f05bf3636aa70ae9a6a9f0286732466f.ppt
- Количество слайдов: 50
 Clinical Research and Healthcare – Where is the Link? 24 February 2004 Orlando, FL
Clinical Research and Healthcare – Where is the Link? 24 February 2004 Orlando, FL
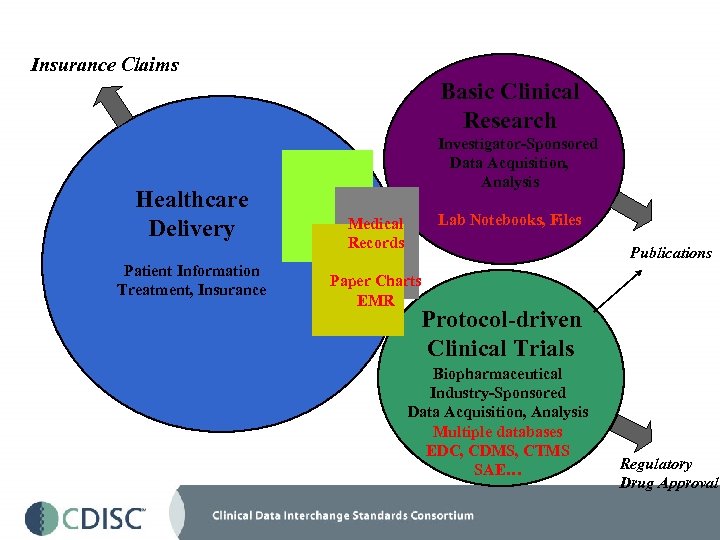 Insurance Claims Basic Clinical Research Healthcare Delivery Patient Information Treatment, Insurance Investigator-Sponsored Data Acquisition, Analysis Lab Notebooks, Files Medical Records Publications Paper Charts EMR Protocol-driven Clinical Trials Biopharmaceutical Industry-Sponsored Data Acquisition, Analysis Multiple databases EDC, CDMS, CTMS SAE… Regulatory Drug Approval
Insurance Claims Basic Clinical Research Healthcare Delivery Patient Information Treatment, Insurance Investigator-Sponsored Data Acquisition, Analysis Lab Notebooks, Files Medical Records Publications Paper Charts EMR Protocol-driven Clinical Trials Biopharmaceutical Industry-Sponsored Data Acquisition, Analysis Multiple databases EDC, CDMS, CTMS SAE… Regulatory Drug Approval
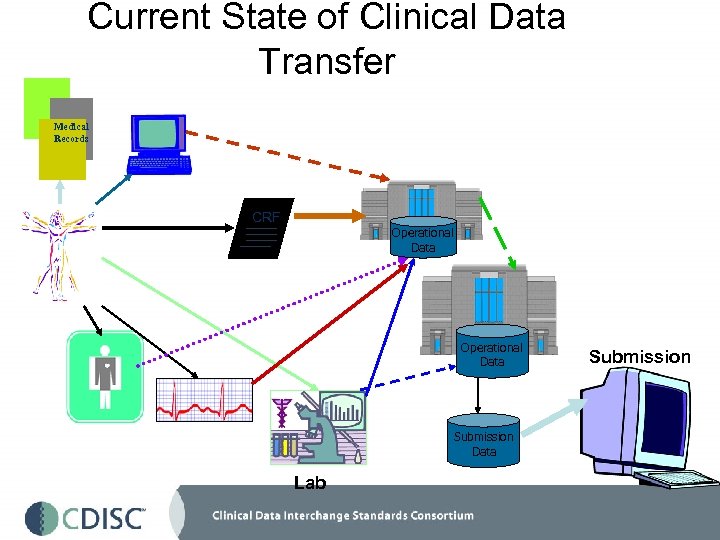 Current State of Clinical Data Transfer Medical Records CRF C Operational Data Submission Data Lab Submission
Current State of Clinical Data Transfer Medical Records CRF C Operational Data Submission Data Lab Submission
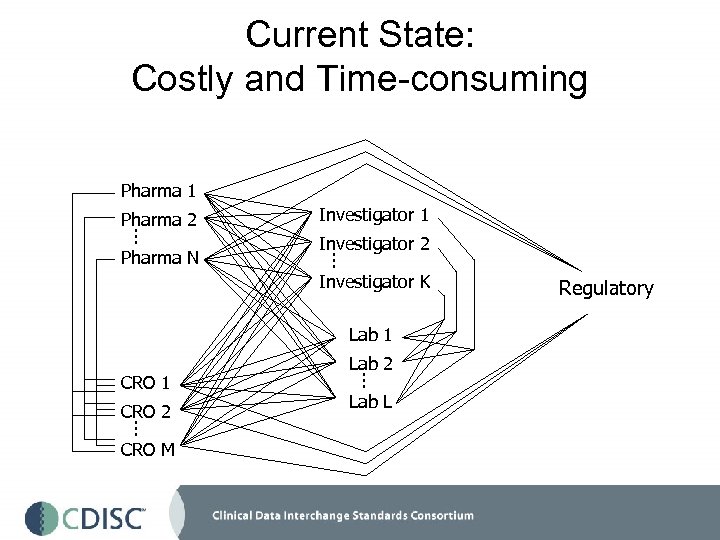 Current State: Costly and Time-consuming Pharma 1 Pharma 2. . . Pharma N Investigator 1 Investigator 2. . . Investigator K Lab 1 CRO 2. . . CRO M Lab 2. . . Lab L Regulatory
Current State: Costly and Time-consuming Pharma 1 Pharma 2. . . Pharma N Investigator 1 Investigator 2. . . Investigator K Lab 1 CRO 2. . . CRO M Lab 2. . . Lab L Regulatory
 The Current State of Clinical Information • Healthcare information is found in paper medical records, in disparate databases, in hospitalbased information systems--- there are islands of data • Clinical research data may exist in additional databases and/or research notebooks • Clinical trial data is collected on 3 -part NCR forms (~90% of trials) or via a multitude of electronic data capture applications
The Current State of Clinical Information • Healthcare information is found in paper medical records, in disparate databases, in hospitalbased information systems--- there are islands of data • Clinical research data may exist in additional databases and/or research notebooks • Clinical trial data is collected on 3 -part NCR forms (~90% of trials) or via a multitude of electronic data capture applications
 Current Technology Issues in Conducting Clinical Trials • Electronic Data Capture (EDC) and/or e. Clinical Trials and new technologies are being piloted (if not adopted) by the major pharma companies. • Sources for electronic data are many and varied (e. g. EDC, IVRS, hand-held diaries, clinical laboratories, AE databases); ready retrieval of electronic data is very important. • Investigative sites are dealing with multiple and varied systems and interfaces.
Current Technology Issues in Conducting Clinical Trials • Electronic Data Capture (EDC) and/or e. Clinical Trials and new technologies are being piloted (if not adopted) by the major pharma companies. • Sources for electronic data are many and varied (e. g. EDC, IVRS, hand-held diaries, clinical laboratories, AE databases); ready retrieval of electronic data is very important. • Investigative sites are dealing with multiple and varied systems and interfaces.
 • Many industry stakeholders have realized that proprietary data structures are not necessarily of value…. instead, focus should be on the molecule---the therapy. • “Innovation depends upon standardization. ” – Dr. Bob O’Neill, Director, Office of Biostatistics, CDER, FDA
• Many industry stakeholders have realized that proprietary data structures are not necessarily of value…. instead, focus should be on the molecule---the therapy. • “Innovation depends upon standardization. ” – Dr. Bob O’Neill, Director, Office of Biostatistics, CDER, FDA
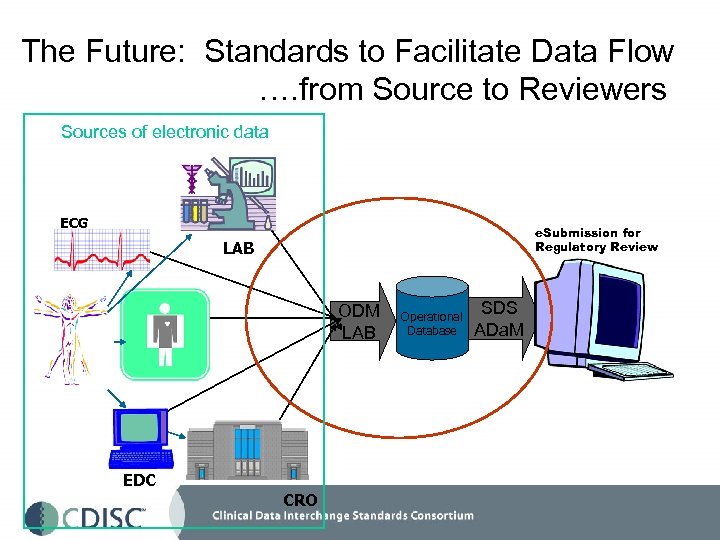 The Future: Standards to Facilitate Data Flow …. from Source to Reviewers Sources of electronic data ECG e. Submission for Regulatory Review LAB ODM LAB EDC CRO Operational Database SDS ADa. M
The Future: Standards to Facilitate Data Flow …. from Source to Reviewers Sources of electronic data ECG e. Submission for Regulatory Review LAB ODM LAB EDC CRO Operational Database SDS ADa. M
 Clinical Data Interchange Standards Consortium CDISC is an open, multidisciplinary, non-profit organization committed to the development of worldwide industry standards to support the electronic acquisition, exchange, submission and archiving of clinical trials data and metadata for medical and biopharmaceutical product development. The CDISC mission is to lead the development of global, vendor-neutral, platform-independent standards to improve data quality and accelerate product development in our industry.
Clinical Data Interchange Standards Consortium CDISC is an open, multidisciplinary, non-profit organization committed to the development of worldwide industry standards to support the electronic acquisition, exchange, submission and archiving of clinical trials data and metadata for medical and biopharmaceutical product development. The CDISC mission is to lead the development of global, vendor-neutral, platform-independent standards to improve data quality and accelerate product development in our industry.
 CDISC History • Started as ‘grass roots’ volunteer group in Fall 1997 with 25 attendees at first meeting • Invited to form DIA SIAC in 1998 • Independent, non-profit organization in February 2000 • ~130 Corporate Memberships, of which ~50 are Corporate Sponsors (e. g. global pharmaceutical companies, CROs and technology providers) • Originally two working teams (Nomenclature and Modeling); Modeling split modeling into four teams; Nomenclature to Glossary Group • Models developed by consensus-based process
CDISC History • Started as ‘grass roots’ volunteer group in Fall 1997 with 25 attendees at first meeting • Invited to form DIA SIAC in 1998 • Independent, non-profit organization in February 2000 • ~130 Corporate Memberships, of which ~50 are Corporate Sponsors (e. g. global pharmaceutical companies, CROs and technology providers) • Originally two working teams (Nomenclature and Modeling); Modeling split modeling into four teams; Nomenclature to Glossary Group • Models developed by consensus-based process
 Evolution of CDISC Standards Data Sources • Site CRFs • Laboratories • Contract Research Organizations • Development Partners Operational Data Interchange & Archive: ODM, LAB Operational Database • Study Data • Audit Trail • Metadata ODM = Operational Data Model/Std LAB = Laboratory Data Model/Std Submission Data Interchange & Archive: SDS, ADa. M Submission Data • CRT/Domain Datasets • Analysis Datasets • Metadata SDS = Submission Domain Standards ADa. M = Analysis Data Models
Evolution of CDISC Standards Data Sources • Site CRFs • Laboratories • Contract Research Organizations • Development Partners Operational Data Interchange & Archive: ODM, LAB Operational Database • Study Data • Audit Trail • Metadata ODM = Operational Data Model/Std LAB = Laboratory Data Model/Std Submission Data Interchange & Archive: SDS, ADa. M Submission Data • CRT/Domain Datasets • Analysis Datasets • Metadata SDS = Submission Domain Standards ADa. M = Analysis Data Models
 Progress on the Standards • LAB Standard: Production Version 1. 0. 1 available; approved HL 7 RIM message – V 1. 0 plus microbiology extension – Four methods of implementation • ODM Standard: Production Version 1. 2 available as an XML schema – Define. xml in progress with SDS metadata • SDS Standard: Version 3. 0 available – Version 3. 1 anticipated to be HL 7 Informative Document referenced by FDA Guidance by Q 2 2004 • ADa. M: Selected analysis dataset models
Progress on the Standards • LAB Standard: Production Version 1. 0. 1 available; approved HL 7 RIM message – V 1. 0 plus microbiology extension – Four methods of implementation • ODM Standard: Production Version 1. 2 available as an XML schema – Define. xml in progress with SDS metadata • SDS Standard: Version 3. 0 available – Version 3. 1 anticipated to be HL 7 Informative Document referenced by FDA Guidance by Q 2 2004 • ADa. M: Selected analysis dataset models
 CDISC SDS Standard • Submission Domain Standards Version 3. 1 – Incorporates changes based upon results of Version 3. 0 FDA pilot – Scheduled to be balloted through HL 7 and referenced in FDA Guidance as of April 2004; final documents anticipated by June 2004 – Provides means to submit Case Report Tabulation data to FDA in standard form, thus facilitating • Review of submissions using standard tools • Population of cross-trial database (Janus model) Plan is to reference as specification (HL 7 -approved CDISC SDS V 3. 1) in Guidance for FDA Implementation of International Conference on Harmonization (ICH) e. Common Technical Document (e. CTD).
CDISC SDS Standard • Submission Domain Standards Version 3. 1 – Incorporates changes based upon results of Version 3. 0 FDA pilot – Scheduled to be balloted through HL 7 and referenced in FDA Guidance as of April 2004; final documents anticipated by June 2004 – Provides means to submit Case Report Tabulation data to FDA in standard form, thus facilitating • Review of submissions using standard tools • Population of cross-trial database (Janus model) Plan is to reference as specification (HL 7 -approved CDISC SDS V 3. 1) in Guidance for FDA Implementation of International Conference on Harmonization (ICH) e. Common Technical Document (e. CTD).
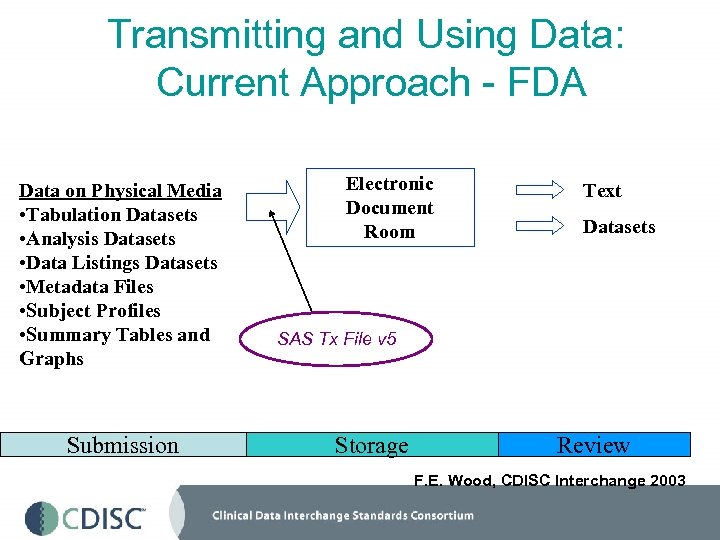 Transmitting and Using Data: Current Approach - FDA Data on Physical Media • Tabulation Datasets • Analysis Datasets • Data Listings Datasets • Metadata Files • Subject Profiles • Summary Tables and Graphs Submission Electronic Document Room Text Datasets SAS Tx File v 5 Storage Review F. E. Wood, CDISC Interchange 2003
Transmitting and Using Data: Current Approach - FDA Data on Physical Media • Tabulation Datasets • Analysis Datasets • Data Listings Datasets • Metadata Files • Subject Profiles • Summary Tables and Graphs Submission Electronic Document Room Text Datasets SAS Tx File v 5 Storage Review F. E. Wood, CDISC Interchange 2003
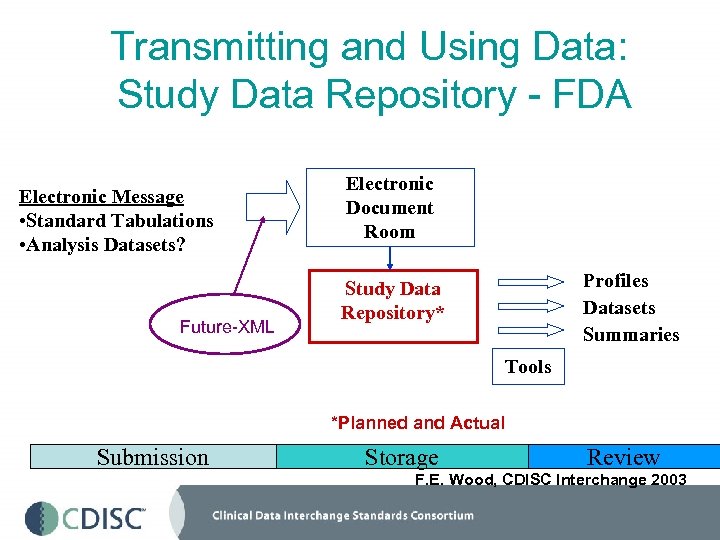 Transmitting and Using Data: Study Data Repository - FDA Electronic Message • Standard Tabulations • Analysis Datasets? Future-XML Electronic Document Room Profiles Datasets Summaries Study Data Repository* Tools *Planned and Actual Submission Storage Review F. E. Wood, CDISC Interchange 2003
Transmitting and Using Data: Study Data Repository - FDA Electronic Message • Standard Tabulations • Analysis Datasets? Future-XML Electronic Document Room Profiles Datasets Summaries Study Data Repository* Tools *Planned and Actual Submission Storage Review F. E. Wood, CDISC Interchange 2003
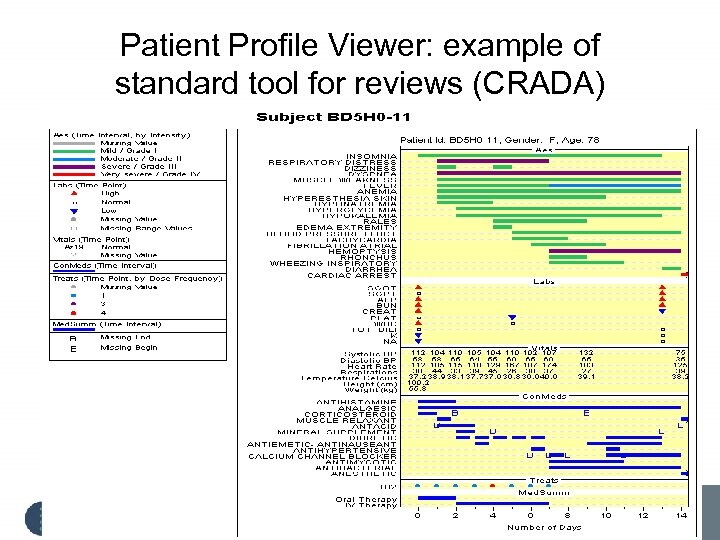 Patient Profile Viewer: example of standard tool for reviews (CRADA)
Patient Profile Viewer: example of standard tool for reviews (CRADA)
 Interchange Standards: Long-term Desired Outcomes • A holistic approach to standards, facilitating data interchange from sites through regulatory submission, utilizing XML • Standards for data acquisition supporting the population of a cross-trial warehouse within FDA • HL 7 -CDISC models harmonized to yield value for both clinical research and healthcare – sharing of information between EMR and clinical trials • Global adoption of CDISC data standards CDISC Meeting with FDA Commissioner, April 2003
Interchange Standards: Long-term Desired Outcomes • A holistic approach to standards, facilitating data interchange from sites through regulatory submission, utilizing XML • Standards for data acquisition supporting the population of a cross-trial warehouse within FDA • HL 7 -CDISC models harmonized to yield value for both clinical research and healthcare – sharing of information between EMR and clinical trials • Global adoption of CDISC data standards CDISC Meeting with FDA Commissioner, April 2003
 Evolution of CDISC Standards Data Sources • Site CRFs • Laboratories • Contract Research Organizations • Development Partners Operational Submission Database Future Uniform Standard Data Interchange • Study Data & Archive: • Audit Trail ODM, LAB SDS, ADa. M • Metadata ODM = Operational Data Model/Std LAB = Laboratory Data Model/Std Submission Data • CRT/Domain Datasets • Analysis Datasets • Metadata SDS = Submission Domain Standards ADa. M = Analysis Data Models
Evolution of CDISC Standards Data Sources • Site CRFs • Laboratories • Contract Research Organizations • Development Partners Operational Submission Database Future Uniform Standard Data Interchange • Study Data & Archive: • Audit Trail ODM, LAB SDS, ADa. M • Metadata ODM = Operational Data Model/Std LAB = Laboratory Data Model/Std Submission Data • CRT/Domain Datasets • Analysis Datasets • Metadata SDS = Submission Domain Standards ADa. M = Analysis Data Models
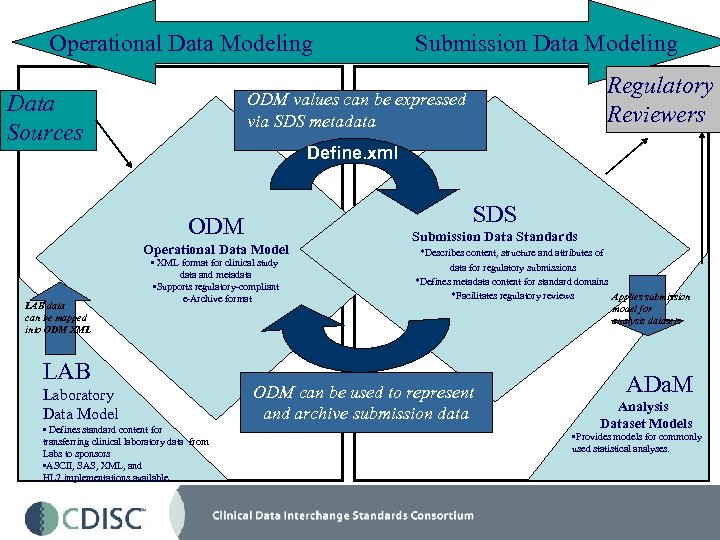 Operational Data Modeling Regulatory Reviewers ODM values can be expressed via SDS metadata Data Sources Define. xml SDS ODM Operational Data Model LAB data can be mapped into ODM XML Submission Data Modeling • XML format for clinical study data and metadata • Supports regulatory-compliant e-Archive format LAB Laboratory Data Model • Defines standard content for transferring clinical laboratory data from Labs to sponsors • ASCII, SAS, XML, and HL 7 implementations available Submission Data Standards *Describes content, structure and attributes of data for regulatory submissions *Defines metadata content for standard domains *Facilitates regulatory reviews Applies submission model for analysis datasets ODM can be used to represent and archive submission data ADa. M Analysis Dataset Models • Provides models for commonly used statistical analyses.
Operational Data Modeling Regulatory Reviewers ODM values can be expressed via SDS metadata Data Sources Define. xml SDS ODM Operational Data Model LAB data can be mapped into ODM XML Submission Data Modeling • XML format for clinical study data and metadata • Supports regulatory-compliant e-Archive format LAB Laboratory Data Model • Defines standard content for transferring clinical laboratory data from Labs to sponsors • ASCII, SAS, XML, and HL 7 implementations available Submission Data Standards *Describes content, structure and attributes of data for regulatory submissions *Defines metadata content for standard domains *Facilitates regulatory reviews Applies submission model for analysis datasets ODM can be used to represent and archive submission data ADa. M Analysis Dataset Models • Provides models for commonly used statistical analyses.
 CDISC and HL 7 • Formal Association as of 2001 • Technical Committee (Regulated Clinical Research and Information Management – RCRIM) co-chaired by FDA, CDISC and HL 7 • Benefits • Capitalizes on HL 7 standards development experience base • Leverages HL 7 accreditation and HHS acknowledgement as SDO; FDA support • Expands value and influence of clinical trials standards efforts through harmonization • Provides opportunity for common informatics platform for healthcare and clinical research
CDISC and HL 7 • Formal Association as of 2001 • Technical Committee (Regulated Clinical Research and Information Management – RCRIM) co-chaired by FDA, CDISC and HL 7 • Benefits • Capitalizes on HL 7 standards development experience base • Leverages HL 7 accreditation and HHS acknowledgement as SDO; FDA support • Expands value and influence of clinical trials standards efforts through harmonization • Provides opportunity for common informatics platform for healthcare and clinical research
 Shared Goals; Individual Contributions • Shared Purpose (CDISC, FDA/HHS and HL 7) – To improve the quality of public health – To have one overarching standard model for data interchange for healthcare information and clinical trial/clinical research data • Domain Expertise – CDISC, FDA and the Pharmaceutical Industry – Regulated clinical research data acquisition, review and archive requirements – HL 7 – Healthcare information exchange standards and methodology; accreditation processes
Shared Goals; Individual Contributions • Shared Purpose (CDISC, FDA/HHS and HL 7) – To improve the quality of public health – To have one overarching standard model for data interchange for healthcare information and clinical trial/clinical research data • Domain Expertise – CDISC, FDA and the Pharmaceutical Industry – Regulated clinical research data acquisition, review and archive requirements – HL 7 – Healthcare information exchange standards and methodology; accreditation processes
 Regulated Clinical Research Information Management TC (RCRIM) Initiated as Clinical Trials SIG in Jan. 2001 TC approved in April 2002 Joint Leadership: HL 7, CDISC, FDA Active Membership: FDA, CDISC, HL 7 & others • Facilitates development of common standards across variety of organizations to improve information management during clinical research and regulatory evaluation • Defines messages, document structures, and terminology to support collection, storage, distribution, integration and analysis of clinical research data • Assures related or supportive standards produced by other HL 7 groups are robust enough to be used in regulated clinical research
Regulated Clinical Research Information Management TC (RCRIM) Initiated as Clinical Trials SIG in Jan. 2001 TC approved in April 2002 Joint Leadership: HL 7, CDISC, FDA Active Membership: FDA, CDISC, HL 7 & others • Facilitates development of common standards across variety of organizations to improve information management during clinical research and regulatory evaluation • Defines messages, document structures, and terminology to support collection, storage, distribution, integration and analysis of clinical research data • Assures related or supportive standards produced by other HL 7 groups are robust enough to be used in regulated clinical research
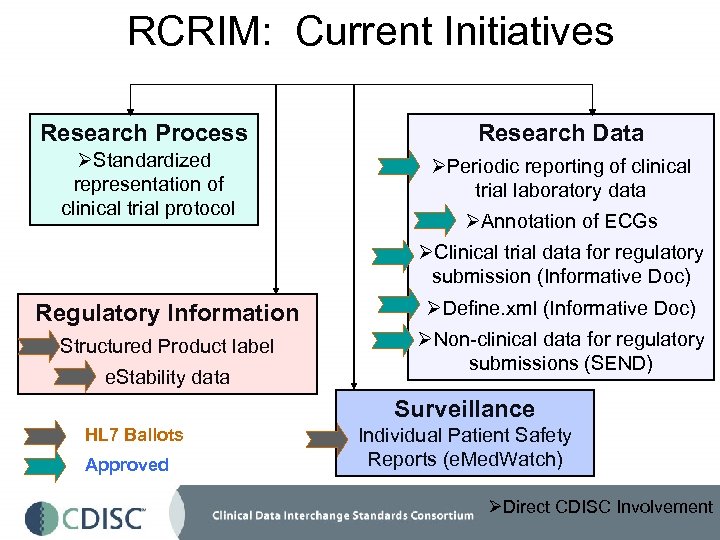 RCRIM: Current Initiatives Research Process Research Data ØStandardized representation of clinical trial protocol ØPeriodic reporting of clinical trial laboratory data ØAnnotation of ECGs ØClinical trial data for regulatory submission (Informative Doc) Regulatory Information ØDefine. xml (Informative Doc) Structured Product label ØNon-clinical data for regulatory submissions (SEND) e. Stability data Surveillance HL 7 Ballots Approved Individual Patient Safety Reports (e. Med. Watch) ØDirect CDISC Involvement
RCRIM: Current Initiatives Research Process Research Data ØStandardized representation of clinical trial protocol ØPeriodic reporting of clinical trial laboratory data ØAnnotation of ECGs ØClinical trial data for regulatory submission (Informative Doc) Regulatory Information ØDefine. xml (Informative Doc) Structured Product label ØNon-clinical data for regulatory submissions (SEND) e. Stability data Surveillance HL 7 Ballots Approved Individual Patient Safety Reports (e. Med. Watch) ØDirect CDISC Involvement
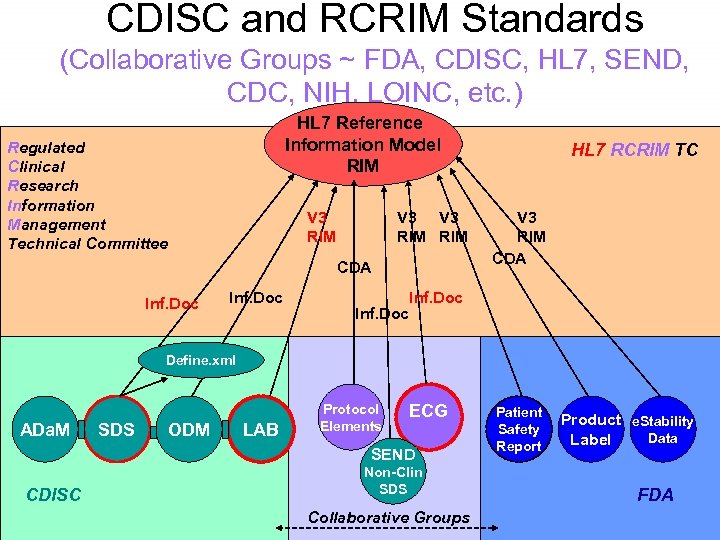 CDISC and RCRIM Standards (Collaborative Groups ~ FDA, CDISC, HL 7, SEND, CDC, NIH, LOINC, etc. ) HL 7 Reference Information Model RIM Regulated Clinical Research Information Management Technical Committee V 3 RIM CDA Inf. Doc HL 7 RCRIM TC V 3 RIM CDA Inf. Doc Define. xml ADa. M SDS ODM LAB Protocol Elements ECG SEND CDISC Non-Clin SDS Collaborative Groups Patient Safety Report Product e. Stability Data Label FDA
CDISC and RCRIM Standards (Collaborative Groups ~ FDA, CDISC, HL 7, SEND, CDC, NIH, LOINC, etc. ) HL 7 Reference Information Model RIM Regulated Clinical Research Information Management Technical Committee V 3 RIM CDA Inf. Doc HL 7 RCRIM TC V 3 RIM CDA Inf. Doc Define. xml ADa. M SDS ODM LAB Protocol Elements ECG SEND CDISC Non-Clin SDS Collaborative Groups Patient Safety Report Product e. Stability Data Label FDA
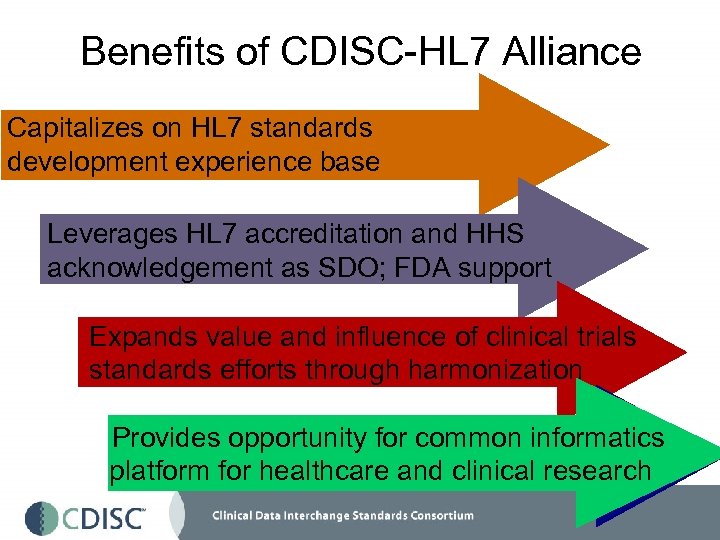 Benefits of CDISC-HL 7 Alliance Capitalizes on HL 7 standards development experience base Leverages HL 7 accreditation and HHS acknowledgement as SDO; FDA support Expands value and influence of clinical trials standards efforts through harmonization Provides opportunity for common informatics platform for healthcare and clinical research
Benefits of CDISC-HL 7 Alliance Capitalizes on HL 7 standards development experience base Leverages HL 7 accreditation and HHS acknowledgement as SDO; FDA support Expands value and influence of clinical trials standards efforts through harmonization Provides opportunity for common informatics platform for healthcare and clinical research
 Proof-of Concept Project • Designed to demonstrate compatibility between HL 7 RIM -derived Clinical Document Architecture (CDA) and CDISC Operational Data Model (ODM) – Single source entry for medical records and clinical trials – e. Source documentation contributes to patient chart, not the reverse (i. e. extraction from EMR); reduces regulatory issues – Minimizes redundancy in data entry, thus improving data integrity/quality – Can potentially be driven from electronic protocol • First stage POC initiated through Duke Hospital and DCRI • Have commitments from two major technology partners, an academic institution and an academic research organization; establishing more pharma support for this breakthrough project (five interested in ‘ringside seat’)
Proof-of Concept Project • Designed to demonstrate compatibility between HL 7 RIM -derived Clinical Document Architecture (CDA) and CDISC Operational Data Model (ODM) – Single source entry for medical records and clinical trials – e. Source documentation contributes to patient chart, not the reverse (i. e. extraction from EMR); reduces regulatory issues – Minimizes redundancy in data entry, thus improving data integrity/quality – Can potentially be driven from electronic protocol • First stage POC initiated through Duke Hospital and DCRI • Have commitments from two major technology partners, an academic institution and an academic research organization; establishing more pharma support for this breakthrough project (five interested in ‘ringside seat’)
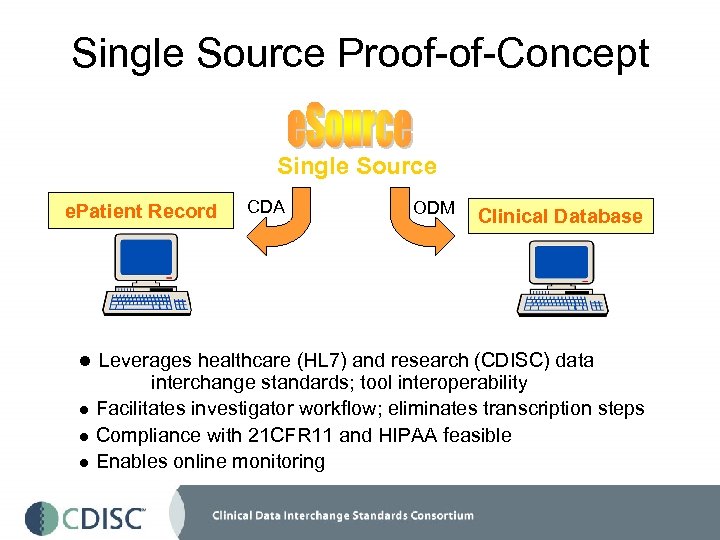 Single Source Proof-of-Concept Single Source e. Patient Record CDA ODM Clinical Database l Leverages healthcare (HL 7) and research (CDISC) data interchange standards; tool interoperability l Facilitates investigator workflow; eliminates transcription steps l Compliance with 21 CFR 11 and HIPAA feasible l Enables online monitoring
Single Source Proof-of-Concept Single Source e. Patient Record CDA ODM Clinical Database l Leverages healthcare (HL 7) and research (CDISC) data interchange standards; tool interoperability l Facilitates investigator workflow; eliminates transcription steps l Compliance with 21 CFR 11 and HIPAA feasible l Enables online monitoring
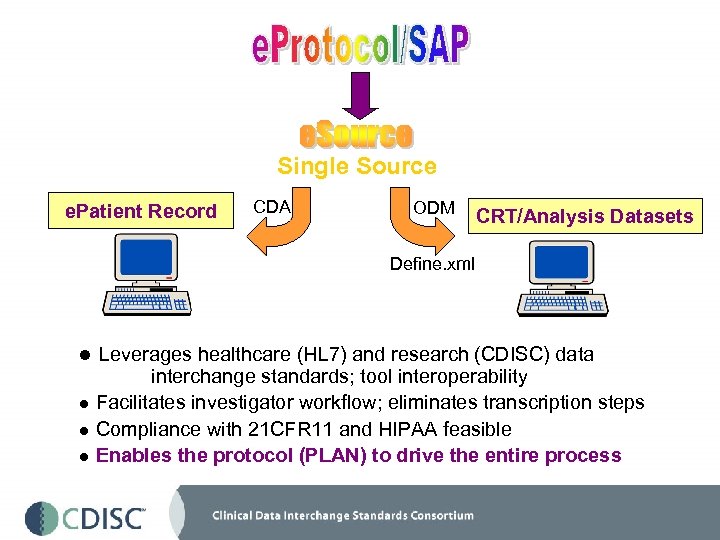 Single Source e. Patient Record CDA ODM CRT/Analysis Datasets Define. xml l Leverages healthcare (HL 7) and research (CDISC) data interchange standards; tool interoperability l Facilitates investigator workflow; eliminates transcription steps l Compliance with 21 CFR 11 and HIPAA feasible l Enables the protocol (PLAN) to drive the entire process
Single Source e. Patient Record CDA ODM CRT/Analysis Datasets Define. xml l Leverages healthcare (HL 7) and research (CDISC) data interchange standards; tool interoperability l Facilitates investigator workflow; eliminates transcription steps l Compliance with 21 CFR 11 and HIPAA feasible l Enables the protocol (PLAN) to drive the entire process
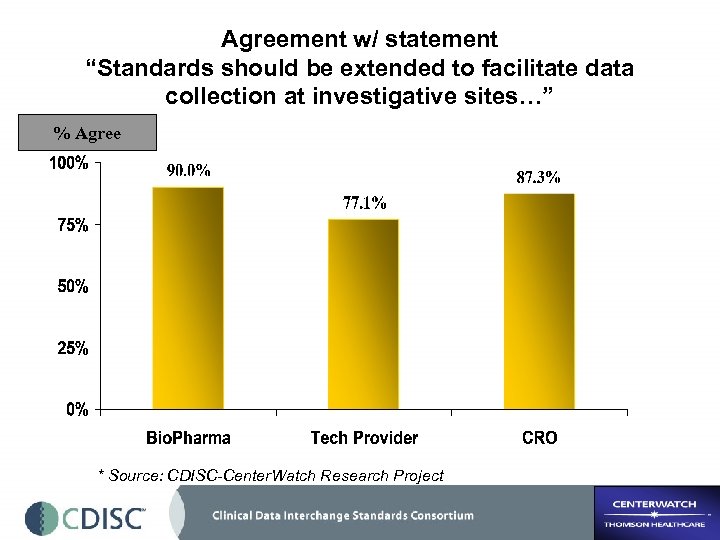 Agreement w/ statement “Standards should be extended to facilitate data collection at investigative sites…” % Agree * Source: CDISC-Center. Watch Research Project
Agreement w/ statement “Standards should be extended to facilitate data collection at investigative sites…” % Agree * Source: CDISC-Center. Watch Research Project
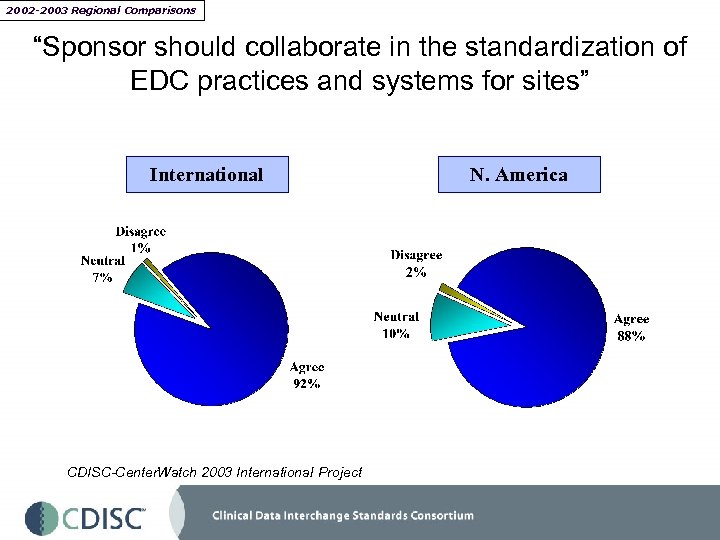 2002 -2003 Regional Comparisons “Sponsor should collaborate in the standardization of EDC practices and systems for sites” International CDISC-Center. Watch 2003 International Project N. America
2002 -2003 Regional Comparisons “Sponsor should collaborate in the standardization of EDC practices and systems for sites” International CDISC-Center. Watch 2003 International Project N. America
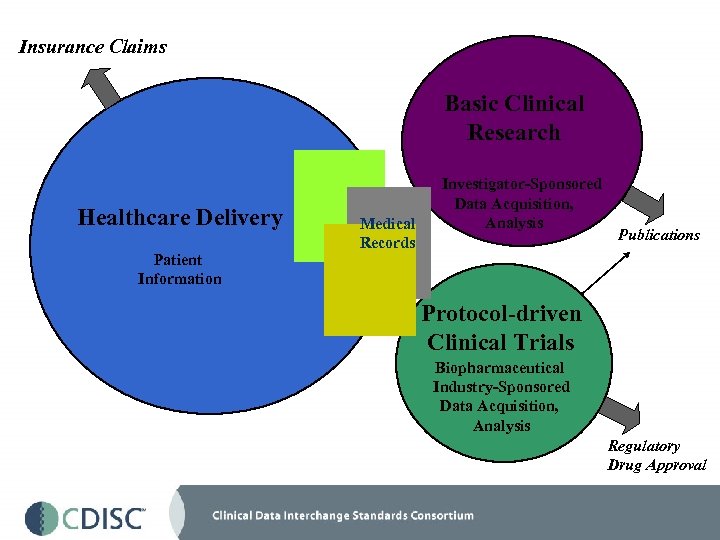 Insurance Claims Basic Clinical Research Healthcare Delivery Patient Information Investigator-Sponsored Data Acquisition, Analysis Medical Publications Records Protocol-driven Clinical Trials Biopharmaceutical Industry-Sponsored Data Acquisition, Analysis Regulatory Drug Approval
Insurance Claims Basic Clinical Research Healthcare Delivery Patient Information Investigator-Sponsored Data Acquisition, Analysis Medical Publications Records Protocol-driven Clinical Trials Biopharmaceutical Industry-Sponsored Data Acquisition, Analysis Regulatory Drug Approval
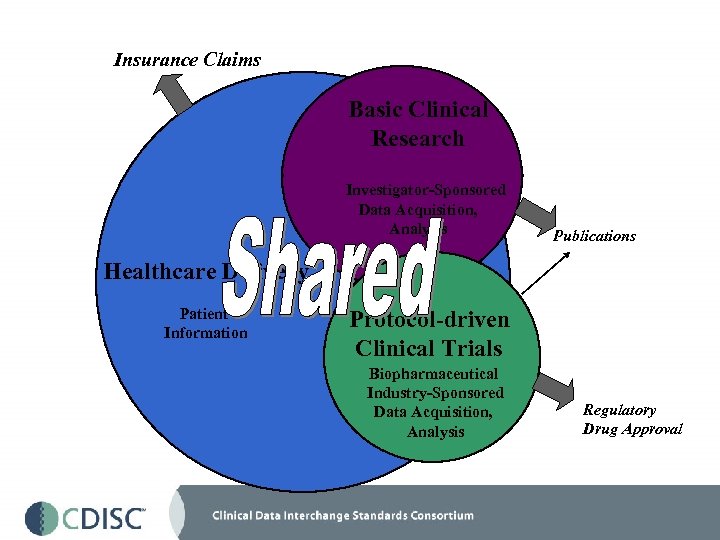 Insurance Claims Basic Clinical Research Investigator-Sponsored Data Acquisition, Analysis Publications Healthcare Delivery Patient Information Protocol-driven Clinical Trials Biopharmaceutical Industry-Sponsored Data Acquisition, Analysis Regulatory Drug Approval
Insurance Claims Basic Clinical Research Investigator-Sponsored Data Acquisition, Analysis Publications Healthcare Delivery Patient Information Protocol-driven Clinical Trials Biopharmaceutical Industry-Sponsored Data Acquisition, Analysis Regulatory Drug Approval
 Information and Contacts • For standards and information, see www. cdisc. org • Quarterly updates available via e-mail; contact Shirley Williams swilliams@cdisc. org • Technical questions: Julie Evans jevans@cdisc. org or Public Discussion Forum • Education and Membership: Frank Newby fnewby@cdisc. org • Rebecca Kush: rkush@cdisc. org
Information and Contacts • For standards and information, see www. cdisc. org • Quarterly updates available via e-mail; contact Shirley Williams swilliams@cdisc. org • Technical questions: Julie Evans jevans@cdisc. org or Public Discussion Forum • Education and Membership: Frank Newby fnewby@cdisc. org • Rebecca Kush: rkush@cdisc. org
 Knowing is not enough; we must apply. Willing is not enough; we must do. - Goethe- CDISC could not be what it is without all of our kind supporters who ‘apply’ and ‘do’…. THANK YOU!
Knowing is not enough; we must apply. Willing is not enough; we must do. - Goethe- CDISC could not be what it is without all of our kind supporters who ‘apply’ and ‘do’…. THANK YOU!




 The CDISC Vision “The exchange of all clinical trial data between any two parties will be achieved by the application of the appropriate CDISC data models and standards. ”
The CDISC Vision “The exchange of all clinical trial data between any two parties will be achieved by the application of the appropriate CDISC data models and standards. ”
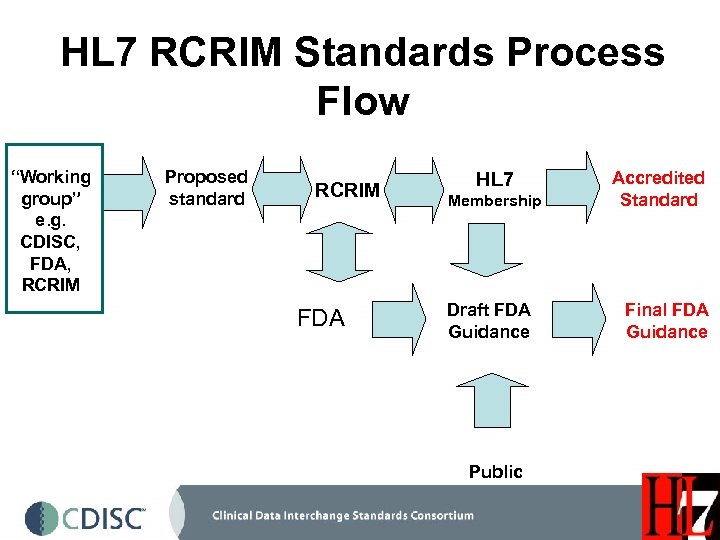 HL 7 RCRIM Standards Process Flow “Working group” e. g. CDISC, FDA, RCRIM Proposed standard RCRIM FDA HL 7 Membership Draft FDA Guidance Public Accredited Standard Final FDA Guidance
HL 7 RCRIM Standards Process Flow “Working group” e. g. CDISC, FDA, RCRIM Proposed standard RCRIM FDA HL 7 Membership Draft FDA Guidance Public Accredited Standard Final FDA Guidance
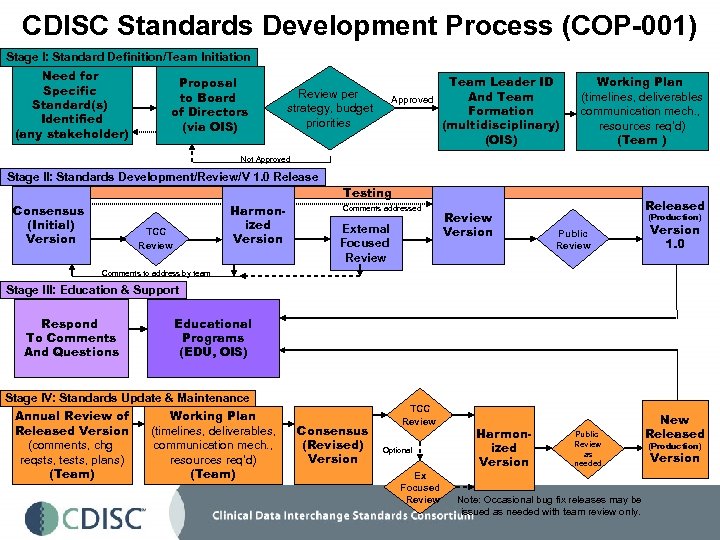 CDISC Standards Development Process (COP-001) Stage I: Standard Definition/Team Initiation Need for Specific Standard(s) Identified (any stakeholder) Proposal to Board of Directors (via OIS) Team Leader ID And Team Approved Formation (multidisciplinary) (OIS) Review per strategy, budget priorities Working Plan (timelines, deliverables communication mech. , resources req’d) (Team ) Not Approved Stage II: Standards Development/Review/V 1. 0 Release Testing Consensus (Initial) Version TCC Review Harmonized Version Comments addressed External Focused Review Version Released (Production) Public Review Version 1. 0 Comments to address by team Stage III: Education & Support Respond To Comments And Questions Educational Programs (EDU, OIS) Stage IV: Standards Update & Maintenance Working Plan Annual Review of Released Version (timelines, deliverables, communication mech. , (comments, chg reqsts, tests, plans) resources req’d) (Team) Consensus (Revised) Version TCC Review Optional Ex Focused Review Harmonized Version Public Review as needed Note: Occasional bug fix releases may be issued as needed with team review only. New Released (Production) Version
CDISC Standards Development Process (COP-001) Stage I: Standard Definition/Team Initiation Need for Specific Standard(s) Identified (any stakeholder) Proposal to Board of Directors (via OIS) Team Leader ID And Team Approved Formation (multidisciplinary) (OIS) Review per strategy, budget priorities Working Plan (timelines, deliverables communication mech. , resources req’d) (Team ) Not Approved Stage II: Standards Development/Review/V 1. 0 Release Testing Consensus (Initial) Version TCC Review Harmonized Version Comments addressed External Focused Review Version Released (Production) Public Review Version 1. 0 Comments to address by team Stage III: Education & Support Respond To Comments And Questions Educational Programs (EDU, OIS) Stage IV: Standards Update & Maintenance Working Plan Annual Review of Released Version (timelines, deliverables, communication mech. , (comments, chg reqsts, tests, plans) resources req’d) (Team) Consensus (Revised) Version TCC Review Optional Ex Focused Review Harmonized Version Public Review as needed Note: Occasional bug fix releases may be issued as needed with team review only. New Released (Production) Version
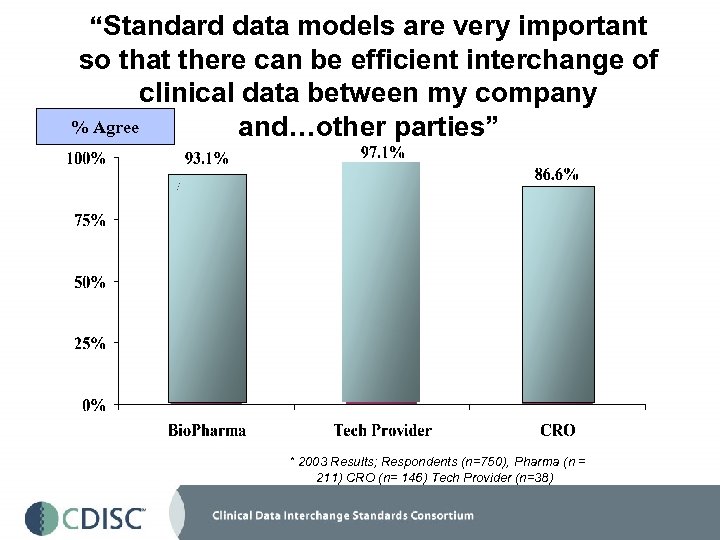 “Standard data models are very important so that there can be efficient interchange of clinical data between my company % Agree and…other parties” * 2003 Results; Respondents (n=750), Pharma (n = 211) CRO (n= 146) Tech Provider (n=38)
“Standard data models are very important so that there can be efficient interchange of clinical data between my company % Agree and…other parties” * 2003 Results; Respondents (n=750), Pharma (n = 211) CRO (n= 146) Tech Provider (n=38)
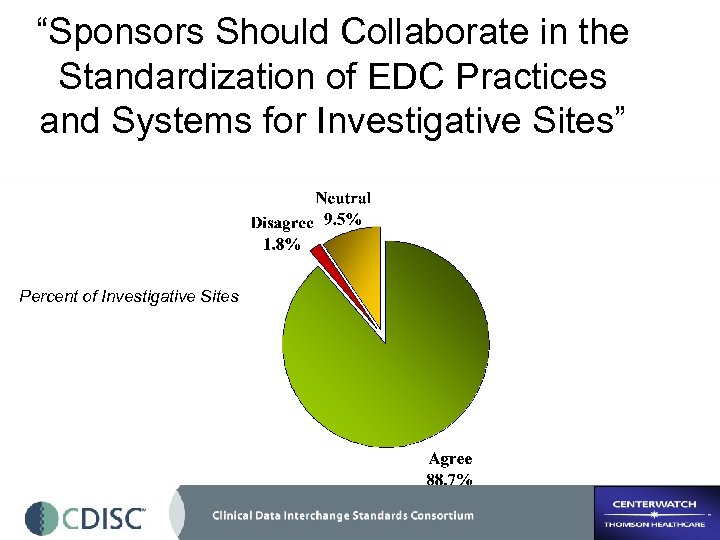 “Sponsors Should Collaborate in the Standardization of EDC Practices and Systems for Investigative Sites” Percent of Investigative Sites
“Sponsors Should Collaborate in the Standardization of EDC Practices and Systems for Investigative Sites” Percent of Investigative Sites
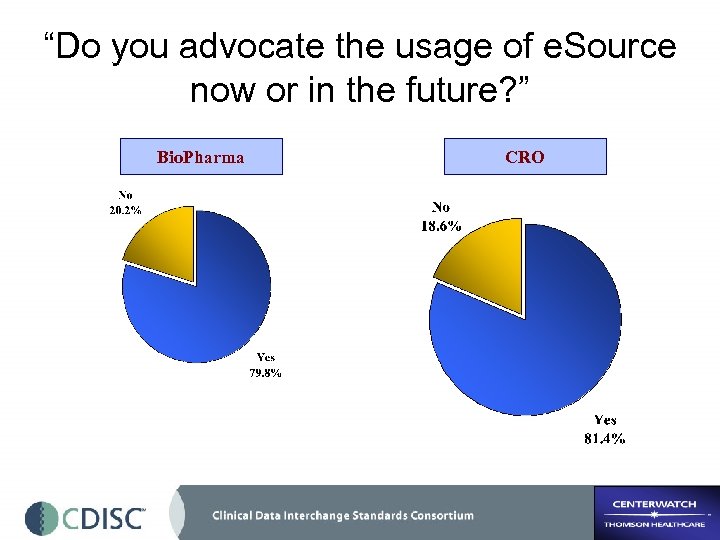 “Do you advocate the usage of e. Source now or in the future? ” Bio. Pharma CRO
“Do you advocate the usage of e. Source now or in the future? ” Bio. Pharma CRO
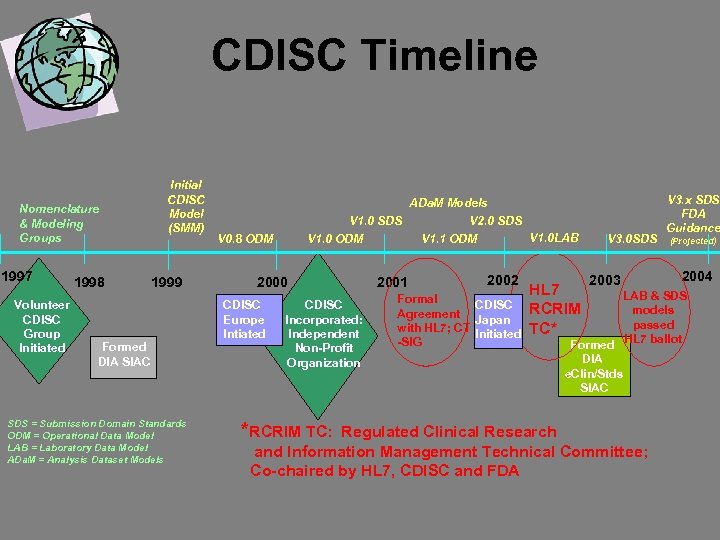 CDISC Timeline Nomenclature & Modeling Groups 1997 Volunteer CDISC Group Initiated 1998 Initial CDISC Model (SMM) 1999 Formed DIA SIAC SDS = Submission Domain Standards ODM = Operational Data Model LAB = Laboratory Data Model ADa. M = Analysis Dataset Models ADa. M Models V 1. 0 SDS V 2. 0 SDS V 1. 0 LAB V 1. 0 ODM V 1. 1 ODM V 0. 8 ODM 2000 CDISC Europe Intiated CDISC Incorporated: Independent Non-Profit Organization *RCRIM TC: 2001 2002 Formal CDISC Agreement Japan with HL 7; CT Initiated -SIG HL 7 RCRIM TC* V 3. 0 SDS 2003 V 3. x SDS FDA Guidance (Projected) 2004 LAB & SDS models passed HL 7 ballot Formed DIA e. Clin/Stds SIAC Regulated Clinical Research and Information Management Technical Committee; Co-chaired by HL 7, CDISC and FDA
CDISC Timeline Nomenclature & Modeling Groups 1997 Volunteer CDISC Group Initiated 1998 Initial CDISC Model (SMM) 1999 Formed DIA SIAC SDS = Submission Domain Standards ODM = Operational Data Model LAB = Laboratory Data Model ADa. M = Analysis Dataset Models ADa. M Models V 1. 0 SDS V 2. 0 SDS V 1. 0 LAB V 1. 0 ODM V 1. 1 ODM V 0. 8 ODM 2000 CDISC Europe Intiated CDISC Incorporated: Independent Non-Profit Organization *RCRIM TC: 2001 2002 Formal CDISC Agreement Japan with HL 7; CT Initiated -SIG HL 7 RCRIM TC* V 3. 0 SDS 2003 V 3. x SDS FDA Guidance (Projected) 2004 LAB & SDS models passed HL 7 ballot Formed DIA e. Clin/Stds SIAC Regulated Clinical Research and Information Management Technical Committee; Co-chaired by HL 7, CDISC and FDA
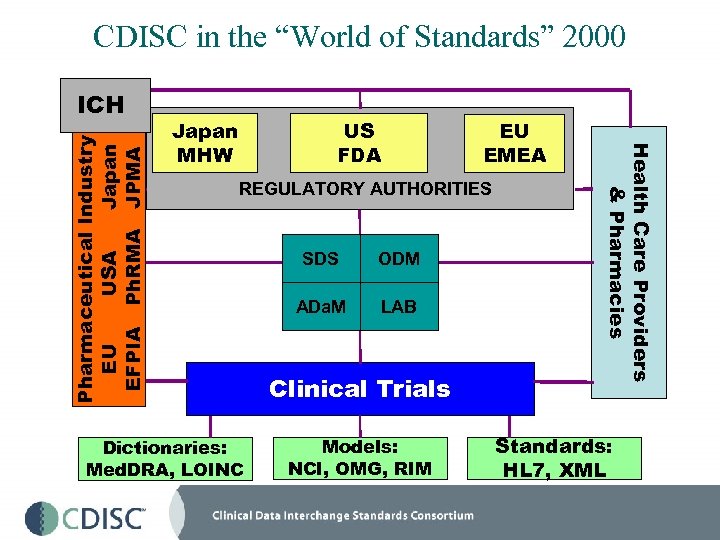 CDISC in the “World of Standards” 2000 Japan MHW US FDA EU EMEA REGULATORY AUTHORITIES Dictionaries: Med. DRA, LOINC SDS ODM ADa. M LAB Clinical Trials Models: NCI, OMG, RIM Health Care Providers & Pharmacies Pharmaceutical Industry EU USA Japan EFPIA Ph. RMA JPMA ICH Standards: HL 7, XML
CDISC in the “World of Standards” 2000 Japan MHW US FDA EU EMEA REGULATORY AUTHORITIES Dictionaries: Med. DRA, LOINC SDS ODM ADa. M LAB Clinical Trials Models: NCI, OMG, RIM Health Care Providers & Pharmacies Pharmaceutical Industry EU USA Japan EFPIA Ph. RMA JPMA ICH Standards: HL 7, XML
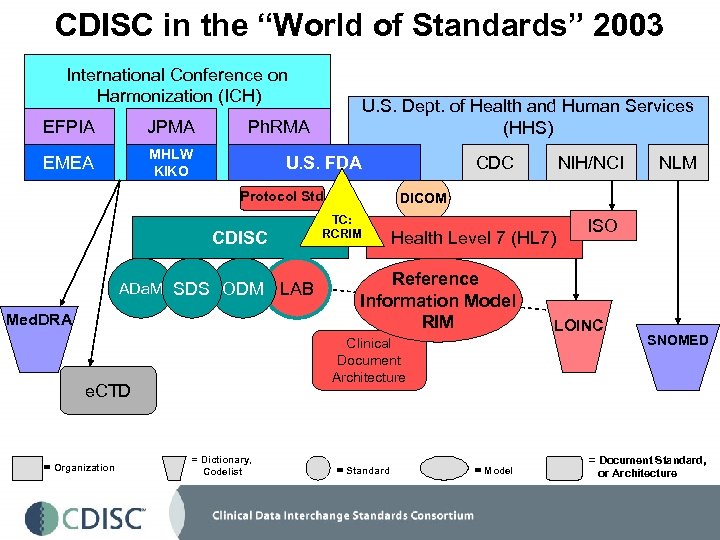 CDISC in the “World of Standards” 2003 International Conference on Harmonization (ICH) EFPIA JPMA EMEA MHLW KIKO U. S. Dept. of Health and Human Services (HHS) Ph. RMA U. S. FDA Protocol Std ADa. M SDS ODM Med. DRA Health Level 7 (HL 7) Reference Information Model RIM Clinical Document Architecture e. CTD = Organization LAB = Dictionary, Codelist NIH/NCI NLM DICOM TC: RCRIM CDISC CDC = Standard = Model ISO LOINC SNOMED = Document Standard, or Architecture
CDISC in the “World of Standards” 2003 International Conference on Harmonization (ICH) EFPIA JPMA EMEA MHLW KIKO U. S. Dept. of Health and Human Services (HHS) Ph. RMA U. S. FDA Protocol Std ADa. M SDS ODM Med. DRA Health Level 7 (HL 7) Reference Information Model RIM Clinical Document Architecture e. CTD = Organization LAB = Dictionary, Codelist NIH/NCI NLM DICOM TC: RCRIM CDISC CDC = Standard = Model ISO LOINC SNOMED = Document Standard, or Architecture
 Speaking of health benefits/opportunities to be realized from making more effective use of IT…. • I think that CDISC will be a big part of moving FDA onto an electronic information architecture where we can realize all of these opportunities. I think this will have a profound and positive impact on our drug review process, allowing us to design trials that can be less expensive and still tell us more about the risks and benefits of a new medical product. And I think that the most significant and perhaps enduring legacy to your efforts could be the very immediate and significant impact it has on improving the lives of patients. – Mark B. Mc. Clellan, MD, Ph. D, FDA Commissioner
Speaking of health benefits/opportunities to be realized from making more effective use of IT…. • I think that CDISC will be a big part of moving FDA onto an electronic information architecture where we can realize all of these opportunities. I think this will have a profound and positive impact on our drug review process, allowing us to design trials that can be less expensive and still tell us more about the risks and benefits of a new medical product. And I think that the most significant and perhaps enduring legacy to your efforts could be the very immediate and significant impact it has on improving the lives of patients. – Mark B. Mc. Clellan, MD, Ph. D, FDA Commissioner
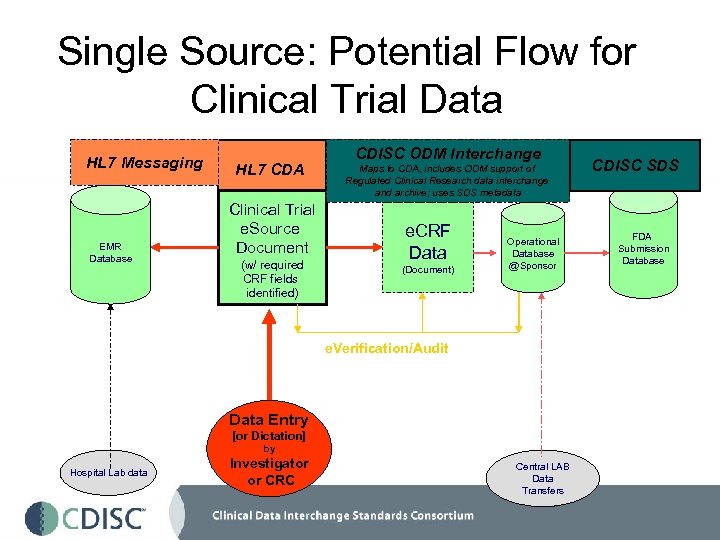 Single Source: Potential Flow for Clinical Trial Data HL 7 Messaging EMR Database HL 7 CDA Clinical Trial e. Source Document (w/ required CRF fields identified) CDISC ODM Interchange Maps to CDA, includes ODM support of Regulated Clinical Research data interchange and archive; uses SDS metadata e. CRF Data (Document) Operational Database @Sponsor e. Verification/Audit Data Entry [or Dictation] by Hospital Lab data Investigator or CRC Central LAB Data Transfers CDISC SDS FDA Submission Database
Single Source: Potential Flow for Clinical Trial Data HL 7 Messaging EMR Database HL 7 CDA Clinical Trial e. Source Document (w/ required CRF fields identified) CDISC ODM Interchange Maps to CDA, includes ODM support of Regulated Clinical Research data interchange and archive; uses SDS metadata e. CRF Data (Document) Operational Database @Sponsor e. Verification/Audit Data Entry [or Dictation] by Hospital Lab data Investigator or CRC Central LAB Data Transfers CDISC SDS FDA Submission Database
 Submission Domain Standards (SDS) Version 3 Interventions Events Findings Other Exposure AE Labs Incl Excl* Demog Con. Meds Disposition Vitals Subj Char* RELATES* Extra Data* Subst Use* * New in Version 3 Med. Hist Phys. Exam ECG QS*, MB* CP*, DV* Comments* Study Design* Study Sum*
Submission Domain Standards (SDS) Version 3 Interventions Events Findings Other Exposure AE Labs Incl Excl* Demog Con. Meds Disposition Vitals Subj Char* RELATES* Extra Data* Subst Use* * New in Version 3 Med. Hist Phys. Exam ECG QS*, MB* CP*, DV* Comments* Study Design* Study Sum*


