e8d08da1a4d6c4bafb22c8cb1f9ffcf2.ppt
- Количество слайдов: 25
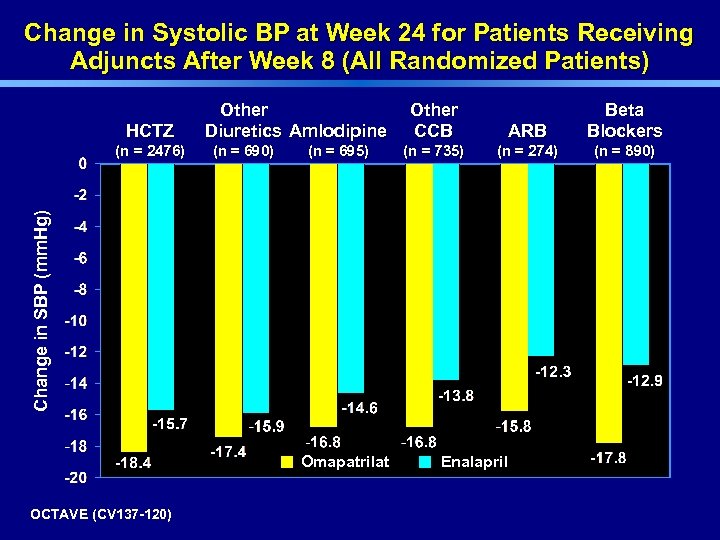
Change in Systolic BP at Week 24 for Patients Receiving Adjuncts After Week 8 (All Randomized Patients) HCTZ (n = 690) (n = 695) Other CCB ARB Beta Blockers (n = 735) (n = 274) (n = 890) Change in SBP (mm. Hg) (n = 2476) Other Diuretics Amlodipine Omapatrilat OCTAVE (CV 137 -120) Enalapril
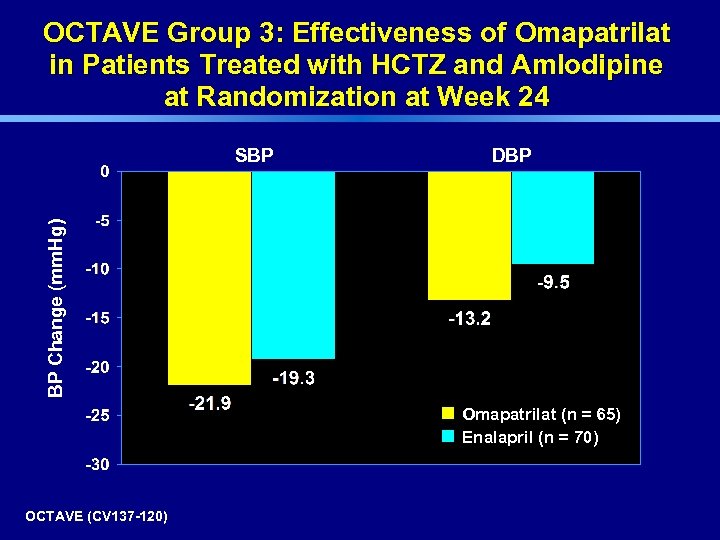
OCTAVE Group 3: Effectiveness of Omapatrilat in Patients Treated with HCTZ and Amlodipine at Randomization at Week 24 DBP BP Change (mm. Hg) SBP Omapatrilat (n = 65) Enalapril (n = 70) OCTAVE (CV 137 -120)
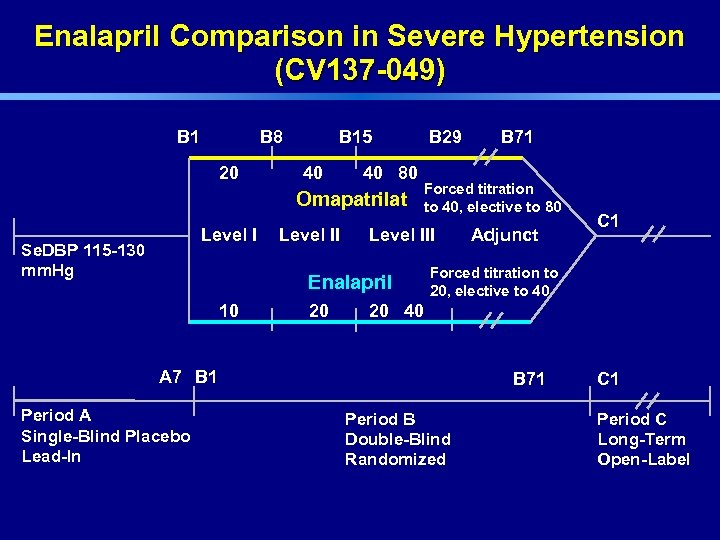
Enalapril Comparison in Severe Hypertension (CV 137 -049) B 1 B 8 20 B 15 40 40 80 Omapatrilat Level I Se. DBP 115 -130 mm. Hg Level II B 29 Forced titration to 40, elective to 80 Level III Enalapril 10 20 Adjunct C 1 Forced titration to 20, elective to 40 20 40 A 7 B 1 Period A Single-Blind Placebo Lead-In B 71 Period B Double-Blind Randomized C 1 Period C Long-Term Open-Label
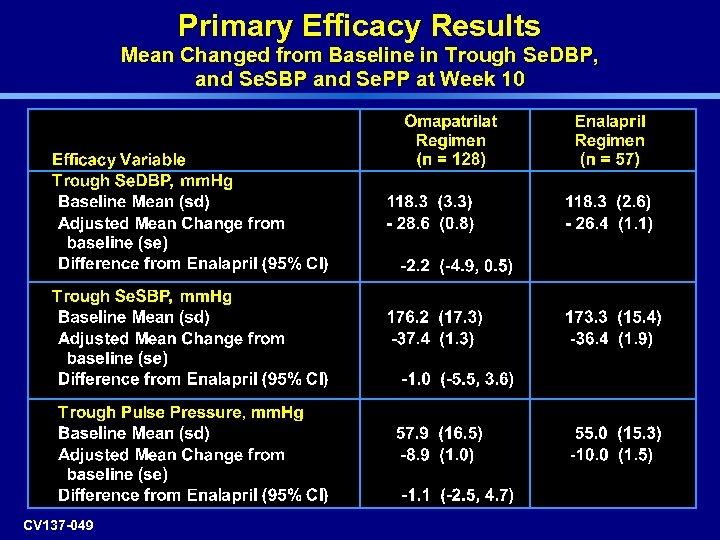
Primary Efficacy Results Mean Changed from Baseline in Trough Se. DBP, and Se. SBP and Se. PP at Week 10 CV 137 -049
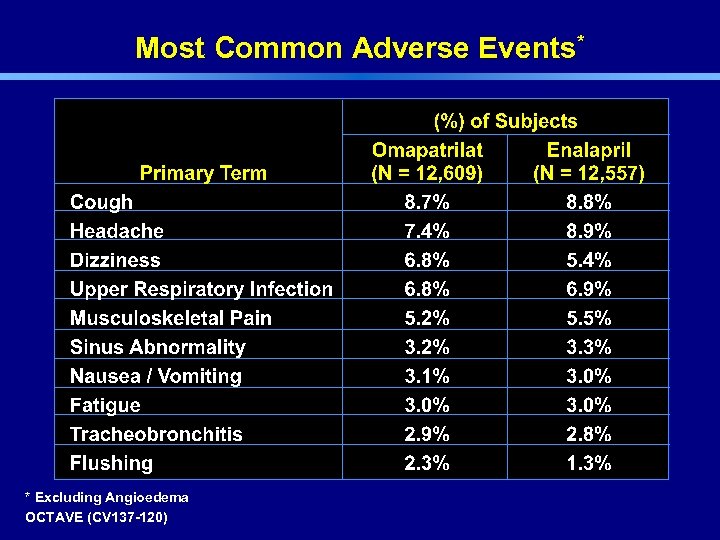
Most Common Adverse Events* * Excluding Angioedema OCTAVE (CV 137 -120)

Definition of Hospitalization for Heart Failure n OVERTURE Trial – Included all hospitalizations attributable to heart failure as adjudicated by Endpoint Committee which required IV treatment and had a duration > 24 hours n SOLVD Treatment Trial – Included all hospitalizations attributable to heart failure by the investigator regardless of treatment or duration OVERTURE (CV 137 -068)
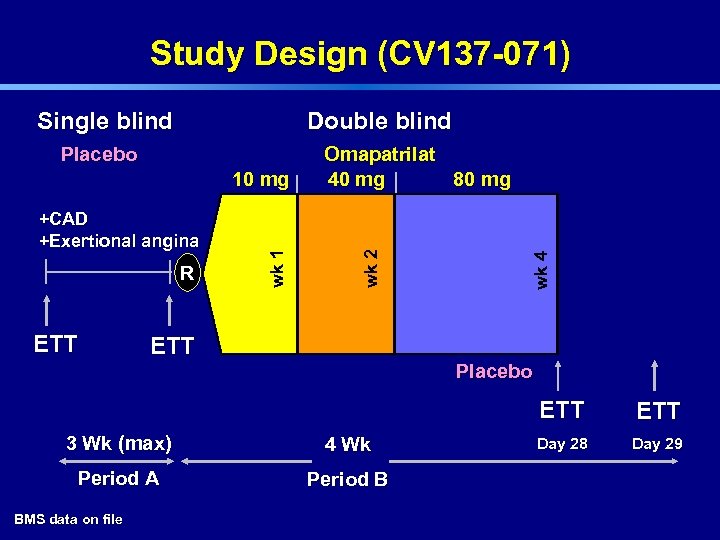
Study Design (CV 137 -071) Double blind +CAD +Exertional angina R ETT wk 1 10 mg Omapatrilat 40 mg 80 mg wk 4 Placebo wk 2 Single blind ETT Placebo ETT 3 Wk (max) 4 Wk Period A Period B BMS data on file ETT Day 28 Day 29
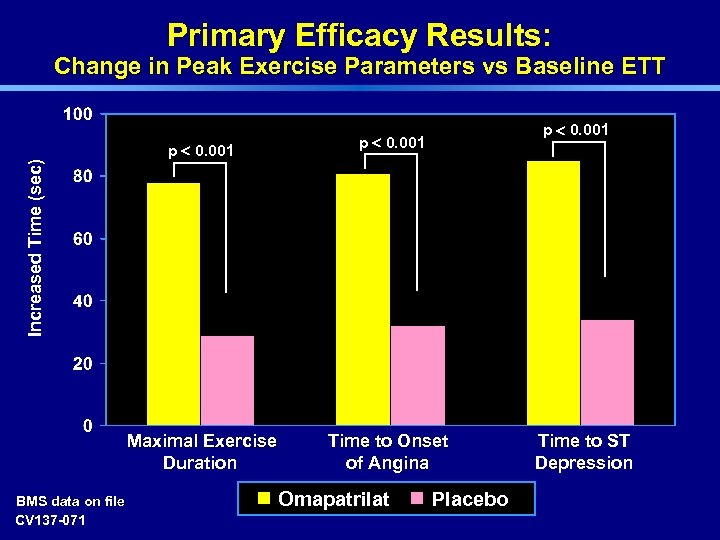
Primary Efficacy Results: Change in Peak Exercise Parameters vs Baseline ETT p < 0. 001 Increased Time (sec) Maximal Exercise Duration BMS data on file CV 137 -071 p < 0. 001 Time to Onset of Angina Omapatrilat Placebo Time to ST Depression

Diabetic Patients (CV 137 -046) Type II diabetics with microalbuminuria (30 -300 mg/gram creatinine) or overt nephropathy (> 300 mg/gram creatinine) DBP 85 -110 mm. Hg or Se. SBP 130 -180 mm. Hg omapatrilat 20 mg 40 mg 80 mg amlodipine 2. 5 mg wk 4 10 mg wk 8 Elective titration Randomization 2 week placebo lead-in 12 week Double-blind
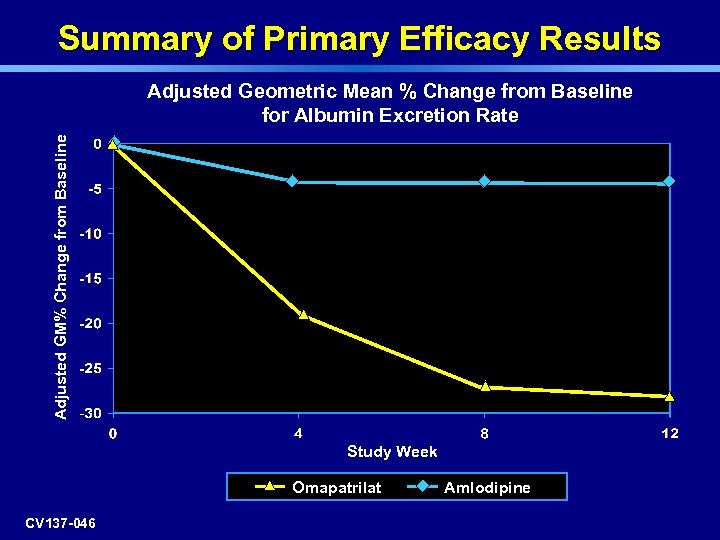
Summary of Primary Efficacy Results Adjusted GM% Change from Baseline Adjusted Geometric Mean % Change from Baseline for Albumin Excretion Rate Study Week Omapatrilat CV 137 -046 Amlodipine
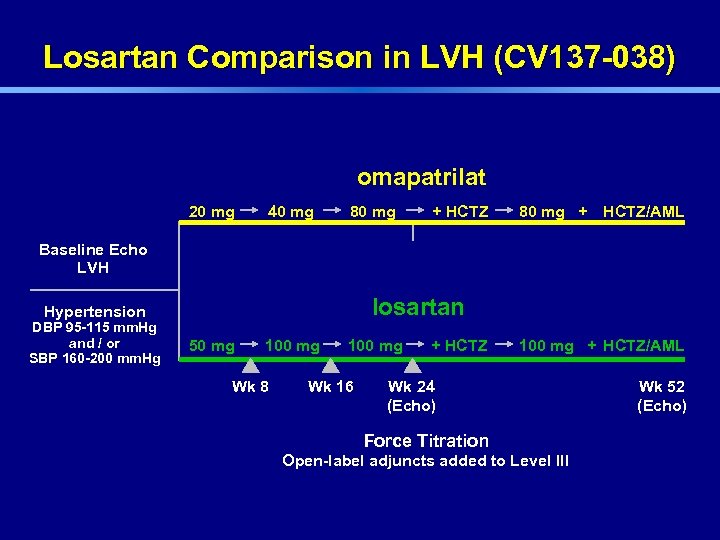
Losartan Comparison in LVH (CV 137 -038) omapatrilat 20 mg 40 mg 80 mg + HCTZ/AML Baseline Echo LVH losartan Hypertension DBP 95 -115 mm. Hg and / or SBP 160 -200 mm. Hg 50 mg 100 mg Wk 8 100 mg Wk 16 + HCTZ 100 mg + HCTZ/AML Wk 24 (Echo) Force Titration Open-label adjuncts added to Level III Wk 52 (Echo)
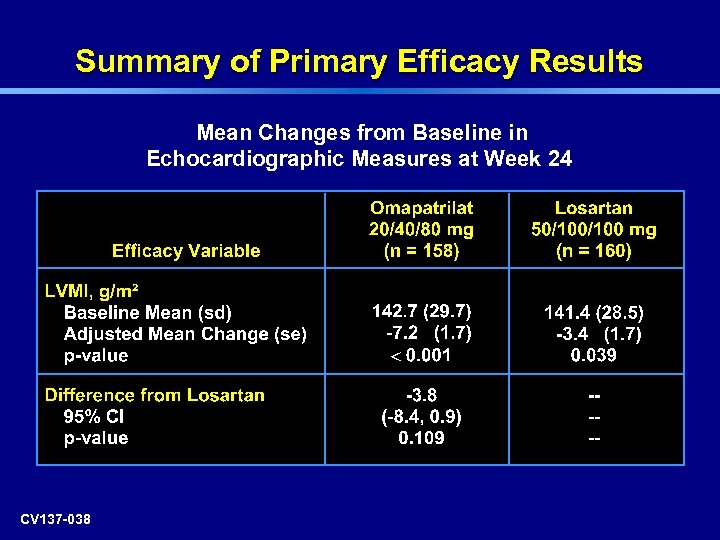
Summary of Primary Efficacy Results Mean Changes from Baseline in Echocardiographic Measures at Week 24 CV 137 -038
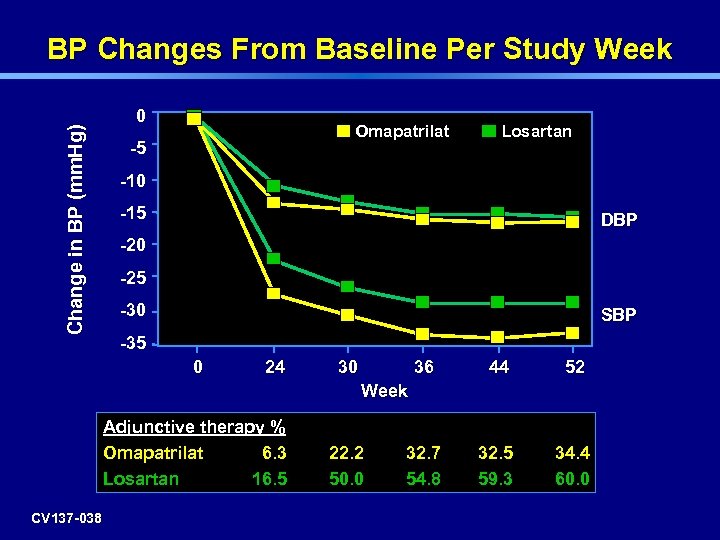
Change in BP (mm. Hg) BP Changes From Baseline Per Study Week 0 Omapatrilat -5 Losartan -10 -15 DBP -20 -25 -30 SBP -35 0 24 30 36 44 52 32. 7 54. 8 32. 5 59. 3 34. 4 60. 0 Week Adjunctive therapy % Omapatrilat 6. 3 Losartan 16. 5 CV 137 -038 22. 2 50. 0

CHOIRS Background (Conduit Hemodynamics of Omapatrilat International Research Study) n Elevated pulse pressure, an indirect measure of increased vascular stiffness, associated with: – Myocardial infarction, stroke – Development and progression of heart failure – Increased mortality n Current epidemic of uncontrolled systolic hypertension due to a lack of treatments that reduce arterial stiffness n Natriuretic peptides have a favorable effect on large arteries in basic studies although their effects in humans have not been elevated
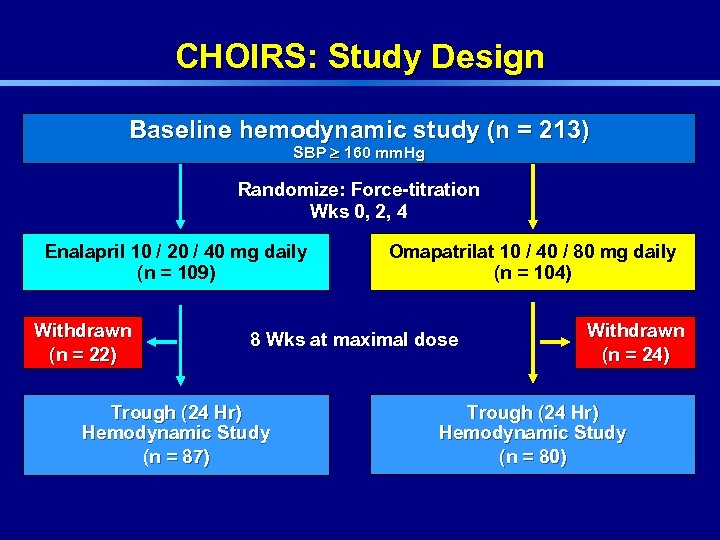
CHOIRS: Study Design Baseline hemodynamic study (n = 213) SBP 160 mm. Hg Randomize: Force-titration Wks 0, 2, 4 Enalapril 10 / 20 / 40 mg daily (n = 109) Withdrawn (n = 22) Omapatrilat 10 / 40 / 80 mg daily (n = 104) 8 Wks at maximal dose Trough (24 Hr) Hemodynamic Study (n = 87) Withdrawn (n = 24) Trough (24 Hr) Hemodynamic Study (n = 80)
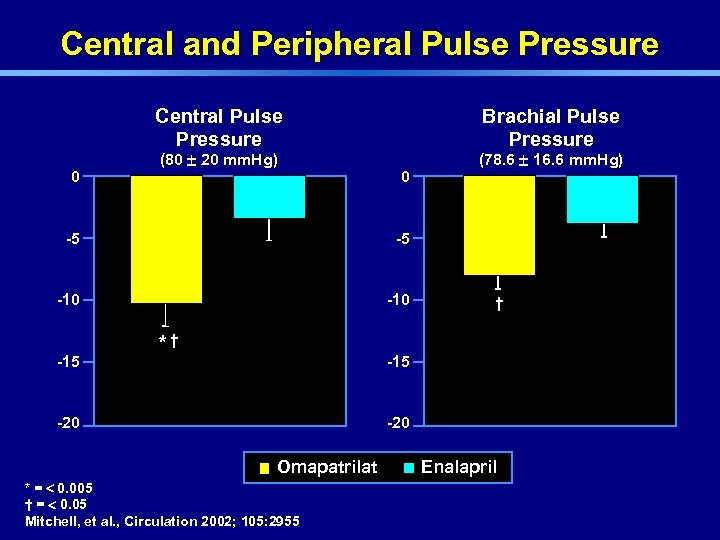
Central and Peripheral Pulse Pressure Central Pulse Pressure 0 (80 ± 20 mm. Hg) Brachial Pulse Pressure 0 -5 -5 -10 (78. 6 ± 16. 6 mm. Hg) -15 *† † -15 -20 Omapatrilat * = < 0. 005 † = < 0. 05 Mitchell, et al. , Circulation 2002; 105: 2955 Enalapril

Omapatrilat Target Population Patients with: n A high risk of major cardiovascular events* – Cardiovascular disease (e. g. , MI, CHF) – Target organ damage (e. g. , LVH, proteinuria) – 3 or more cardiovascular risk factors – Diabetes or renal disease and n Hypertension that is difficult to control with existing medications Use with special caution in black patients and current smokers *Based on WHO-ISH guidelines
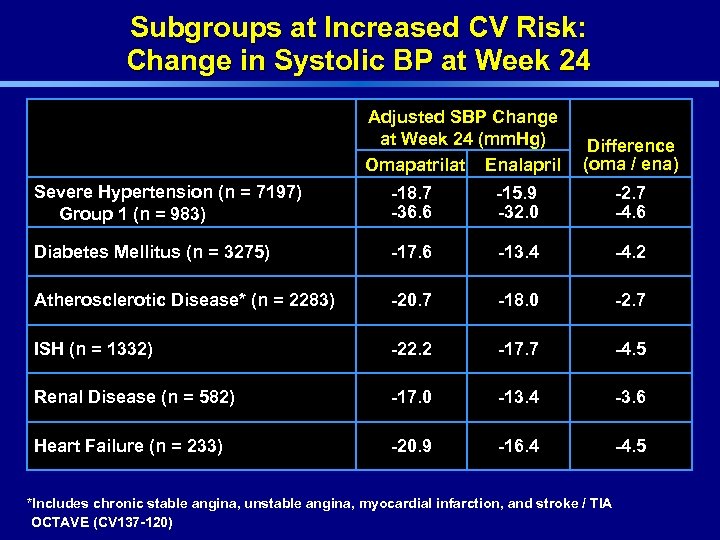
Subgroups at Increased CV Risk: Change in Systolic BP at Week 24 Adjusted SBP Change at Week 24 (mm. Hg) Omapatrilat Enalapril Difference (oma / ena) Severe Hypertension (n = 7197) Group 1 (n = 983) -18. 7 -36. 6 -15. 9 -32. 0 -2. 7 -4. 6 Diabetes Mellitus (n = 3275) -17. 6 -13. 4 -4. 2 Atherosclerotic Disease* (n = 2283) -20. 7 -18. 0 -2. 7 ISH (n = 1332) -22. 2 -17. 7 -4. 5 Renal Disease (n = 582) -17. 0 -13. 4 -3. 6 Heart Failure (n = 233) -20. 9 -16. 4 -4. 5 *Includes chronic stable angina, unstable angina, myocardial infarction, and stroke / TIA OCTAVE (CV 137 -120)
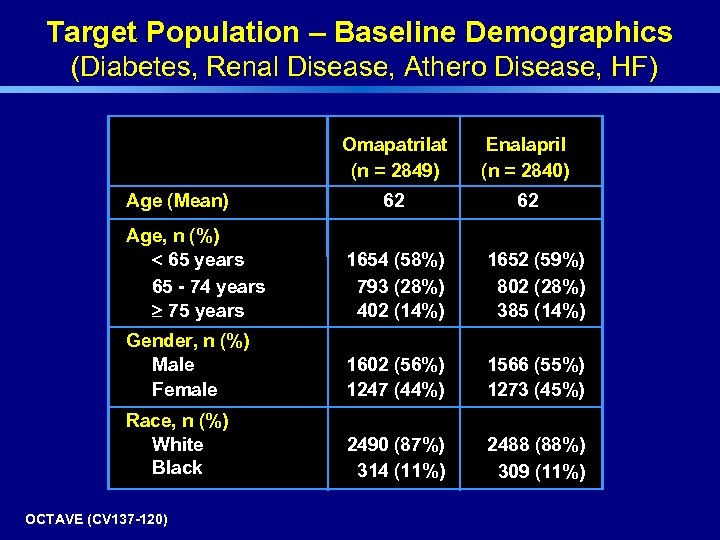
Target Population – Baseline Demographics (Diabetes, Renal Disease, Athero Disease, HF) Omapatrilat (n = 2849) Age (Mean) Enalapril (n = 2840) 62 62 Age, n (%) < 65 years 65 - 74 years 75 years 1654 (58%) 793 (28%) 402 (14%) 1652 (59%) 802 (28%) 385 (14%) Gender, n (%) Male Female 1602 (56%) 1247 (44%) 1566 (55%) 1273 (45%) Race, n (%) White Black 2490 (87%) 314 (11%) 2488 (88%) 309 (11%) OCTAVE (CV 137 -120)
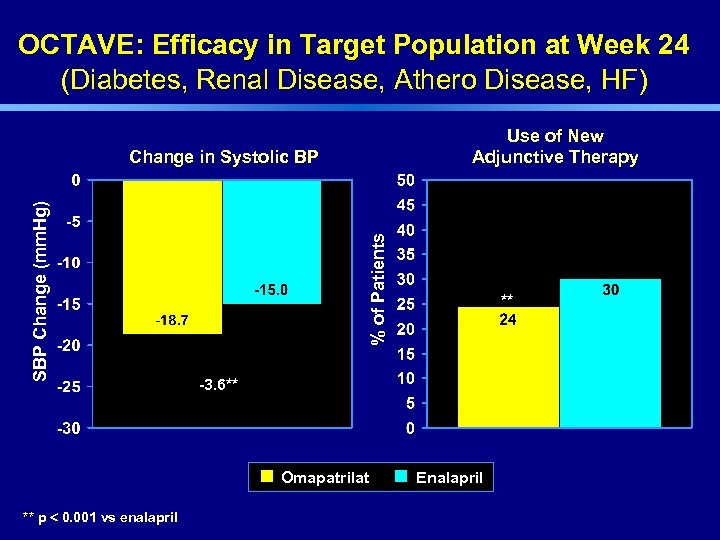
OCTAVE: Efficacy in Target Population at Week 24 (Diabetes, Renal Disease, Athero Disease, HF) Use of New Adjunctive Therapy % of Patients SBP Change (mm. Hg) Change in Systolic BP -3. 6** Omapatrilat ** p < 0. 001 vs enalapril ** Enalapril
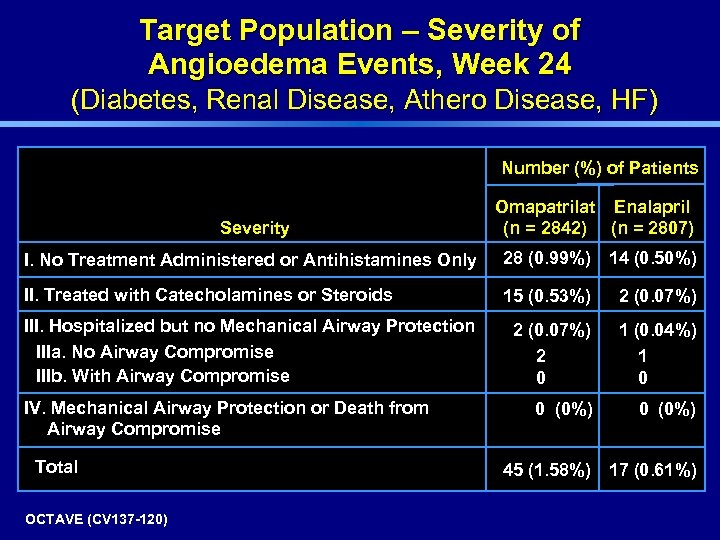
Target Population – Severity of Angioedema Events, Week 24 (Diabetes, Renal Disease, Athero Disease, HF) Number (%) of Patients Severity Omapatrilat Enalapril (n = 2842) (n = 2807) I. No Treatment Administered or Antihistamines Only 28 (0. 99%) 14 (0. 50%) II. Treated with Catecholamines or Steroids 15 (0. 53%) 2 (0. 07%) 2 0 1 (0. 04%) 1 0 0 (0%) III. Hospitalized but no Mechanical Airway Protection IIIa. No Airway Compromise IIIb. With Airway Compromise IV. Mechanical Airway Protection or Death from Airway Compromise Total OCTAVE (CV 137 -120) 45 (1. 58%) 17 (0. 61%)
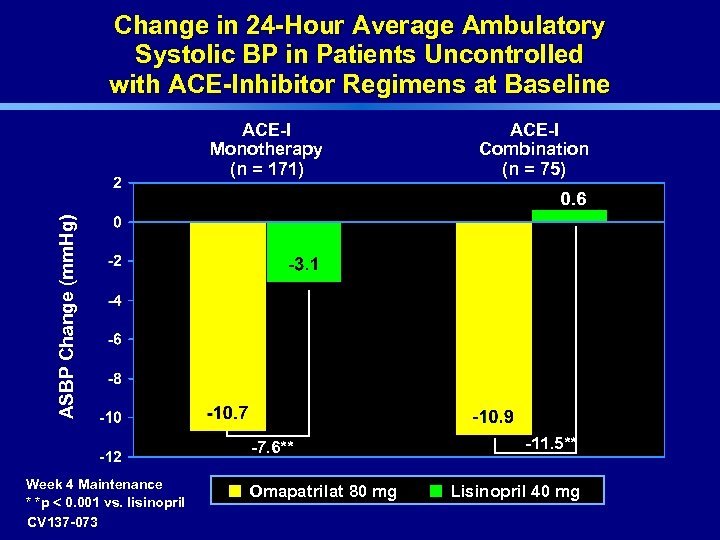
Change in 24 -Hour Average Ambulatory Systolic BP in Patients Uncontrolled with ACE-Inhibitor Regimens at Baseline ACE-I Combination (n = 75) ASBP Change (mm. Hg) ACE-I Monotherapy (n = 171) -7. 6** Week 4 Maintenance * *p < 0. 001 vs. lisinopril CV 137 -073 Omapatrilat 80 mg -11. 5** Lisinopril 40 mg

Difficult to Control Patients Untreated patients with severe hypertension Patients uncontrolled with a regimen not including an ACE-I Patients uncontrolled with a regimen including an ACE-I Omapatrilat provides consistent benefit in BP reduction over enalapril in each of these difficult to control populations.
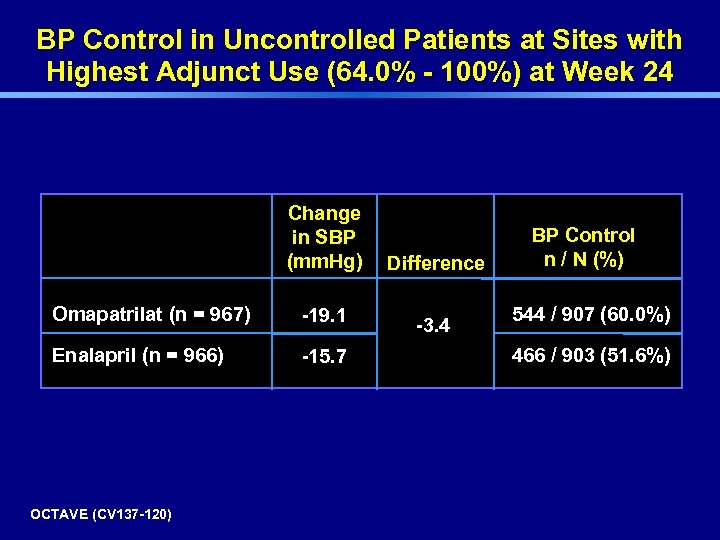
BP Control in Uncontrolled Patients at Sites with Highest Adjunct Use (64. 0% - 100%) at Week 24 Change in SBP (mm. Hg) Omapatrilat (n = 967) -19. 1 Enalapril (n = 966) -15. 7 OCTAVE (CV 137 -120) Difference -3. 4 BP Control n / N (%) 544 / 907 (60. 0%) 466 / 903 (51. 6%)
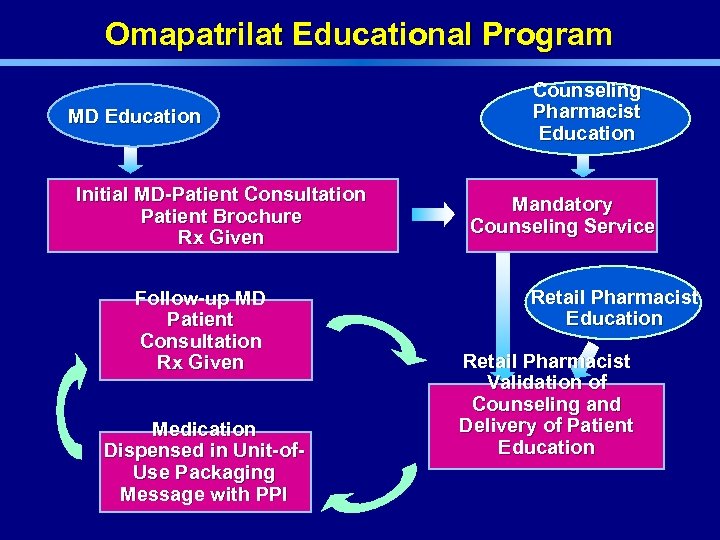
Omapatrilat Educational Program MD Education Initial MD-Patient Consultation Patient Brochure Rx Given Follow-up MD Patient Consultation Rx Given Medication Dispensed in Unit-of. Use Packaging Message with PPI Counseling Pharmacist Education Mandatory Counseling Service Retail Pharmacist Education Retail Pharmacist Validation of Counseling and Delivery of Patient Education
e8d08da1a4d6c4bafb22c8cb1f9ffcf2.ppt