5cfd0abac0e9b4418fd549ff2e98f2ab.ppt
- Количество слайдов: 44

Cleaning, Packaging and Sterilization of Instruments Provided by: Mc. Kesson Corporation Mc. Kesson Medical-Surgical Richmond, VA

Continuing Education Program Following established protocols for instrument processing is an important aspect of modern health care as it helps to minimize the patient’s risk for infection of the surgical site. This program reviews the most current recommended practices for instrument processing and is approved through two national sterile processing organizations. IAHCSMM and CBSPD offer certification programs for sterile processing personnel

INSTRUMENT PROCESSING There are six (6) recommended steps for Instrument Processing: 1. 2. 3. 4. 5. 6. Cleaning Inspection Packaging Sterilization Storage & Delivery Quality Assurance

STEP 1 - CLEANING Cleaning of instruments should be performed in a designated area, immediately after the surgical procedure. Quick cleaning removes blood or debris much easier, and can minimize corrosion, pitting or instrument staining.

STEP 1 - CLEANING Whenever possible, it is best to mechanically clean instruments with an ultrasonic or washer using a neutral p. H detergent. Mechanical cleaning has been proven to be up to 16 times more effective over manual cleaning.

STEP 1 - CLEANING Ultrasonic cleaners use high frequency sound waves to create microscopic bubbles that collapse. This “cavitation” process is very effective in getting into the serrations, box locks and crevices of instruments.

STEP 1 - CLEANING After cleaning, thoroughly rinse instruments with tap water to ensure loosened debris and detergents are removed. If the tap water is of poor quality, consider using treated water as a final rinse to avoid instrument staining.

STEP 2 - INSPECTION Each instrument must be critically inspected after each cleaning for residual debris or damage. Replace instruments as needed and never sterilize a “dirty” instrument.

STEP 2 - INSPECTION In addition, check each instrument for proper function and lubricate those that have “metal to metal” action. Instruments with stiff joints could be a sign of inadequate cleaning.

STEP 3 - PACKAGING Instrument packaging should be done in a clean, low contamination area using only FDA approved materials, such as: • Sterilization pouches • Sterilization wrappers • Sterilization containers

STEP 3 - PACKAGING Sterilization pouches are for packaging single instruments and small, light weight items. Paper/plastic pouches allow the user to see the packaged instruments and are available in heat-seal & self-seal design for use with all sterilization processes.
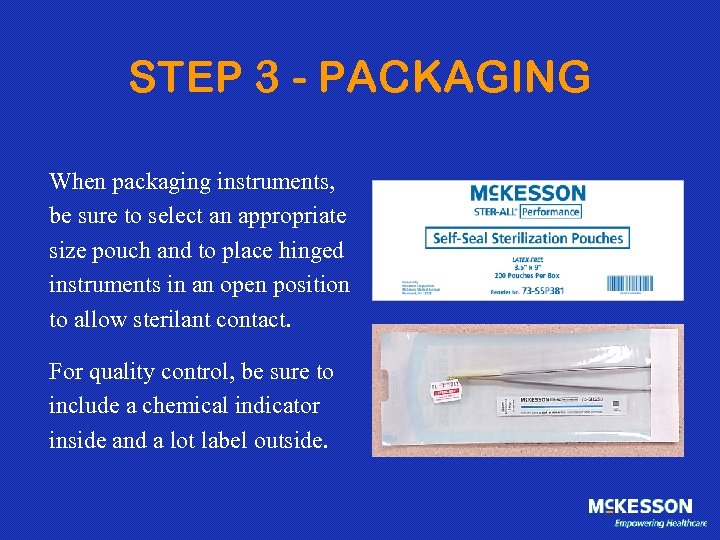
STEP 3 - PACKAGING When packaging instruments, be sure to select an appropriate size pouch and to place hinged instruments in an open position to allow sterilant contact. For quality control, be sure to include a chemical indicator inside and a lot label outside.

STEP 3 - PACKAGING To assist sterilization and aid drying, always place pouches facing each other and on edge using a pouch divider. Inexpensive pouch dividers are available for small and large sterilizers.

STEP 3 - PACKAGING Sterilization wrap is used for packaging instrument cassettes and/or trays. To allow sterilant to reach the instruments and to not trap air inside the package, cassettes and trays should be perforated. Wrap should be snug to prevent low spots that could collect condensate, but not too tight to cause strike through.

STEP 3 - PACKAGING Instrument trays should be double wrapped to maintain sterility per the wrap mfg’s instructions for use. Instrument trays should be wrapped in a way that allows for sterile presentation of the processed items.

STEP 3 - PACKAGING Wrapped packs should be sealed with 1”, 3/4” or ½” sterilization indicator tape. For quality control, be sure to include a chemical indicator inside and a lot label outside. Class 5 Class 4

STEP 3 - PACKAGING Wrapped packs (being heavier) should be placed on the lower shelf of the sterilizer. Loading heavier items on the bottom shelf and lighter items on the top, enables condensate to drain out without wetting other items in the load. To maintain sterility, packages need to be visibly dry before transporting to a sterile storage area

STEP 3 - PACKAGING Sterilization containers can be used to process instruments or sets. Containers offer excellent protection during storage and transport to the sterile field. As with wrapped trays, loading containers on the bottom shelf and lighter items on the top, enables condensate to drain away from lighter packages. Some rigid containers can be stacked during sterilization; however, all can be stacked during storage to save space

STEP 4 - STERILIZATION Steam sterilization is CDC recommended as the process of choice whenever possible. Steam under pressure is considered safe, fast and the most cost effective sterilization method for health care facilities. Gravity displacement and pre-vacuum are the two common types of steam sterilizers

STEP 4 - STERILIZATION Steam sterilizers are Class II medical devices subject to FDA approval per a 510(k) clearance letter. They are available in a wide variety of shapes and sizes for sterilization of heat-stable instruments.

STEP 4 - STERILIZATION Always process according to the sterilizer or instrument manufacturer’s validated Instructions For Use. Some common sterilizer cycle recommendations are: • 250°F/121°C for 30 minutes • 270°F/132°C for 10 minutes • 273°F/135°C for 4 minutes

STEP 4 - STERILIZATION Not all instruments can be processed at common or “normal” cycle times. For example, SYNTHES General graphic cases: • Gravity sterilizer: 132 -135°C for 22 minutes • Pre-vacuum sterilizer: 132 -135°C for 8 minutes

STEP 5 – Storage & Delivery Sterile items should be stored in a manner that reduces the potential for contamination. The shelf-life of sterile packages is event related and depends on the quality of the packaging material, storage conditions and amount of handling.

STEP 5 – Storage & Delivery Sterile packages should always be handled with care. Avoid dragging, crushing, bending, compressing or puncturing, as this will compromise the sterility of the contents. Be sure to inspect sterile packages before distributing. Do not use any package that is damaged, wet or opened.

STEP 5 – Storage & Delivery Sterility maintenance bags (dust covers) may be used to protect items that could be subjected to environmental challenges or multiple handling before use. Should be 2 -3 mils thick and applied ASAP after sterilization and after the item has cooled.

STEP 6 – Quality Assurance Chemical indicators should be placed on the outside/inside of each package to visibly show they have been processed. Biological indicators should be run at least weekly to verify sterilization and recorded as part of your formal Infection Control – QA program.

STEP 6 – Quality Assurance Chemical Indicators are available in tape, card and strip formats for use with all processes. These devices change color as indicated, for the process they are monitoring.

STEP 6 – Quality Assurance Most large steam sterilizers are pre-vacuum and require an air removal check before using. This daily test is done with a Bowie-Dick test pack. Most table top sterilizers are gravity displacement and do not require this special test.

STEP 6 – Quality Assurance Test Procedure: Place a Bowie-Dick test pack on the lowest shelf, over the drain in an empty chamber at 273°F for 3. 5 or 4 minutes. After processing, the indicator sheet inside the test pack must show a uniform color change to pass. A shortened cycle omitting dry time should be run first to properly heat up the sterilizer.

STEP 6 – Quality Assurance Biological Indicators are processed along with a load for sterilizer verification. For steam sterilizers, users purchase a 55 -60°C incubator and biological indicator vials. Sterilizer failure is noted if the BI grows during incubation. Growth is observed by a color change in the media from purple to yellow.

STEP 6 – Quality Assurance Biological Indicators can also be sent to an outside Lab for 3 rd party documentation. Mail-in systems use BI strips and pre-paid mailer envelopes. Users simply run the BI strip and then mail. A Lab Report is returned and placed on a “password protected” website: www. checkyourtest. com

STEP 6 – Quality Assurance While sterilizers can and do mechanically fail, human error is the leading cause of sterilizer failure. • • Cold start Wrong cycle Overloading Improper packaging After processing, the BI strip is mailed to an outside Laboratory for incubation. After 24 hrs, a formal Lab Report is returned to verify steam sterilization

Mc. Kesson thanks you for taking the time to review… The six (6) recommended steps for Instrument processing: 1. 2. 3. 4. 5. 6. Cleaning Inspection Packaging Sterilization Storage & Delivery Quality Assurance

CE Certificate Available This continuing education program was developed and produced by SPSmedical Supply Corp. for the Mc. Kesson Corporation. Customers who view this program may request a complimentary CE Certificate from SPSmedical.

SPSmedical Supply Corp. The largest sterilizer testing Lab in North America with over 48 sterilizers, SPSmedical serves on 10 AAMI sterilization standards committees & provides seminar Speakers to health care organizations. For technical questions and CE Certificates, please call: 1 -800 -722 -1529 or E-mail: education@spsmedical. com

References ANSI/AAMI ST 35: 2003 Safe handling and biological decontamination of reusable medical devices in health care facilities and in nonclinical settings ANSI/AAMI ST 33: 1996 Guidelines for the selection and use of reusable rigid container systems for EO & Steam sterilization in HC facilities, 2 ed

References ANSI/AAMI ST 46: 2002 Steam sterilization and sterility assurance in health care facilities ANSI/AAMI ST 37: 1996 Flash sterilization – Steam sterilization of patient care items for immediate care use

References ANSI/AAMI ST 60: 1996 ANSI/AAMI ST 66: 1999 Sterilization of health care products-Chemical indicators- Part 1: General requirements Sterilization of health care products-Chemical indicators- Part 2: Class 2 indicators for air removal test sheets and packs

Mc. Kesson Brand Sterility Assurance Products

Mc. Kesson Brand Sterility Assurance Products To assist users in meeting their sterility assurance needs, contact your local Mc. Kesson Representative and ask for the following Mc. Kesson brand quality products: PACKAGING • Self-Seal & Heat-Seal Pouches • Disposable Sterilization Wrap • Sterilization Indicator Tape • Record Keeping System MONITORING • Chemical Indicators • Biological Indicators • B-D and BI Test Packs • Incubators/Record Books

Mc. Kesson Brand Sterile Packaging Products Self-Seal Pouches Heat-Seal Pouches 73 -SSP 381 73 -SSP 382 73 -SSP 383 73 -SSP 387 73 -SSP 391 73 -HSP 250 73 -HSP 254 73 -HSP 255 73 -HSP 256 73 -HSP 257 3. 5” x 9” 5. 25” x 10” 7. 5” x 13” 8” x 16” 12” x 18” 4” x 8” 5. 25” x 10” 5. 25” x 15” 6” x 10” 7. 5” x 13”

Mc. Kesson Brand Sterile Packaging Products Sterilization Wrap Sterilization Tape 73 -HDW 1515 73 -HDW 1818 73 -HDW 2020 73 -HDW 2424 73 -HDW 3030 73 -ST 048 73 -ST 036 73 -BT 048 73 -BT 036 73 -GT 036 15” x 15” 18” x 18” 20” x 20” 24” x 24” 30” x 30” Label Applicator Load Record Label Load Record Card Records Envelope 73 -MDG 001 73 -URL 012 73 -DLC 250 73 -LEB 100 3/4” x 60 yds 1” x 60 yds

Mc. Kesson Brand Sterilization Monitoring Products Chemical Indicators Chemical Integrators 73 -SIS 250 4” Steam Strip 73 -SIL 250 8” Steam Strip 73 -GIL 250 8” EO Strip 73 -DMS 250 8” Dual Strip 73 -SSI 1000 73 -STP 025 Daily Air Removal Test 73 -SBD 030 Bowie-Dick Test Pack Steam (100 pk) Steam (1, 000 pk) Steam Test Pack

Mc. Kesson Brand Sterilization Monitoring Products Biological Indicators 73 -SCS 025 73 -SCS 100 73 -SBT 025 73 -SBT 255 73 -PCS 025 73 -NDB 055 73 -NDB 060 Steam vial (25 bx) Steam vial (100 bx) Steam Test Pack w/25 controls Steam Test Pack w/5 controls Peracetic Acid Culture Set (for STERIS System 1) 60°C Dry Block Incubator, 16 mm 60°C Dry Block Incubator, 11 mm 73 -PP 012 73 -PP 052 Mail-In System (12 bx) Mail-In System (52 bx)
5cfd0abac0e9b4418fd549ff2e98f2ab.ppt