ae28a88e0cfde27b78a28b8ad722d8d1.ppt
- Количество слайдов: 53

Chemical Foundations

Nature’s Chemical Language • Rattlebox moth

Nature’s Chemical Language • Play many roles – Us – Other organisms – Physical environment

Elements • Fundamental forms of matter • Can’t be broken down further • 92 occur naturally on Earth • 25 are essential for life

Most Common Elements in Living Organisms • Oxygen • Hydrogen • Carbon • Nitrogen
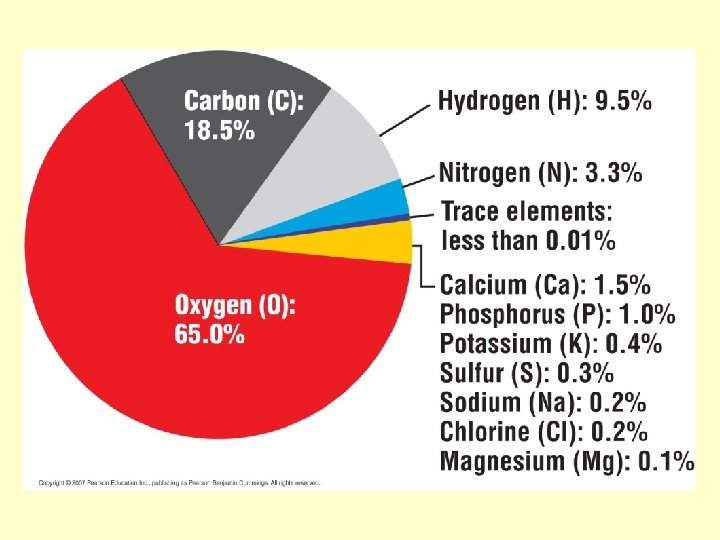

Trace Elements • Additives to food • Dietary deficiencies • Essential to health


What is an Atom? • Smallest particle that is still an element • Subatomic particles: – Protons (+) – Electrons (-) – Neutrons (no charge)

Examples of Atoms electron proton neutron Hydrogen Helium

Atomic Number • • # of protons All atoms of an element the same Atomic # H = 1 Atomic # C = 6
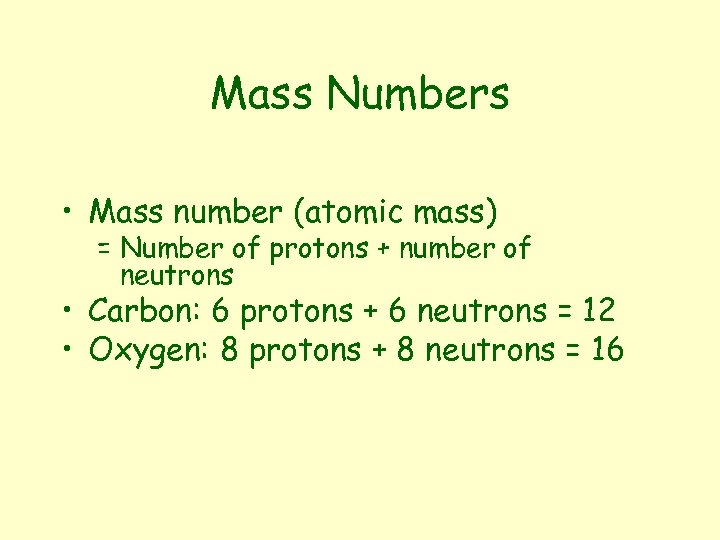
Mass Numbers • Mass number (atomic mass) = Number of protons + number of neutrons • Carbon: 6 protons + 6 neutrons = 12 • Oxygen: 8 protons + 8 neutrons = 16
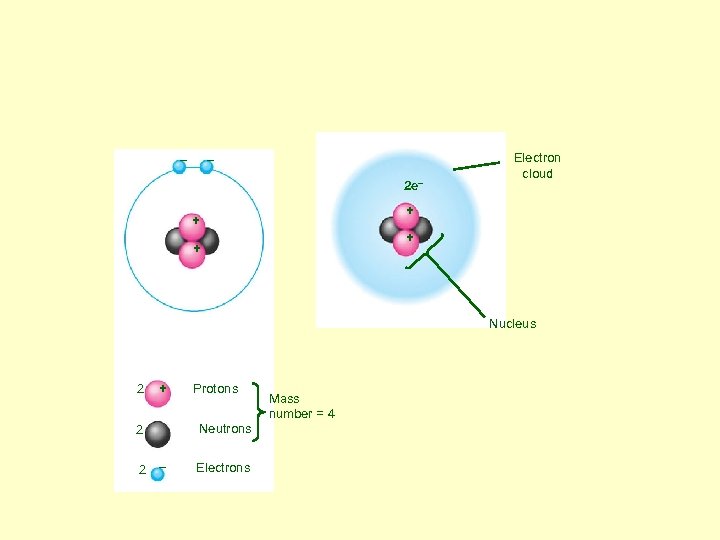
– – 2 e– Electron cloud + + Nucleus 2 + Neutrons 2 2 Protons – Electrons Mass number = 4
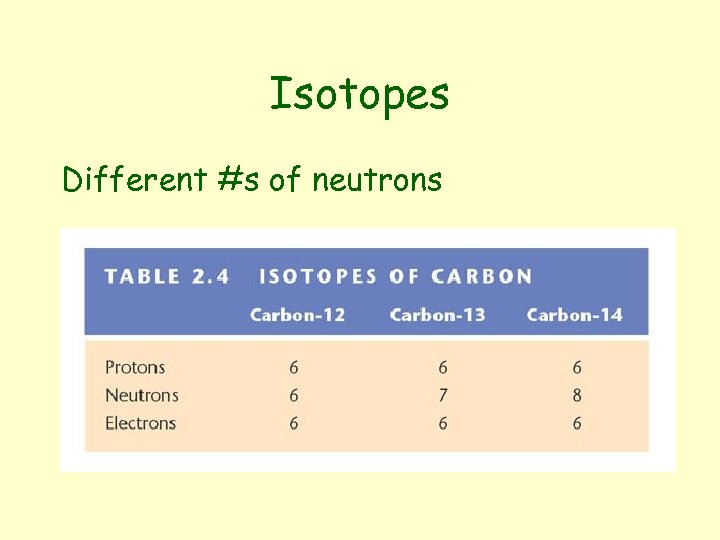
Isotopes Different #s of neutrons

Radioisotopes • Unstable nucleus • Emits energy and particles • Radioactive decay – fixed rate

Uses of Radioisotopes • Radiometric/carbon dating – Ratio of C 14 to C 12 • Tracers • Radiation therapy • Dangers
Uses of Radioisotopes

Will Atoms Interact? Depends on: # and arrangement of e-
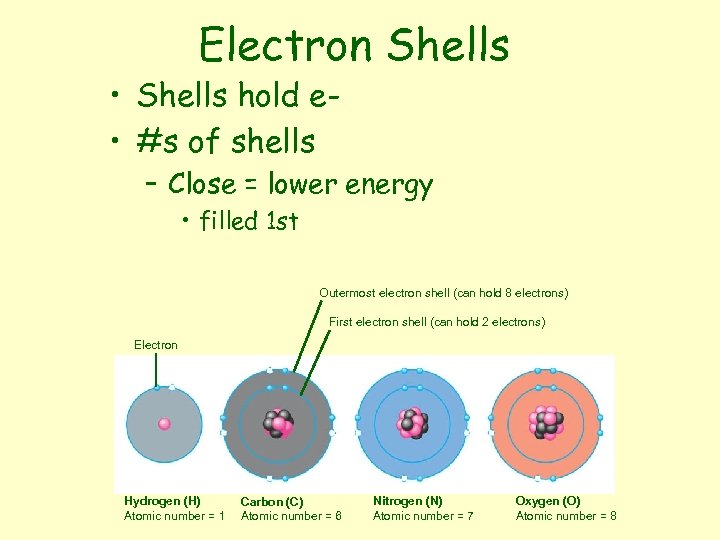
Electron Shells • Shells hold e • #s of shells – Close = lower energy • filled 1 st Outermost electron shell (can hold 8 electrons) First electron shell (can hold 2 electrons) Electron Hydrogen (H) Atomic number = 1 Carbon (C) Atomic number = 6 Nitrogen (N) Atomic number = 7 Oxygen (O) Atomic number = 8

Electron Vacancies • Unfilled shells = likely rxn • H, C, O, N – unfilled outer shells CARBON 6 p+ , 6 e- NITROGEN 7 p+ , 7 e- HYDROGEN 1 p+ , 1 e-

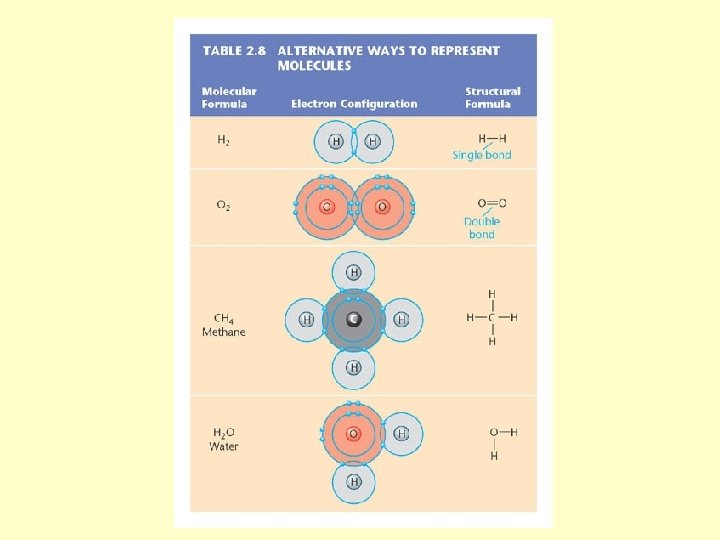
Chemical Bonds, Molecules, & Compounds • • Bonds = electron interactions Form molecules Compounds e. g. . - H 2 O Molecules e. g. - O 2

Chemical Formulas • Symbols • Glucose is C 6 H 12 O 6 – 6 carbons – 12 hydrogens – 6 oxygens

Chemical Formulas • Chemical equation shows rxn Reactants ---> Products • Start and finish with = # of atoms • HCL + Na. OH Na. Cl + H 2 O

Important Types of Bonds • Ionic Bonds (salt, Na. Cl) • Covalent Bonds (methane CH 3) • Hydrogen Bonds (water)

Ionic Bonding • • Atom loses e- -> (+) cation Atom gains e- -> (-) anion Difference charge? Results?

Formation of Na. Cl • Sodium atom (Na) – Outer shell e- ? • Chlorine atom (Cl) – Outer shell e- ? • Transfers e • Ions together as Na. Cl
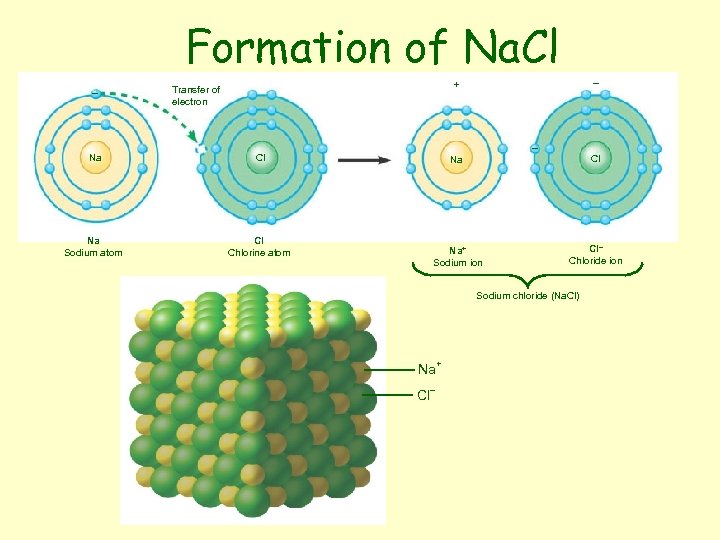
Formation of Na. Cl – – + Transfer of electron Na Na Sodium atom Cl Chlorine atom – Cl Na+ Sodium ion Cl– Chloride ion Sodium chloride (Na. Cl) Na+ Cl–
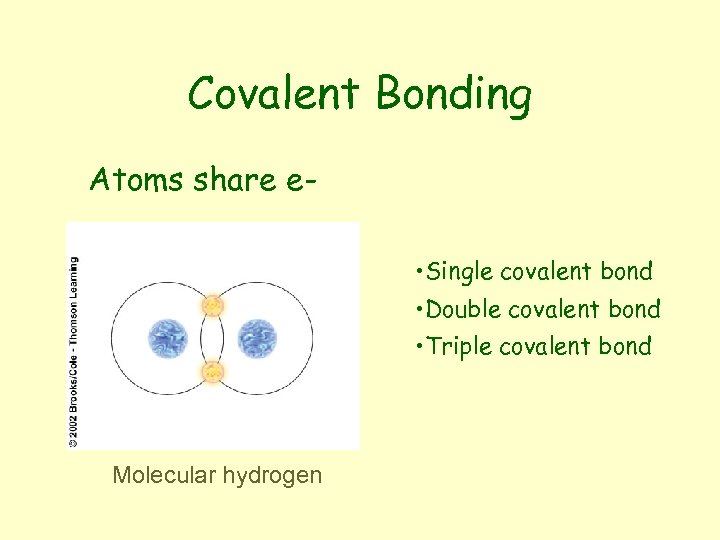
Covalent Bonding Atoms share e • Single covalent bond • Double covalent bond • Triple covalent bond Molecular hydrogen

Polar or Nonpolar Bonds? • Nonpolar: share electrons equally • Polar: unequal sharing – Water – Will electrons spend more time with the nucleus of O or H?
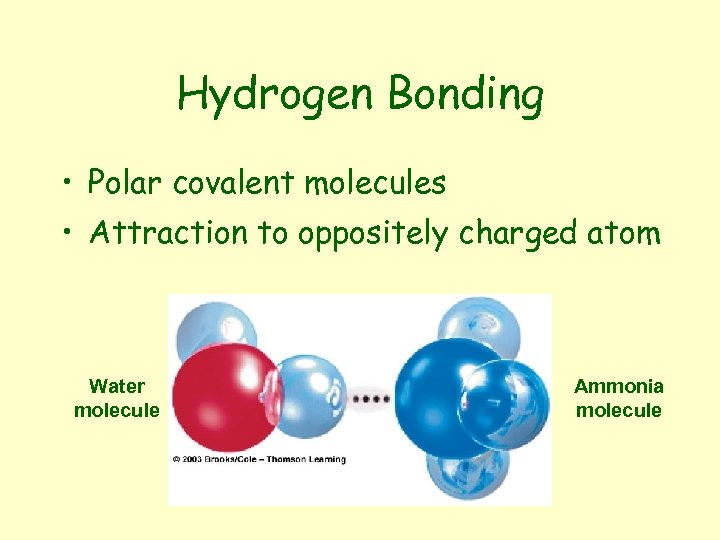
Hydrogen Bonding • Polar covalent molecules • Attraction to oppositely charged atom Water molecule Ammonia molecule
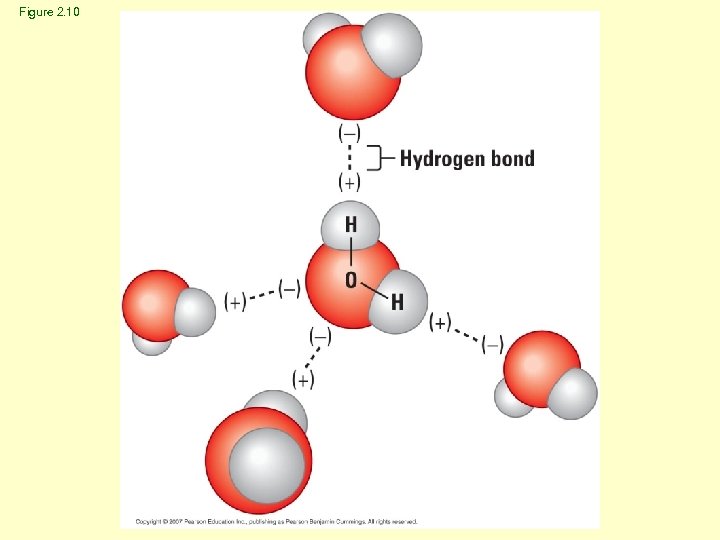
Figure 2. 10

Water • No net charge • Oxygen has a slight ______ charge • Hydrogen has a slight ______ charge O H H

Properties of Water • Temperature stabilizing – Absorbs more heat – Evaporation – Forms crystal-lattice structure – Wonders of ice!

Extra Photo 02. 11 x 2


Properties of Water • Cohesive – Due to hydrogen bonds – Increases surface tension – Vital property to many organisms • Great solvent

Figure 2. 12
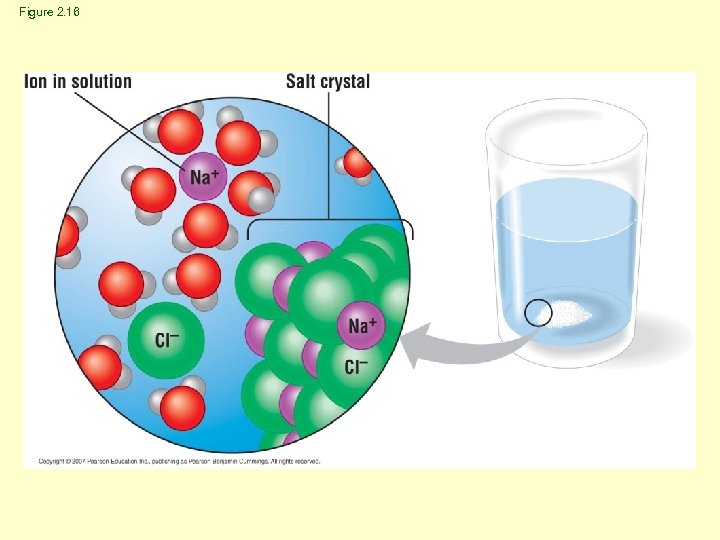
Figure 2. 16

Properties of Water • Bonds to hydrophilic substances – Bonds to polar molecules • Repels hydrophobic ones – Repels nonpolar molecules

Hydrogen Ions: + H • Unbound protons • Biological effects • Form when water ionizes

The p. H Scale • Measures H+ concentration of fluid • Log scale (1 = 10 X change) Highest H+ Lowest H+ 0 -----------7 ----------14 Acidic Neutral Basic

Examples of p. H • Pure water p. H of 7. 0 • Acidic – Stomach acid: p. H 1. 0 - 3. 0 – Lemon juice: p. H 2. 3 • Basic – Seawater: p. H 7. 8 - 8. 3 – Baking soda: p. H 9. 0

Acids & Bases • Acids – Donate H+ when dissolved in water – Acidic solutions have p. H < 7 • Bases – Accept H+ when dissolved in water – Acidic solutions have p. H > 7
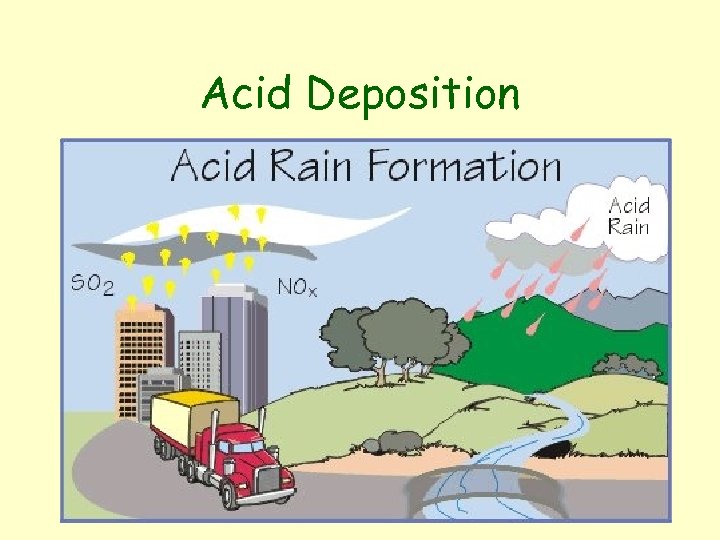
Acid Deposition
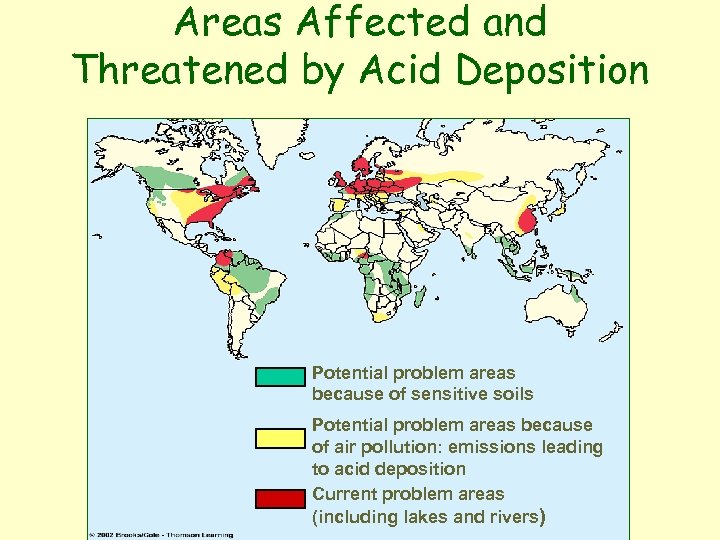
Areas Affected and Threatened by Acid Deposition Potential problem areas because of sensitive soils Potential problem areas because of air pollution: emissions leading to acid deposition Current problem areas (including lakes and rivers)

Extra Photo 02. 18 x 2

Figure 2. 18

Acid Rain

Extra Photo 02. 18 x 1

Buffer Systems • Minimize shifts in p. H • Partnership between weak acid and weak base

Blood p. H • Carbonic Acid-Bicarbonate Buffer System • When blood p. H rises, carbonic acid dissociates to form bicarbonate and H+ H 2 C 03 -----> HC 03 - + H+ • When blood p. H drops, bicarbonate binds H+ to form carbonic acid HC 03 - + H+ -----> H 2 C 03

Question of the Day My new laundry detergent proudly states “phosphate free”, why should I be interested if my detergent has phosphate? Should I continue to buy this detergent? Is it better? Safer?
ae28a88e0cfde27b78a28b8ad722d8d1.ppt