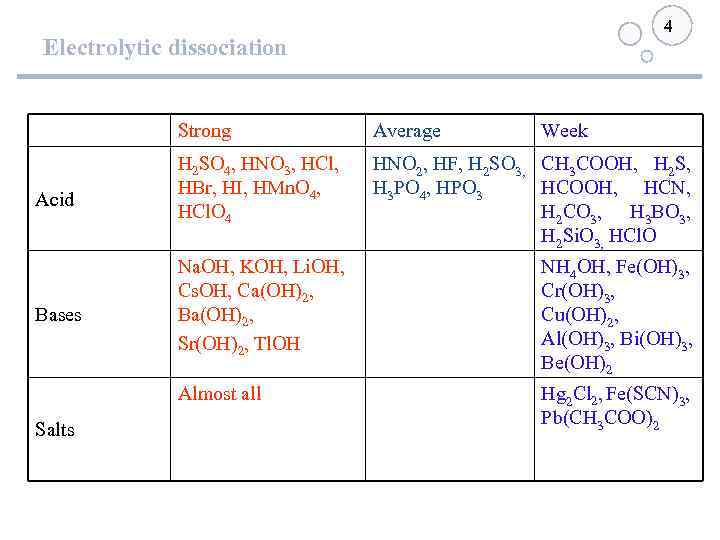
 4 Electrolytic dissociation Strong Bases Salts H 2 SO 4, HNO 3, HCl, HBr, HI, HMn. O 4, HCl. O 4 HNO 2, HF, H 2 SO 3, СН 3 СООН, H 2 S, H 3 PO 4, HPO 3 НСООН, НСN, H 2 CO 3, H 3 BO 3, H 2 Si. O 3, HCl. O Na. OH, KOH, Li. OH, Cs. OH, Ca(OH)2, Ba(OH)2, Sr(OH)2, Tl. OH NН 4 ОН, Fe(OH)3, Cr(OH)3, Cu(OH)2, Al(OH)3, Bi(OH)3, Be(OH)2 Almost all Acid Average Week Нg 2 Сl 2, Fe(SCN)3, Pb(СН 3 СОО)2
4 Electrolytic dissociation Strong Bases Salts H 2 SO 4, HNO 3, HCl, HBr, HI, HMn. O 4, HCl. O 4 HNO 2, HF, H 2 SO 3, СН 3 СООН, H 2 S, H 3 PO 4, HPO 3 НСООН, НСN, H 2 CO 3, H 3 BO 3, H 2 Si. O 3, HCl. O Na. OH, KOH, Li. OH, Cs. OH, Ca(OH)2, Ba(OH)2, Sr(OH)2, Tl. OH NН 4 ОН, Fe(OH)3, Cr(OH)3, Cu(OH)2, Al(OH)3, Bi(OH)3, Be(OH)2 Almost all Acid Average Week Нg 2 Сl 2, Fe(SCN)3, Pb(СН 3 СОО)2
![p. H : definition р. Н = –lg[H+], so [H+] = 10– р. Н, p. H : definition р. Н = –lg[H+], so [H+] = 10– р. Н,](https://present5.com/presentation/150785993_417189257/image-9.jpg) p. H : definition р. Н = –lg[H+], so [H+] = 10– р. Н, р. OН = –lg[OH–], so[OH–] = 10– р. OН, p. H + p. OH = 14 7
p. H : definition р. Н = –lg[H+], so [H+] = 10– р. Н, р. OН = –lg[OH–], so[OH–] = 10– р. OН, p. H + p. OH = 14 7