2c16dc8c4269fe4e6d889ee76c5ede5e.ppt
- Количество слайдов: 59

Chemical Bonding

IONIC BONDS • Ionic bonds - bonds formed via the electrostatic attraction between oppositely charged ions. + r • Coulomb’s law 0 = dielectric constant = 8. 84 x 10 -9 farads m-1 farad = coulomb volt-1 • Most structures in minerals contain ionic bonds.

COVALENT BONDS • Covalent bonds - bonds formed by sharing of electrons. • Many gaseous species and organic compounds contain covalent bonds, e. g. , N 2. • A few minerals, such as diamond and graphite, contain exclusively 100% covalent bonds. • In many minerals covalent bonds occur. The sharing of electrons in these bonds may not be equal. That is, covalent bonds may have some ionic character, and vice versa.

Covalent Bonds Sharing electrons such as in Si-O, S-O, P-O, C-O and C-C Double bonds when 2 orbitals shared, e. g. V=O, U=O, C=O Triple bonds when 3 orbitals shared; e. g. N=N

COVALENT BONDING Lewis dot structures Ar occurs as a monoatomic substance because its valence shell (1 s and 3 p orbitals) is completely full. An oxygen atom has an unhappy electron configuration:
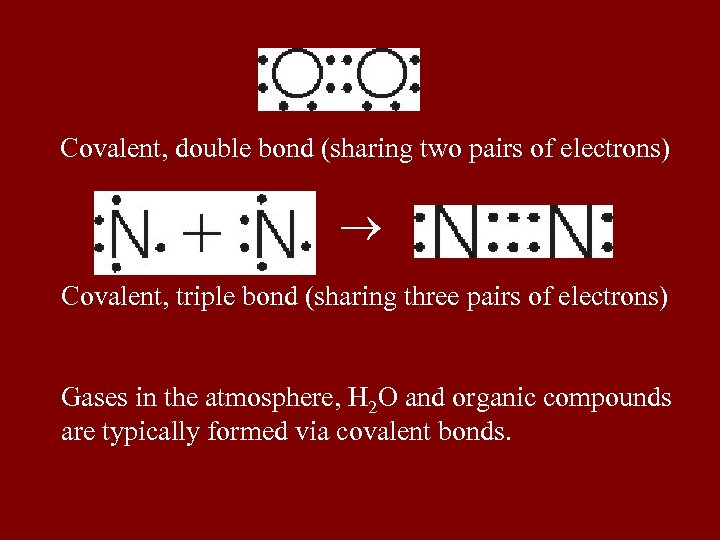
Covalent, double bond (sharing two pairs of electrons) Covalent, triple bond (sharing three pairs of electrons) Gases in the atmosphere, H 2 O and organic compounds are typically formed via covalent bonds.
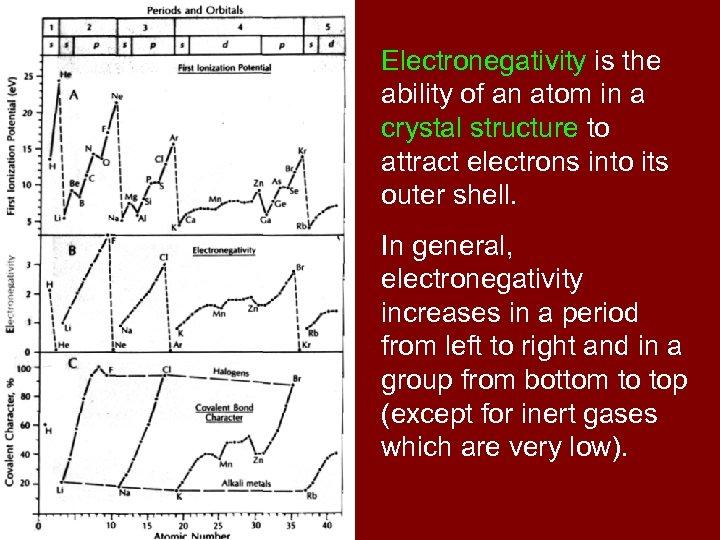
Electronegativity is the ability of an atom in a crystal structure to attract electrons into its outer shell. In general, electronegativity increases in a period from left to right and in a group from bottom to top (except for inert gases which are very low).
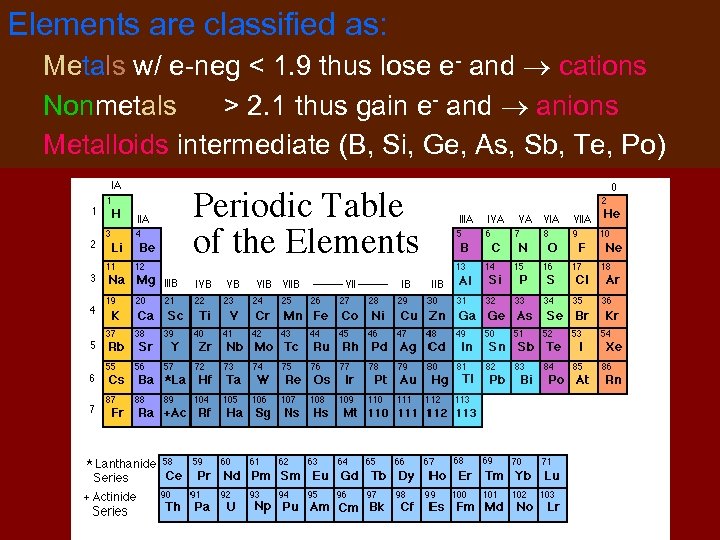
Elements are classified as: Metals w/ e-neg < 1. 9 thus lose e- and cations Nonmetals > 2. 1 thus gain e- and anions Metalloids intermediate (B, Si, Ge, As, Sb, Te, Po)

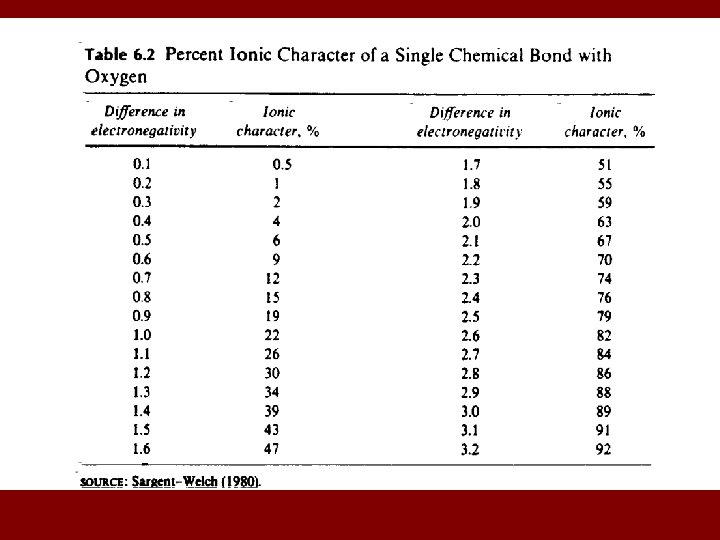
MEASURE OF IONIC/COVALENT CHARACTER • Electronegativity ( ) - measures the ability of an atom in a compound to attract electrons to itself. • Metals have low values and non-metals have high values. • We use differences in electronegativity to determine degree of ionic character of a bond. • Example: Cs. F Cs = 0. 7; F = 4. 0 F - Cs = 3. 3 > 92% ionic; < 8% covalent

OTHER EXAMPLES Zn = 1. 6; S = 2. 5 S - Zn = 2. 5 - 1. 6 = 0. 9 19% ionic; 81% covalent H 2 H = 2. 1 H - H = 0 0% ionic; 100% covalent CCl 4 C = 2. 5; Cl = 3. 0 Cl - C = 3. 0 - 2. 5 = 0. 5 6% ionic; 94% covalent

COVALENT BONDING IN MINERALS • Sulfides are the most important group of covalently bonded minerals. • Pyrite (Fe. S 2) - similar structure to halite, with Fe on the Na sites, and S 2 on Cl sites (see figure). • Bond distance between S atoms is 2. 10 Å, which is less than 2 rionic = 3. 12 Å for S 2 -! This implies strong covalent bonding. • Fe = 1. 8; S = 2. 5; Fe - S = 0. 7 12% ionic, 88% covalent

HYBRIDIZATION OF ORBITALS • Fe-S 2 bonds are a good example of hybridization. • Fe is octahedrally coordinated by S 2, but s, p and d orbitals by themselves do not allow octahedral coordination. • However, if we combine two d, one s, and three p orbitals mathematically, we can get 6 new, hybrid d 2 sp 3 orbitals which point to the corners of an octahedron.

WHY DO WE CARE IF A BOND IS IONIC OR COVALENT? • All physical and chemical properties of a compound depend on the character of the bonds. • Example: Solubility in water A general rule is that, compounds with a high percentage of ionic bonds have high solubilities in aqueous solution. e. g. , Na. Cl Cl - Na = 3. 0 - 0. 9 = 2. 1 67% ionic, relatively soluble in water C(diamond) C - C = 0 0% ionic, nearly insoluble in water

Chemical Bonds 3) Metallic Bonding Atoms are closely packed in metal structures. "touching" nearest neighbors A high density of valence e- around any given atom & also a high density of neighbor atoms around the loose valence e. The effect is to show such a general attraction for these ethat they become free to maintain an electrical neutrality: Electron gas = good conductivity Also, good ductility, relatively soft, metallic lustre.

Consider a hypothetical one-dimensional metal composed of lithium atoms, i. e. Li→ Li – Li – Li, etc. The one-dimensional metal can be extended to three dimensions using the principles illustrated on the next slide. Ψ represents an eigen function, or distribution function, which gives the electron density in the x-direction in the following diagram.
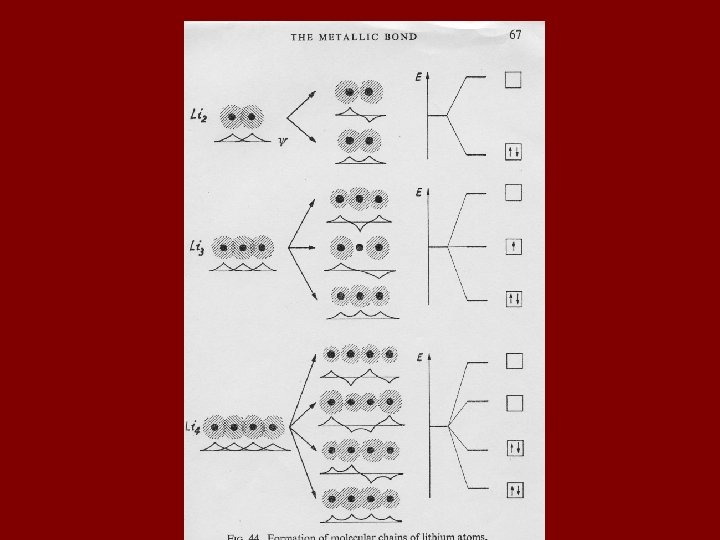
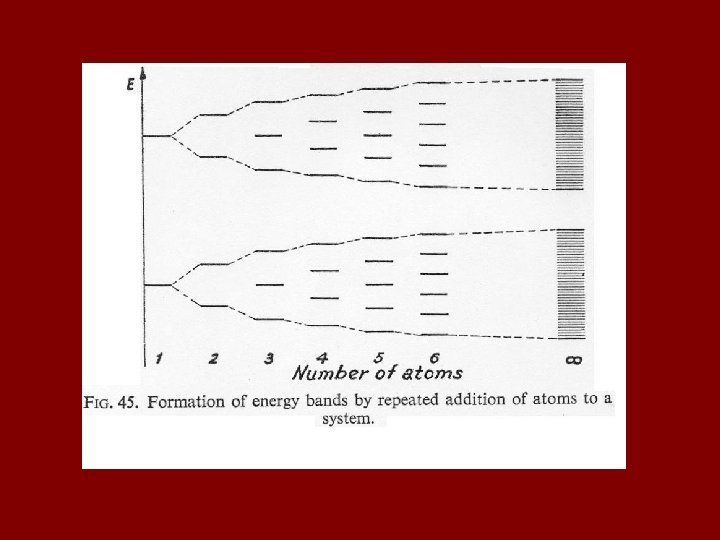

Chemical Bonds 4) Van der Waals Bonds Weakest bond Usually between neutral molecules (even large ones like graphite sheets) Produced by polar or partial polar covalent bonds or polar atoms Weakness of the bond is apparent in graphite cleavage cov Vd. W Condensed oxygen
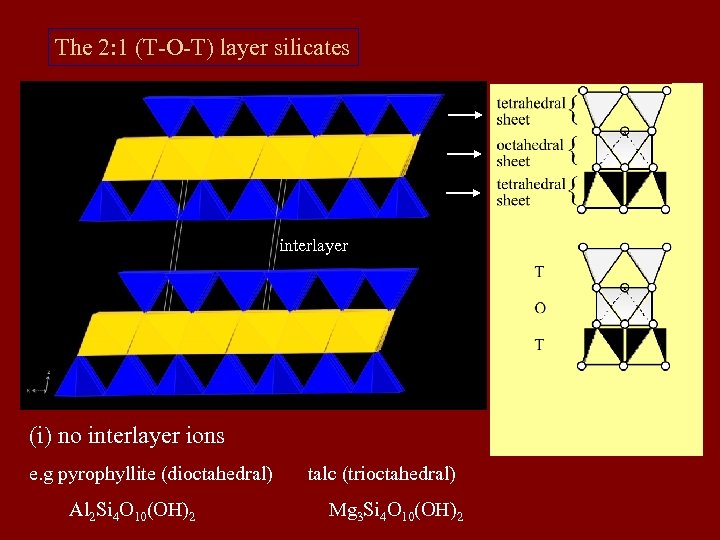
The 2: 1 (T-O-T) layer silicates interlayer (i) no interlayer ions e. g pyrophyllite (dioctahedral) Al 2 Si 4 O 10(OH)2 talc (trioctahedral) Mg 3 Si 4 O 10(OH)2

STRUCTURE OF WATER • Ionic character: O - H = 3. 5 - 2. 1 = 1. 4 39% ionic; 61% covalent - water is polar covalent. • Water is a polar substance, meaning there is a positive and a negative pole to the molecule. the water dipole 109° +

(5) HYDROGEN BONDING • Because each water molecule has a positive and a negative end, these can attract one another to form a hydrogen bond.
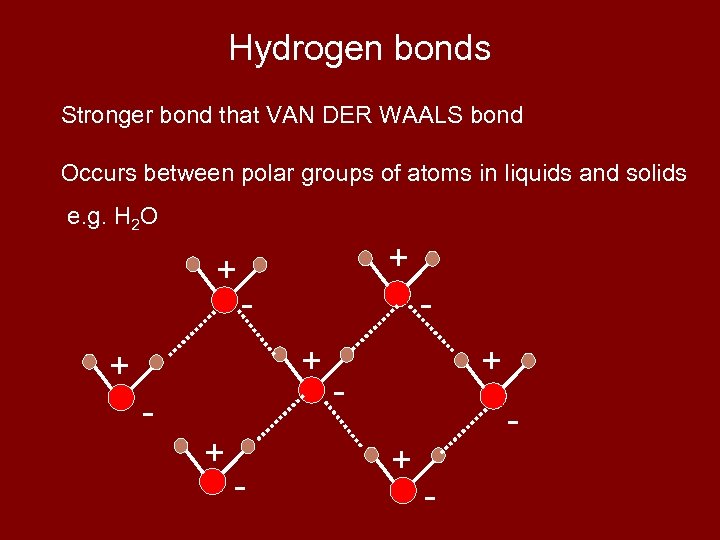
Hydrogen bonds Stronger bond that VAN DER WAALS bond Occurs between polar groups of atoms in liquids and solids e. g. H 2 O + + - + - + -

ION HYDRATION • Also because of the polar nature of water, ions will be surrounded by water dipoles (hydrated) in solution. • Hydration isolates the ions from their neighbors and neutralizes the attractive forces that hold minerals together.

Theories of Bonding • Molecular orbital theory: Most applicable to organics • Crystal field theory: Based consideration of electronic structure • Ligand field theory: Based consideration of electronic structure and symmetry of ligand orbitals
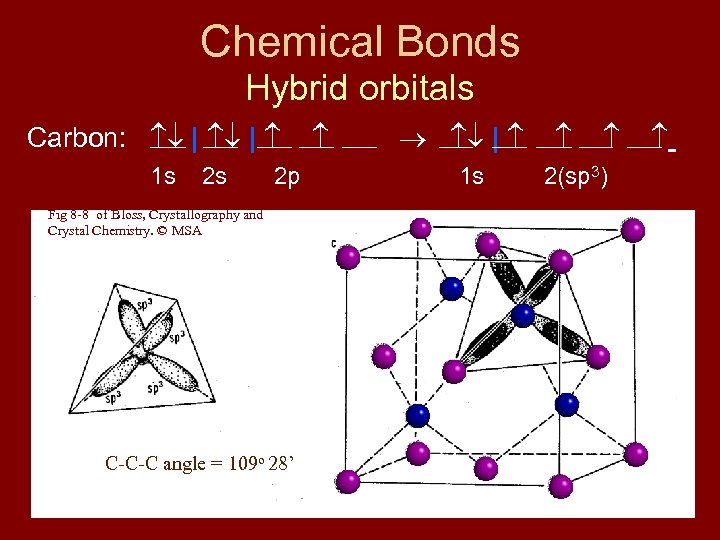
Chemical Bonds Hybrid orbitals Carbon: | | 1 s 2 s 2 p Fig 8 -8 of Bloss, Crystallography and Crystal Chemistry. © MSA C-C-C angle = 109 o 28’ | 1 s 2(sp 3)

Chemical Bonds Hybrid orbitals Alternatively: Carbon: | | 1 s 2 s 2 p | 1 s 2(sp 2) | 2 p The 3 2(sp 2) orbitals are coplanar & 120 o apart
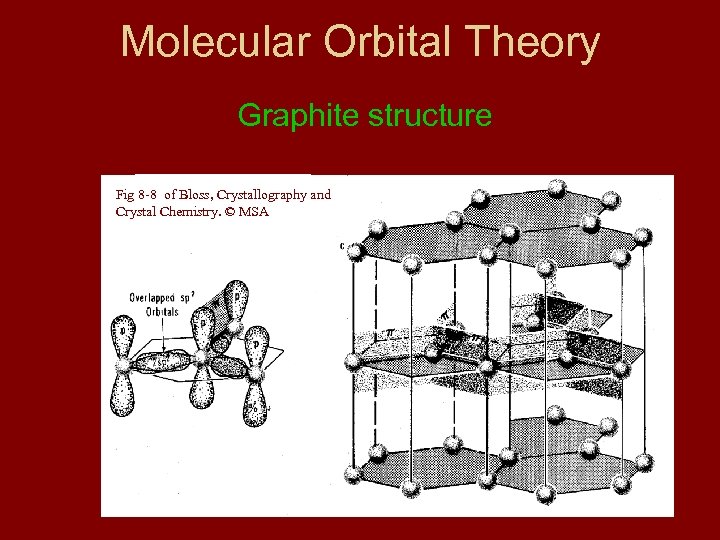
Molecular Orbital Theory Graphite structure Fig 8 -8 of Bloss, Crystallography and Crystal Chemistry. © MSA

• σ-type (sigma) bonds – orbitals are symmetrical about a line joining them • π-type (pi) bonds – orbitals generally lie in a plane • σ*-type • π *-type } degenerate orbital levels


Crystal-Field and Ligand-Field Theories Ligand-field theory can be considered to be an outgrowth of crystal-field theory. The latter considers the electronic structure of the bond between a transition metal and a coordinating anion (e. g. oxygen), which is called a ligand. The method uses group theory as applied to symmetry operations to determine the splitting of energy levels due to the symmetry properties of electrical fields in the crystal. This electrical field is called the crystal field and is caused by the symmetry of the electrical charges of the anions, dependent on their number and distance from the cation.

However, consider a transition metal cation in octahedral coordination. As the cation has lost its outer valence electrons, bonding is concerned with the d-subshell elections, whose orbitals are shown on the next slide. The dxy, dyz, and dzx orbitals lie between the Cartesian x, y and z axes, whereas the dz 2 and dx 2 -y 2 orbitals lie on the x, y and z axes. There is repulsion between the electrons of the ligands in octahedral coordination and those in the dz 2 and dx 2 -y 2, in effect raising their energy level. The dxy, dyz, and dzx orbitals, lying between the Cartesian axes, have less repulsion and, hence, are at lower energy levels. The result is splitting of the d subshell energy level, with bonding formed with the lower energy orbitals, which coincide with the corners of the octahedron.
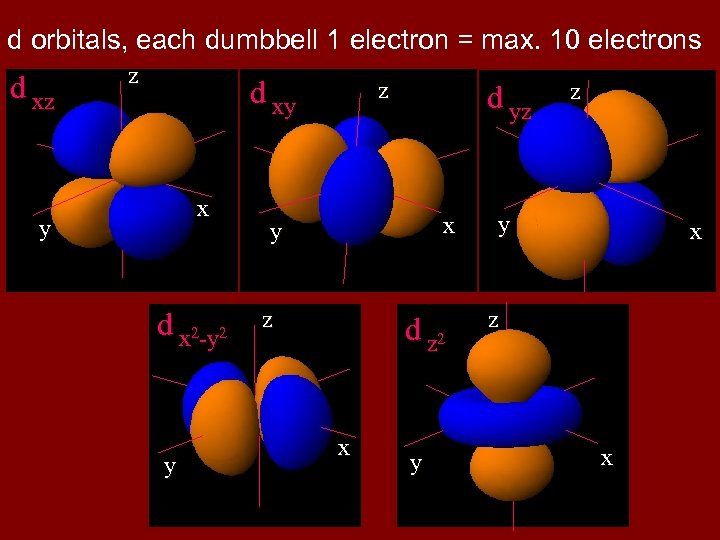
d orbitals, each dumbbell 1 electron = max. 10 electrons d xz z d xy x y d x 2 -y 2 y z d yz x y z d z 2 x y z y x z x
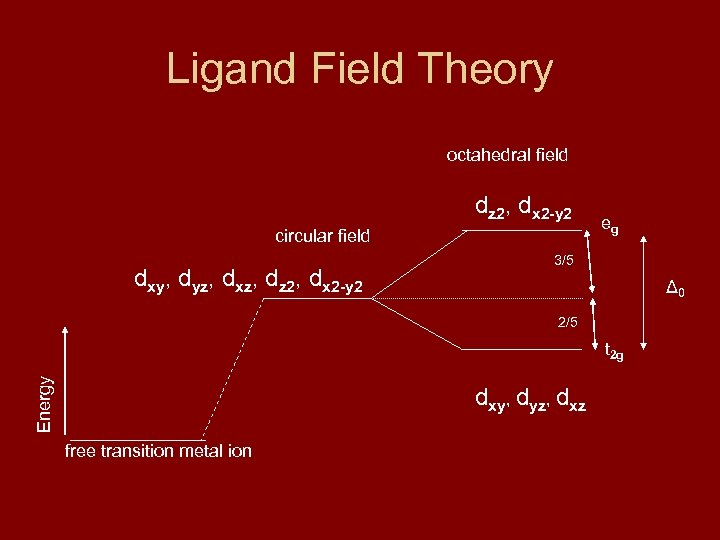
Ligand Field Theory octahedral field dz 2, dx 2 -y 2 circular field dxy, dyz, dxz, dz 2, dx 2 -y 2 eg 3/5 Δ 0 2/5 Energy t 2 g dxy, dyz, dxz free transition metal ion

In the preceding slide, Δ 0 is the energy difference between the higher energy eg level and the lower energy t 2 g level. The names given the levels are based on symmetries in group theory. Since in all there are five orbitals, the energy difference between the two level is split into 3/5 and 2/5 for the higher energy and lower energy levels, respectively. As shown below, the same sort of splitting occurs in tetrahedral coordination.

In the presence of crystal-field splitting, the electrons can assume “high spin” or “low spin” configurations. Consider Mn 2+, which has the electron configuration: 1 s 22 p 63 s 23 p 63 d 5 The splitting may occur in either of two ways shown on the next slide.


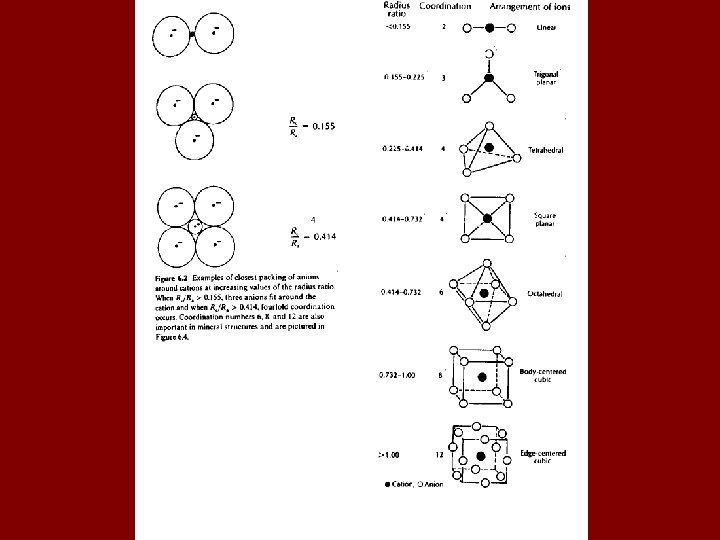
IONIC CRYSTAL STRUCTURES • Ions form a crystal such that they are “closest packed”. • This is a consequence of Coulomb’s law. • Ions arrange themselves such that interionic distances are minimized. • If we assume ions are hard, incompressible spheres (like billiard balls), we can use the concept of radius ratio as a key to explaining crystal structures.

COORDINATION NUMBER • Coordination number - Number of anions surrounding a given cation. • The coordination number can be estimated by the ratio of the radius of the cation to the radius of the anion (Rc/Ra). • As the radius ratio increases, more anions can be fit exactly around a cation. • Coordination numbers of 5, 7, 9, 10 and 11 are very rare in mineral structures.

Coordination Polyhedra Consider coordination of anions about a central cation Halite Na Cl Cl [6]Na. Cl
![Coordination Polyhedra Na • Could do the opposite, Na [6]Cl but conventionally Na Cl Coordination Polyhedra Na • Could do the opposite, Na [6]Cl but conventionally Na Cl](https://present5.com/presentation/2c16dc8c4269fe4e6d889ee76c5ede5e/image-42.jpg)
Coordination Polyhedra Na • Could do the opposite, Na [6]Cl but conventionally Na Cl Na choose the cation Na • Can predict the coordination by considering the radius ratio: RC/RA Cations are generally smaller than anions so begin with maximum ratio = 1. 0
![Common cation-coordination numbers in structures with close-packing of anions: [12] = Rc/Ra = 1 Common cation-coordination numbers in structures with close-packing of anions: [12] = Rc/Ra = 1](https://present5.com/presentation/2c16dc8c4269fe4e6d889ee76c5ede5e/image-43.jpg)
Common cation-coordination numbers in structures with close-packing of anions: [12] = Rc/Ra = 1 cubocathedron [8] = cube [6] = octahedron [4] = tetrahedron [3] = trigonal planar [2] = linear decrease

IONIC RADII • Determined from X-ray studies of interatomic distances between ions in crystals. • Ions are not really hard spheres. They can be deformed by electrical charges surrounding them. Thus, ionic radius is dependent on CN. • Example: Na CN 4 6 8 9 radius (Å) 1. 07 1. 10 1. 24 1. 40 • Ionic radii are important because ions with similar radii can substitute for one another in minerals.

TRENDS IN IONIC RADII • The ionic radii of isoelectronic series decrease with increasing atomic number for both cations and anions. • The radii of ions with the same charge in a group increase with increasing atomic number, i. e. , downward in the periodic table. • The radii of ions of the same element decrease with increasing positive charge and increase with increasing negative charge. • The radii of ions with charges of +2 and +3 among the transition metals of the fourth period decrease with increasing atomic number, implying a contraction of the electron cloud as the 3 d orbitals are filled. • Ions of different elements may have similar radii: Na+, Ca 2+, Cd 2+, Y 3+, Tl 3+, REE 3+, Th 4+ ~ 1. 00 Å (CN = 6); Hf 4+, Zr 4+ ~ 0. 80 Å (CN = 8); Fe 3+, Co 3+, Ni 3+ ~ 0. 65 Å (CN = 6)/ Li+/Mg 2+ over cross relationship)

CRYSTAL STRUCTURES We can describe ionic crystal structures as cation coordination polyhedra, outlined by the location of the anions around the cations. The nature of this polyhedron follows the radius ratio: I. One and the same cation can occur in different coordination numbers. II. The smaller the coordination number, the smaller the ionic radius. III. The smaller the average ionic radius, the smaller the average coordination number of the cation.

Pauling’s Rules for Ionic Crystals Deal with the energy state of the crystal structure 1 st Rule The cation-anion distance = radii Can use RC/RA to determine the coordination number of the cation This is our previous discussion on coordination polyhedra

Pauling’s Rules for Ionic Crystals 2 nd Rule First note that the strength of an electrostatic bond = valence / CN Na+ in Na. Cl is in [6] coordination For Na+ the strength = +1 divided by 6 = + 1/6 Cl Cl Na

Pauling’s Rules for Ionic Crystals 2 nd Rule: “the electrostatic valency principle”, An ionic structure will be stable to the extent that the sum of the strengths of electrostatic bonds that reach an anion from adjacent cations = the charge of that anion + 1/6 Na + 6 ( + 1/6 ) = +1 (sum from Na) charge of Cl = -1 These charges are equal in magnitude so the structure is stable Na + 1/6 Na - 1/ 6 Cl Na + 1/6
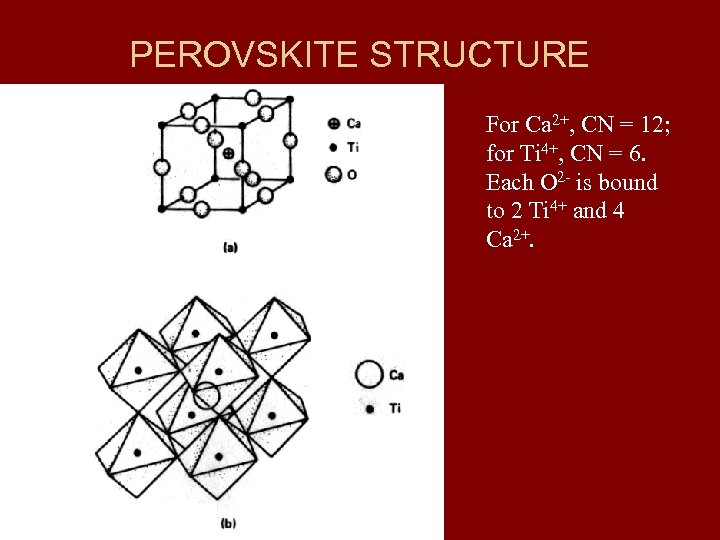
PEROVSKITE STRUCTURE For Ca 2+, CN = 12; for Ti 4+, CN = 6. Each O 2 - is bound to 2 Ti 4+ and 4 Ca 2+.

APPLICATION OF PAULING’S RULE 2 TO PEROVSKITE • Ti-O bond strength = 4/6 • Ca-O bond strength = 2/12 = 1/6 • Total bond strength reaching each oxygen ion is: 4/6 x 2 + 1/6 x 4 = 12/6 = 2 = -2 (from Ti) (from Ca) (charge on O) • Thus, Pauling’s 2 nd rule is satisfied.

GOLDSCHMIDT’S RULES 1. The ions of one element can extensively replace those of another in ionic crystals if their radii differ by less than approximately 15%. 2. Ions whose charges differ by one unit substitute readily for one another provided electrical neutrality of the crystal is maintained. If the charges differ by more than one unit, substitution is generally slight. 3. When two different ions can occupy a particular position in a crystal lattice, the ion with the higher ionic potential forms a stronger bond with the anions surrounding the site (ionic potential = charge of cation divided by the ionic radius for

RINGWOOD’S MODIFICATION OF GOLDSCHMIDT’S RULES 4. Substitutions may be limited, even when the size and charge criteria are satisfied, when the competing ions have different electronegativities and form bonds of different ionic character. This rule was proposed in 1955 by A. E. Ringwood to explain discrepancies with respect to the first three Goldschmidt rules. For example, Na+ and Cu+ have the same radius and charge, but do not substitute for one another.

COUPLED SUBSTITUTIONS Can Th 4+ substitute for Ce 3+ in monazite (Ce. PO 4)? Rule 1: When CN = 9, r. Th 4+ = 1. 17 Å, r. Ce 3+ = 1. 23Å. OK Rule 2: Only 1 charge unit difference. OK Rule 3: Ionic potential (Th 4+) = 4/1. 17 = 3. 42; ionic potential (Ce 3+) = 3/1. 23 = 2. 44, so Th 4+ is preferred! Rule 4: Th = 1. 3; Ce = 1. 1. OK But we must have a coupled substitution to maintain neutrality: Th 4+ + Si 4+ Ce 3+ + P 5+

OTHER EXAMPLES OF COUPLED SUBSTITUTION Plagioclase: Na. Al. Si 3 O 8 - Ca. Al 2 Si 2 O 8 Na+ + Si 4+ Ca 2+ + Al 3+ Gold and arsenic in pyrite (Fe. S 2): Au+ + As 3+ 2 Fe 2+ REE and Na in apatite (Ca 5(PO 4)3 F): REE 3+ + Na+ 2 Ca 2+

THREE TYPES OF TRACEELEMENT SUBSTITUTION 1) CAMOUFLAGE 2) CAPTURE 3) ADMISSION

CAMOUFLAGE This is the most obviuous kind of trace element substitution in which the substituting element has the same ionic charge and the same or nearly identical ionic radius as the major element. An example is the substitution of Ni 2+ (0. 72Å) for Mg 2+ (0. 65Å) in forsterite Mg 2 Si. O 4.

CAPTURE • Occurs when a minor element enters a crystal preferentially to the major element because it has a higher ionic potential than the major element. • For example, K-feldspar captures Ba 2+ (1. 44 Å; Z/r = 1. 39) or Sr 2+ (1. 21 Å; Z/r = 1. 65) in place of K+ (1. 46 Å, Z/r = 0. 68). • Requires coupled substitution to balance charge: K+ + Si 4+ Sr 2+ (Ba 2+) + Al 3+

ADMISSION • Involves entry of a foreign ion with an ionic potential less than that of the major ion. • Example Rb+ (1. 57 Å; Z/r = 0. 637) for K+ (1. 46 Å, Z/r = 0. 68) in K-feldspar. • The minor ion is preferred.
2c16dc8c4269fe4e6d889ee76c5ede5e.ppt