bb77f5b3fe9a51d9f2123c90c2a799a9.ppt
- Количество слайдов: 6

Chicken Soup for the Busy Coordinator (January 2010) New online DSRB Supplementary Report Forms

New Report Forms There are 6 new online DSRB Supplementary Forms. Study Amendment Form Study Status Report Study Non-Compliance / Deviation Report UPIRTSO Reports Other Study Notifications HSA Clinical Trial Certificate Records

Supplementary Report Forms can be found under “My Studies” Click ‘View’ to see the documents you have attached, or ‘Create’ to generate reports for DSRB submission.
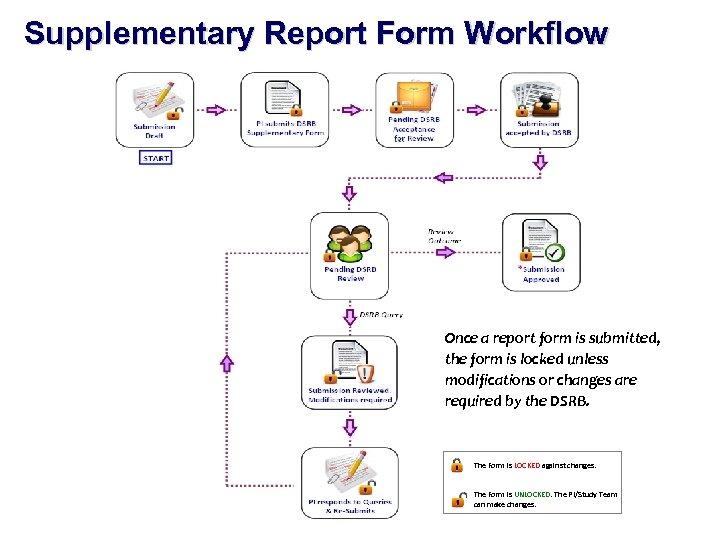
Supplementary Report Form Workflow Once a report form is submitted, the form is locked unless modifications or changes are required by the DSRB. The Form is LOCKED against changes. The Form is UNLOCKED. The PI/Study Team can make changes.

Important Note on Study Amendment Form Workflow For study amendments that include changes in the • original objectives • innovation and scientific methodology • alignment of the study to the institutions’ research objectives • image and standards of the research study These will be directed to the Department and Institutional Representatives for approval prior to submission to DSRB.

For Feedback, Suggestions or Questions on B 2 B Online : Email: B 2 BAdmin@nhg. com. sg For Feedback or Suggestions for CRCS : Email: crcs@nhg. com. sg Clinical Research Coordinator Society c/o Research & Development Office 6 Commonwealth Lane #04 -01/02 GMTI Building Singapore 149547
bb77f5b3fe9a51d9f2123c90c2a799a9.ppt