be1f336d43b1dd02e71289c3c29b89ea.ppt
- Количество слайдов: 25
 Challenges in production and use of radiopharmaceuticals in Kenya: a review Rogers Atebe MPharm(Industrial Pharmacy) Year II Department of Pharmaceutics and Pharmacy Practice School of Pharmacy, University of Nairobi [Presentation for the Annual PSK Scientific Conference: 3 rd June 2016]
Challenges in production and use of radiopharmaceuticals in Kenya: a review Rogers Atebe MPharm(Industrial Pharmacy) Year II Department of Pharmaceutics and Pharmacy Practice School of Pharmacy, University of Nairobi [Presentation for the Annual PSK Scientific Conference: 3 rd June 2016]
 Radiopharmaceuticals • Radiopharmaceuticals - Unique medicinal formulations containing radioisotope(s) • mainly used in major clinical areas for diagnosis and/or therapy • Diagnosis/ imaging: – Essential for mapping physiological function and metabolic activity giving specific information about organ function and dysfunction; mapping functional morphology of organs noninvasively – Have high specificity when tagged to molecules of high affinity for specific tissues such as monoclonal antibodies, macroaggregated albumin, methylene diphosphonate; – For example - detection and staging of some types of cancers. 2
Radiopharmaceuticals • Radiopharmaceuticals - Unique medicinal formulations containing radioisotope(s) • mainly used in major clinical areas for diagnosis and/or therapy • Diagnosis/ imaging: – Essential for mapping physiological function and metabolic activity giving specific information about organ function and dysfunction; mapping functional morphology of organs noninvasively – Have high specificity when tagged to molecules of high affinity for specific tissues such as monoclonal antibodies, macroaggregated albumin, methylene diphosphonate; – For example - detection and staging of some types of cancers. 2
 example • Positron emission Tomography(PET)- a very specific and targeted imaging technique widely used globally – Employing radiopharmaceuticals in several disease diagnosis – Commonly used radiopharmaceutical is 18 Ffluorodeoxyglucose(18 F-FDG) – Many Kenyans trooping to India for this imaging • At least 70 -80% of cancers are diagnosed in late stages due to: lack of awareness; inadequate diagnostic facilities; lack of treatment facilities; high cost of treatment mainly due to high poverty index. 3
example • Positron emission Tomography(PET)- a very specific and targeted imaging technique widely used globally – Employing radiopharmaceuticals in several disease diagnosis – Commonly used radiopharmaceutical is 18 Ffluorodeoxyglucose(18 F-FDG) – Many Kenyans trooping to India for this imaging • At least 70 -80% of cancers are diagnosed in late stages due to: lack of awareness; inadequate diagnostic facilities; lack of treatment facilities; high cost of treatment mainly due to high poverty index. 3
 …… • Radiopharmaceuticals find use in treatment of several diseases including some types of cancers • Either used alone or in combination with otherapies • In Kenya, cancer causes more deaths than HIV, TB and malaria combined. • About 30 % of cancers are: – curable if detected early; – treatable with prolonged survival if detected early; • 30% of cancer patients can be provided with adequate symptom management and palliative care 4
…… • Radiopharmaceuticals find use in treatment of several diseases including some types of cancers • Either used alone or in combination with otherapies • In Kenya, cancer causes more deaths than HIV, TB and malaria combined. • About 30 % of cancers are: – curable if detected early; – treatable with prolonged survival if detected early; • 30% of cancer patients can be provided with adequate symptom management and palliative care 4
 Levels of production for radiopharmaceuticals • Level 1: Purchase and delivery of final dose form; no manipulation [hospital] • Level 2: Minimal manipulation from prepared and approved reagents (kits) [radiopharmacy/hotlab] – E. g. Milking 99 Mo generator for 99 MTc; Labeling of blood cells • Level 3: Compounding from parent ingredients – 3 a: diagnostic radiopharmaceuticals – 3 b: therapeutic radiopharmaceuticals – 3 c: synthesis/radiolabeling e. g 18 F-FDG for PET 5
Levels of production for radiopharmaceuticals • Level 1: Purchase and delivery of final dose form; no manipulation [hospital] • Level 2: Minimal manipulation from prepared and approved reagents (kits) [radiopharmacy/hotlab] – E. g. Milking 99 Mo generator for 99 MTc; Labeling of blood cells • Level 3: Compounding from parent ingredients – 3 a: diagnostic radiopharmaceuticals – 3 b: therapeutic radiopharmaceuticals – 3 c: synthesis/radiolabeling e. g 18 F-FDG for PET 5
 Some operational definitions • Radiopharmaceutical preparation: a ready-to-use form of the radiopharmaceutical • Radionuclide generator: for elution of daughter radionuclide for radiopharmaceutical preparation • Radiopharmaceutical precursor: required in radiolabeling process to result into a radiopharmaceutical preparation • Kit for radiopharmaceutical preparation: a sterilized, validated product to which the appropriate radionuclide is added (or into which it is diluted) before medical use 6
Some operational definitions • Radiopharmaceutical preparation: a ready-to-use form of the radiopharmaceutical • Radionuclide generator: for elution of daughter radionuclide for radiopharmaceutical preparation • Radiopharmaceutical precursor: required in radiolabeling process to result into a radiopharmaceutical preparation • Kit for radiopharmaceutical preparation: a sterilized, validated product to which the appropriate radionuclide is added (or into which it is diluted) before medical use 6
 Primary production of radiopharmaceuticals • Radionuclide required to make radiopharmaceuticals produced mostly artificially via either nuclear reactors, Nuclear fission production, cyclotrons or linear accelerator processes • e. g 99 Mo for elution of 99 MTc useful in production of γ-rays for imaging obtained from fission production • Equipment installation is capital intensive including building of bunkers for safety concerns e. g recent commissioning of a > Ksh 2 Billion linear accelerator in KNH • Lack of skilled personnel to operate equipment and use technologies- Lack of radiopharmacists, oncologists, medical Physicists etc 7
Primary production of radiopharmaceuticals • Radionuclide required to make radiopharmaceuticals produced mostly artificially via either nuclear reactors, Nuclear fission production, cyclotrons or linear accelerator processes • e. g 99 Mo for elution of 99 MTc useful in production of γ-rays for imaging obtained from fission production • Equipment installation is capital intensive including building of bunkers for safety concerns e. g recent commissioning of a > Ksh 2 Billion linear accelerator in KNH • Lack of skilled personnel to operate equipment and use technologies- Lack of radiopharmacists, oncologists, medical Physicists etc 7
![Primary considerations for production • Pharmaceutical production guidelines [c. GMP] • Safety concern: radiations Primary considerations for production • Pharmaceutical production guidelines [c. GMP] • Safety concern: radiations](https://present5.com/presentation/be1f336d43b1dd02e71289c3c29b89ea/image-8.jpg) Primary considerations for production • Pharmaceutical production guidelines [c. GMP] • Safety concern: radiations are potentially hazardous – IAEA guidelines & national regulations – General principles of radioprotection: • Justification principle • Optimization principle (ALARP) • Limitation of exposure to radiation (legal) • Time limitation principle [minimize] • Distance limitation principle [maximize] • Shielding principle (heavy concrete, lead brick walls; tungsten) • Time constraints: natural radioactive decay based on halflives…e. g. t 1/2 for 99 Mo is 66 hrs and 6 hrs for 99 MTc • Regulatory framework ( production and use) 8
Primary considerations for production • Pharmaceutical production guidelines [c. GMP] • Safety concern: radiations are potentially hazardous – IAEA guidelines & national regulations – General principles of radioprotection: • Justification principle • Optimization principle (ALARP) • Limitation of exposure to radiation (legal) • Time limitation principle [minimize] • Distance limitation principle [maximize] • Shielding principle (heavy concrete, lead brick walls; tungsten) • Time constraints: natural radioactive decay based on halflives…e. g. t 1/2 for 99 Mo is 66 hrs and 6 hrs for 99 MTc • Regulatory framework ( production and use) 8
![Quality assurance [c. GMP & c. GRPP] c. GMP c. GRPP Current Good manufacturing Quality assurance [c. GMP & c. GRPP] c. GMP c. GRPP Current Good manufacturing](https://present5.com/presentation/be1f336d43b1dd02e71289c3c29b89ea/image-9.jpg) Quality assurance [c. GMP & c. GRPP] c. GMP c. GRPP Current Good manufacturing practices Design and development Delineated responsibilities Correct raw and packaging materials Batch release QP Self inspection and quality audits Shelf life quality • Current good radiopharmacy practice (c. GRPP) • Specific QA considerations based on material characteristics • Low volumes • Possibility of administration before full QC tests are done • Process validation is key 9
Quality assurance [c. GMP & c. GRPP] c. GMP c. GRPP Current Good manufacturing practices Design and development Delineated responsibilities Correct raw and packaging materials Batch release QP Self inspection and quality audits Shelf life quality • Current good radiopharmacy practice (c. GRPP) • Specific QA considerations based on material characteristics • Low volumes • Possibility of administration before full QC tests are done • Process validation is key 9
 GMP • Procurement, – Quality and sources of correct raw and packaging materials – Transportation and storage – Labeling requirements (radioactive!) • Facility set up and management based on operational level – Licenses: PPB, NEMA, Radiation Board, IAEA – Environmental and operational safety • Receiving, QC testing – Radiochemical & radionuclidic purity, sterility, apyrogenicity • Storage, dispensing (aseptic conditions) 10
GMP • Procurement, – Quality and sources of correct raw and packaging materials – Transportation and storage – Labeling requirements (radioactive!) • Facility set up and management based on operational level – Licenses: PPB, NEMA, Radiation Board, IAEA – Environmental and operational safety • Receiving, QC testing – Radiochemical & radionuclidic purity, sterility, apyrogenicity • Storage, dispensing (aseptic conditions) 10
 GMP • Functional QA System – Critical role of qualification, validation and calibration; aseptic procedure – Actual testing • Most after release and use – Batch release • Parametric release • Waste disposal – Full identification of waste and site – Short half-life separate from long half-life – Methods: (i)dilute & disperse, (ii)delay & decay, (iii)concentrate & contain 11
GMP • Functional QA System – Critical role of qualification, validation and calibration; aseptic procedure – Actual testing • Most after release and use – Batch release • Parametric release • Waste disposal – Full identification of waste and site – Short half-life separate from long half-life – Methods: (i)dilute & disperse, (ii)delay & decay, (iii)concentrate & contain 11
 GMP • Production – – Premises material of construction Processing: validated processes Radioisotope labs and “hot cells” Radioprotection principles • Personnel – Pharmacist, nuclear technologist/radiographer, nuclear physician, nuclear physicist, biologist, nurse – Basic training [IAEA curriculum] • Mathematics of radiation, radioprotection and regulation, radiation biology, radiopharmaceutical chemistry, clinical uses of radiopharmaceuticals 12
GMP • Production – – Premises material of construction Processing: validated processes Radioisotope labs and “hot cells” Radioprotection principles • Personnel – Pharmacist, nuclear technologist/radiographer, nuclear physician, nuclear physicist, biologist, nurse – Basic training [IAEA curriculum] • Mathematics of radiation, radioprotection and regulation, radiation biology, radiopharmaceutical chemistry, clinical uses of radiopharmaceuticals 12
 Labeling as per GMP • More stringent compared to conventional pharmaceuticals • A statement that the product is radioactive or the international symbol for radioactivity • Name of the radiopharmaceutical preparation; • Indication that the preparation is for diagnostic or for therapeutic use; including Route of administration; • Total radioactivity present at a stated date and, where necessary, time; and for solutions, a statement of the radioactivity in a suitable volume (for example, in MBq per ml of the solution) may be given instead; Total volume of solutions at time of measuring activity. • Expiry date and, where necessary, time[where half-life is less than 30 days]; • Batch (lot) number assigned by the manufacturer 13
Labeling as per GMP • More stringent compared to conventional pharmaceuticals • A statement that the product is radioactive or the international symbol for radioactivity • Name of the radiopharmaceutical preparation; • Indication that the preparation is for diagnostic or for therapeutic use; including Route of administration; • Total radioactivity present at a stated date and, where necessary, time; and for solutions, a statement of the radioactivity in a suitable volume (for example, in MBq per ml of the solution) may be given instead; Total volume of solutions at time of measuring activity. • Expiry date and, where necessary, time[where half-life is less than 30 days]; • Batch (lot) number assigned by the manufacturer 13
 Outer package labeling • Add: – any special storage requirements with respect to temperature and light; – where applicable, the name and concentration of any added microbial preservatives or, where necessary, that no antimicrobial preservative has been added; • Radiation Protection Programs for the Transport of Radioactive Material Safety Guide (IAEA, Vienna, 2007). Consult the IAEA website (http: //wwwns. iaea. org/standards/) for the current guidance 14
Outer package labeling • Add: – any special storage requirements with respect to temperature and light; – where applicable, the name and concentration of any added microbial preservatives or, where necessary, that no antimicrobial preservative has been added; • Radiation Protection Programs for the Transport of Radioactive Material Safety Guide (IAEA, Vienna, 2007). Consult the IAEA website (http: //wwwns. iaea. org/standards/) for the current guidance 14
 • Documentation GMP – Material flow, handling and control records – BMR with emphasis on date and time(for half-life of less than 30 days) – Equipment records – Personnel personal, medical, training and radiation monitoring records – Qualification, validation, calibration records – Disposal records including any spillages 15
• Documentation GMP – Material flow, handling and control records – BMR with emphasis on date and time(for half-life of less than 30 days) – Equipment records – Personnel personal, medical, training and radiation monitoring records – Qualification, validation, calibration records – Disposal records including any spillages 15
 Regulatory issues • Radiation Protection Act; Cap 243 Laws of Kenya [Commencement: 1 st November, 1984] • The Pharmacy and Poisons Act; Cap 244 Laws of Kenya [Commencement: 1 st May, 1957] 16
Regulatory issues • Radiation Protection Act; Cap 243 Laws of Kenya [Commencement: 1 st November, 1984] • The Pharmacy and Poisons Act; Cap 244 Laws of Kenya [Commencement: 1 st May, 1957] 16
 Cap 244 Interpretation “drug” includes any medicine, medicinal (s 2) preparation or therapeutic substance; “medicinal substance” means any medicine, product, article, or substance which is claimed to be useful for any of the following purposes— (a) treating, preventing or alleviating disease or symptoms of disease; (b) diagnosing disease or ascertaining the existence, degree or extent of a physiological condition; or (c) preventing or interfering with the normal operation of a physiological function whether permanently or temporarily and whether by way of terminating, reducing, postponing or increasing or accelerating the operation of the function in human beings or animals; Radiopharmaceuticals covered in broad definition: • Diagnostic role (imaging) • Therapeutic role 17
Cap 244 Interpretation “drug” includes any medicine, medicinal (s 2) preparation or therapeutic substance; “medicinal substance” means any medicine, product, article, or substance which is claimed to be useful for any of the following purposes— (a) treating, preventing or alleviating disease or symptoms of disease; (b) diagnosing disease or ascertaining the existence, degree or extent of a physiological condition; or (c) preventing or interfering with the normal operation of a physiological function whether permanently or temporarily and whether by way of terminating, reducing, postponing or increasing or accelerating the operation of the function in human beings or animals; Radiopharmaceuticals covered in broad definition: • Diagnostic role (imaging) • Therapeutic role 17
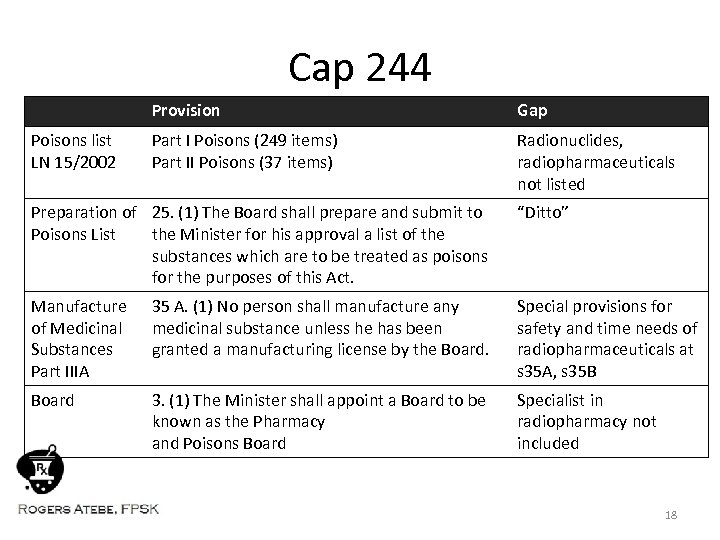 Cap 244 Provision Poisons list LN 15/2002 Gap Part I Poisons (249 items) Part II Poisons (37 items) Radionuclides, radiopharmaceuticals not listed Preparation of 25. (1) The Board shall prepare and submit to Poisons List the Minister for his approval a list of the substances which are to be treated as poisons for the purposes of this Act. “Ditto” Manufacture of Medicinal Substances Part IIIA 35 A. (1) No person shall manufacture any medicinal substance unless he has been granted a manufacturing license by the Board. Special provisions for safety and time needs of radiopharmaceuticals at s 35 A, s 35 B Board 3. (1) The Minister shall appoint a Board to be known as the Pharmacy and Poisons Board Specialist in radiopharmacy not included 18
Cap 244 Provision Poisons list LN 15/2002 Gap Part I Poisons (249 items) Part II Poisons (37 items) Radionuclides, radiopharmaceuticals not listed Preparation of 25. (1) The Board shall prepare and submit to Poisons List the Minister for his approval a list of the substances which are to be treated as poisons for the purposes of this Act. “Ditto” Manufacture of Medicinal Substances Part IIIA 35 A. (1) No person shall manufacture any medicinal substance unless he has been granted a manufacturing license by the Board. Special provisions for safety and time needs of radiopharmaceuticals at s 35 A, s 35 B Board 3. (1) The Minister shall appoint a Board to be known as the Pharmacy and Poisons Board Specialist in radiopharmacy not included 18
 Challenges • Highly specialized facilities design: high investment costs • Qualified specialized equipment • Demand for aseptic technique (equipment, trained staff) – Designated clean room requirements and hot cells • Small scale: difficult and expensive to implement GMP & lack of economies of scale • Highly technical: requires highly specialized trained manpower • Use of controlled materials /procedures ( international and national regulations) 19
Challenges • Highly specialized facilities design: high investment costs • Qualified specialized equipment • Demand for aseptic technique (equipment, trained staff) – Designated clean room requirements and hot cells • Small scale: difficult and expensive to implement GMP & lack of economies of scale • Highly technical: requires highly specialized trained manpower • Use of controlled materials /procedures ( international and national regulations) 19
 Challenges • Radiological and pharmaceutical safety • Adaptation of GMP to radioactive products • Possibility of administration before full QC tests are done • Human Capacity for cancer treatment in Kenya (public sector) – Currently: 4 radiation oncologists, 6 medical oncologists, 4 pediatric oncologists, 5 radiation therapy technologists, 3 oncology nurses and 2 medical physicists. NO RADIOPHARMACIST • Disposal of radioactive waste 20
Challenges • Radiological and pharmaceutical safety • Adaptation of GMP to radioactive products • Possibility of administration before full QC tests are done • Human Capacity for cancer treatment in Kenya (public sector) – Currently: 4 radiation oncologists, 6 medical oncologists, 4 pediatric oncologists, 5 radiation therapy technologists, 3 oncology nurses and 2 medical physicists. NO RADIOPHARMACIST • Disposal of radioactive waste 20
 Overcoming the challenges • Mainstreaming radiopharmaceuticals into the pharmaceutical regulatory framework under cap 244 • Training (curriculum development) and recognition nuclear pharmacists as a specialty under pharmacy law. • Create centralised radiopharmaceuticals manufacturing approach for economies of scale • Seek / collaborate for technical support, including skills training, from external agencies e. g IAEA • Enthuse development partners to identify radiopharmaceuticals as a need for LMICs [seat of 70% of all cancer cases globally] • Incentives for innovation in pharmaceutical manufacturing sector • Team approach in nuclear medicine practice, and integration of all healthcare professionals involved 21
Overcoming the challenges • Mainstreaming radiopharmaceuticals into the pharmaceutical regulatory framework under cap 244 • Training (curriculum development) and recognition nuclear pharmacists as a specialty under pharmacy law. • Create centralised radiopharmaceuticals manufacturing approach for economies of scale • Seek / collaborate for technical support, including skills training, from external agencies e. g IAEA • Enthuse development partners to identify radiopharmaceuticals as a need for LMICs [seat of 70% of all cancer cases globally] • Incentives for innovation in pharmaceutical manufacturing sector • Team approach in nuclear medicine practice, and integration of all healthcare professionals involved 21
 Conclusion • Manufacture of radiopharmaceuticals from the precursor ingredients requires additional skills and special competencies as well as facilities as prescribed by the International Atomic Energy Agency (IAEA), the global regulator of use of nuclear technologies. • Kenya lags behind in harnessing the benefits of use of radiopharmaceuticals • There is therefore need for national regulations to augment the IAEA regulations. • As a country we require regulations in the area of GMP guidelines including defining the qualified personnel in this field. • Furthere is need to mainstream radiopharmaceuticals under the control of the Pharmacy and Poisons Act, cap 244 of the Laws of Kenya. 22
Conclusion • Manufacture of radiopharmaceuticals from the precursor ingredients requires additional skills and special competencies as well as facilities as prescribed by the International Atomic Energy Agency (IAEA), the global regulator of use of nuclear technologies. • Kenya lags behind in harnessing the benefits of use of radiopharmaceuticals • There is therefore need for national regulations to augment the IAEA regulations. • As a country we require regulations in the area of GMP guidelines including defining the qualified personnel in this field. • Furthere is need to mainstream radiopharmaceuticals under the control of the Pharmacy and Poisons Act, cap 244 of the Laws of Kenya. 22
 References • • • Amugune, B. (2016). UIP 710 Radiopharmaceuticals notes (p. 7). Dar, M. A. , Masoodi, M. H. , & Farooq, S. (2015). Medical uses of Radiopharmaceuticals, 3(8), 24– 29. Elsinga, P. , Todde, S. , Penuelas, I. , & et al. (2010). Guidance on current good radiopharmacy practice (c. GRPP) for the small-scale preparation of radiopharmaceuticals. European Journal of Nuclear Medicine and Molecular Imaging, 37(5), 1049– 62. http: //doi. org/10. 1007/s 00259 -010 -1407 -3 European Commission. (2008). Manufacture of Radiopharmaceuticals - Annex 3. Eudra. Lex, (December 2007), 1 – 8. IAEA. (2006). Radiopharmaceuticals Production and Availability. Radiopharmaceuticals: Production and Availability. IP. (2008). RADIOPHARMACEUTICALS Final text for addition to The International Pharmacopoeia. In International Pharmacopoeia (Vol. 2, pp. 1– 28). Retrieved from http: //www. who. int/medicines/publications/pharmacopoeia/Radgenmono. pdf Malvi, R. , Bajpai, R. , & Jain, S. (2012). A Review on Therapeutic Approach of Radiopharmaceutical in Health Care System, 3(3), 487– 492. Votano, J. , Parham, M. , & Hall, L. (2008). Operational Guidance on Hospital Radiopharmacy: a Safe and Effective Approach. Chemistry & , 75. Retrieved from http: //onlinelibrary. wiley. com/doi/10. 1002/cbdv. 200490137/abstract WHO. (2003). Annex 3 Guidelines on Good Manufacturing Practices for radiopharmaceutical products, 4(908), 26– 35. WHO. (2007). Quality assurance of Pharmaceuticals Vol 2, 2 nd Edition, 2, 2 nd upd, 418. 23
References • • • Amugune, B. (2016). UIP 710 Radiopharmaceuticals notes (p. 7). Dar, M. A. , Masoodi, M. H. , & Farooq, S. (2015). Medical uses of Radiopharmaceuticals, 3(8), 24– 29. Elsinga, P. , Todde, S. , Penuelas, I. , & et al. (2010). Guidance on current good radiopharmacy practice (c. GRPP) for the small-scale preparation of radiopharmaceuticals. European Journal of Nuclear Medicine and Molecular Imaging, 37(5), 1049– 62. http: //doi. org/10. 1007/s 00259 -010 -1407 -3 European Commission. (2008). Manufacture of Radiopharmaceuticals - Annex 3. Eudra. Lex, (December 2007), 1 – 8. IAEA. (2006). Radiopharmaceuticals Production and Availability. Radiopharmaceuticals: Production and Availability. IP. (2008). RADIOPHARMACEUTICALS Final text for addition to The International Pharmacopoeia. In International Pharmacopoeia (Vol. 2, pp. 1– 28). Retrieved from http: //www. who. int/medicines/publications/pharmacopoeia/Radgenmono. pdf Malvi, R. , Bajpai, R. , & Jain, S. (2012). A Review on Therapeutic Approach of Radiopharmaceutical in Health Care System, 3(3), 487– 492. Votano, J. , Parham, M. , & Hall, L. (2008). Operational Guidance on Hospital Radiopharmacy: a Safe and Effective Approach. Chemistry & , 75. Retrieved from http: //onlinelibrary. wiley. com/doi/10. 1002/cbdv. 200490137/abstract WHO. (2003). Annex 3 Guidelines on Good Manufacturing Practices for radiopharmaceutical products, 4(908), 26– 35. WHO. (2007). Quality assurance of Pharmaceuticals Vol 2, 2 nd Edition, 2, 2 nd upd, 418. 23
 Acknowledgements • Dr Beatrice Amugune, Ph. D – School of Pharmacy University of Nairobi • Prof Kimani Kuria, Ph. D – School of Pharmacy, University of Nairobi 24
Acknowledgements • Dr Beatrice Amugune, Ph. D – School of Pharmacy University of Nairobi • Prof Kimani Kuria, Ph. D – School of Pharmacy, University of Nairobi 24
 THANK YOU 25
THANK YOU 25


