64ea04becc85c131a18b90940c383dd6.ppt
- Количество слайдов: 29
 Chemistry 125: Lecture 39 January 13, 2010 Reaction Order Information, Understanding Changes in Bond Dissociation Energies, and Predicting Rate Constants This For copyright notice see final page of this file
Chemistry 125: Lecture 39 January 13, 2010 Reaction Order Information, Understanding Changes in Bond Dissociation Energies, and Predicting Rate Constants This For copyright notice see final page of this file
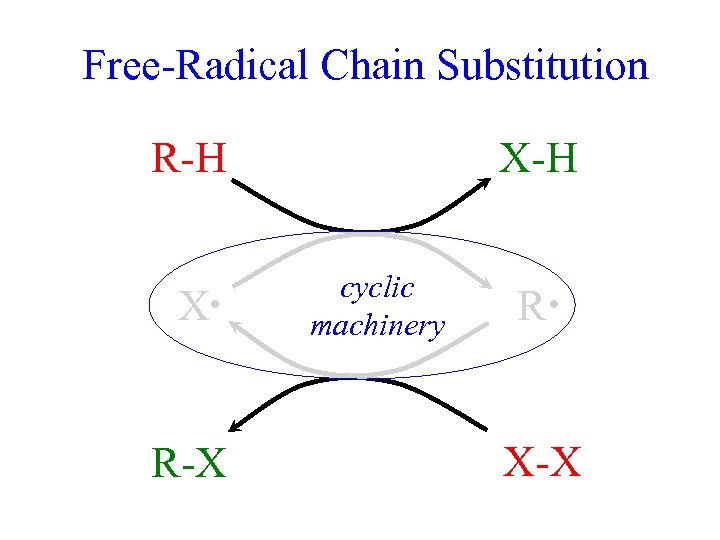 Free-Radical Chain Substitution R-H X • R-X X-H cyclic machinery R • X-X
Free-Radical Chain Substitution R-H X • R-X X-H cyclic machinery R • X-X
 Possibility of Halogenation (Mechanism for Reasonable Rate) (Equilibrium) H 3 C • X • H 3 C-H + X 2 HX H 3 CX HX X 2 + 3 Cost Step 1 F 105 136 142 136 37 31 37 Cl ” 103 163 103 58 2 58 88 151 46 Br ” 46 17 88 71 141 36 I ” 36 34 71 Return Step 2 115 84 72 58 Profit 78 251 26 187 26 160 22 129 109 24 9 12 How can we predict activation energy & k?
Possibility of Halogenation (Mechanism for Reasonable Rate) (Equilibrium) H 3 C • X • H 3 C-H + X 2 HX H 3 CX HX X 2 + 3 Cost Step 1 F 105 136 142 136 37 31 37 Cl ” 103 163 103 58 2 58 88 151 46 Br ” 46 17 88 71 141 36 I ” 36 34 71 Return Step 2 115 84 72 58 Profit 78 251 26 187 26 160 22 129 109 24 9 12 How can we predict activation energy & k?
 Digression: on Reaction Order The kinetic analogue of the Law of Mass Action (i. e. dependance of rate on concentrations) can provide insight about reaction mechanism.
Digression: on Reaction Order The kinetic analogue of the Law of Mass Action (i. e. dependance of rate on concentrations) can provide insight about reaction mechanism.
 Could use a single tap “twice” as large Rate (amount per second) Doubled Rate Chemists can also change [Concentration]
Could use a single tap “twice” as large Rate (amount per second) Doubled Rate Chemists can also change [Concentration]
![Rate “Laws”: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Rate “Laws”: Kinetic Order Rate = d [Prod] / d t = k concentration(s)?](https://present5.com/presentation/64ea04becc85c131a18b90940c383dd6/image-6.jpg) Rate “Laws”: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Dependent on Mechanism Discovered by Experiment Simple One-Step Reactions 0 th Order: Rate = k
Rate “Laws”: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Dependent on Mechanism Discovered by Experiment Simple One-Step Reactions 0 th Order: Rate = k
 0 th Order Kinetics Would more sheep give a faster rate? Photo: Antonio Vidigal by permission “Substrate” Catalyst e. g. enzyme NO! (saturation) Rate [Catalyst]1 [Substrate]0 But if the catalysis was not initially recognized.
0 th Order Kinetics Would more sheep give a faster rate? Photo: Antonio Vidigal by permission “Substrate” Catalyst e. g. enzyme NO! (saturation) Rate [Catalyst]1 [Substrate]0 But if the catalysis was not initially recognized.
![Rate “Laws”: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Rate “Laws”: Kinetic Order Rate = d [Prod] / d t = k concentration(s)?](https://present5.com/presentation/64ea04becc85c131a18b90940c383dd6/image-8.jpg) Rate “Laws”: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Dependent on Mechanism Discovered by Experiment Simple One-Step Reactions 0 th Order: Rate = k 1 st Order: Rate = k [A] (Reasonable)
Rate “Laws”: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Dependent on Mechanism Discovered by Experiment Simple One-Step Reactions 0 th Order: Rate = k 1 st Order: Rate = k [A] (Reasonable)
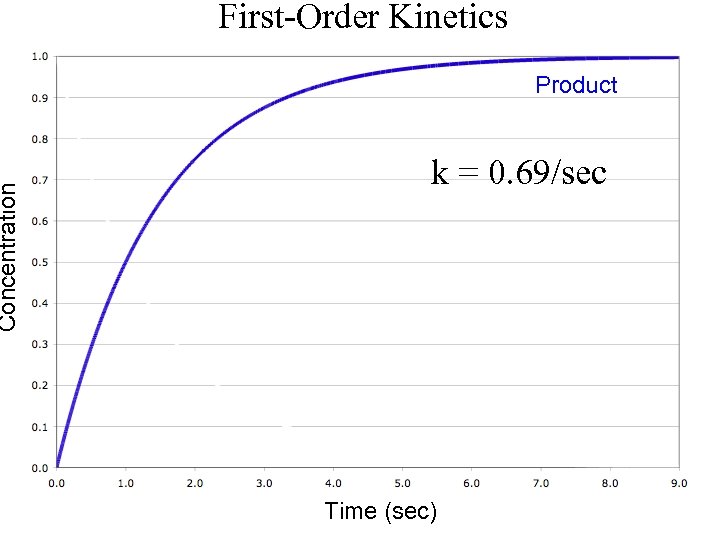 Concentration First-Order Kinetics Product k = 0. 69/sec Time (sec)
Concentration First-Order Kinetics Product k = 0. 69/sec Time (sec)
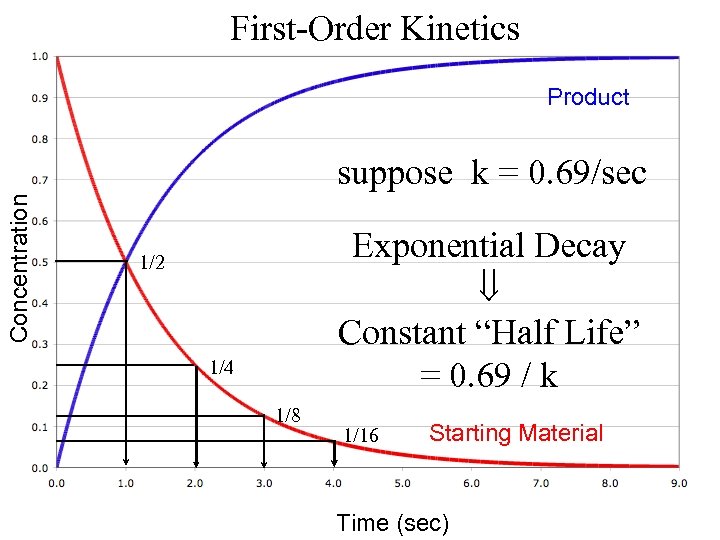 First-Order Kinetics Product Concentration suppose k = 0. 69/sec Exponential Decay Constant “Half Life” = 0. 69 / k 1/2 1/4 1/8 1/16 Starting Material Time (sec)
First-Order Kinetics Product Concentration suppose k = 0. 69/sec Exponential Decay Constant “Half Life” = 0. 69 / k 1/2 1/4 1/8 1/16 Starting Material Time (sec)
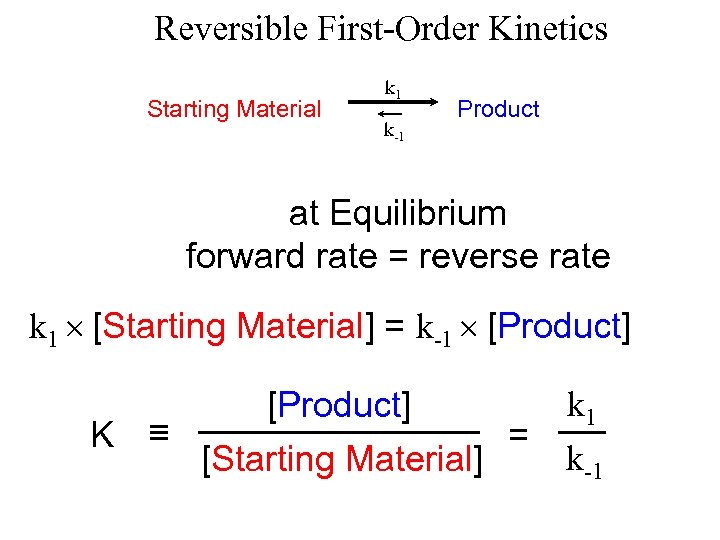 Reversible First-Order Kinetics Starting Material k 1 k-1 Product at Equilibrium forward rate = reverse rate k 1 [Starting Material] = k-1 [Product] K [Product] = [Starting Material] k 1 k-1
Reversible First-Order Kinetics Starting Material k 1 k-1 Product at Equilibrium forward rate = reverse rate k 1 [Starting Material] = k-1 [Product] K [Product] = [Starting Material] k 1 k-1
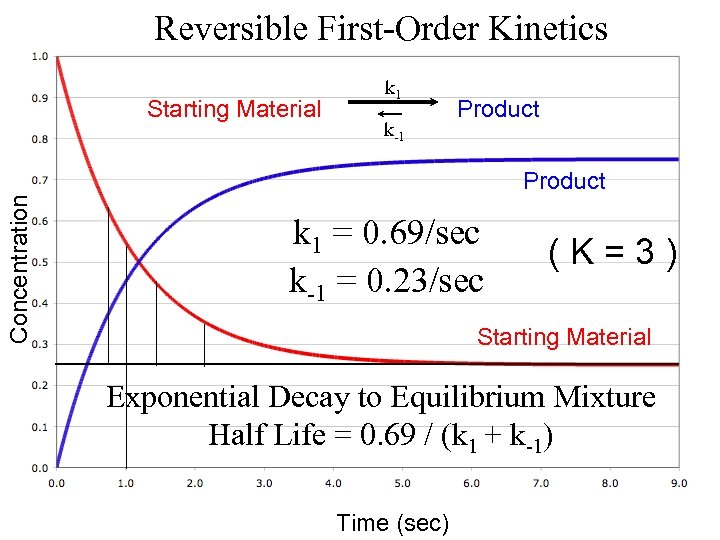 Reversible First-Order Kinetics Starting Material k 1 k-1 Product Concentration Product k 1 = 0. 69/sec k-1 = 0. 23/sec ( K = 3 ) Starting Material Exponential Decay to Equilibrium Mixture Half Life = 0. 69 / (k 1 + k-1) Time (sec)
Reversible First-Order Kinetics Starting Material k 1 k-1 Product Concentration Product k 1 = 0. 69/sec k-1 = 0. 23/sec ( K = 3 ) Starting Material Exponential Decay to Equilibrium Mixture Half Life = 0. 69 / (k 1 + k-1) Time (sec)
![Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)?](https://present5.com/presentation/64ea04becc85c131a18b90940c383dd6/image-13.jpg) Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Dependent on Mechanism Discovered by Experiment Simple One-Step Reactions 0 th Order: Rate = k 1 st Order: Rate = k [A] [B] a catalyst or [B] >> [A] 2 nd Order: Rate = k [A]2 or Rate = k [A] [B] “ 1 st Order in A” k If [B] is (effectively) constant “Pseudo” 1 st Order
Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Dependent on Mechanism Discovered by Experiment Simple One-Step Reactions 0 th Order: Rate = k 1 st Order: Rate = k [A] [B] a catalyst or [B] >> [A] 2 nd Order: Rate = k [A]2 or Rate = k [A] [B] “ 1 st Order in A” k If [B] is (effectively) constant “Pseudo” 1 st Order
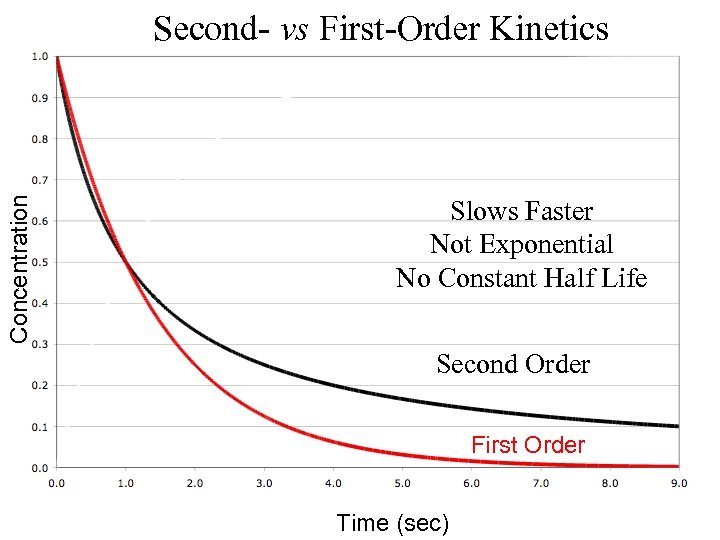 Concentration Second- vs First-Order Kinetics Slows Faster Not Exponential No Constant Half Life Second Order First Order Time (sec)
Concentration Second- vs First-Order Kinetics Slows Faster Not Exponential No Constant Half Life Second Order First Order Time (sec)
![Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)?](https://present5.com/presentation/64ea04becc85c131a18b90940c383dd6/image-15.jpg) Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Dependent on Mechanism Discovered by Experiment Complex Reactions The Rate-Limiting Step Who Cares? Rapid pre“equilibrium” with starting material reactive intermediate (low concentration)
Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Dependent on Mechanism Discovered by Experiment Complex Reactions The Rate-Limiting Step Who Cares? Rapid pre“equilibrium” with starting material reactive intermediate (low concentration)
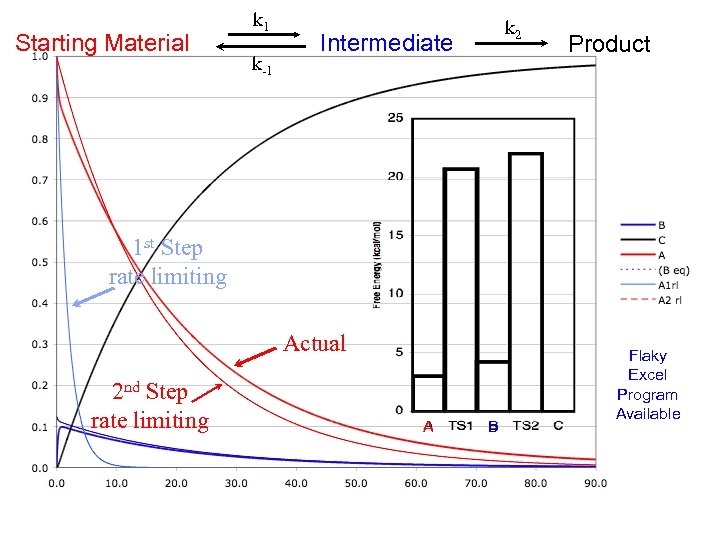 Starting Material k 1 k-1 k 2 Intermediate Product 1 st Step rate limiting Actual 2 nd Step rate limiting A B Flaky Excel Program Available
Starting Material k 1 k-1 k 2 Intermediate Product 1 st Step rate limiting Actual 2 nd Step rate limiting A B Flaky Excel Program Available
![Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)?](https://present5.com/presentation/64ea04becc85c131a18b90940c383dd6/image-17.jpg) Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Dependent on Mechanism Discovered by Experiment Complex Reactions The Rate-Limiting Step Fractional Order Importance of e. g. Rate = k [A] [B]1/4 “Dominant” species B 4 dominant / B reactive in B 4 4 B [dominant species] quantity added (how much you think you have) Others follow Law of Mass Action.
Rate Laws: Kinetic Order Rate = d [Prod] / d t = k concentration(s)? Dependent on Mechanism Discovered by Experiment Complex Reactions The Rate-Limiting Step Fractional Order Importance of e. g. Rate = k [A] [B]1/4 “Dominant” species B 4 dominant / B reactive in B 4 4 B [dominant species] quantity added (how much you think you have) Others follow Law of Mass Action.
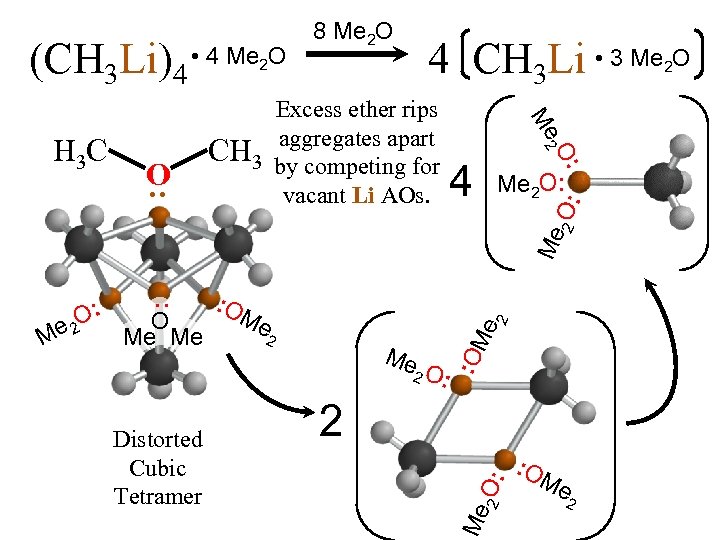 4 CH 3 Li • 3 Me O 2 Me 2 O: Me 4 O: 2 : O O Me : H 3 C Excess ether rips aggregates apart CH 3 by competing for vacant Li AOs. : 2 2 O: (CH 3 Li)4 • 4 Me O 8 Me 2 O 2 Me 2 O: Distorted Cubic Tetramer Me 2 2 O: O Me Me : O M e 2 Me O: : O Me 2
4 CH 3 Li • 3 Me O 2 Me 2 O: Me 4 O: 2 : O O Me : H 3 C Excess ether rips aggregates apart CH 3 by competing for vacant Li AOs. : 2 2 O: (CH 3 Li)4 • 4 Me O 8 Me 2 O 2 Me 2 O: Distorted Cubic Tetramer Me 2 2 O: O Me Me : O M e 2 Me O: : O Me 2
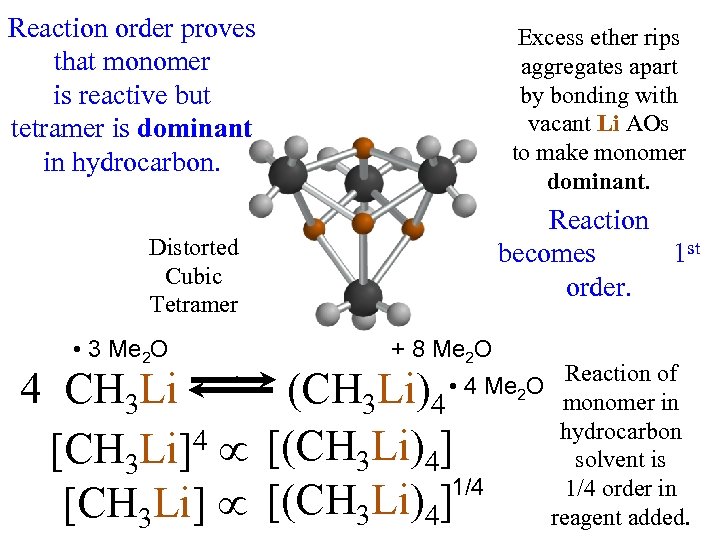 Reaction order proves that monomer is reactive but tetramer is dominant in hydrocarbon. Excess ether rips aggregates apart by bonding with vacant Li AOs to make monomer dominant. Reaction becomes 1 st order. Distorted Cubic Tetramer • 3 Me 2 O 4 CH 3 Li [CH 3 Li]4 K = [CH 3 Li] + 8 Me 2 O (CH 3 Li)4 [(CH 3 Li)4] 1/4 [(CH 3 Li)4] [CH 3 Li]4 • 4 Me 2 O Reaction of monomer in hydrocarbon solvent is 1/4 order in reagent added.
Reaction order proves that monomer is reactive but tetramer is dominant in hydrocarbon. Excess ether rips aggregates apart by bonding with vacant Li AOs to make monomer dominant. Reaction becomes 1 st order. Distorted Cubic Tetramer • 3 Me 2 O 4 CH 3 Li [CH 3 Li]4 K = [CH 3 Li] + 8 Me 2 O (CH 3 Li)4 [(CH 3 Li)4] 1/4 [(CH 3 Li)4] [CH 3 Li]4 • 4 Me 2 O Reaction of monomer in hydrocarbon solvent is 1/4 order in reagent added.
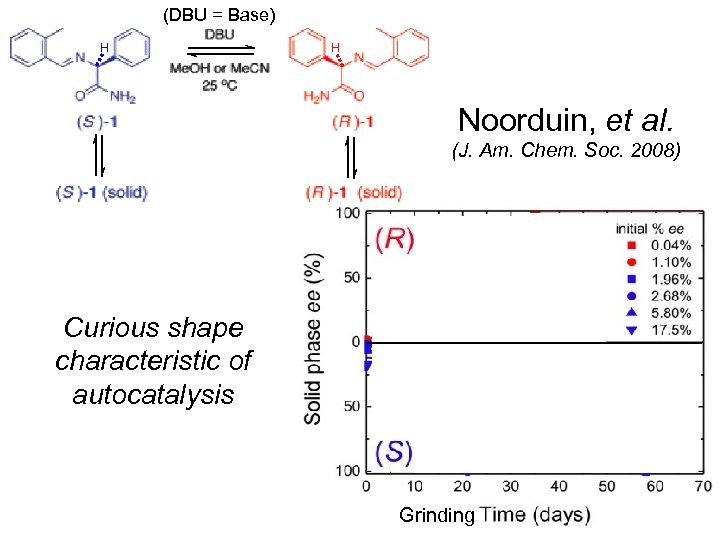 (DBU = Base) H H Noorduin, et al. (J. Am. Chem. Soc. 2008) Curious shape characteristic of autocatalysis Grinding
(DBU = Base) H H Noorduin, et al. (J. Am. Chem. Soc. 2008) Curious shape characteristic of autocatalysis Grinding
 Back to Bond Dissociation Energies for Predicting Rate Constants Free-Radical Substitution for Simplicity [minimal solvent influence]
Back to Bond Dissociation Energies for Predicting Rate Constants Free-Radical Substitution for Simplicity [minimal solvent influence]
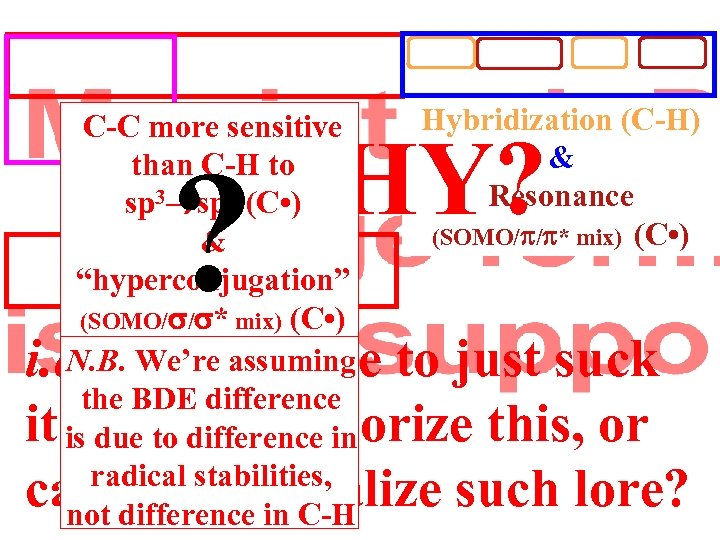 Ellison II Hybridization (C-H) C-C more sensitive than C-H to Overlap (C-X) sp 3 sp 2 (C • ) && E-Match (C-X) “hyperconjugation” (SOMO/ / * mix) (C • ) N. B. We’re assuming the BDE difference is due to difference in radical stabilities, not difference in C-H WHY? ? & Resonance (SOMO/ / * mix) (C • ) i. e. Do we have to just suck it up and memorize this, or can we rationalize such lore?
Ellison II Hybridization (C-H) C-C more sensitive than C-H to Overlap (C-X) sp 3 sp 2 (C • ) && E-Match (C-X) “hyperconjugation” (SOMO/ / * mix) (C • ) N. B. We’re assuming the BDE difference is due to difference in radical stabilities, not difference in C-H WHY? ? & Resonance (SOMO/ / * mix) (C • ) i. e. Do we have to just suck it up and memorize this, or can we rationalize such lore?
 R-X Bond Dissociation Energies (kcal/mole) R-H > R-Cl > R-Br > R-I X Phenyl (and vinyl) have good overlap; sp 2 C-X bonds. (Stabilization of starting material strengthens bond. See above) Allyl (and benzyl) are “resonance stabilized” radicals. (Stabilization of product radical weakens bonds. See above) R
R-X Bond Dissociation Energies (kcal/mole) R-H > R-Cl > R-Br > R-I X Phenyl (and vinyl) have good overlap; sp 2 C-X bonds. (Stabilization of starting material strengthens bond. See above) Allyl (and benzyl) are “resonance stabilized” radicals. (Stabilization of product radical weakens bonds. See above) R
 R-X Bond Dissociation Energies (kcal/mole) R-H > R-Cl > R-Br > R-I X Modest variation with R from methyl to t-butyl R
R-X Bond Dissociation Energies (kcal/mole) R-H > R-Cl > R-Br > R-I X Modest variation with R from methyl to t-butyl R
 BDE relative to CH 3 X (kcal/mole) R-X BDE : Alkyl Variation in Detail If this trend is due to radical stabilization by substitution, other X-R bond strengths should show the same trend. “sp 2 sigma bond to C (vs. H) preferentially stabilizes the more-substituted radicals. ” “Probably a bit of stabilization from SOMO overlap with * C-H and C-H” (not nearly as much as with * C=C and C=C in allyl or benzyl) X (C-C overlap more sensitive to hybridization than C-H overlap) Cf. Jones & Fleming, p. 479 H-R Cf. Jones & Fleming, pp. 478 -9 R
BDE relative to CH 3 X (kcal/mole) R-X BDE : Alkyl Variation in Detail If this trend is due to radical stabilization by substitution, other X-R bond strengths should show the same trend. “sp 2 sigma bond to C (vs. H) preferentially stabilizes the more-substituted radicals. ” “Probably a bit of stabilization from SOMO overlap with * C-H and C-H” (not nearly as much as with * C=C and C=C in allyl or benzyl) X (C-C overlap more sensitive to hybridization than C-H overlap) Cf. Jones & Fleming, p. 479 H-R Cf. Jones & Fleming, pp. 478 -9 R
 BDE relative to CH 3 X (kcal/mole) R-X BDE : Alkyl Variation in Detail If this trend is due to radical stabilization by substitution, other X-R bond strengths should show the same trend. X t-Butyl-R seems to show similar radical stabilization by substitution, but… H-R t Bu-R R
BDE relative to CH 3 X (kcal/mole) R-X BDE : Alkyl Variation in Detail If this trend is due to radical stabilization by substitution, other X-R bond strengths should show the same trend. X t-Butyl-R seems to show similar radical stabilization by substitution, but… H-R t Bu-R R
 BDE relative to CH 3 X (kcal/mole) R-X BDE : Alkyl Variation in Detail If this trend is due to radical stabilization by substitution, other X-R bond strengths should show the same trend. X Me-R Et-R i Pr-R Molecular Mechanics Strain Energies H-R t Bu-R R
BDE relative to CH 3 X (kcal/mole) R-X BDE : Alkyl Variation in Detail If this trend is due to radical stabilization by substitution, other X-R bond strengths should show the same trend. X Me-R Et-R i Pr-R Molecular Mechanics Strain Energies H-R t Bu-R R
 Exam Dates Wednesday, Feb. 3 Friday, Feb 26 Monday, April 5 Friday, May 7 (9 am) Grad TAs Senior Peer Tutors Eugene Douglas Hayley Israel TBA Andrew Moir Connie Wang
Exam Dates Wednesday, Feb. 3 Friday, Feb 26 Monday, April 5 Friday, May 7 (9 am) Grad TAs Senior Peer Tutors Eugene Douglas Hayley Israel TBA Andrew Moir Connie Wang
 End of Lecture 39 Jan. 13, 2010 Copyright © J. M. Mc. Bride 2010. Some rights reserved. Except for cited third-party materials, and those used by visiting speakers, all content is licensed under a Creative Commons License (Attribution-Non. Commercial-Share. Alike 3. 0). Use of this content constitutes your acceptance of the noted license and the terms and conditions of use. Materials from Wikimedia Commons are denoted by the symbol . Third party materials may be subject to additional intellectual property notices, information, or restrictions. The following attribution may be used when reusing material that is not identified as third-party content: J. M. Mc. Bride, Chem 125. License: Creative Commons BY-NC-SA 3. 0
End of Lecture 39 Jan. 13, 2010 Copyright © J. M. Mc. Bride 2010. Some rights reserved. Except for cited third-party materials, and those used by visiting speakers, all content is licensed under a Creative Commons License (Attribution-Non. Commercial-Share. Alike 3. 0). Use of this content constitutes your acceptance of the noted license and the terms and conditions of use. Materials from Wikimedia Commons are denoted by the symbol . Third party materials may be subject to additional intellectual property notices, information, or restrictions. The following attribution may be used when reusing material that is not identified as third-party content: J. M. Mc. Bride, Chem 125. License: Creative Commons BY-NC-SA 3. 0


