61e34073ceea68bcf24808be3601c12a.ppt
- Количество слайдов: 125

Chapter 4 Application of Designing Safer Chemicals 四川大学化学学院 2002年 7月26日 1

4. 1 Isosteric Replacement of Carbon with Silicon (用硅对碳进 行等电排置换) in the Design of Safer Chemicals l 4. 2 Designing Biodegradable l Chemicals l 4. 3 Designing Aquatically Safer Chemicals l References 四川大学化学学院

4. 1 Isosteric Replacement of Carbon with Silicon in the Design of Safer Chemicals (用硅对碳进行等电排置换) 3

Silicon is an Isostere of Carbon The degradation and oxidative metabolism of organic silicon compounds 四川大学化学学院 Differences between Silicon compounds and Carbon compounds Examples of isosteric replacement of carbon with silicon for the design of safer chemicals

4. 1. 1 Silicon is an Isosteric atom of Carbon Common Features of Silicon and Carbon n grouped in column 4 A of the Periodic Table n having many chemical similarities : tetravalent, tetrahedral, and form stable bonds with carbon. n 金刚石 四川大学化学学院 碳化硅 2002年 7月26日 5

Silicon is an Isosteric atom of Carbon Organic derivatives of silicon generally have no intrinsic toxicity, in contrast with the other Group 4 A elements germanium (Ge), tin (Sn), and lead (Pd). From a toxicity perspective, silicon is the only Group 4 A element that is a suitable replacement for carbon. In addition, silicon is an abundant, inexpensive element and one that is available in a variety of forms. 四川大学化学学院

Examples: Urethanes (尿烷), natural analogs of the neurotransmitter (神经传递质) acetylcholine (乙酰胆碱 )(1), were found to be antagonists (拮抗药)of (1) with identical dose-response curves. Interestingly, silane (silicon) was much less toxic to mice than urethane (carbon), and exhibited muscle relaxant properties. 四川大学化学学院 2002年 7月26日 7
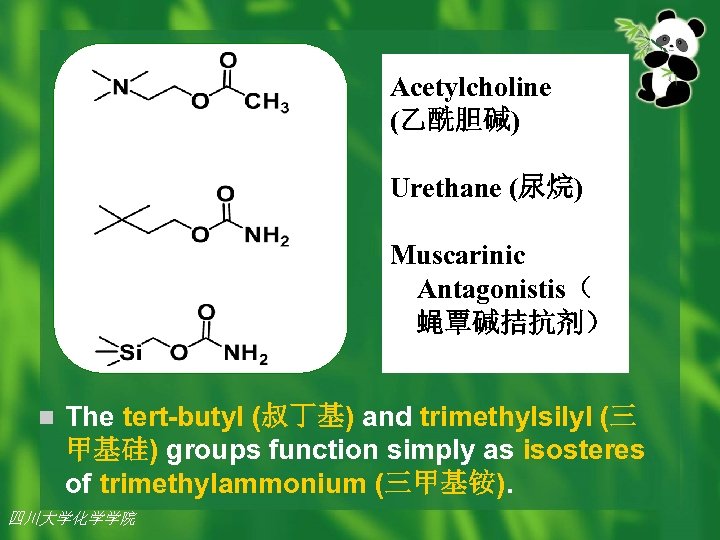
Acetylcholine (乙酰胆碱) Urethane (尿烷) Muscarinic Antagonistis( 蝇覃碱拮抗剂) n The tert-butyl (叔丁基) and trimethylsilyl (三 甲基硅) groups function simply as isosteres of trimethylammonium (三甲基铵). 四川大学化学学院

Carbamate (氨基甲酸酯) insecticide and its silicon analog were found to have similar toxicity to the house fly. Carbamate Silicon substituted Isostere of carbamate (More degradable, less toxic to human and environment) 四川大学化学学院

4. 1. 2 Differences between Silicon and Carbon Compounds Silicon is the element most similar to carbon, however it is not a generic replacement for all carbon atoms and certain strict limitations apply. Double bonds to silicon, and three-membered rings containing silicon, are unstable to air and moisture. Single bonds from silicon to heteroatoms such as nitrogen and oxygen are strong but can hydrolyze readily. 四川大学化学学院

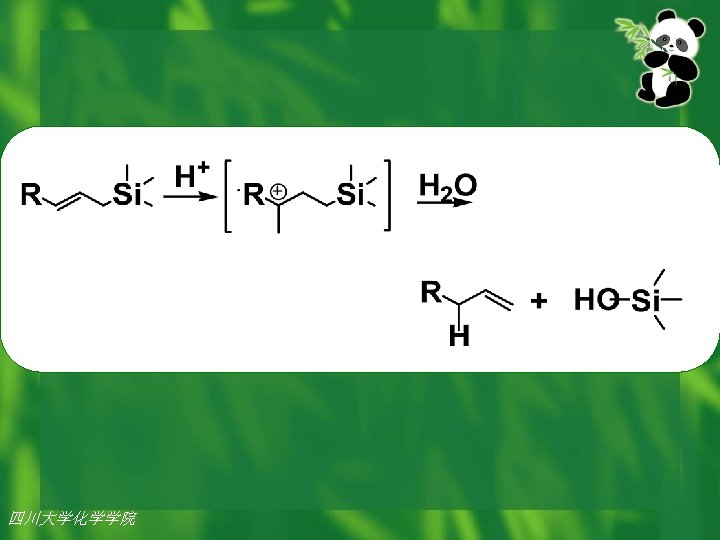
A subtle yet influential difference in atomic size exists for silicon and carbon , important differences in chemical reactivity also exist. When silicon is proximal (最接近的) to unsaturation, as in a vinyl (乙烯基) or allyl (丙稀 基) silane, the compounds are stable, but unlike their carbon analogs, they are subject to acid catalyzed silicon-carbon bond cleavage. Breaking of C-Si bond is easy. Herein lies a potential avenue (方法) for the design of environmentally degradable products. 四川大学化学学院
四川大学化学学院

4. 1. 3 The degradation and oxidative metabolism of organic silicon compound An important component of designing safer chemicals is predicting their environment fate, and both abiotic (非生物) degradation and biological oxidation can play a role. Designing chemicals that will biodegrade to innocuous products is highly desirable, and isosteric substitution of carbon with silicon in many cases may enhance abiotic degradation and biological oxidation. 四川大学化学学院

Abiotic Degradation n Currently the major environmental source of organosilane is silicone polymer (聚硅 酮)(siloxanes硅氧烷), primarily polymers of 1, 1 -dimethyl silanediol. n Siloxanes were once thought to be environmentally stable, but are now known to be depolymerized in the presence of water and soil. 四川大学化学学院 2002年 7月26日 14
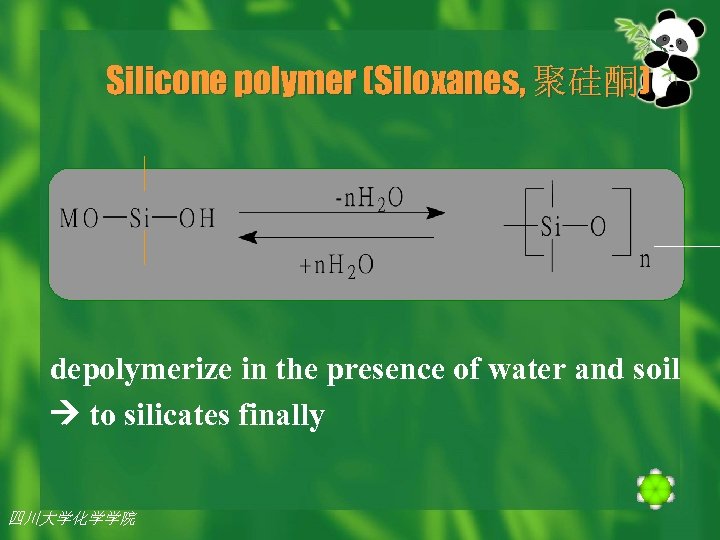
Silicone polymer (Siloxanes, 聚硅酮) depolymerize in the presence of water and soil to silicates finally 四川大学化学学院

4. 1. 4 Examples for the Design of Safer Chemicals Using Silicon Substitution for Carbon Example 1: Silane Analogs of DDT Example 2:Organosilane Fungicides (杀真菌剂) 四川大学化学学院

Silane Analogs of DDT n In an early effort to design a more benign version of DDT, a number of silane analogs such as the DDD analog were prepared with the anticipation that these would be less environmentally persistent. 四川大学化学学院 2002年 7月26日 DDT 17

Silane Analogs of DDT 四川大学化学学院

Silane Analogs of DDT n The presence of the readily oxidized siliconhydrogen bond would have been one source of instability, both in the environment and in vivo. n More significant for this research, however, was an SAR study that found the overall size of the DDT molecule strongly correlated with bioactivity, implicating the atomic size of the central silicon for the lack of insect toxicity for DDD and congeners(同 类). 四川大学化学学院 2002年 7月26日 19

Organosilane Fungicides n As a novel entry into the class of triazole (三唑) fungicides (杀真菌剂), Meberg and coworkers prepared a series of silane analogs. n One of these, flusilazole (氟苯代硅三唑) proved to be a highly effective crop fungicide(谷类防真菌) and is now a major commercial product. 四川大学化学学院 2002年 7月26日 20
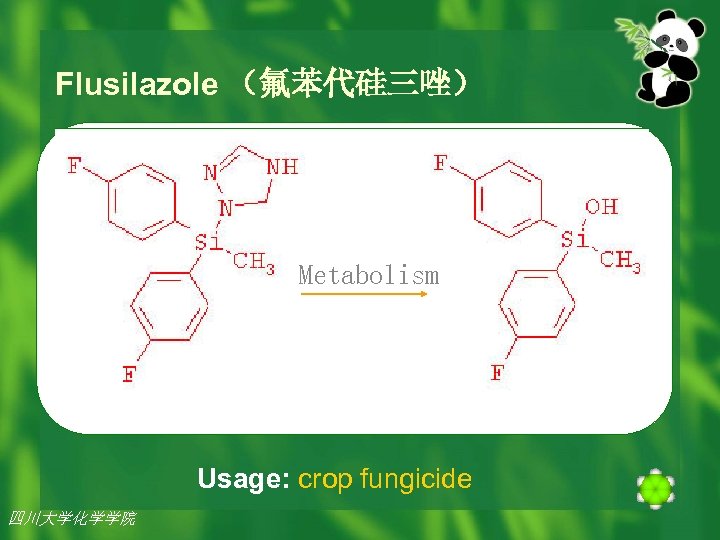
Flusilazole (氟苯代硅三唑) Metabolism Usage: crop fungicide 四川大学化学学院

4. 1 Isosteric Replacement of Carbon with Silicon (用硅对碳进行等 电排置换)in the Design of Safer Chemicals l 4. 2 Designing Biodegradable Chemicals l 4. 3 Designing Aquatically Safer Chemicals l References l 四川大学化学学院 2002年 7月26日 22

4. 2 Designing Biodegradable Chemicals 23

The safer chemical eliminates the production and release of more persistent and potentially hazardous substitutes through molecular design to enhance biodegradability to non-toxic products. Enhanced biodegradability is also a worthy goal precisely because pollution cannot always be prevented at the source. 四川大学化学学院 2002年 7月26日 24
The Microbial Basis of Biodegradation Group Contribution Method for Predicting Biodegradability 四川大学化学学院 Chemical Structure and Biodegradability Examples of Designing Biodegradable Chemicals

Hazards could not always be known or predicted: Chemicals: (resist biodegradation) exert toxic effect to biota (生物区), hardly to predict their potential toxic effect at the time of release to the environment. Moreover, bioaccumulative toxicity criteria (safe), but chronic (慢性的) or other unforeseen toxic effect (unknown). Increasing the safety of chemicals Increasing the treatability of the waste generated 四川大学化学学院 2002年 7月26日 26

4. 2. 1 The Microbial Basis of biodegradation Animals: excrete chemicals that they cannot metabolize; Plants: tend to convert chemicals into water insoluble. Microbial populations: are characterized by catabolic (代 谢 分 解 的 ) versatility, rapid growth in the presence of food, high metabolic activity and species diversity. The eventual mineralization of organic compounds (their conversion to inorganic substances such as CO 2 and water) can be attributed predominantly to microbial degradation.

The key role in biodegradation: An abundance of evidence exists to show that microorganisms are responsible for the degradation of many organic chemicals cannot be altered significantly by higher organism. Microorganisms(primarily bacteria 细菌 and fungi 真菌)are by far the most important agents of biodegradation in nature. 四川大学化学学院

Microbial degradation is the major loss mechanism for most organic chemicals in aquatic(水的) and terrestrial( 陆地) environments, and is the cornerstone of the modern wastewater treatment plant. SCU 成都活水公园 四川大学化学学院 2002年 7月26日 29

四川大学化学学院 2002年 7月26日 30

The process of biodegradation 1. An organic compound must first enter the microbial cell through the cell wall & cytoplasmic ( 细胞质) membrane. This may occur by passive diffusion or with the assistance of specific transport systems. Especially: for aquatic and terrestrial (陆生的) environments ——low levels of organic substrate and other nutrients. For example: large polymeric substrates: proteins, polysaccharides (多糖), biodegraded to smaller chemicals by extracellular enzymes (细胞外酶). 四川大学化学学院

The process of biodegradation 2. Once inside the cell, the reactions that a compound may undergo are determined by its molecular structure, hundreds of transformations have been described in the literature, but almost all can be classified broadly as: ü ü reductive; ü hydrolytic; ü 四川大学化学学院 oxidative; conjugative reactions

The process of biodegradation What process a special compound will undergo inside the cell depends strongly on its molecular structure. In addition, The catabolic pathways employed by microbial populations are also diverse and vary with the environmental conditions. 四川大学化学学院 2002年 7月26日 33

The strategy of microbial degradation But despite the immense structural variety of naturally occurring as well as anthropogenic (人类引 起的) compounds, their utilization by microorganisms always involves the same basic strategy. That strategy is stepwise degradation to yield one or more intermediate products capable of entering the central pathways of metabolism. The overall objective is always to produce carbon and energy for growth. 四川大学化学学院

The strategy of microbial degradation Persistent and toxic intermediates occasionally arise from potential biodegradation of a compound, but this is the exception rather than the rule. Naturally occurring organic compounds are degradable via pathways that represent evolutionary adaptations to prevailing conditions. 四川大学化学学院 2002年 7月26日 35

Gratuitous metabolism (幸运代谢) Many man-made chemicals are identical or similar to naturally occurring substances, but human activities have also produced structures never seen or at least infrequently encountered in nature. Many of these, nonetheless, can be attacked by microorganism by virtue of a phenomena referred to as Gratuitous Metabolism or Fortuitous Metabolism. Cause:degradative enzymes generally are not absolutely specific for their natural substances. 四川大学化学学院
The Microbial Basis of Biodegradation Group Contribution Method for Predicting Biodegradability 四川大学化学学院 Chemical Structure and Biodegradability Examples of Designing Biodegradable Chemicals 2002年 7月26日 37

4. 2. 2 Relationship between chemical structure and bio-degradability The bio-degradability of a substance, which is one of the properties of the substance, depends strongly on its chemical structure. Studies, Research and Environmental Monitoring : Small changes in molecular structure can appreciably alter a chemical's susceptibility to biodegradation! 四川大学化学学院

Relationship between chemical structure and bio-degradability The following molecular features generally increase resistance to aerobic biodegradation: 1. Halogens; especially chlorine and fluorine; 2. Chain branching(支链物质), especially quaternary carbon(季碳)and tertiary nitrogen, or extensive branching such as in surfactants derived from tri-or tetrapropylene; 3. Nitro, nitroso(亚硝基), azo(偶氮基), arylamino groups(芳氨基); 四川大学化学学院 2002年 7月26日 39

Relationship between chemical structure and bio-degradability Polycyclic residues(多环残基)(such as in polycyclic aromatic hydrocarbons(多环芳香烃) or PAHs(稠环芳烃)), especially with more than 3 fused rings; 5. Heterocyclic residues(杂环残基); e. g. , pyridine rings(吡啶环); 6. Aliphatic ether (C-O-C) bonds(脂肪族醚键). 7. High-substituted compounds. 4. 四川大学化学学院

The Cause for resistance to biodegradability For the most part, the features listed above affect the ability of the compound to serve as an inducer or substrate, or both, of degradative enzymes and cellular transport systems. For example, Addition of a chlorine atom to a phenyl ring makes the ring less susceptible to attack by oxygenase enzymes, which utilize a form of electrophilic oxygen as a cosubstrate (共存底物). Strongly electronwithdrawing (吸电子) substituents such as halogens are therefore to be avoided in chemical design if possible. 四川大学化学学院

Note: This list is not exhaustive(详尽的), nor should it be inferred that the presence of even a single atom or group from the list necessarily renders a compound recalcitrant (反抗的). Moreover, in most cases the mechanism by which increased resistance to biodegradation is conferred is not known in detail. But this should not blind us to the fact that sufficient information is available to allow application of these principles in chemical design. 四川大学化学学院 2002年 7月26日 42

The chemical structures which favor biodegradability Biodegradability is usually enhanced: by the presence of potential sites of enzymatic hydrolysis (e. g. , esters, amides); 2. by the introduction of oxygen in the form of hydroxyl (羟基), aldehydic (醛基) or carboxylic ( 羧基) groups; 3. by the presence of un-substituted linear alkyl chains (especially >4 carbons) and phenyl rings, which represent possible sites for attack by oxygenases. 1.

The chemical structures which favor biodegradability The first step of biodegradation is some kind of oxidation reaction. And this step is almost always rate limiting. The second of these three factors is particularly important because the first step in the biodegradation of many compounds (e. g. , hydrocarbons) is the enzymatic insertion of oxygen into the structure. 四川大学化学学院

The importance of inserting oxygen in the molecule More generally, if the first biodegradative step is some form of oxidation, it seems logical to expect that biodegrability will be enhanced if the synthetic chemist has in effect already carried it (oxygen inserted) out during molecular design. 四川大学化学学院

The solubility and bio-degradability The aqueous solubility of the molecules alter significantly the biodegradability. The possible effects of solubility on biodegradability are as the following: 四川大学化学学院

The solubility and bio-degradability 1: Microbial bioavailability (微生物生物利用度) Ø Insoluble chemicals tend to adsorb in activated sludge (淤泥), sediments (沉积物) and soil (土壤). Most studies have shown that this tends to reduce the rate of biodegradation. Ø Under the same conditions, the inclusion of groups which increase the solubility of a insoluble chemical may increase its biodegradability. 四川大学化学学院

The solubility and bio-degradability 2: Rate of solubilization (溶解速度) Ø Most studies have shown that for solid with very low solubility, only the dissolved or dispersed phase is available to microorganisms. Ø Therefore, the rate of dissolution of a solid in water may control the rate of biodegradation. Ø Many microorganisms excrete biosurfactants (e. g. , rhamnolipids, 鼠李糖脂) that enhance the rate of solubilizition. 四川大学化学学院

The solubility and bio-degradability 3:Low aqueous concentration ü Some studies have shown that for chemicals soluble to the extent of only a few micrograms per liter or less, this concentration may be too low for optimal function (无法发挥其最佳功 能) of cellular enzymes (细胞酶) or transport systems (传输系统). Thus the biodegradability is limited. 四川大学化学学院

The solubility and bio-degradability At the present it can be stated that : n (i) highly substituted structures are likely to be less rapidly biodegraded than much simpler compounds; and n (ii) for very insoluble chemicals, replacement of a given functional group with one that increases solubility may also result in enhanced biodegradability. 四川大学化学学院 2002年 7月26日 50
The Microbial Basis of Biodegradation Group Contribution Method for Predicting Biodegradability 四川大学化学学院 Chemical Structure and Biodegradability Examples of Designing Biodegradable Chemicals 2002年 7月26日 51

4. 2. 4 Examples of Designing Biologically Safer Chemicals 四川大学化学学院

Linear Alkylbenzene Sulfonates (线性烷基苯磺酸, LAS) Dialkyl Quaternaries (二烷基季铵) Alkylphenol Ethoxylates (烷基酚乙氧基化物, APES) 四川大学化学学院

1: Linear Alkylbenzene Sulfonates (线性烷基苯磺酸盐, LAS) 1884: Lever & Co starts producing Sunlight soap. 1900: Sunlight Flakes became Lux Flakes(力士皂片). 1930: Lever & Co - Unilever(联合利华) 1940 s: ABS. At first the alkyl chains were derived from a kerosene (煤油) fraction or animal fats. 四川大学化学学院
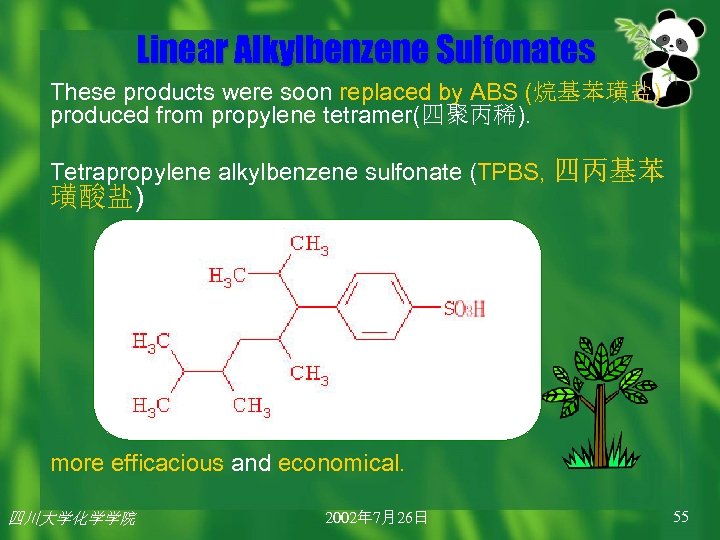
Linear Alkylbenzene Sulfonates These products were soon replaced by ABS (烷基苯璜盐) produced from propylene tetramer(四聚丙稀). Tetrapropylene alkylbenzene sulfonate (TPBS, 四丙基苯 璜酸盐) more efficacious and economical. 四川大学化学学院 2002年 7月26日 55
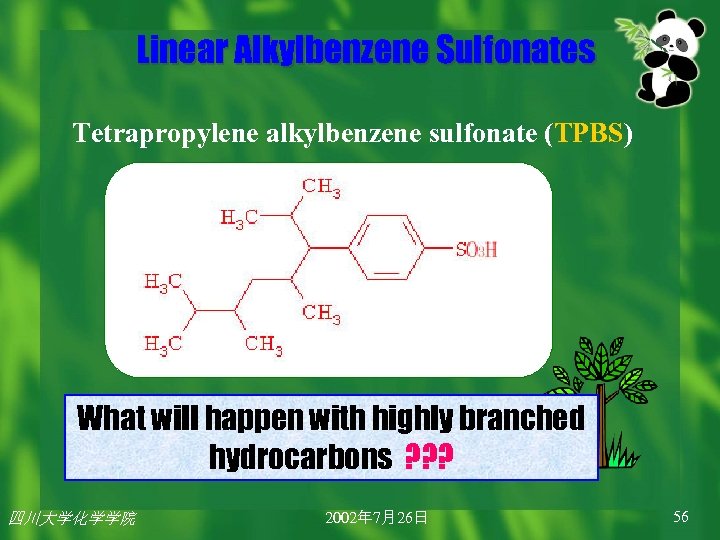
Linear Alkylbenzene Sulfonates Tetrapropylene alkylbenzene sulfonate (TPBS) What will happen with highly branched hydrocarbons ? ? ? 四川大学化学学院 2002年 7月26日 56

Environmental problem TPBS (四丙基苯璜酸盐): Highly branched products incompletely biodegraded in municipal sewage ( 城市污水) treatment systems. TPBS was degraded by only about 50 % in sewage treatment units and as a result excessive foaming occurred in activated sludge aeration (通 风) tanks, as well as in receiving rivers. 四川大学化学学院 2002年 7月26日 57

The foaming was far worse than that caused by proteinaceous (蛋白质的) material in sewage prior to the introduction of synthetic surfactants and in extreme cases sewage-works operators were killed by asphyxiation(窒息) after falling into foaming tanks from walkways made slippery by the foam. . . Because of its incomplete biodegradation, the concentration of TPBS in river waters was as high as 2 mg L-1, and water tended to foam when coming out of the tap. " How to solve this problem ? ? ? 四川大学化学学院 2002年 7月26日 58

Using linear Alkylbenzene Sulfonates (LAS) to replace TPBS: the structure Eventually methods were developed that permitted the economical manufacture of a more environmentally acceptable product LAS, LAS surfactants could be completely degradated in municipal sewage treatment systems. 四川大学化学学院

4. 1 Isosteric Replacement of Carbon with Silicon (用硅对碳进行等 电排置换)in the Design of Safer Chemicals l 4. 2 Designing Biodegradable Chemicals l 4. 3 Designing Aquatically Safer Chemicals l References l 四川大学化学学院 2002年 7月26日 60

4. 3 Designing Aquatically Safer Chemicals 61
Toxic effects of Chemicals to Aquatic species • Aquatic species have their own unique roles in ecosystems and are important for the subsistence of other species, including consumer and predator( 捕食者). • Among other things, aquatic species comprise the components of food chains that lead to man. 四川大学化学学院 2002年 7月26日 62

Toxic effects of Chemicals to Aquatic species • Green algae (绿藻) are the primary producers in aquatic ecosystems in that, through photosynthesis, they produce oxygen and synthesize carbohydrates and other foodstuffs. 四川大学化学学院

Toxic effects of Chemicals to Aquatic species n Chemicals that are toxic to aquatic species may therefore put an ecosystem at unreasonable risk of harm, and can lead to disruption of some food chains. The survival of terrestrial species, including humans, is at least partially dependent upon aquatic organisms. 四川大学化学学院 2002年 7月26日 64

Toxic effects of Chemicals to Aquatic species n There are two general types of chemicalinduced lethality (致命性,毁坏性) in aquatic organisms: non-specific (i. e. , narcosis); specific. The majority of chemicals that are toxic to aquatic species are toxic by narcosis(麻醉). 四川大学化学学院 2002年 7月26日 65

Narcosis Toxicity The mechanistic basis of narcosis toxicity (麻醉 致毒): l A chemical diffuses across the biological membranes of an aquatic organism (e. g. , gills in fish). l Once a high enough concentration is reached within the cells or in cellular membranes, it will cause nonspecific perturbations in cellular function. l These perturbations can lead to death if a sufficient concentration of a chemical has diffused into or across cellular membranes. 四川大学化学学院

Narcosis Toxicity n Chemicals with only a narcotic mode of toxic action are those that do not react with cellular macromolecules, and represent a variety of chemical classes including chlorinated hydrocarbons, alcohols, ethers, ketones, weak organic acids and bases, and simple aromatic nitro-compounds, to name just a few. 四川大学化学学院 2002年 7月26日 67

Narcosis Toxicity Because cellular membranes have a higher lipid content, they are more readily penetrated by non-polar, lipid-soluble chemicals than by lipid-insoluble polar chemicals. Thus, the relative toxic potency of a nonpolar substance that acts through a narcosis mechanism is a function of its lipophilicity. Narcosis, in principle, corresponds mechanistically to the same non-specific mode of action, induced by gaseous anesthetic drugs. Narcosis toxicity represents baseline or minimum toxicity. 四川大学化学学院

Specific or Reactive toxicity In contrast to chemicals that are only toxic by narcosis, some chemicals are toxic to aquatic organisms as a result of a chemical reaction or a specific interaction between the chemical (or its metabolite) with a critical cellular macromolecule. The chemicals exhibit excess toxicity to that of narcosis and is named specific or reactive toxicity. 四川大学化学学院

Specific or Reactive toxicity u For example, excess toxicity can be expected to result if a chemical (or its metabolite) can covalently bond to critical protein molecules (e. g. , enzymes, DNA) or interact covalently with cellular receptor sites. u Examples of the types of chemicals that may exhibit excess (specific-type) toxicity to aquatic organisms are cyanogens(氰), electrophiles (e. g. , alkylating agents), and chemicals metabolized to electrophilic chemicals. 四川大学化学学院 2002年 7月26日 70
1. Use of Structure-Activity Relationships to Predict Aquatic Toxicity 2. Modification of Physicochemical Properties and Structure 四川大学化学学院 3. Modification of Chemical Structure via Chemical Class

4. 3. 1 Use of Structure-Activity Relationships to Predict Aquatic Toxicity 四川大学化学学院

What is SARs ? ? ? SARs refer to the ability of a group of analogous chemicals to produce a particular biological effect, and the influence that the structural differences between the chemicals have on relative potency in producing the biological effect. QSAR(定量构效关系): physicochemistry properties 四川大学化学学院 2002年 7月26日 73

Use of Structure-Activity Relationships to Predict Aquatic Toxicity In 1976, the United States Congress passed the Toxic Substances Control Act (TSCA, 毒物控制提案 ). One of the major objectives of TSCA is to characterize and understand the risks a new chemical poses to humans and the environment before the chemical is introduced into commerce, so that such risk can be minimized or prevented. 四川大学化学学院

Use of Structure-Activity Relationships to Predict Aquatic Toxicity n In order to identify risks posed by new chemicals for which no data are available, and to do so under the strict time constraints prescribed by TSCA, the EPA bases many of its toxicity assessments on structure-activity relationships (SARs). n QSAR(定量构效关系)-physicochemistry properties: n octanol-water partition coefficient (log P or log Kow), water solubility, dissolution constant, relative molecular weight, etc. n n 四川大学化学学院 2002年 7月26日 75

4. 3. 2 Adjusting the Structure and Physical Chemical Properties 四川大学化学学院

Related Physical Chemical Properties n n n (1) Octanol-Water Partition Coefficient (log P 或 log Kow) (2) Water Solubility (3) Molecular Size and Weight (4) Ion Pair (5) Zwitterions (两性离子) (6) Chelation (螯合作用) 四川大学化学学院 2002年 7月26日 77

(1) Octanol-Water Partition Coefficient (log P或log Kow) The octanol-water Partition Coefficient is a term used to express a substance's lipophilicity. It is the phsicochemistry property most frequently used to estimate the aquatic toxicity of organic chemicals. This is because log. P often correlates well with biological activity. water Octanol log P 四川大学化学学院

Octanol-Water Partition Coefficient For Nonionic Organic Substance that only exhibit a narcotic mode of toxic action: Ø log P≤ 5, its acute toxicity and chronic toxicity will increase exponentially with increasing lipophilicity (not for dyes, polymers, and surfacts); Ø log P> 5,toxicity following acute exposure decreases exponentially log P: 5 -8,only chronic toxicity log P: ≥ 8, no toxicity 四川大学化学学院

Octanol-Water Partition Coefficient "Narcotic" chemicals such as aliphatic alcohols, chlorinated benzenes, ketenes, disulfides, and "reactive" chemicals such as acrylates (丙烯酸盐), esters acute aquatic toxicity up to log P of 6. However, some reactive chemicals, such as, aliphatic amines (脂肪胺) surfactants log P values greater than 8 may still exhibit acute toxicity. 四川大学化学学院 2002年 7月26日 80

Octanol-Water Partition Coefficient On the other hand, chemicals with low log P values are not sufficiently lipophilic to enter the cellular membranes of aquatic organisms, and therefore, are not bioavailable and are of low toxicity. For example, chemicals with molecular weights of less than 200 daltons and have a log P of 2 or lower have low toxicity to aquatic species, i. e. , acute median lethal concentration (LC 50) values greater than 100 mg/L. 四川大学化学学院

For chemicals whose aquatic toxicity is due to narcosis, chronic and acute aquatic toxicity Chemists can reduce the potential for aquatic toxicity by designing chemicals. l molecular weights of less than 200 daltons, log P < 2, l log P > 8 regardless of molecular weight. To decrease log P, polar substitutes such as carboxylic, alcoholic, or other water soluble groups can be added to chemicals, and the resultant log P of the modified substance can be calculated. On the other hand, log P can be increased by adding hydrophobic groups such as halogens (卤素), phenyl rings, and alkyl groups. 四川大学化学学院

(2) Water Solubility As a general rule, chemicals having water solubility less than 1 ppb are essentially non-toxic to aquatic species due to low bioavailability. For example, tertiary amyl alcohol (叔戊醇) is 98 g/L more water soluble than its isomer n-pentanol (正戊醇). CH 3 CH 2 C(OH)(CH 3)2 a+98 g/L 四川大学化学学院 CH 3 CH 2 CH 2 OH a g/L

(3) Molecular Size and Weight If the new chemical's molecular weight is increased, the chemical will be less toxic to aquatic organisms while holding all other factors constant. At a molecular weight of 1000 Daltons, uptake will be negligible because the chemical will not diffuse across the respiratory membranes of aquatic organisms. Chemicals with minimum cross-section diameters greater than 1 nm are too large for passive diffusion and uptake through the respiratory membranes of aquatic organisms. 四川大学化学学院

Natural phthalocyanine dyes (天然酞菁染料) n minimum cross-sectional diameters >> 1 nm. n low acute and chronic toxicity to aquatic organisms The Design Principle *to increase minimum cross-sectional diameters *to increase the molecule size 四川大学化学学院

(4)Ion Pair Some chemical salts exist as strong ion pairs when an ion and its counter ion are associated strongly with one another. These chemicals dissociate weakly or not at all in water, consequently, may have low water solubility and low aquatic toxicity. If a soluble and charged chemical can be converted to a strong ion pair and still retain its usefulness in an use application, then the resulting substance will be less toxic to aquatic organisms. 四川大学化学学院

For example, some pesticides are formulated with either cationic or anionic surfactants. If the same pesticides could be formulated with the cationic surfactant: anionic surfactant strong ion pair (assuming a 1: 1 ratio), then the toxicity of the surfactants, could be reduced more than 100 -fold. Here the formation of a strong ion pair to disperse chemical pesticides result in a safer commercial product, as opposed to a surfactant/dispersant that is toxic to aquatic organisms. 四川大学化学学院

(5) Zwitterions (两性离子) Zwitterions are substances that contain positively and negatively charged groups. 酸性蓝 1号 四川大学化学学院

Zwitterions generally have low toxicity to aquatic organisms, provided that the substance has balanced charges and does not have surfactant properties. In general, most zwitterions are not surfactants, and the toxicity to fish and water leas(水蚤) is low. However, algal (海藻的) toxicity has been observed at less than 10 mg/L for some chemicals. 四川大学化学学院

(6) Chelation (螯合作用) Chelation is the process in which a metal cation becomes bound within an organic molecule. In chelation, two or more polar electron- donating atoms (e. g. , oxygen, nitrogen, etc. ) within an organic molecule form a complex with a multivalent metal (e. g. , Co 2+, Ni 2+, Cu 2+, Zn 2+, Mg 2+, Ca 2+) to form a ringlike structure. Chelation can also occur through sulfur atoms with the formation of a stable four-membered ring. 四川大学化学学院

Substances that can chelate polyvalent metals are often toxic to algae (藻类) in soft water situations because they deprive the algae of essential nutrient elements, e. g. , Ca 2+, Mg 2+, or Fe 2+. Therefore, algal toxicity can be reduced if a chelator is bound with a polyvalent metal before exposure to algae takes place or if the chemical is released to surface waters which have moderate to high hardness. 四川大学化学学院

4. 3. 3 Molecular Modification 四川大学化学学院

1、Narcosis and Excess Toxicity n Examples of chemicals that are toxic by specific mechanisms include: electrophiles such as epoxides, alkyl halides, acrylates, aldehydes, certain esters; dinitrobenzenes; and thiols, to name a few. n The electrophilic chemicals may combine covalently with nucleophiles located in receptors of intracellular macromolecules. This bonding results in a cellular change that is difficult to reverse, and result in irreversible toxicity. 四川大学化学学院

Excess Toxicity The aquatic toxicity of a chemical that is toxic from a specific mechanism is usually considerably greater than that predicted for the substance using a QSAR equation developed for chemicals that are toxic by narcosis. Excess Toxicity= TAT-TAP TAT:tested experimentally TAP: predicted by QSAR 四川大学化学学院

2、 Reduce excess toxicity by modification l The toxicity of chemicals that are toxic by a specific mechanism can be decreased by making structural modifications that sterically hinder them such that they can no longer reach or act with their target sites (i. e. , reducing their toxicity to only narcosis). l 四川大学化学学院

u For example, sterically hindered cyclic aliphatic amine is 25 -fold less toxic to fish than the QSAR predicted value for aliphatic amines. 四川大学化学学院
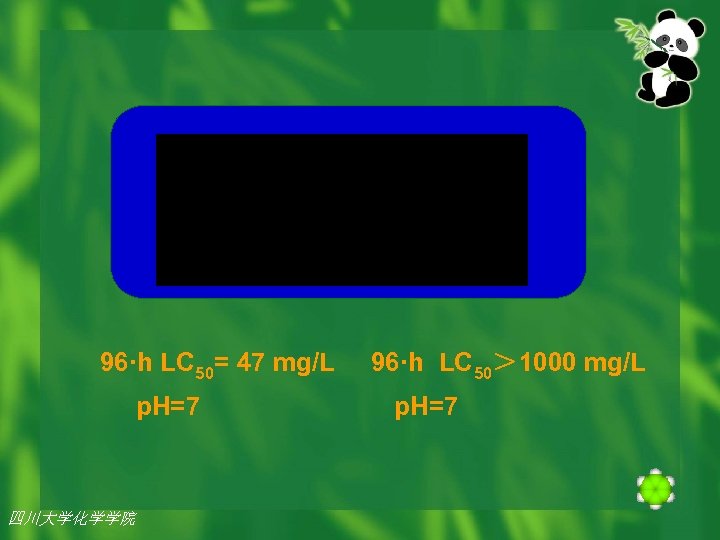
96·h LC 50= 47 mg/L p. H=7 四川大学化学学院 96·h LC 50> 1000 mg/L p. H=7

CH 2=CHCOOCH 2 OH LC 50 = 4. 8 mg/L H 2 C=C(CH 3)COOCH 2 OH LC 50 = 227 mg/L 四川大学化学学院 2002年 7月26日 98

3、Dyes Organic dyes may be divided into four groups : 1. nonionic (neutral dyes); 2. anionic (negatively charged dyes or acid dyes); 3. cationic (positive charged) ; 4. amphoteric (mixture of positive and negative charges on the same molecule). 四川大学化学学院

Dyes For dyes which are charged (anionic, cationic, and amphoteric), no correlation (QSAR) has been found between aquatic toxicity and physicochemical properties. Therefore, instead of using QSARs, scientists predict aquatic toxicity using the nearest analog SAR method for which data are available. 四川大学化学学院 2002年 7月26日 100

(1)Nonionic Dyes (Neutral Dyes) The theory of narcosis and excess toxicity discussed in the preceding paragraphs also applies to neutral dyes. To design safer neutral dyes: a water solubility below 1 ppb; l adding polar substituents l (If a neutral dye is required to be soluble) l molecular weight >1000 daltons; l minimum cross-section diameter > 1 nm. 四川大学化学学院
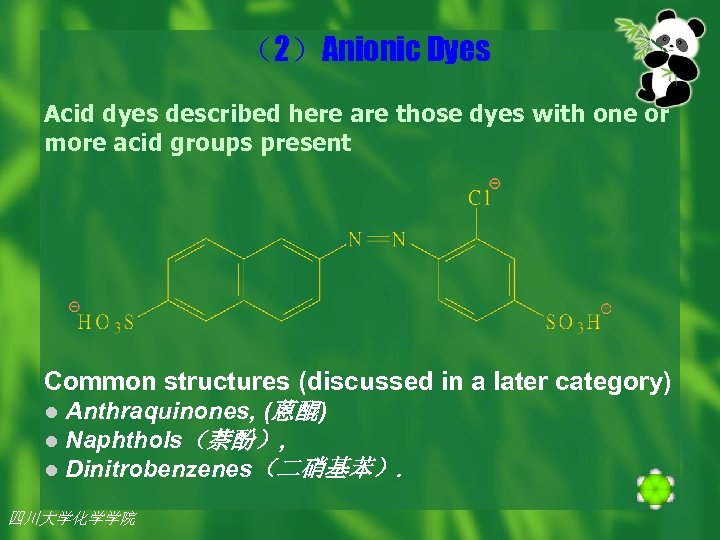
(2)Anionic Dyes Acid dyes described here are those dyes with one or more acid groups present Common structures (discussed in a later category) l Anthraquinones, (蒽醌) l Naphthols(萘酚), l Dinitrobenzenes(二硝基苯). 四川大学化学学院

Anionic Dyes Molecular weights > 1000 daltons: no systemic toxicity was observed. Molecular weights less than 1000 daltons: (monoand di-acid dyes) show moderate toxicity toward fish, invertebrates, and algae. Dyes with azo bonds have been associated with moderate aquatic toxicity. Acid dyes with > 3 acid groups, show low toxicity to fish. 四川大学化学学院

The design of Anionic Dyes The best way to design mono- and di- acid dyes is to increase the molecular weight to more than 1000 daltons. This results in reduced toxicity to fish and invertebrates, regardless of toxic functional groups (i. e. , dinitro, phenols, nthraquinones) as long as they are not surface-active. If the molecular weights are less than 1000 daltons, then add three or more acid groups. Acid dyes with three or more acid groups show low toxicity toward fish and aphnids, and only indirect toward green algae. 四川大学化学学院

(3)Metalized Acid Dyes A metalized dye is an acid dye which is combined with a metal. Frequently, iron, copper, cobalt, aluminum, nickel, chromium, and zinc are the metals associated with acid dyes. These dye molecules are combined to metals but sometimes contain residual uncomplexed free metal (Al, Cr, Co), that is toxic to aquatic organisms. 四川大学化学学院

M = Al, Cr, Co, etc. , toxic to aquatic organisms. 四川大学化学学院

The design of Metalized Acid Dyes Increasing the molecular weight above 1000 daltons will usually decrease aquatic toxicity. In addition, chemists should use metals such as iron, copper, or zinc, and avoid the use of aluminum, chromium and cobalt. Dyes should be made to eliminate residual unchelated metal in the overall dye product. 四川大学化学学院

(4)Cationic Dyes Cationic dyes are a chemically diverse group of positively charged dyes ranging in molecular weight from 200 to greater than 1000 daltons. Localized (定域化) cationic dyes. The positive charge may appear on a carbon, nitrogen, phosphorus, oxygen, or sulfur atom. Some cationic dyes are associated with metal complexes, and water solubility vary depending on their counter ion. 四川大学化学学院

Delocalized (离域化) cationic dyes positive charge is not located on a particular atom, often as a result of conjugation between heteroatoms such as N, S, O, P. For Delocalized cationic dyes: Molecular weights >(=)1000 daltons exhibit lower toxicity (unable to be taken up by aquatic organisms). l Contain primary nitrogen are generally less toxic than those that contains secondary nitrogens, which are less toxic than those that contain tertiary nitrogens. l 四川大学化学学院

To design delocalized cationic dyes : increase molecular weights to more than 1000 daltons. l limit the extent of substitution on the nitrogens. l To design localized cationic dyes : limit the number of localized positive charges or redesigning the dye to be amphoteric(两性 的). 四川大学化学学院

(5)Amphoteric Dyes (两性染料) Amphoteric dyes l behave as an acid or a base, are often characterized by the presence of positive and negative charges within the same molecule l The toxicity of amphoteric dyes depends on the ratio of cations to anions in the dye. If the ratio favors the anion, then the dye will have lower toxicity (i. e. , nearer to the acid dyes). 四川大学化学学院
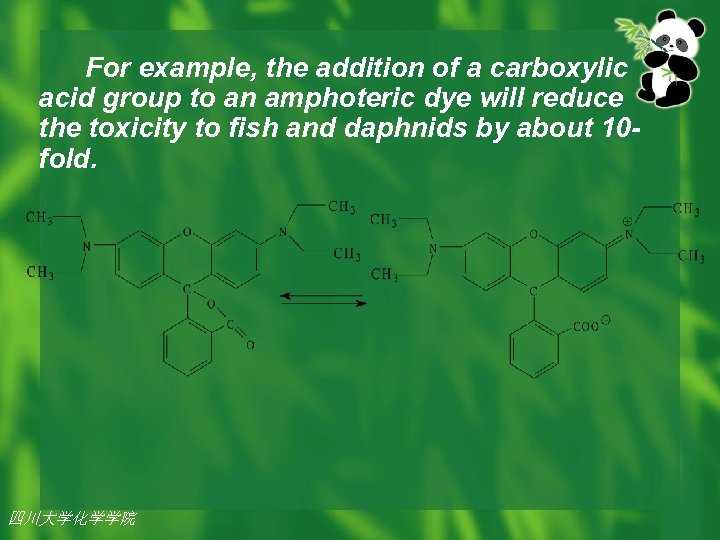
For example, the addition of a carboxylic acid group to an amphoteric dye will reduce the toxicity to fish and daphnids by about 10 fold. 四川大学化学学院

总结设计对水生生物更加安全的染料化学品应 遵循的原则 阴离子数大于阳离子数(电荷数) 磺酸基优于羧酸基 尽可能使分子量大于1000 尽可能增大分子的最小横截直径 四川大学化学学院

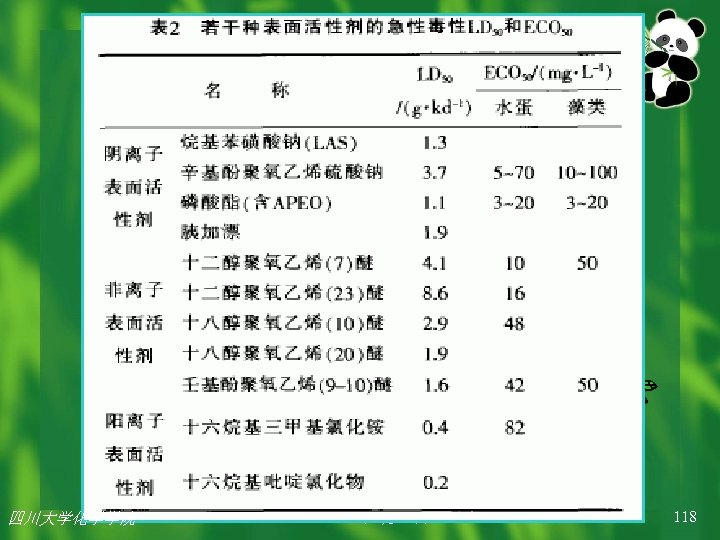
4、 Surfactants: contain both hydrophobic and hydrophilic moieties, which enable the substances to form micelles in water. Surfactants are toxic to aquatic species because they can disrupt the interface between biological membranes (which are chiefly lipid in content) and the aquatic environment (which is water). In simple terms, the aquatic toxicity of surfactants is rigidly due to their ability to disintegrate biological membranes to the extent that their structure-specific components are loosened and are no longer functional. 四川大学化学学院

四川大学化学学院

The relative toxicological potency of a surfactant is dependent on balance between the hydrophobic component and the hydrophilic component. In general, toxicity is higher when the hydrophilic component is relatively larger compared to the hydrophobic component. 四川大学化学学院

Surfactants with melting points of above 35°C will be self-dispersing in warm water only (e. g. , 50°C) and will not function as surfactants at environmental temperatures (20°C). Such surfactants will generally not be acutely toxic to aquatic organisms, and only chronic toxicity is possible. Surfactants with molecular weights less than 1000 daltons exhibit surface active toxicity and can be toxic to aquatic organisms. Surfactants that have molecular weights rather than 1000 Daltons are too large to be absorbed, and consequently exhibit surface toxicity only. 四川大学化学学院
四川大学化学学院 2002年 7月26日 118

5、 Polyanionic Monomer 四川大学化学学院 2002年 7月26日 119

6、Organo-Metallics n Many organometallics such as those used as fouling agents, pesticides, and fungicides are toxic to aquatic organisms. To prevent toxicity of the metal: an organometallic chemical should be designed by increasing: n 1) the log P to greater than 8; n 2) the melting point; n 3) the molecular weight > 1000 daltons. n 四川大学化学学院 2002年 7月26日 120

7、Inorganic Metallics n A variety of inorganic metallic substances are essential for all forms of life. These substances are taken up by living cells and their uptake is strictly regulated because most (or all) of them are toxic when consumed in excess. n However, those inorganic chemicals known to be toxic or cause environmental damage (e. g. , phosphates) can be avoided or not used. 四川大学化学学院 2002年 7月26日 121

参 考 文 献 1. Roger L. Garrett, Designing Safer Chemicls。Stephen C Devito and Roger L. Garrett. edited American Chemical Society, Washington, D. C. 1996,2 2. Stephen C Devito, Designing Safer Chemicls. Stephen C Devito and Roger L. Garrett. edited American Chemical Society, Washington, D. C. 1996, 16. 3. 王连生,韩塑暌,等编著, 分子结构、性质与活性,化学 业出版社, 1997. 122

第四章 业 作 1. 由碳、硅元素的化学性质,试讨论用硅取代 碳的可行性及硅取代物的优点? 2. 用辛醇-水分配系数表征有毒化学品毒性的一 般准则是什么? 3. 除了辛醇-水分配系数,还有那些性质可以表 征有毒化学品毒性? 4. 从分子量、分子体积角度看,应该怎样设计 更加安全的化学品? 四川大学化学学院

第四章 作 业 5. 可降解的化合物在化学结构和物理性质上有何 特点? 6. 常见可降解和不可降解的基团有哪些? 7. 根据染料的基团特性,有机染料可分为几类 ?设计对水生生物更加安全的染料化学品要遵 循怎样的原则? 四川大学化学学院 2002年 7月26日 124

小论文 n 题目 (5选1,见绿色化学课程网站) n 作者 n 中英文摘要 n 引言 n 正文 n 结论 n 参考文献 四川大学化学学院 2002年 7月26日 125
61e34073ceea68bcf24808be3601c12a.ppt