bc2d5e7b3c077dac520b627b8548097f.ppt
- Количество слайдов: 33
 Chapter 15 Air Pollution and Stratospheric Ozone Depletion
Chapter 15 Air Pollution and Stratospheric Ozone Depletion
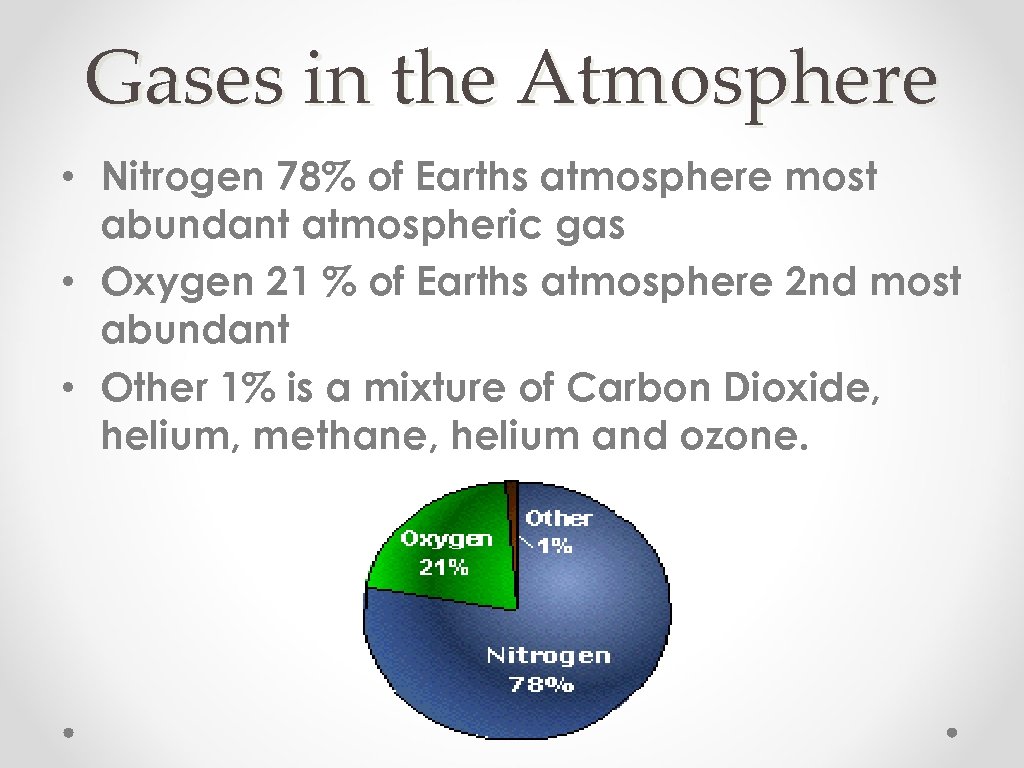 Gases in the Atmosphere • Nitrogen 78% of Earths atmosphere most abundant atmospheric gas • Oxygen 21 % of Earths atmosphere 2 nd most abundant • Other 1% is a mixture of Carbon Dioxide, helium, methane, helium and ozone.
Gases in the Atmosphere • Nitrogen 78% of Earths atmosphere most abundant atmospheric gas • Oxygen 21 % of Earths atmosphere 2 nd most abundant • Other 1% is a mixture of Carbon Dioxide, helium, methane, helium and ozone.
 Air Pollution © Air pollution- the introduction of chemicals, particulate matter, or microorganisms into the atmosphere at concentrations high enough to harm plants, animals, and materials such as buildings, or to alter ecosystems. Major Air Pollutants © © © Sulfur Dioxide Nitrogen Oxides Carbon Oxides © © © Particulate Matter Volatiles Organic Compounds Ozone © © Lead Mercury
Air Pollution © Air pollution- the introduction of chemicals, particulate matter, or microorganisms into the atmosphere at concentrations high enough to harm plants, animals, and materials such as buildings, or to alter ecosystems. Major Air Pollutants © © © Sulfur Dioxide Nitrogen Oxides Carbon Oxides © © © Particulate Matter Volatiles Organic Compounds Ozone © © Lead Mercury
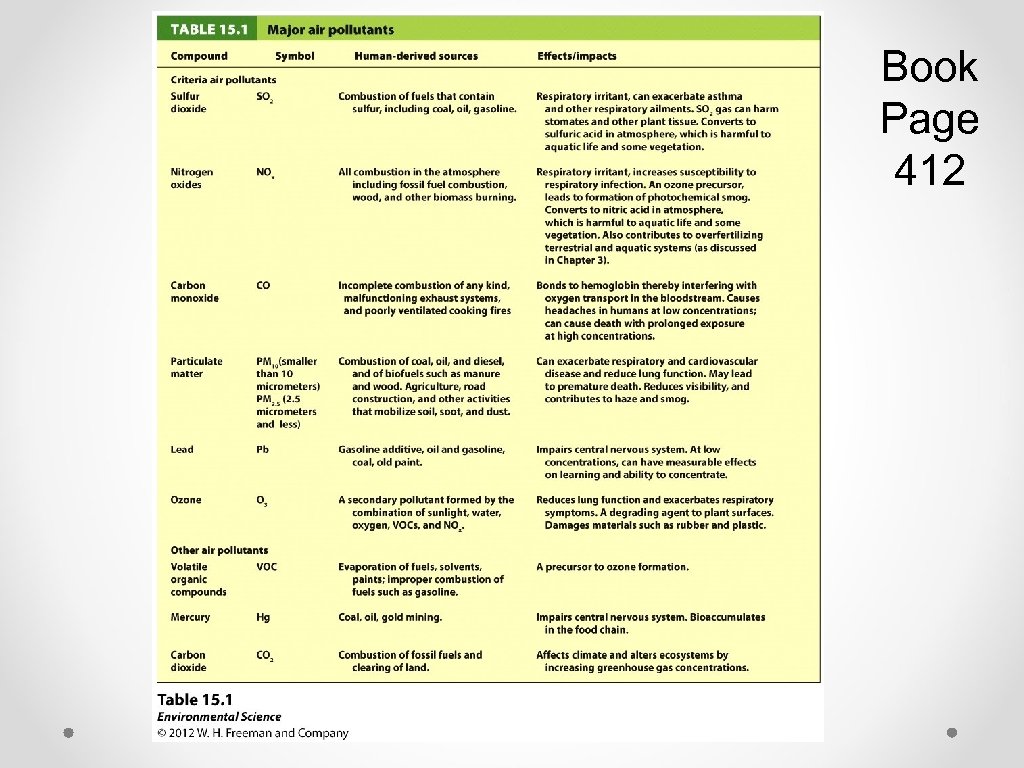 Book Page 412
Book Page 412
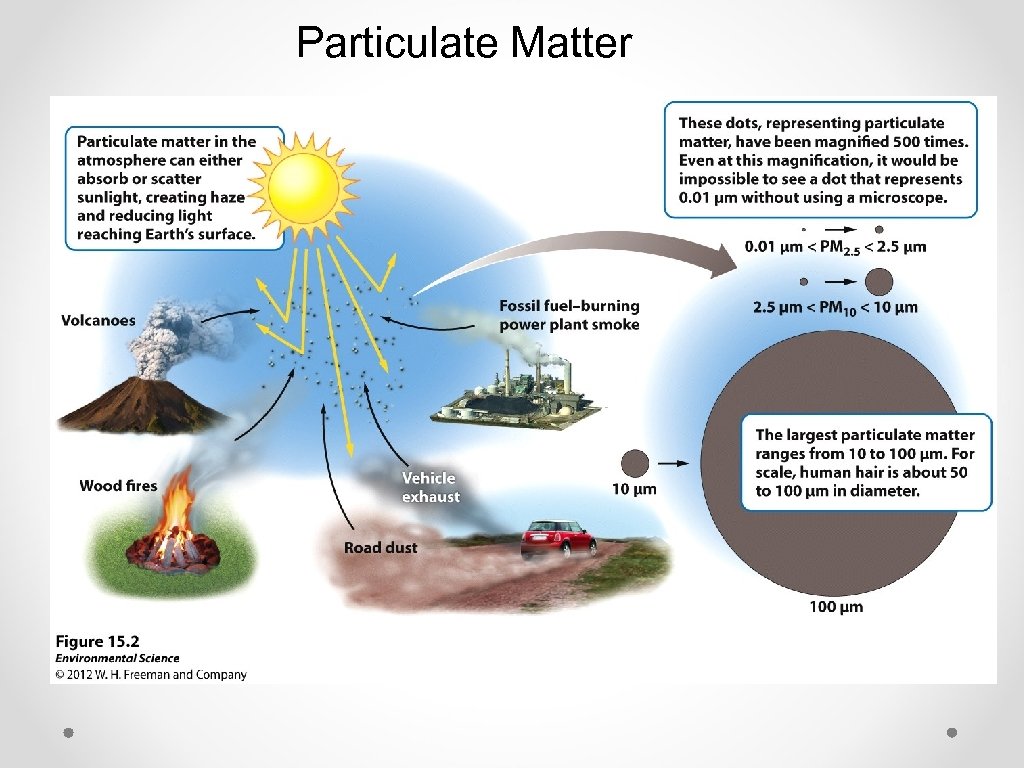 Particulate Matter
Particulate Matter
 Natural Sources of Air Pollution © © Volcanoes Lightning Forest fires Plants
Natural Sources of Air Pollution © © Volcanoes Lightning Forest fires Plants
 Anthropogenic Sources of Air Pollution © © On-road vehicles Power plants Industrial processes Waste disposal
Anthropogenic Sources of Air Pollution © © On-road vehicles Power plants Industrial processes Waste disposal
 Primary Pollutants © Primary pollutants- polluting compounds that come directly out of the smoke-stack, exhaust pip, or natural emission source. © Examples: CO, CO 2, SO 2, NOx, and most suspended particulate matter. Secondary Pollutants © © Secondary pollutants- pollutants that have undergone transformation in the presence of sunlight, water, oxygen, or other compounds. Examples: ozone, sulfate and nitrate
Primary Pollutants © Primary pollutants- polluting compounds that come directly out of the smoke-stack, exhaust pip, or natural emission source. © Examples: CO, CO 2, SO 2, NOx, and most suspended particulate matter. Secondary Pollutants © © Secondary pollutants- pollutants that have undergone transformation in the presence of sunlight, water, oxygen, or other compounds. Examples: ozone, sulfate and nitrate
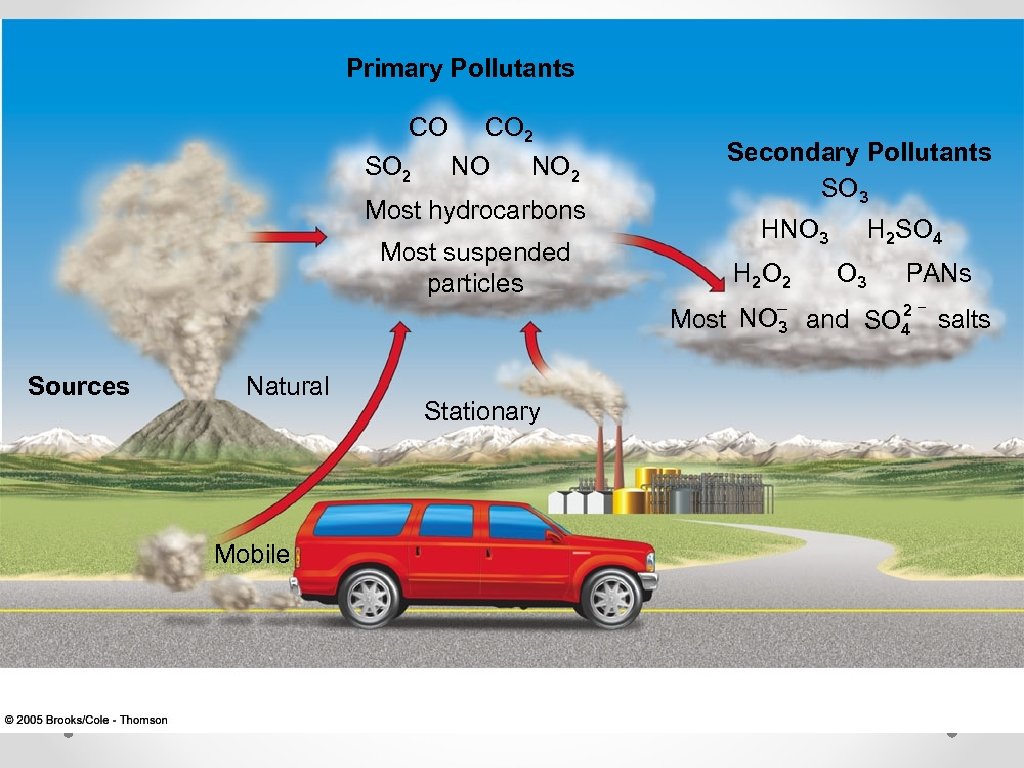 Primary Pollutants CO SO 2 CO 2 NO Secondary Pollutants SO 3 NO 2 Most hydrocarbons HNO 3 Most suspended particles H 2 O 2 Most Sources Natural Mobile Stationary NO– 3 H 2 SO 4 O 3 and PANs 2– SO 4 salts
Primary Pollutants CO SO 2 CO 2 NO Secondary Pollutants SO 3 NO 2 Most hydrocarbons HNO 3 Most suspended particles H 2 O 2 Most Sources Natural Mobile Stationary NO– 3 H 2 SO 4 O 3 and PANs 2– SO 4 salts
 Smog Photochemical smog • When nitrogen oxides (NO and NO 2 coming from cars and industry) react with volatile organic hydrocarbons due to UV radiation (sunlight) and produce a mix of primary and secondary pollutants sunlight Cars + NO 2 + hydrocarbons Pollution (smog) Also Called GROUND LEVEL OZONE
Smog Photochemical smog • When nitrogen oxides (NO and NO 2 coming from cars and industry) react with volatile organic hydrocarbons due to UV radiation (sunlight) and produce a mix of primary and secondary pollutants sunlight Cars + NO 2 + hydrocarbons Pollution (smog) Also Called GROUND LEVEL OZONE
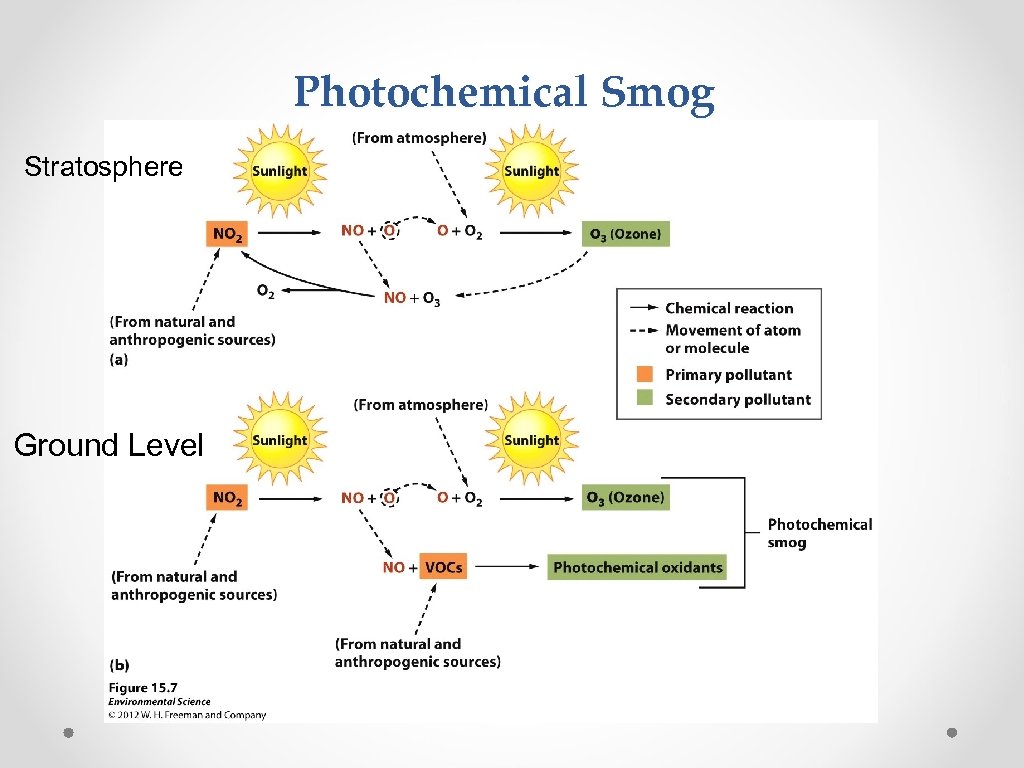 Photochemical Smog Stratosphere Ground Level
Photochemical Smog Stratosphere Ground Level
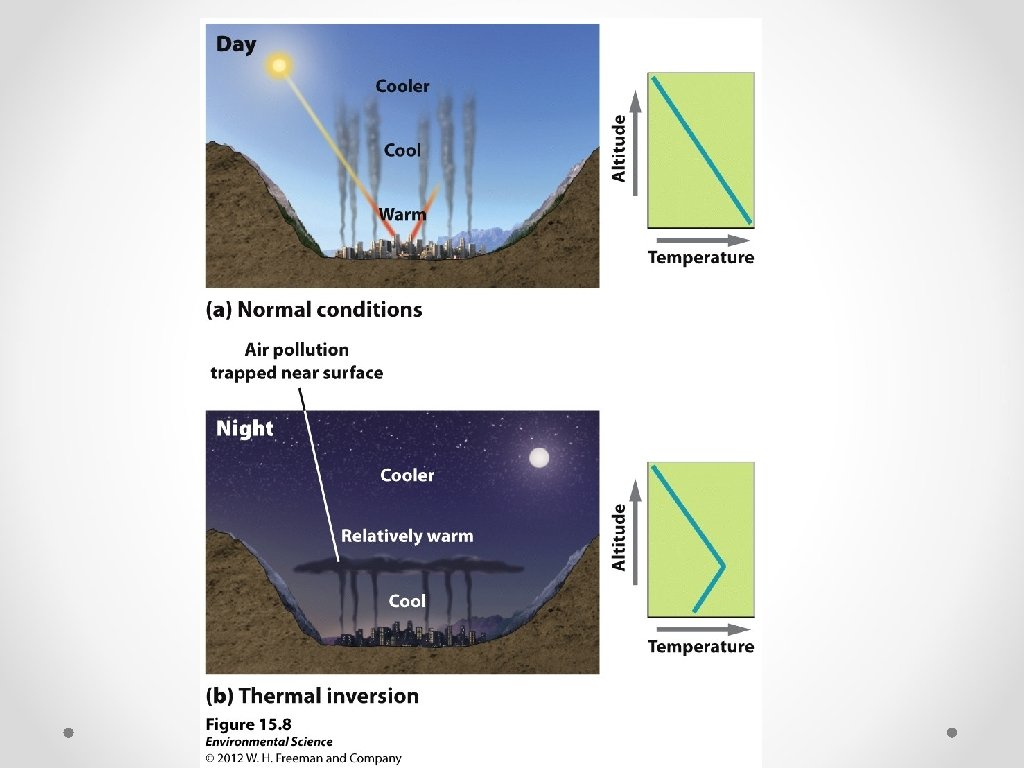
 Thermal Inversions © © Thermal Inversion- when a relatively warm layer of air at mid-altitude covers a layer of cold, dense air below. The warm inversion layer traps emissions that then accumulate beneath it.
Thermal Inversions © © Thermal Inversion- when a relatively warm layer of air at mid-altitude covers a layer of cold, dense air below. The warm inversion layer traps emissions that then accumulate beneath it.
 Acid Deposition © © Acid deposition- occurs when NO 2 and SO 2 are released into the atmosphere and combine with atmospheric oxygen and water. These form the secondary pollutants nitric acid (HNO 3) and sulfuric acid (H 2 SO 4). These secondary pollutants further break down into nitrate and sulfate which cause the acid in acid deposition.
Acid Deposition © © Acid deposition- occurs when NO 2 and SO 2 are released into the atmosphere and combine with atmospheric oxygen and water. These form the secondary pollutants nitric acid (HNO 3) and sulfuric acid (H 2 SO 4). These secondary pollutants further break down into nitrate and sulfate which cause the acid in acid deposition.
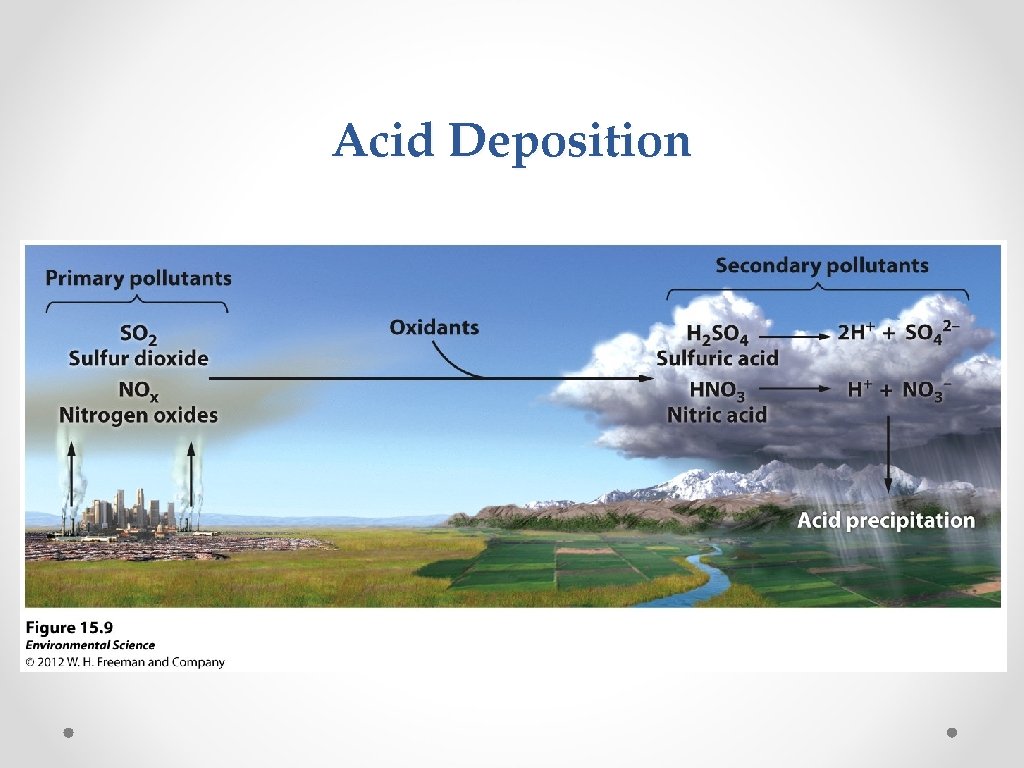 Acid Deposition
Acid Deposition
 Effects of Acid Deposition © © © Lowering the p. H of lake water Decreasing species diversity of aquatic organisms Mobilizing metals (Aluminum) that are found in soils and releasing these into surface waters Damaging statues, monuments, and buildings Affects pipes and releases lead
Effects of Acid Deposition © © © Lowering the p. H of lake water Decreasing species diversity of aquatic organisms Mobilizing metals (Aluminum) that are found in soils and releasing these into surface waters Damaging statues, monuments, and buildings Affects pipes and releases lead
 Ways to Prevent Air Pollution © © © Removing sulfur dioxide from coal by fluidized bed combustion Catalytic converters on cars Scrubbers on smoke stacks Baghouse filters Electrostatic precipitators
Ways to Prevent Air Pollution © © © Removing sulfur dioxide from coal by fluidized bed combustion Catalytic converters on cars Scrubbers on smoke stacks Baghouse filters Electrostatic precipitators
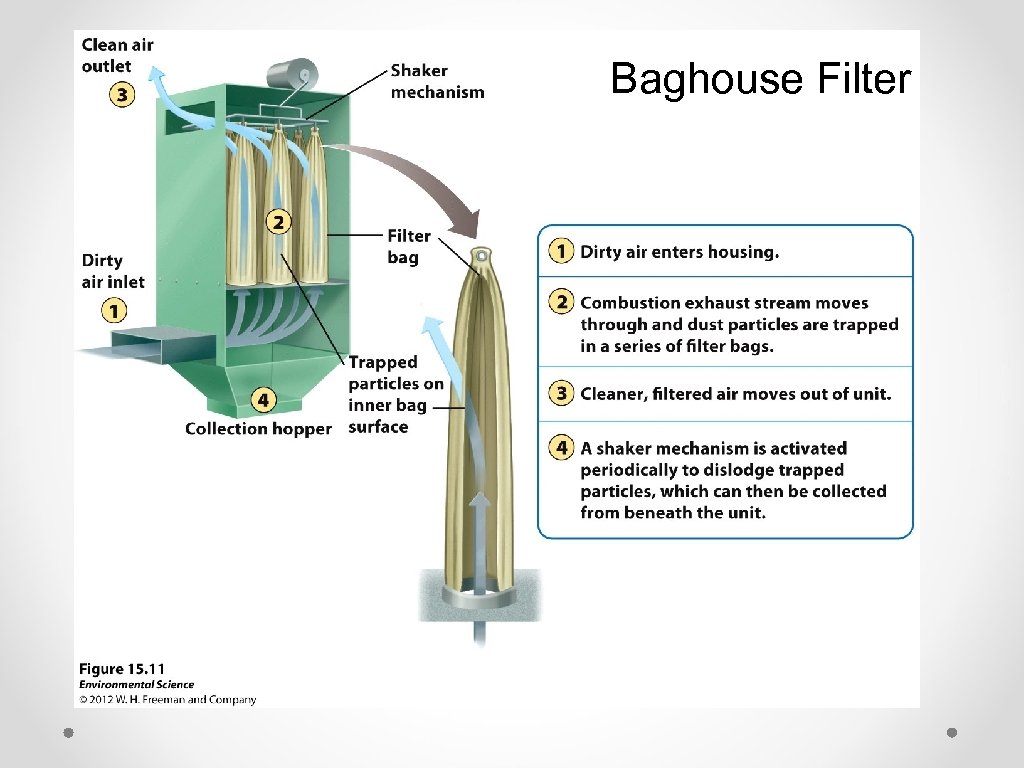 Baghouse Filter
Baghouse Filter
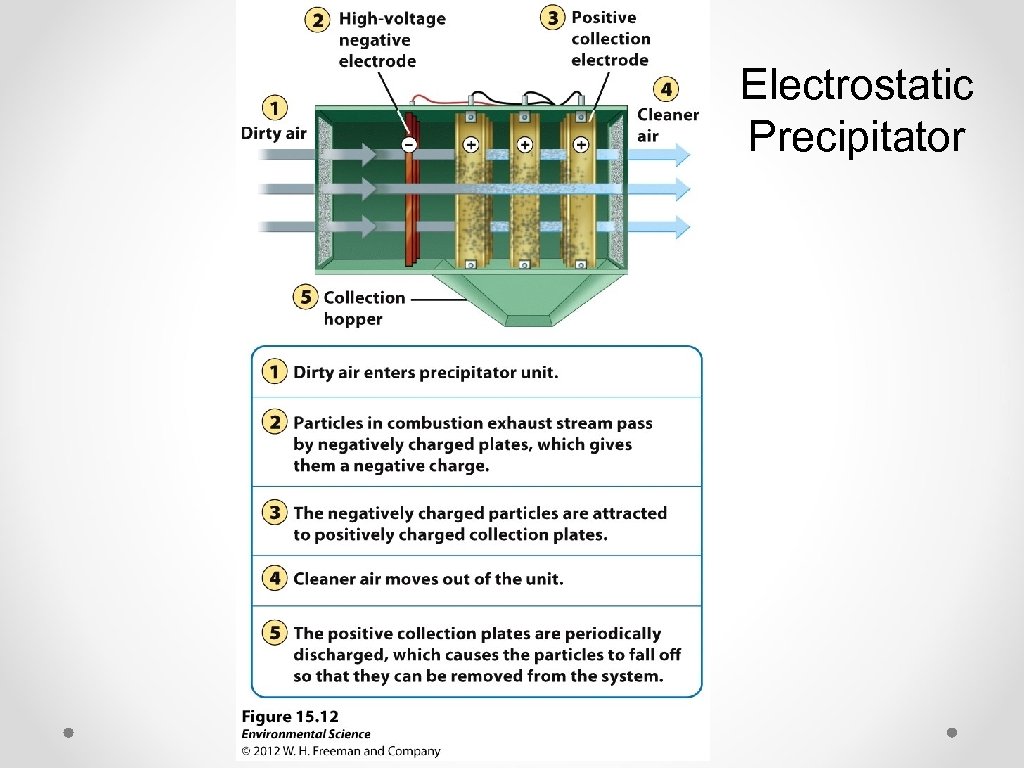 Electrostatic Precipitator
Electrostatic Precipitator
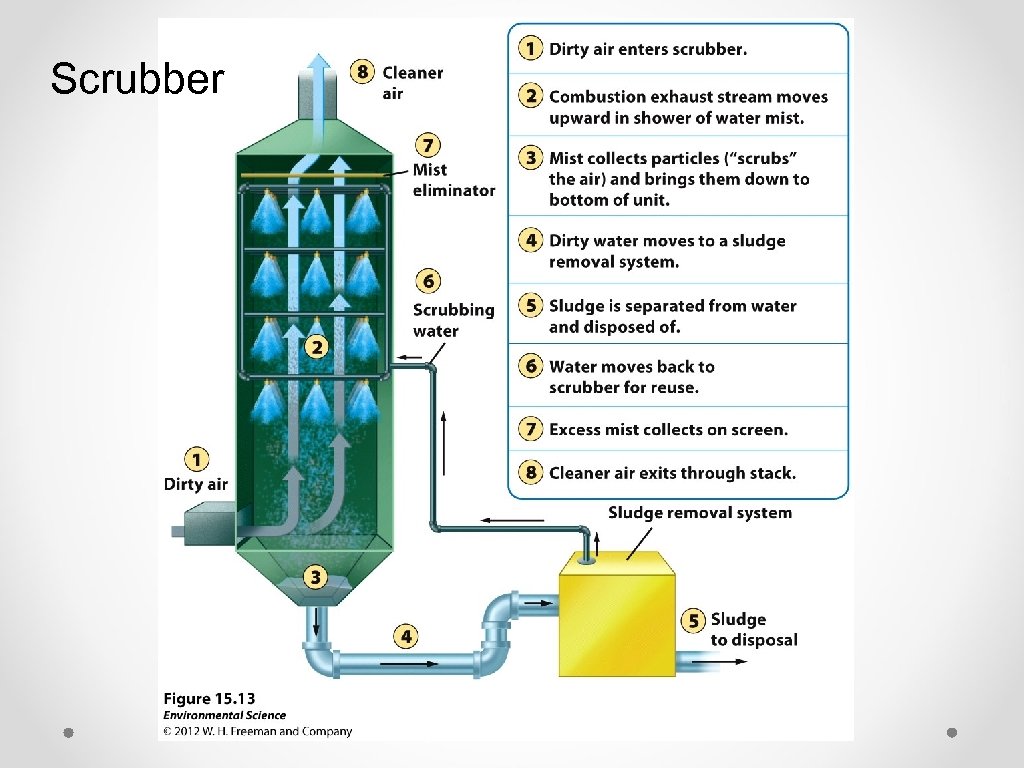 Scrubber
Scrubber
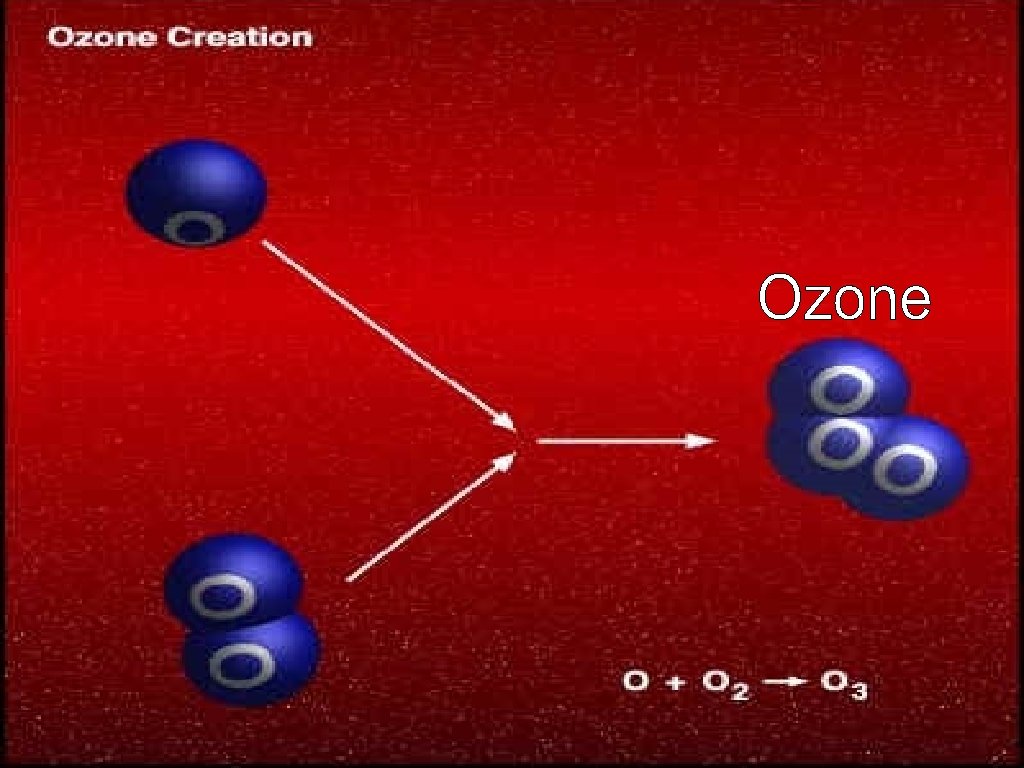
 Stratospheric Ozone © © Ozone is found in the stratosphere Ozone has the ability to absorb ultraviolet radiation and protect life on Earth. Formation of Ozone © © First, UV-C radiation breaks the bonds holding together the O 2 molecule, leaving two free oxygen atoms: O 2 + UV-C -> 2 O The free oxygen atoms then react with other O 2 molecules creating ozone: O 2 + O -> O 3
Stratospheric Ozone © © Ozone is found in the stratosphere Ozone has the ability to absorb ultraviolet radiation and protect life on Earth. Formation of Ozone © © First, UV-C radiation breaks the bonds holding together the O 2 molecule, leaving two free oxygen atoms: O 2 + UV-C -> 2 O The free oxygen atoms then react with other O 2 molecules creating ozone: O 2 + O -> O 3
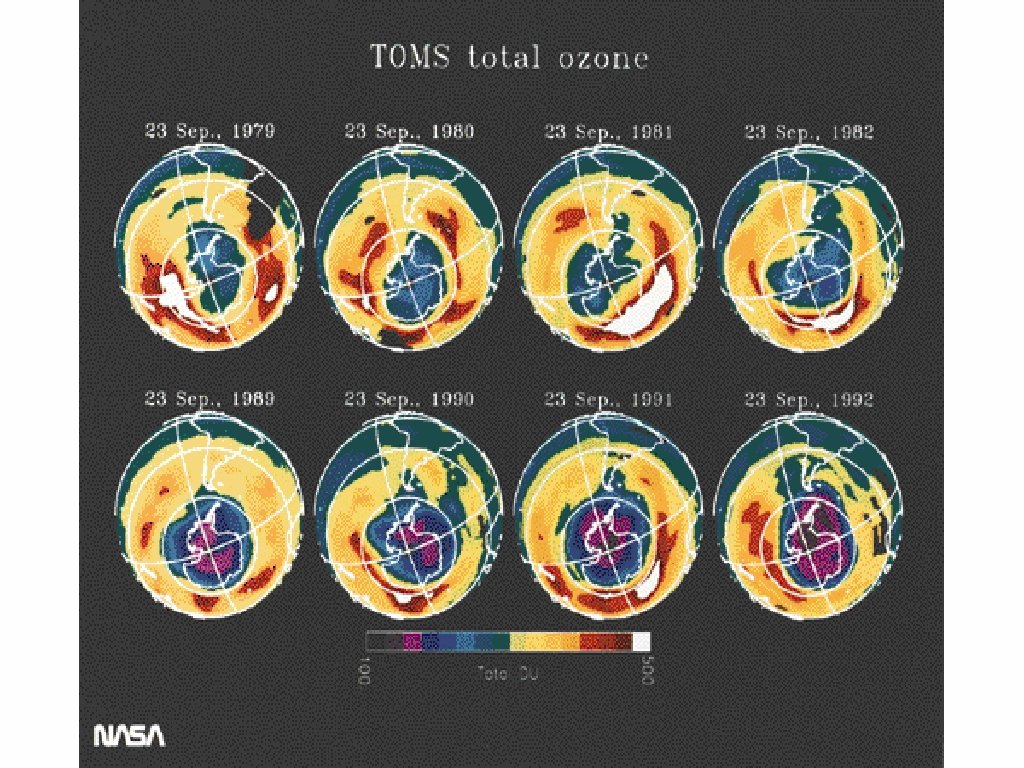
 Stratospheric Ozone is in Danger • Satellites have measured a decrease in O 3 over the past few decades. • Areas over the poles have holes that allow dangerous radiation from the sun to enter earths atmosphere • Human activities have caused this (CFC’s released into the Atmosphere)
Stratospheric Ozone is in Danger • Satellites have measured a decrease in O 3 over the past few decades. • Areas over the poles have holes that allow dangerous radiation from the sun to enter earths atmosphere • Human activities have caused this (CFC’s released into the Atmosphere)
 Where do most come from? Byproducts of foam production, refrigeration, and air conditioning called o o chlorofluorocarbons (CFCs), as well as hydrofluorocarbons (HFCs) and perfluorocarbons (PFCs) Emissions generated by industrial processes and automobiles.
Where do most come from? Byproducts of foam production, refrigeration, and air conditioning called o o chlorofluorocarbons (CFCs), as well as hydrofluorocarbons (HFCs) and perfluorocarbons (PFCs) Emissions generated by industrial processes and automobiles.
 Anthropogenic Contributions to Ozone Destruction © © When CFCs are released into the troposphere they make their way to the stratosphere. The ultraviolet radiation present has enough energy to break the bond connecting chlorine to the CFC molecule. Which can then break apart the ozone molecules. O 3 + Cl -> Cl. O + O 2 One chlorine atom can catalyze the breakdown of as many as 100, 000 ozone molecules before it leaves the stratosphere.
Anthropogenic Contributions to Ozone Destruction © © When CFCs are released into the troposphere they make their way to the stratosphere. The ultraviolet radiation present has enough energy to break the bond connecting chlorine to the CFC molecule. Which can then break apart the ozone molecules. O 3 + Cl -> Cl. O + O 2 One chlorine atom can catalyze the breakdown of as many as 100, 000 ozone molecules before it leaves the stratosphere.
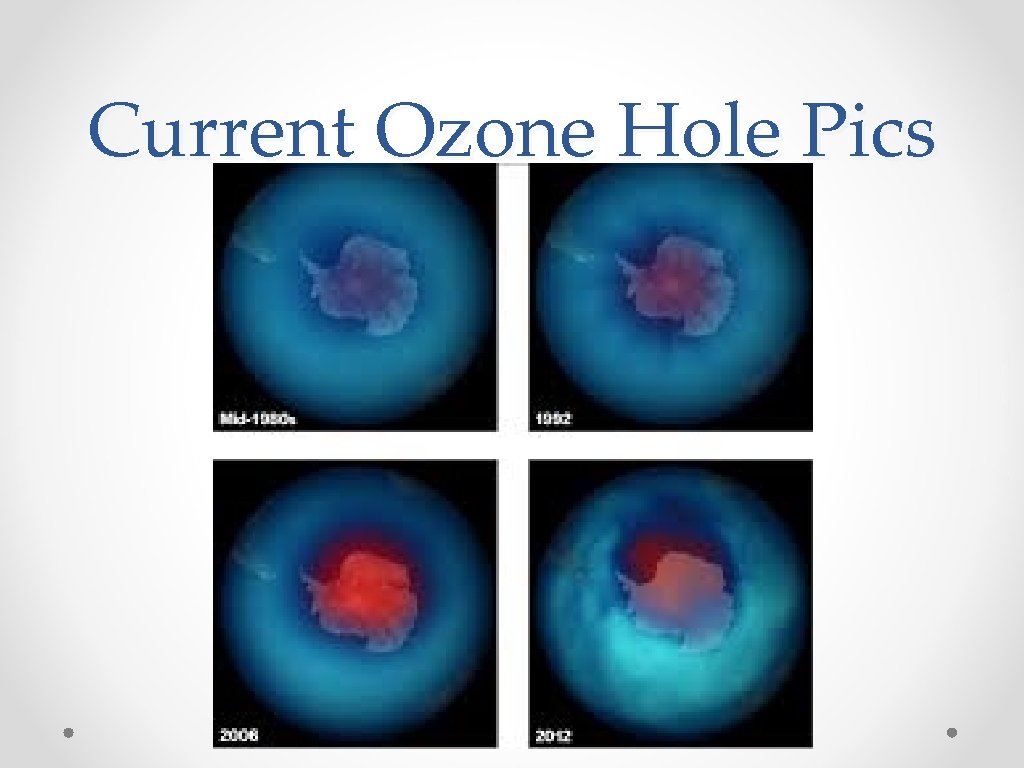 Current Ozone Hole Pics
Current Ozone Hole Pics
 Indoor Air Pollution • Pollution can be 2 -5 times higher inside • Inside cars can be 18 times higher • 18 sources of cancer come from indoor air pollution • Sick building syndrome- term for people getting sick from air pollutants (particularly in new buildings) Top 4 Cigarette Smoke Formaldehyde Radon Particulates
Indoor Air Pollution • Pollution can be 2 -5 times higher inside • Inside cars can be 18 times higher • 18 sources of cancer come from indoor air pollution • Sick building syndrome- term for people getting sick from air pollutants (particularly in new buildings) Top 4 Cigarette Smoke Formaldehyde Radon Particulates
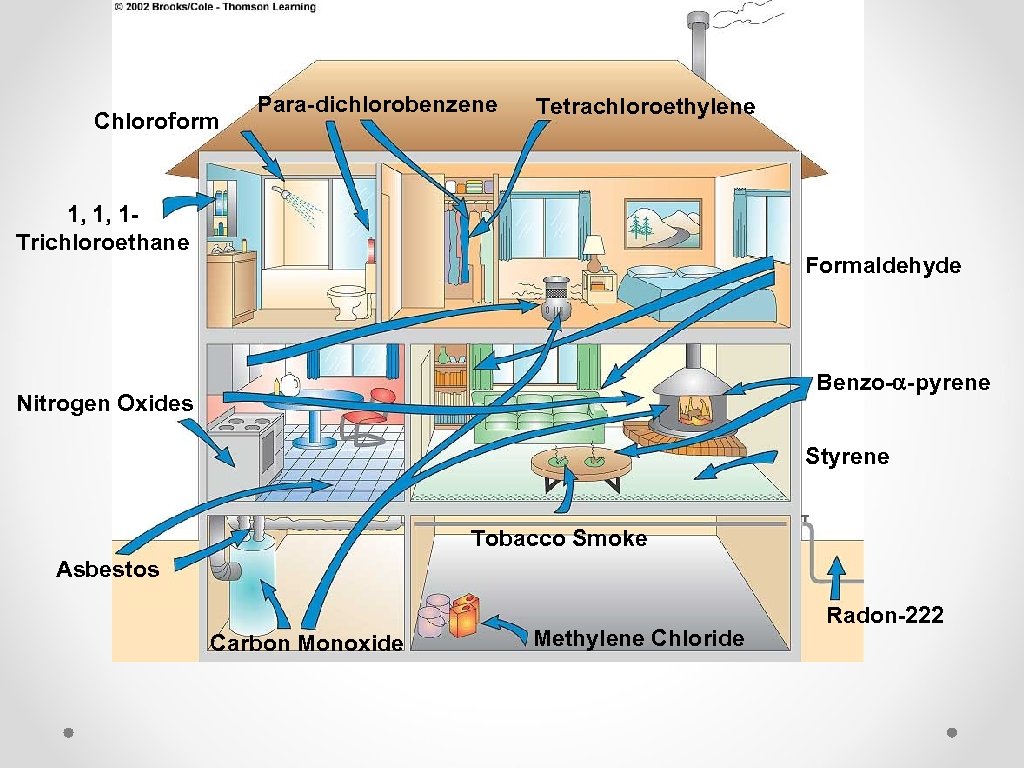 Chloroform Para-dichlorobenzene Tetrachloroethylene 1, 1, 1 Trichloroethane Formaldehyde Benzo-a-pyrene Nitrogen Oxides Styrene Tobacco Smoke Asbestos Carbon Monoxide Methylene Chloride Radon-222
Chloroform Para-dichlorobenzene Tetrachloroethylene 1, 1, 1 Trichloroethane Formaldehyde Benzo-a-pyrene Nitrogen Oxides Styrene Tobacco Smoke Asbestos Carbon Monoxide Methylene Chloride Radon-222
 Indoor Air Pollution Formaldehyde o Colorless gas o Used to make household materials • furniture • Drapes and upholstery • Adhesive in carpeting and wallpaper • insulation
Indoor Air Pollution Formaldehyde o Colorless gas o Used to make household materials • furniture • Drapes and upholstery • Adhesive in carpeting and wallpaper • insulation
 Indoor Air Pollution Radon-222 Naturally occurring radioactive gas Can’t see, taste, or smell Found in underground deposits of minerals Buildings above deposits – enter through cracks and openings o Second leading cause of lung cancer o Need to have house tested and sealed (comes up through the foundation) o o
Indoor Air Pollution Radon-222 Naturally occurring radioactive gas Can’t see, taste, or smell Found in underground deposits of minerals Buildings above deposits – enter through cracks and openings o Second leading cause of lung cancer o Need to have house tested and sealed (comes up through the foundation) o o
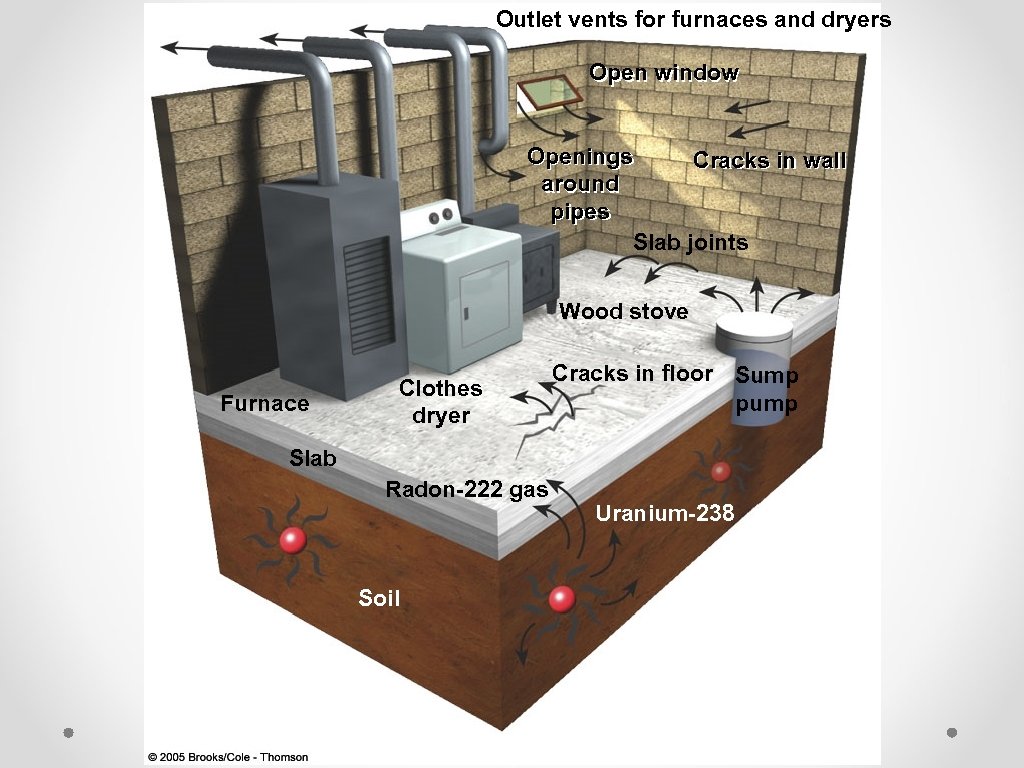 Outlet vents for furnaces and dryers Open window Openings Cracks in wall around pipes Slab joints Wood stove Furnace Clothes dryer Cracks in floor Sump pump Slab Radon-222 gas Soil Uranium-238
Outlet vents for furnaces and dryers Open window Openings Cracks in wall around pipes Slab joints Wood stove Furnace Clothes dryer Cracks in floor Sump pump Slab Radon-222 gas Soil Uranium-238


