding the equivalent of checking your cholesterol level for risk of heart disease
 Chemoprevention of Colon Cancer • Future directions cont. – Combinations; low dose non-selective COX-2 inhibitors + other agents • DFMO + sulindac • Green tea + non-selective COX-2 – Test interventions as more health maintenance rather than just trying to prevent a specific cancer • Studies like the Women’s Health Initiative
Chemoprevention of Colon Cancer • Future directions cont. – Combinations; low dose non-selective COX-2 inhibitors + other agents • DFMO + sulindac • Green tea + non-selective COX-2 – Test interventions as more health maintenance rather than just trying to prevent a specific cancer • Studies like the Women’s Health Initiative
 Colon Cancer Prevention • Currently there are more established/costeffective measures to reduce the burden of colo-rectal cancer; • Chemoprevention of colon cancer currently is not an established approach. • For many reasons pursuing non-invasive interventions for colon cancer risk reduction is important.
Colon Cancer Prevention • Currently there are more established/costeffective measures to reduce the burden of colo-rectal cancer; • Chemoprevention of colon cancer currently is not an established approach. • For many reasons pursuing non-invasive interventions for colon cancer risk reduction is important.


 Examples by Target Organ Kelloff, et al. - Cancer Epidemiol Biomarkers & Prev 2000; 9: 127 -37
Examples by Target Organ Kelloff, et al. - Cancer Epidemiol Biomarkers & Prev 2000; 9: 127 -37
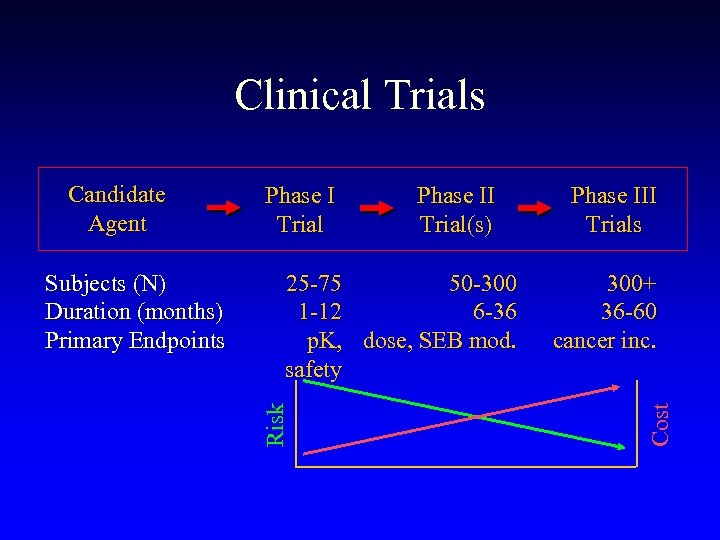 Clinical Trials Phase II Trial(s) 25 -75 50 -300 1 -12 6 -36 p. K, dose, SEB mod. safety Phase III Trials 300+ 36 -60 cancer inc. Cost Subjects (N) Duration (months) Primary Endpoints Phase I Trial Risk Candidate Agent
Clinical Trials Phase II Trial(s) 25 -75 50 -300 1 -12 6 -36 p. K, dose, SEB mod. safety Phase III Trials 300+ 36 -60 cancer inc. Cost Subjects (N) Duration (months) Primary Endpoints Phase I Trial Risk Candidate Agent
 Chemoprevention Targets General • Proliferation • Apoptosis • Circulating growth factors • Immunosurveillance • Metastasis Specific • Tumor necrosis factor • Nuclear factor-kappa B • Activator protein-1 • STAT proteins • Cyclooxygenase-2 • Lipoxygenases
Chemoprevention Targets General • Proliferation • Apoptosis • Circulating growth factors • Immunosurveillance • Metastasis Specific • Tumor necrosis factor • Nuclear factor-kappa B • Activator protein-1 • STAT proteins • Cyclooxygenase-2 • Lipoxygenases
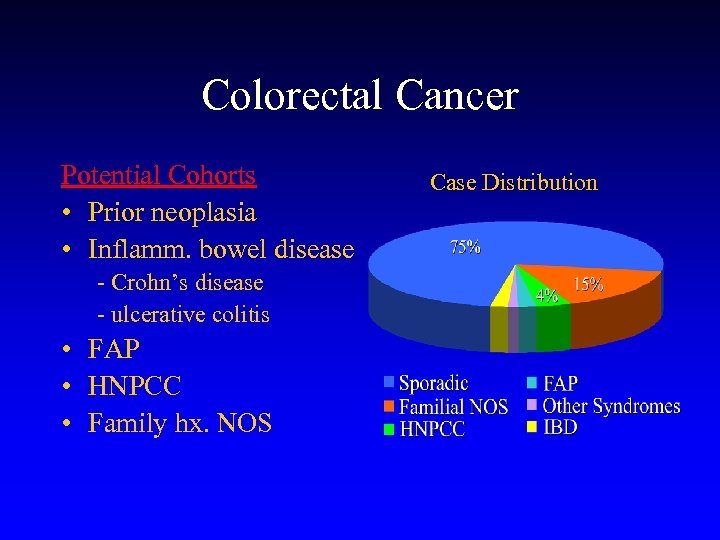 Colorectal Cancer Potential Cohorts • Prior neoplasia • Inflamm. bowel disease - Crohn’s disease - ulcerative colitis • FAP • HNPCC • Family hx. NOS Case Distribution
Colorectal Cancer Potential Cohorts • Prior neoplasia • Inflamm. bowel disease - Crohn’s disease - ulcerative colitis • FAP • HNPCC • Family hx. NOS Case Distribution
 Chemoprevention Agents/Issues with Micronutrients § CARET, ATBC, and NPC Results § Concerning negative results § Micronutrients assumed not to be harmful § Replacement doses vs supraphysiologic (prooxidant effects? ) § Regular dietary consumption vs supplementation § Smokers and gender differences in metabolism
Chemoprevention Agents/Issues with Micronutrients § CARET, ATBC, and NPC Results § Concerning negative results § Micronutrients assumed not to be harmful § Replacement doses vs supraphysiologic (prooxidant effects? ) § Regular dietary consumption vs supplementation § Smokers and gender differences in metabolism


