e990c0d7600f336545d9a88e16b227ee.ppt
- Количество слайдов: 47

Chemistry 125: Lecture 23 October 27, 2010 Valence & Constitutional Structure (1858) Formulae, Models, and Isomers (1860 s) Youthful chemists Couper and Kekulé replaced both radical and type theories with a new approach involving atomic valence and molecular structure, and rooted in the tetravalence and self-linking of carbon. Constitutional valence structures offered the first clear explanation for isomerism, and led to the invention of nomenclature, notation, and molecular models closely related to those in use today. Kekulé’s advocacy of his “sausage” notation seems ludicrous in retrospect, but Crum-Brown’s notation is strikingly modern. Half a century before direct experimental observation became conceivble, most structures of organic molecules were assigned by inspired guessing based on plausibility. Investigation of contemporary molecular models shows how adventuresome students could begin considering the implications of arrangement of atoms in space. For copyright notice see final page of this file

Theory and Types

p. 106 - on the French Type Theory Should the principle which is therein adopted be applied to the common events of life, it will be found that it is simply absurd. Suppose that some one were to systematize the formation of letters into words that formed the contents of a book.

p. 106 - on the French Type Theory Were he to begin by saying that he had discovered a certain word which would serve as a type, and from which by substitution and double decomposition all the others are to be derived, - that he by this means not only could form new words, but new books, and books almost an infinitum, - he would state certainly an empirical truth.

p. 106 - on the French Type Theory At the same time, however, his method would, judged by the light of common sense, be an absurdity. But a principle which common sense brands with absurdity, is philosophically false and a scientific blunder.

p. 107 - on the German Radical Theory I can only remark that it is not merely an unprofitable figure of language, but is injurious to science, inasmuch as it tends to arrest scientific inquiry by adopting the notion that these quasi elements contain some unknown and ultimate power which it is impossible to explain.

p. 107 - on the German Radical Theory It stifles inquiry at the very point where an explanation is demanded, by putting the seal of elements, of ultimate powers, on bodies which are known to be anything but this. Absolutely true but stated undiplomatically, especially for a newcomer.

p. 108 - Look to the Elements Science demands the strict adherence to a principle in direct contradiction to this view. That first principle, without which research cannot advance a step, dare not be ignored; namely, that a whole is simply a derivative of its parts. [? ]

p. 108 - Look to the Elements As a consequence of this, it follows that it is absolutely necessary to scientific unity and research to consider these bodies as entirely derivative, and as containing no secret ultimate power whatever, and that the properties which these so-called quasi elements possess are a direct consequence of the properties of the individual elements of which they are made up.

p. 109 - Focus on Carbon In applying this method, I propose at present to consider the single element carbon. This body is found to have two highly distinguishing characteristics: 1. It combines with equal numbers of hydrogen, chlorine, oxygen, sulfur, &c. 2. It enters into chemical union with itself. These two properties, in my opinion, explain all that is characteristic of organic chemistry.

1858 Tetravalence and Self-Linking of Carbon

C 2 as nucleus H=1 C=6 O=8 Cl = 35. 5

(Cf. Wöhler/S. C. H. Windler hoax)

"Structural" Formulae
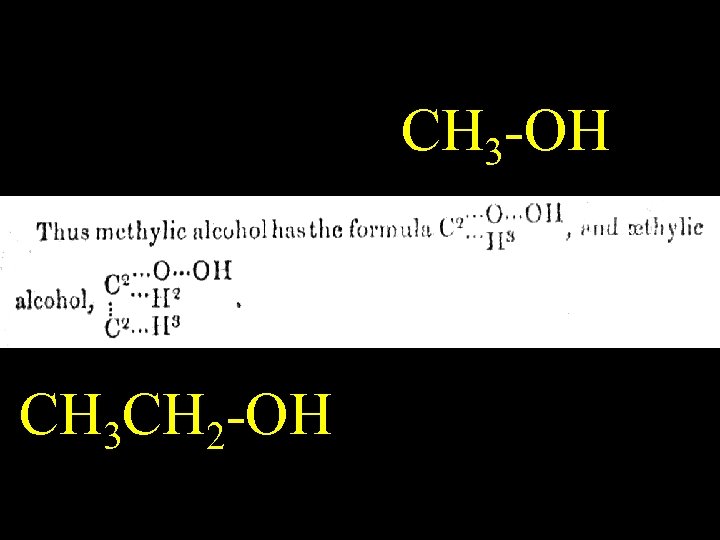
CH 3 -OH CH 3 CH 2 -OH
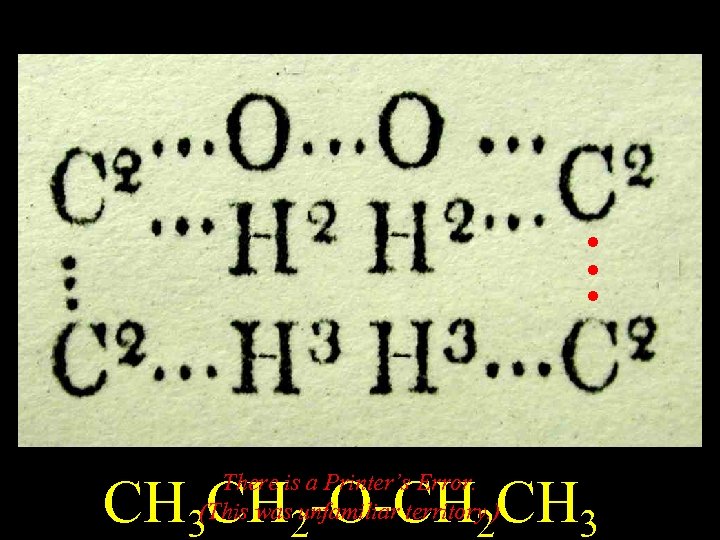
Ethyl Ether . . . There is a Printer’s Error. (This was unfamiliar territory. ) CH 3 CH 2 -O-CH 2 CH 3

Couper C 12
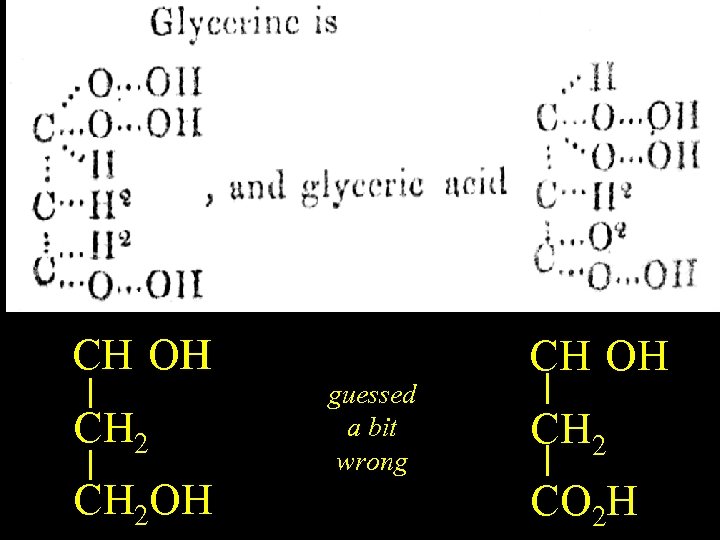
Glycerine CH(OH)2 OH CH 2 OH guessed a bit wrong CH(OH)2 OH CH 2 CO 2 H
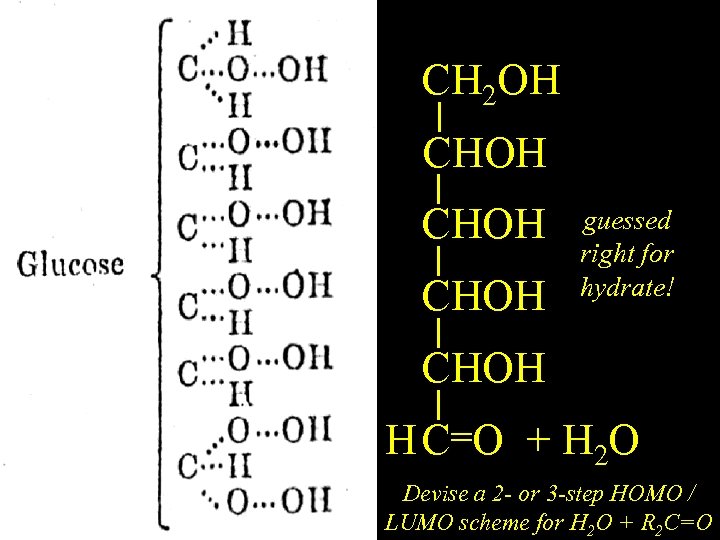
CH 2 OH English Glucose CHOH guessed right for hydrate! CHOH =O + H H CH(OH)2 2 O Devise a 2 - or 3 -step HOMO / LUMO scheme for H 2 O + R 2 C=O
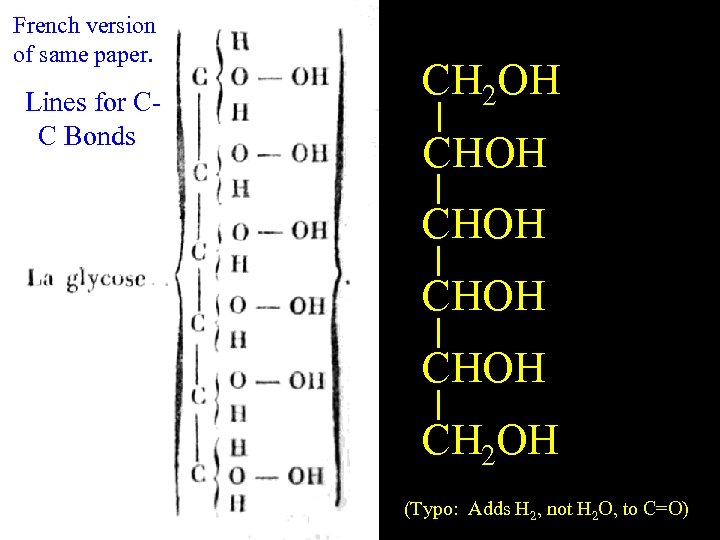
French version of same paper. Lines for CC Bonds CH 2 OH English Glucose CHOH CH 2 OH (Typo: Adds H 2, not H 2 O, to C=O)

Old Aisle Cemetery Kirkintilloch Oct. 28, 2008 Special thanks to our agent Susan Frew

from K. Hafner Angew. Chem. Int. Ed. 18, 641 (1979) Drawing by Kekulé age 13
from K. Hafner Angew. Chem. Int. Ed. 18, 641 (1979) By Kekulé age 18 Giessen for architeture

Liebig's advice to Paris-bound graduate August Kekulé (1851) There you will broaden your horizons, there you will learn a new language, there you will learn to know the life of a great city, but there you will not learn chemistry!

Kekulé in Heidelberg (1856 -58) Performed Research on (CH 3)2 As (Prof. Bunsen's cacodyl radical) in his apartment's kitchen! H H 1857 - Proposed a new type, "Marsh Gas" C H (tetravalence) H 1858 - Proposed self-linking of Carbon

Kekulé Claims Priority (1858) Observations on Mr. Couper's New Chemical Theory "In fact, in two memoirs which have appeared in Liebig's Annalen. . . I have put forward different views, which, in my opinion, should furnish a clearer insight into the constitution of chemical compounds. I may be allowed to indicate that [my first paper] lays down the principle…which I have called the basicity of atoms [we would say 'valence']. If Mr. Couper thinks he has discovered the cause of this difference of basicity in the existence of a special kind of affinity, I am the first to admit that I have no right to contest his priority in this. "

Kekulé Ghent (1865) Structure of Benzene

Kekulé Bonn (1872)

Facts, Ideas, and Words Nomenclature Notation Models

Kekulé on Notation ("Benzene" Paper, 1865) "For greater clarity I am presenting at the end of this note a table giving graphical formulae for most of the substances mentioned. The idea that these formulae are designed to express is rather well known now; so it will not be necessary to dwell upon it. I am keeping the form that I had adopted in 1859 when expressing for the first time my views on the atomic constitution of molecules. This form is nearly identical with that which M. Wurtz used in his beautiful lectures on chemical philosophy. It seems to me preferable to the modifications proposed by MM. Loschmidt and Crum-Brown. "

What SHOULD a Formula Show? Composition: Elements & Number of Atoms Berzelius formulae are adequate for this, but now we also need to show Constitution: Nature & Sequence of Bonds Isomers
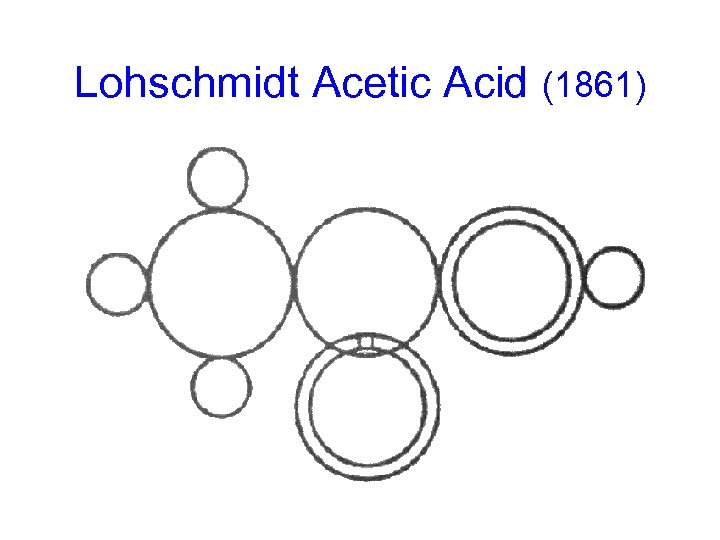
Lohschmidt Acetic Acid (1861)
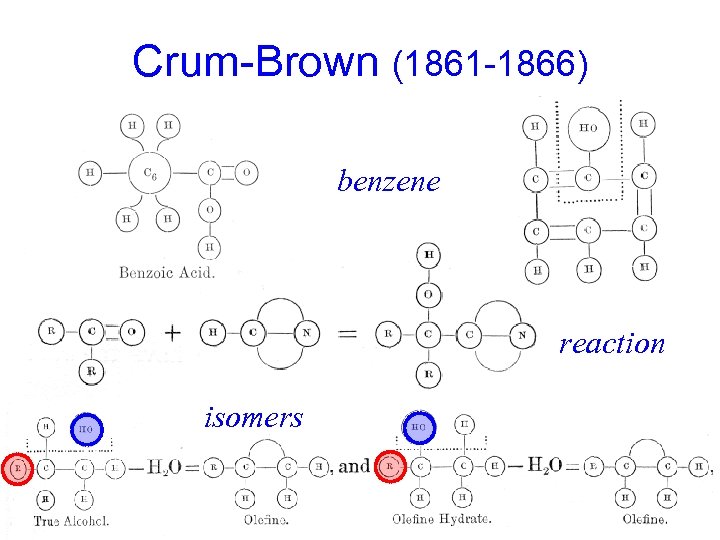
Crum-Brown (1861 -1866) benzene reaction isomers
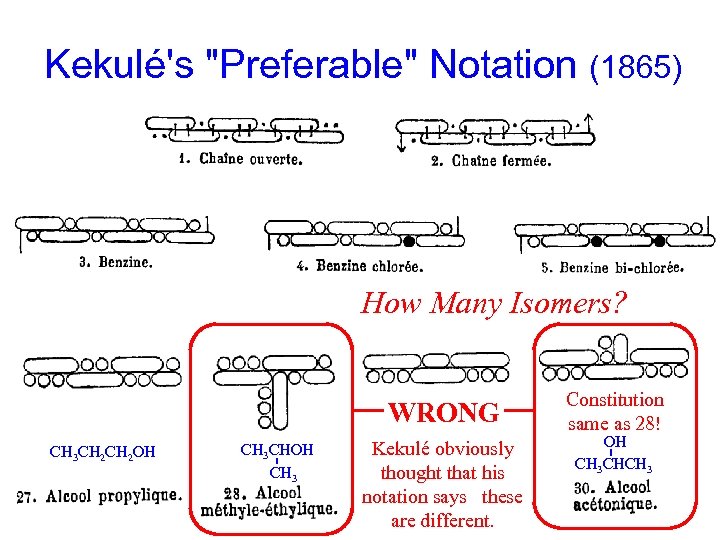
Kekulé's "Preferable" Notation (1865) How Many Isomers? What do you think Kekulé thought? WRONG Constitution same as 28! - 3 3 3 - = O OH Did he think CHOH were pictures of molecules; Kekulé obviously CH these CH CH CHCH CH thought that his what you would see in a “microscope”? CH 3 CH 2 OH 3 notation says these are different. 3 3

Molecular Models

Another class educator will see to Now, if apupil may then be asked at The intelligent thus instructed be giveglance, that in this chemical thearecitation bench, and one of them is at the proximate analysis for carbonate to form carbonicof at he requestedhe has theand the cubesonce alphabet of lime, means acid, before him will indicate the answer will walk the attention oftableclass, arresting quickly to the his in front, and fix it box what has hitherto where thein hisis open, and place on converting memory: “It is composed of onetask intomarked 6, one of the shelvesequivalentpleasant been an irksome a cube a of carbonic acid, (22)–and theory, and two marked atomic one of lime, recreation. The 8 carbon, one, (28). ” Another requested to form oxygen, two. If may be requested to the characteristics? of affinity, the giveof multiple proportions, thea the lime, the will place on the shelf bycube law he ultimate analysis, and ? same means heisomerism, another marked 20, (calcium, ) and &c. , the nomenclature, is furnished with answer: be learned in half composed marked 8, (oxygen, ) these the time may all “Carbonic acid is being the of one atom lime. These cubes being elements heretofore, and learned so required of of carbon and two of oxygen, and ever to and the pupil all not likely contact, be forgotten. of as placed in the lime is composed one atom of “What and we of being asked, calciumhave one now? ” oxygen. ” answers, “Carbonate of lime. ” Gaines Models
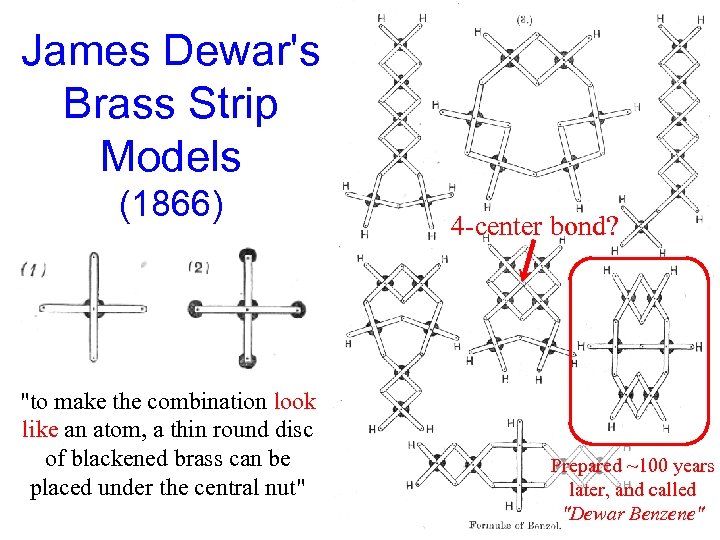
James Dewar's Brass Strip Models (1866) "to make the combination look like an atom, a thin round disc of blackened brass can be placed under the central nut" 4 -center bond? Prepared ~100 years later, and called "Dewar Benzene"

Hofmann Croquet-Ball Models (1865) Established the “standard” colors we still use! “CPK” Model of Alanine an amino acid
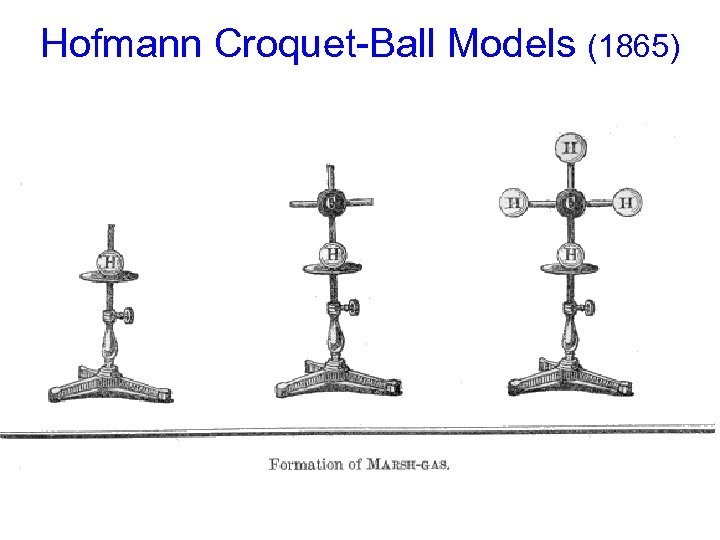
Hofmann Croquet-Ball Models (1865)
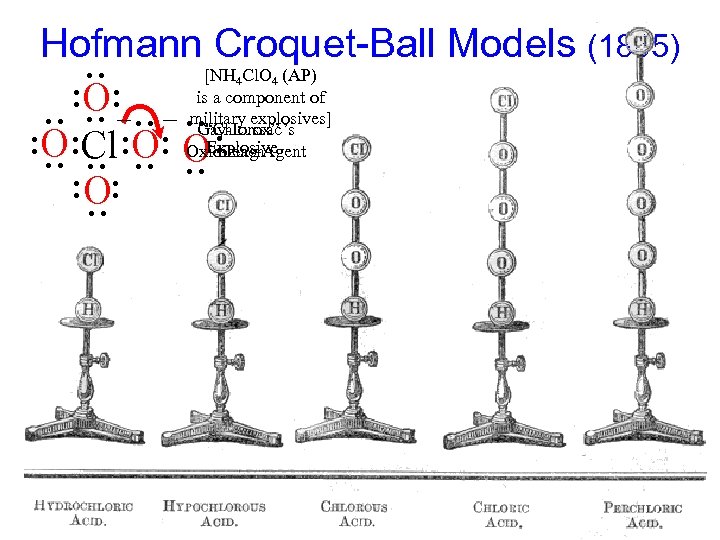
Hofmann Croquet-Ball Models (1865) [NH Cl. O (AP) • • is a component of O _ • • _ • military explosives] • • • “Chlorox” Gay-Lussac’s O Cl O OExplosive Oxidizing Agent bleach • • O • • • • • • 4 4

Hofmann Croquet-Ball Models (1865) Constitutional Models Which Hofmann models Science Museum, London by permission Successive Substitution Products Science Museum, London by permission Isomers? Showdid Hofmann prefer? of Nature & Sequence Bonds NOT Position in Space. Neither!
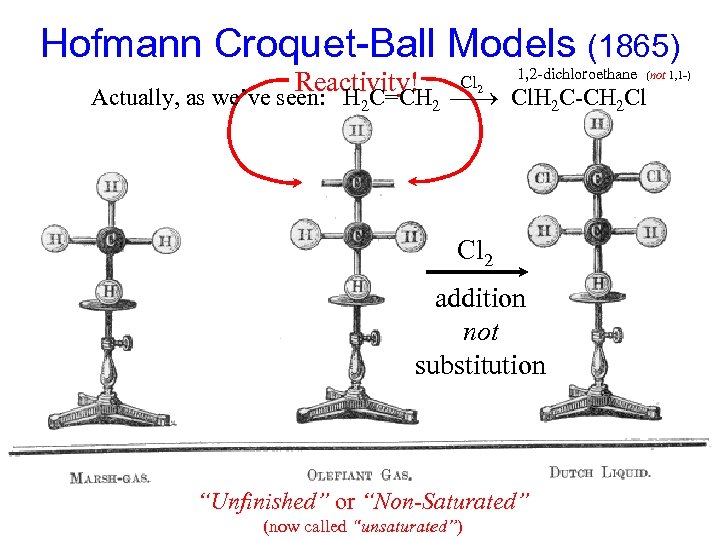
Hofmann Croquet-Ball Models (1865) Reactivity! Cl 2 1, 2 -dichloroethane (not 1, 1 -) Actually, as we’ve seen: H 2 C=CH 2 Cl. H 2 C-CH 2 Cl Cl 2 addition not substitution “Unfinished” or “Non-Saturated” (now called “unsaturated”)
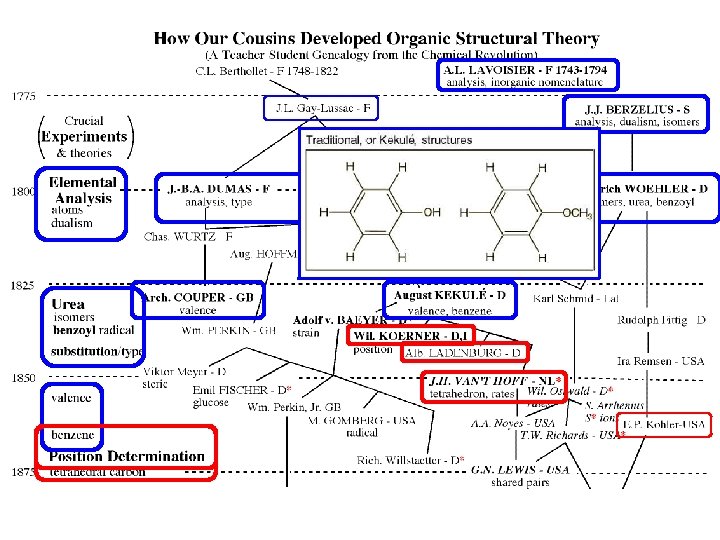
Genealogy Top

Benzene & Molecular Structure Simplest of the "aromatic" compounds 1825 Faraday isolates “bicarburet of hydrogen” ("C 2 H", actually C 6 H 6) from “gas oil”, a 1 gal. per 1000 cu. ft. by-product from preparation of illuminating gas. 1833 s Mitscherlich names it benzine from gumnames it phène” acid) benzoin (via benzoic 1836 Laurent in France from luban jawi. I bring light) fain phaino, He calls C 6 H 5(“frankincense of Java”, Arabic) ) phenyl (we abbreviate it Ph or [incidentally, benzyl is not Ph-, but rather Ph. CH 2 -]

How did Kekulé know Benzene is Hexagonal? On the Constitution of Aromatic Compounds (1866) 1) All aromatic compounds, even the simplest, are significantly richer in carbon than analogous compounds from the class of fatty substances. 2) For aromatic compounds, as for fatty compounds, there are numerous homologous substances; i. e. those whose compositions differ by n CH 2. 3) The simplest aromatic compounds contain at least six carbon atoms. 4) All reaction products from aromatic substances show a certain family similarity, constituting the group of "aromatic" compounds. More vigorous reaction can remove part of the carbon, but the major product contains at least six carbon atoms. . . Decomposition stops at this point, unless there is complete destruction of the organic group. Did he just take a wild guess? (based on tetravalence)
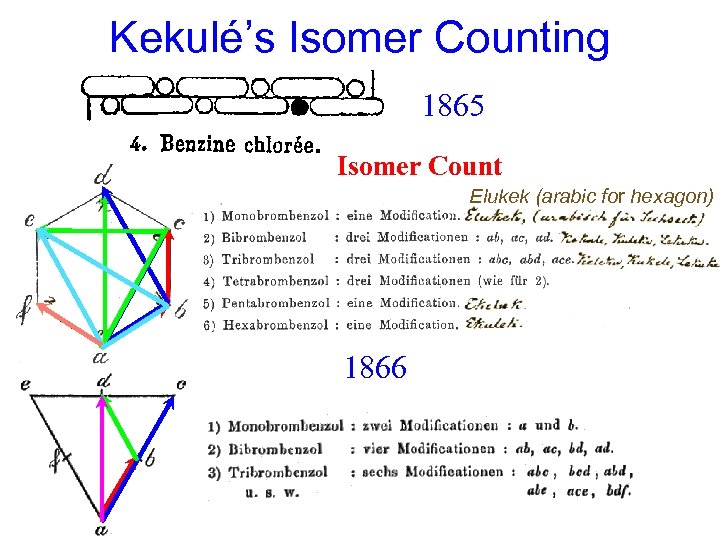
Kekulé’s Isomer Counting 1865 Isomer Count Elukek (arabic for hexagon) 1866

End of Lecture 23 Oct. 27, 2010 Copyright © J. M. Mc. Bride 2009, 2010 reserved. Except for cited third-party materials, and those used by visiting speakers, all content is licensed under a Creative Commons License (Attribution-Non. Commercial-Share. Alike 3. 0). Use of this content constitutes your acceptance of the noted license and the terms and conditions of use. Materials from Wikimedia Commons are denoted by the symbol . Third party materials may be subject to additional intellectual property notices, information, or restrictions. The following attribution may be used when reusing material that is not identified as third-party content: J. M. Mc. Bride, Chem 125. License: Creative Commons BY-NC-SA 3. 0
e990c0d7600f336545d9a88e16b227ee.ppt