4332fef75a9d84a5b5a0d9c28f8a5e6d.ppt
- Количество слайдов: 8

Chemistry Problems Thermodynamics n n n Blase Ferraris (did these problems for Gangluff) Final Project 1996 B and 1997 D
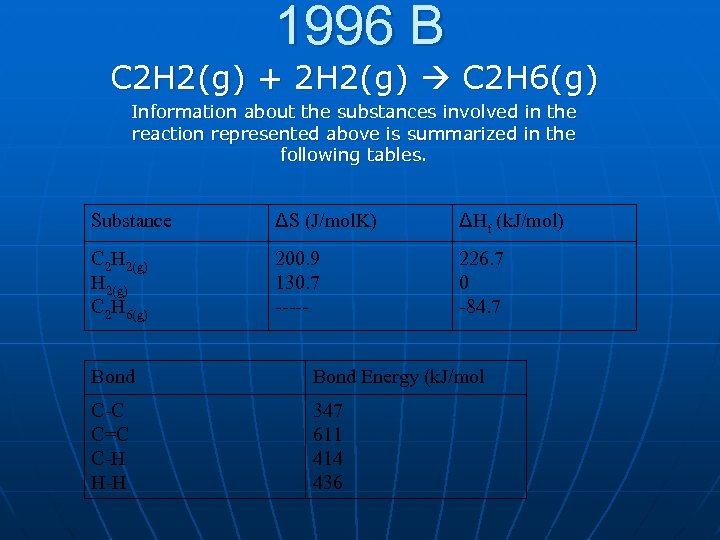
1996 B C 2 H 2(g) + 2 H 2(g) C 2 H 6(g) Information about the substances involved in the reaction represented above is summarized in the following tables. Substance ΔS (J/mol. K) ΔHf (k. J/mol) C 2 H 2(g) C 2 H 6(g) 200. 9 130. 7 ----- 226. 7 0 -84. 7 Bond Energy (k. J/mol C-C C=C C-H H-H 347 611 414 436

A. If the value of the standard entropy change ΔS, for the reaction is -232. 7 joules per mole Kelvin, calculate the standard molar entropy, ΔS, of C 2 H 6 gas. B. Calculate the value of the standard freeenergy change, ΔG, for the reaction. What does the sign of ΔG indicate about the reaction above? C. Calculate the value of the equilibrium constant, K, for the reaction at 298 K. D. Calculate the value of the C C bond energy in C 2 H 2 in kilojuoles per mole.

If the value of the standard entropy change ΔS, for the reaction is -232. 7 joules per mole Kelvin, calculate the standard molar entropy, ΔS, of C 2 H 6 gas. • Use this equation to solve for Entropy • ΔS°rxn= ΔS°products-ΔS°reactants • ΔS°rxn=ΔS°(C 2 H 6)- [ΔS°(C 2 H 2)+2*ΔS°(H 2)] • Just plug and chug • -232. 7= ΔS°(C 2 H 6)- [200. 9+2*130. 7] • Solve for variable • ΔS°(C 2 H 6)= 229. 6 J/K

Calculate the value of the standard free-energy change, ΔG, for the reaction. What does the sign of ΔG indicate about the reaction above? n ΔG=ΔH-TΔS ΔH= ΔHproducts-ΔHreactants ΔH=(- 84. 7 k. J) - (226. 7 k. J) = -311. 4 k. J units are needed for ΔS -232. 7 J/K(from part A) * 1 k. J/1000 J = - 0. 2327 k. J/K ΔG= -311. 4 k. J-T*(- 0. 2327 k. J/K) *note*(use standard temperature (25 C)) ΔG= -311. 4 k. J-(298 K)*(- 0. 2327 k. J/K)= -242. 1 k. J n Negative ΔG° therefore reaction is spontaneous n n n n

Calculate the value of the equilibrium constant, K, for the reaction at 298 K. n n n n Use the following equation: ΔG= -RTln. K Just divide by RT -lnk= ΔG/(RT) Plug and Chug -ln K = -242. 1 ÷ [(8. 31 x 10 -3) (298)] = 97. 7 e 97. 7=k K=3 x 1042

Calculate the value of the C=C bond energy in C 2 H 2 in kilojuoles per mole. n n n ΔH = bonds broken- bonds formed Plug and chug all over again - 311. 4 k. J = [(2) (436) + ΔH (C=C) + (2) (414)] - [(347) + (6) (414)] Solve for variable 820 k. J= ΔH (C=C)

ITS DONE… wait no, one more
4332fef75a9d84a5b5a0d9c28f8a5e6d.ppt