a240e1cae056a88f26f696b90a315ad1.ppt
- Количество слайдов: 47
 Chagas Disease: Clues to the Magnitude of the Problem in Texas “EJ” Hanford, ABD Dr. F. Ben Zhan Dr. Yongmei Lu Dr. Alberto Giordano
Chagas Disease: Clues to the Magnitude of the Problem in Texas “EJ” Hanford, ABD Dr. F. Ben Zhan Dr. Yongmei Lu Dr. Alberto Giordano
 Research Support & Funding Ø Texas State University Center for Geographic Information Science Ø Border Health Initiative Project Effort sponsored in part by the 311 th Human Systems Wing PIA FA 8909 -04 -3 -5000 Brooks City-Base Foundation, Inc.
Research Support & Funding Ø Texas State University Center for Geographic Information Science Ø Border Health Initiative Project Effort sponsored in part by the 311 th Human Systems Wing PIA FA 8909 -04 -3 -5000 Brooks City-Base Foundation, Inc.
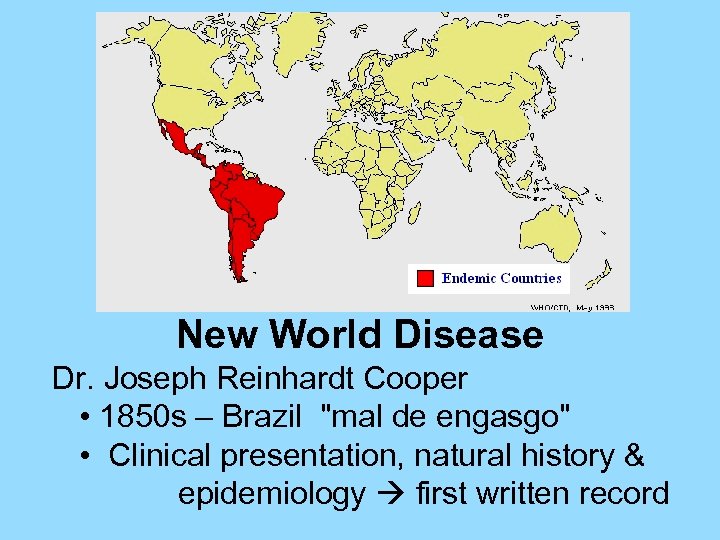 New World Disease Dr. Joseph Reinhardt Cooper • 1850 s – Brazil "mal de engasgo" • Clinical presentation, natural history & epidemiology first written record
New World Disease Dr. Joseph Reinhardt Cooper • 1850 s – Brazil "mal de engasgo" • Clinical presentation, natural history & epidemiology first written record
 Historical Timeline 1909 Dr. Carlos Chagas 1936 Southern Mexico – first recognized case 1955 First indigenous cases in U. S. = 2 in Texas 1970 s Central Mexico 2006 1 st FDA approved 2007 blood-screening test
Historical Timeline 1909 Dr. Carlos Chagas 1936 Southern Mexico – first recognized case 1955 First indigenous cases in U. S. = 2 in Texas 1970 s Central Mexico 2006 1 st FDA approved 2007 blood-screening test
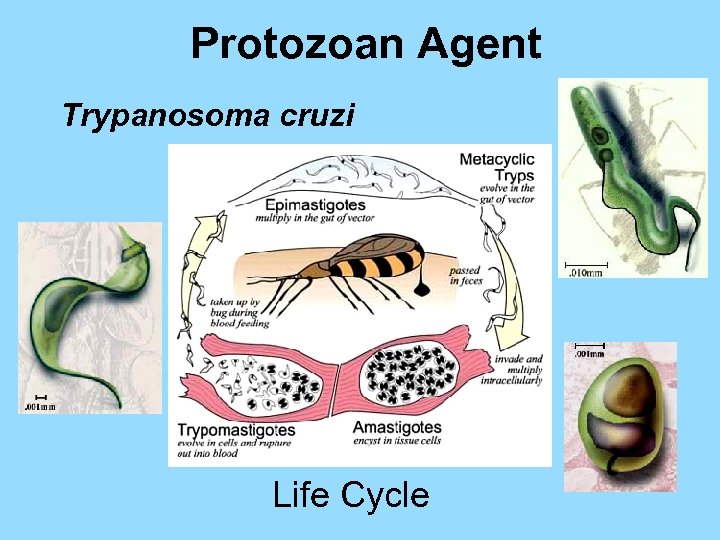 Protozoan Agent Trypanosoma cruzi Life Cycle
Protozoan Agent Trypanosoma cruzi Life Cycle
 Diagnostic Techniques • Clinical Evaluations & … • Demonstration of Parasite – – – Direct microscopic examination Xenodiagnosis Lab animal inoculation Hemoculture (less sensitive) PCR • Serologic Testing – CF, IH, DA, IIF – ELISA (Cross reactions can occur to Leishmaniasis, Blastomycosis, and Toxoplasmosis)
Diagnostic Techniques • Clinical Evaluations & … • Demonstration of Parasite – – – Direct microscopic examination Xenodiagnosis Lab animal inoculation Hemoculture (less sensitive) PCR • Serologic Testing – CF, IH, DA, IIF – ELISA (Cross reactions can occur to Leishmaniasis, Blastomycosis, and Toxoplasmosis)
 Transmission to Humans • Fecal contamination • enters Triatomid bite • through mucosal tissue (eye) • through open wound • within consumed food or water • • • Blood transfusion from infected person Organ donation from infected person Vertical transmission (Congenital multigen. ) Laboratory-acquired (? ) Oral transmission Other ?
Transmission to Humans • Fecal contamination • enters Triatomid bite • through mucosal tissue (eye) • through open wound • within consumed food or water • • • Blood transfusion from infected person Organ donation from infected person Vertical transmission (Congenital multigen. ) Laboratory-acquired (? ) Oral transmission Other ?
 Acute Chagas Disease • • • Initial infection Incubation 1 to 2 weeks May be NO symptoms (98 -99%) May be incorrectly diagnosed Symptoms: Chagoma or Romaña's sign mild fever and/or malaise enlarged lymph nodes, liver, spleen, heart high fever, convulsions & meningoencephalitis • Mortality rate: up to 50% in the young, overall ~ 10% • Diagnosis after incubation: by serodiagnosis or xenodiagnosis • Duration: acute stage may last up to several months
Acute Chagas Disease • • • Initial infection Incubation 1 to 2 weeks May be NO symptoms (98 -99%) May be incorrectly diagnosed Symptoms: Chagoma or Romaña's sign mild fever and/or malaise enlarged lymph nodes, liver, spleen, heart high fever, convulsions & meningoencephalitis • Mortality rate: up to 50% in the young, overall ~ 10% • Diagnosis after incubation: by serodiagnosis or xenodiagnosis • Duration: acute stage may last up to several months
 Latent Stage Chagas Disease • Outwardly asymptomatic • May have subtle changes in – sympathetic & parasympathetic nervous system – internal organs • Duration: decades till death from other cause or till evolution to chronic stage • Diagnosis by serological testing (15% prevalence DNA in sero-neg endemic pop. )
Latent Stage Chagas Disease • Outwardly asymptomatic • May have subtle changes in – sympathetic & parasympathetic nervous system – internal organs • Duration: decades till death from other cause or till evolution to chronic stage • Diagnosis by serological testing (15% prevalence DNA in sero-neg endemic pop. )
 Chronic Chagas Disease • Evolves in 20 to 40 % of infected persons – – Repeated re-infections Genetic polymorphism of T. cruzi Variation in host susceptibility Immuno-compromised by disease / drugs • Cardiomyopathy or Congestive Heart Failure – typically involves right bundle branch block – arrhythmias • Enlarged colon or esophagus • Ischemic stroke • Diagnosis by serological testing & clinical evaluation
Chronic Chagas Disease • Evolves in 20 to 40 % of infected persons – – Repeated re-infections Genetic polymorphism of T. cruzi Variation in host susceptibility Immuno-compromised by disease / drugs • Cardiomyopathy or Congestive Heart Failure – typically involves right bundle branch block – arrhythmias • Enlarged colon or esophagus • Ischemic stroke • Diagnosis by serological testing & clinical evaluation
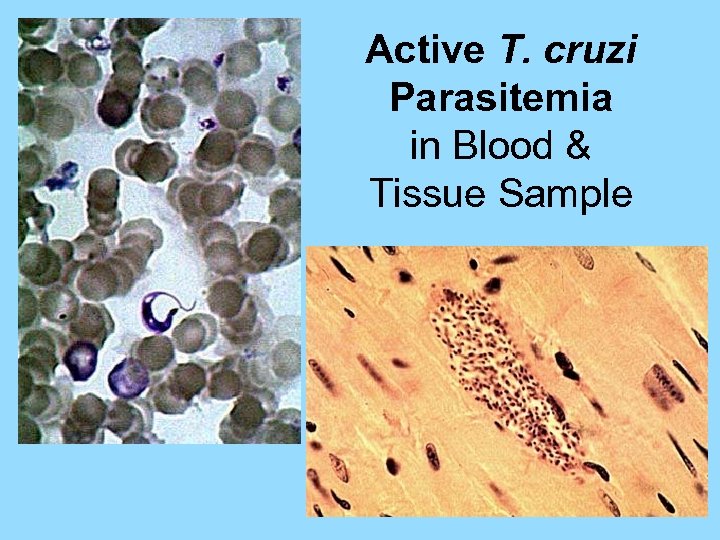 Active T. cruzi Parasitemia in Blood & Tissue Sample
Active T. cruzi Parasitemia in Blood & Tissue Sample
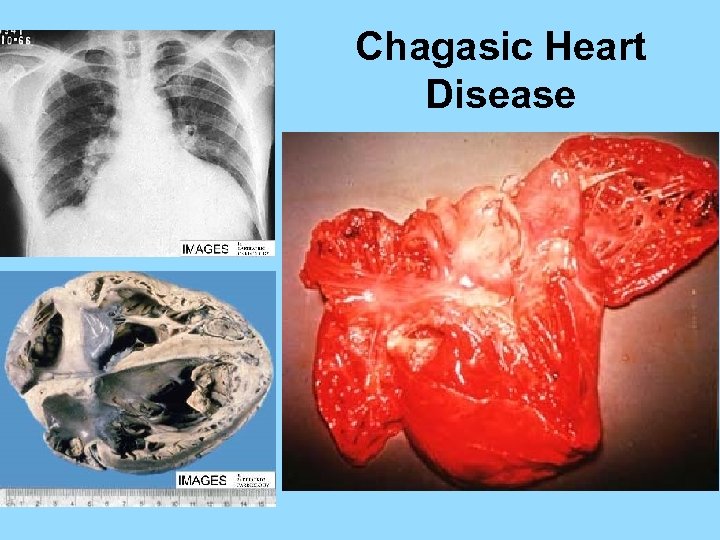 Chagasic Heart Disease
Chagasic Heart Disease
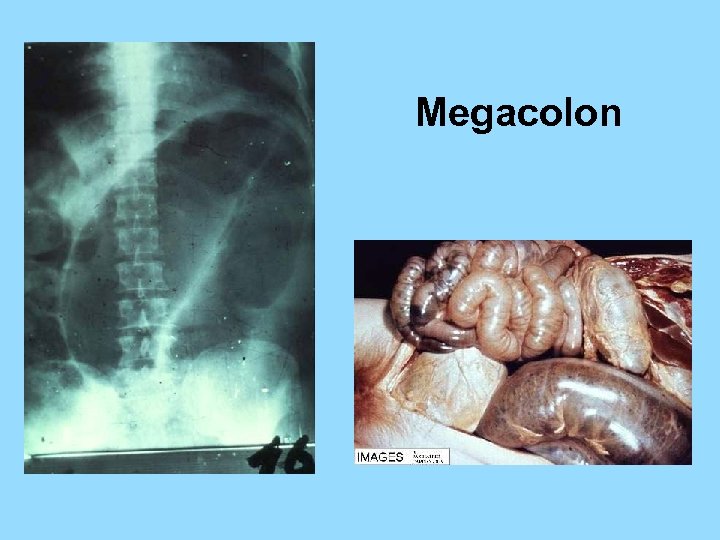 Megacolon
Megacolon
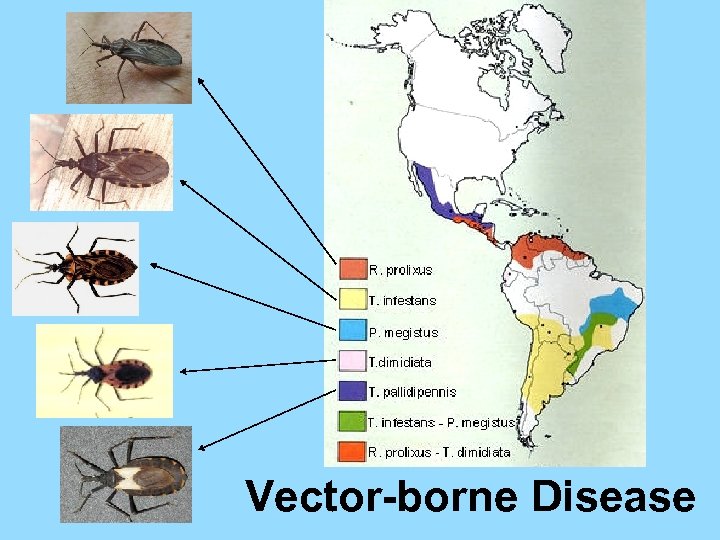 Vector-borne Disease
Vector-borne Disease
 Vinchuca … Kissing bug… Cone-nose…. by any other name… Triatomid species Eggs In-star nymph stages Adult (winged) Disease Vectors: Nymphs & Adults 1 cm 1 inch
Vinchuca … Kissing bug… Cone-nose…. by any other name… Triatomid species Eggs In-star nymph stages Adult (winged) Disease Vectors: Nymphs & Adults 1 cm 1 inch
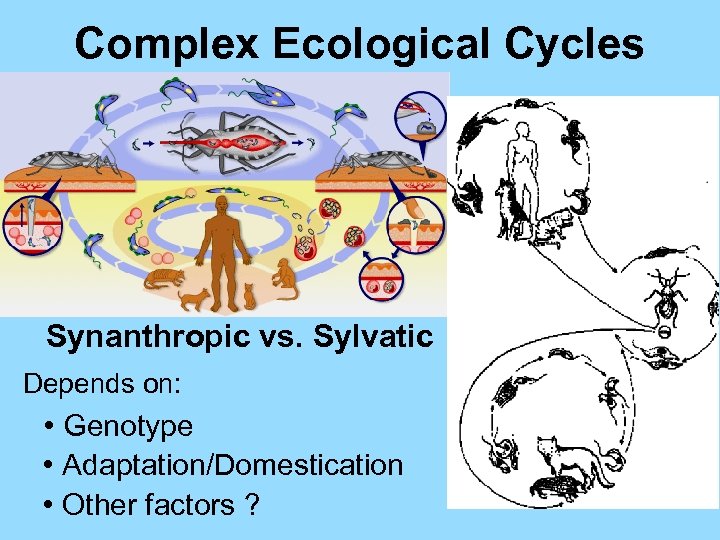 Complex Ecological Cycles Synanthropic vs. Sylvatic Depends on: • Genotype • Adaptation/Domestication • Other factors ?
Complex Ecological Cycles Synanthropic vs. Sylvatic Depends on: • Genotype • Adaptation/Domestication • Other factors ?
 Chagas Disease in Humans • Become infected for life • Potentially progresses through 3 stages Acute Latent/Indeterminate Chronic • Fatal in acute and/or chronic stages & debilitating • Higher risk for immuno-compromised persons • MYTH = a disease only of the rural poor • NO vaccine • NO cure • US FDA approved ELISA blood-screening test
Chagas Disease in Humans • Become infected for life • Potentially progresses through 3 stages Acute Latent/Indeterminate Chronic • Fatal in acute and/or chronic stages & debilitating • Higher risk for immuno-compromised persons • MYTH = a disease only of the rural poor • NO vaccine • NO cure • US FDA approved ELISA blood-screening test
 But NOT in Texas ? ? • Lower virulence • Lower vector density • Different vector habits – Less frequent domestication – Significantly longer feed-defecation response • Lack of ‘trypanosomiasis consciousness’ – Fail to diagnose nondescript acute infections
But NOT in Texas ? ? • Lower virulence • Lower vector density • Different vector habits – Less frequent domestication – Significantly longer feed-defecation response • Lack of ‘trypanosomiasis consciousness’ – Fail to diagnose nondescript acute infections
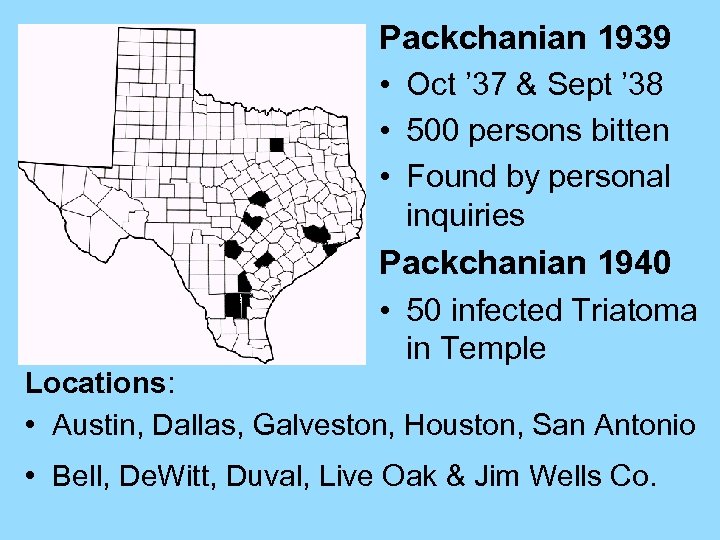 Packchanian 1939 • Oct ’ 37 & Sept ’ 38 • 500 persons bitten • Found by personal inquiries Packchanian 1940 • 50 infected Triatoma in Temple Locations: • Austin, Dallas, Galveston, Houston, San Antonio • Bell, De. Witt, Duval, Live Oak & Jim Wells Co.
Packchanian 1939 • Oct ’ 37 & Sept ’ 38 • 500 persons bitten • Found by personal inquiries Packchanian 1940 • 50 infected Triatoma in Temple Locations: • Austin, Dallas, Galveston, Houston, San Antonio • Bell, De. Witt, Duval, Live Oak & Jim Wells Co.
 Wood 1941 & 1942 • Bug ‘epidemic’ in Quemado Valley • Residents bitten • House infested in Sanderson (Terrell) “these suckers have sure dealt them misery” Locations: • Maverick, Terrell & Bandera Co.
Wood 1941 & 1942 • Bug ‘epidemic’ in Quemado Valley • Residents bitten • House infested in Sanderson (Terrell) “these suckers have sure dealt them misery” Locations: • Maverick, Terrell & Bandera Co.
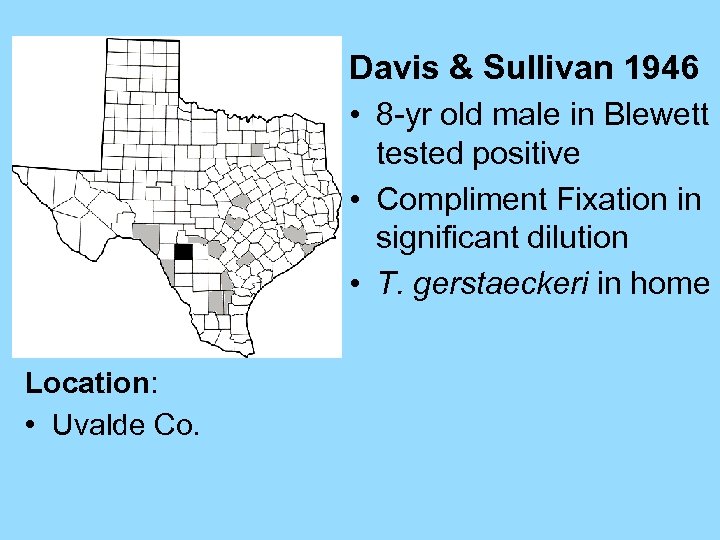 Davis & Sullivan 1946 • 8 -yr old male in Blewett tested positive • Compliment Fixation in significant dilution • T. gerstaeckeri in home Location: • Uvalde Co.
Davis & Sullivan 1946 • 8 -yr old male in Blewett tested positive • Compliment Fixation in significant dilution • T. gerstaeckeri in home Location: • Uvalde Co.
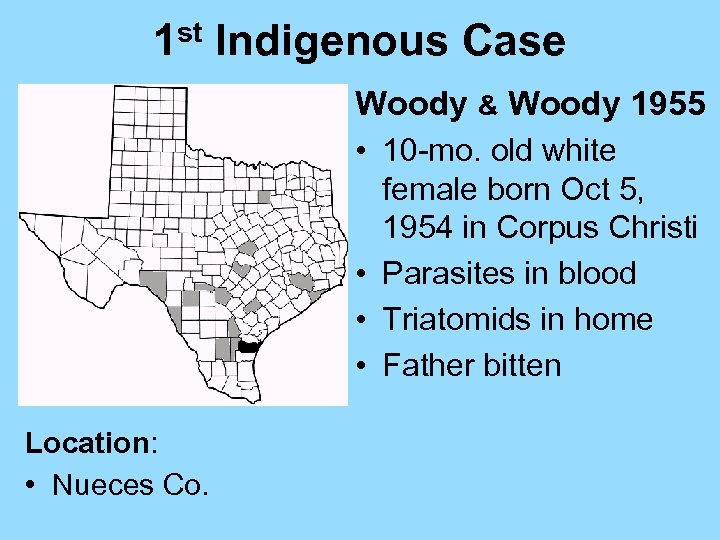 1 st Indigenous Case Woody & Woody 1955 • 10 -mo. old white female born Oct 5, 1954 in Corpus Christi • Parasites in blood • Triatomids in home • Father bitten Location: • Nueces Co.
1 st Indigenous Case Woody & Woody 1955 • 10 -mo. old white female born Oct 5, 1954 in Corpus Christi • Parasites in blood • Triatomids in home • Father bitten Location: • Nueces Co.
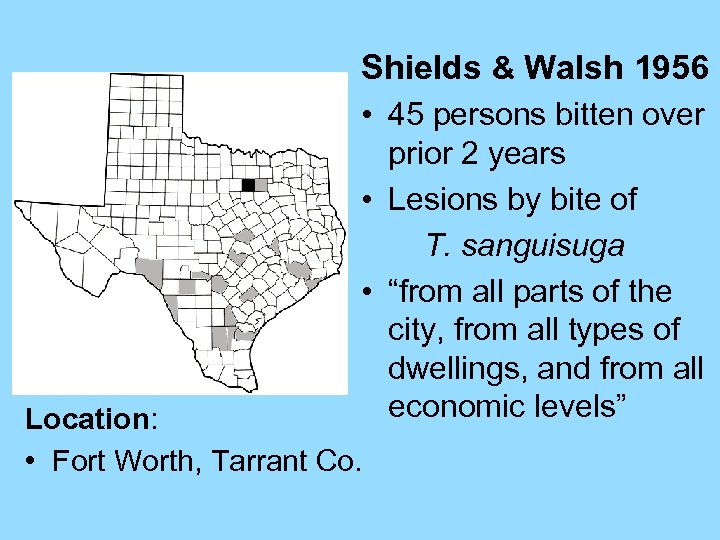 Shields & Walsh 1956 • 45 persons bitten over prior 2 years • Lesions by bite of T. sanguisuga • “from all parts of the city, from all types of dwellings, and from all economic levels” Location: • Fort Worth, Tarrant Co.
Shields & Walsh 1956 • 45 persons bitten over prior 2 years • Lesions by bite of T. sanguisuga • “from all parts of the city, from all types of dwellings, and from all economic levels” Location: • Fort Worth, Tarrant Co.
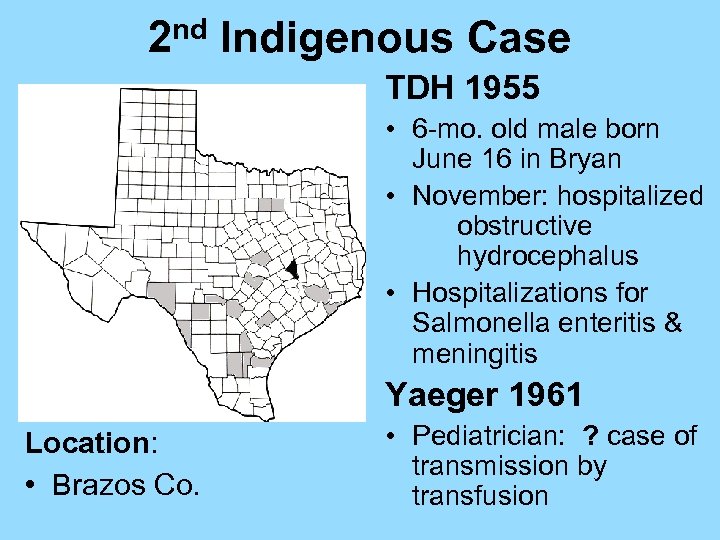 nd 2 Indigenous Case TDH 1955 • 6 -mo. old male born June 16 in Bryan • November: hospitalized obstructive hydrocephalus • Hospitalizations for Salmonella enteritis & meningitis Yaeger 1961 Location: • Brazos Co. • Pediatrician: ? case of transmission by transfusion
nd 2 Indigenous Case TDH 1955 • 6 -mo. old male born June 16 in Bryan • November: hospitalized obstructive hydrocephalus • Hospitalizations for Salmonella enteritis & meningitis Yaeger 1961 Location: • Brazos Co. • Pediatrician: ? case of transmission by transfusion
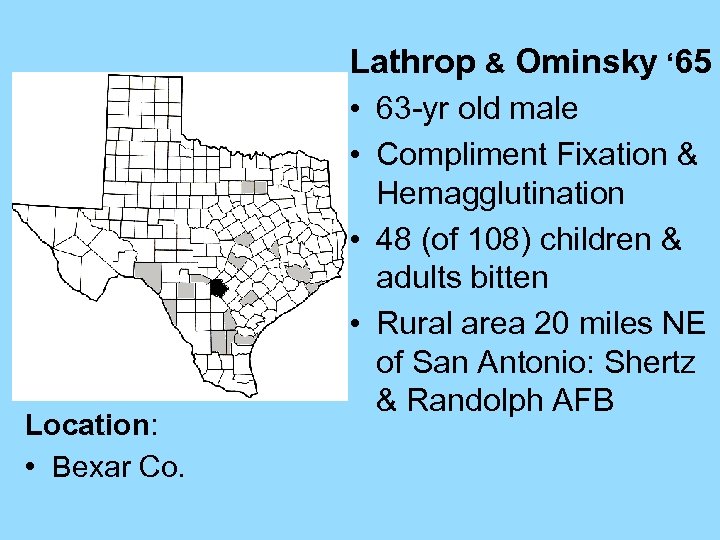 Lathrop & Ominsky ‘ 65 • 63 -yr old male • Compliment Fixation & Location: • Bexar Co. Hemagglutination • 48 (of 108) children & adults bitten • Rural area 20 miles NE of San Antonio: Shertz & Randolph AFB
Lathrop & Ominsky ‘ 65 • 63 -yr old male • Compliment Fixation & Location: • Bexar Co. Hemagglutination • 48 (of 108) children & adults bitten • Rural area 20 miles NE of San Antonio: Shertz & Randolph AFB
 Woody et al. 1965 • 117 bitten in Coastal • • Location: • Nueces Co. • • Bend & Corpus Christi 7 weakly positive to positive titers (ages 5. 5 – 72) but no clinical evidence 2 infection chagomas (no positive test) T. cruzi not isolated 1 st indigenous case still tested positive
Woody et al. 1965 • 117 bitten in Coastal • • Location: • Nueces Co. • • Bend & Corpus Christi 7 weakly positive to positive titers (ages 5. 5 – 72) but no clinical evidence 2 infection chagomas (no positive test) T. cruzi not isolated 1 st indigenous case still tested positive
 Faust 1978 Location: • Potter Co. • 38 -yr old male fatality • Oct ’ 76 vacation in Caracas, Venezuela & Caribbean • 2 Amarillo Hospitals: 1 st Admit = May 11 -13, ’ 77 2 nd Admit = May 26 -July 1 x-ray: cardiomegaly, ECG poor L ventricle function Diagnosis: cardiomyopathy, origin undetermined • Died at home, July 5 • CF & HA tests confirmed on July 25, 1977
Faust 1978 Location: • Potter Co. • 38 -yr old male fatality • Oct ’ 76 vacation in Caracas, Venezuela & Caribbean • 2 Amarillo Hospitals: 1 st Admit = May 11 -13, ’ 77 2 nd Admit = May 26 -July 1 x-ray: cardiomegaly, ECG poor L ventricle function Diagnosis: cardiomyopathy, origin undetermined • Died at home, July 5 • CF & HA tests confirmed on July 25, 1977
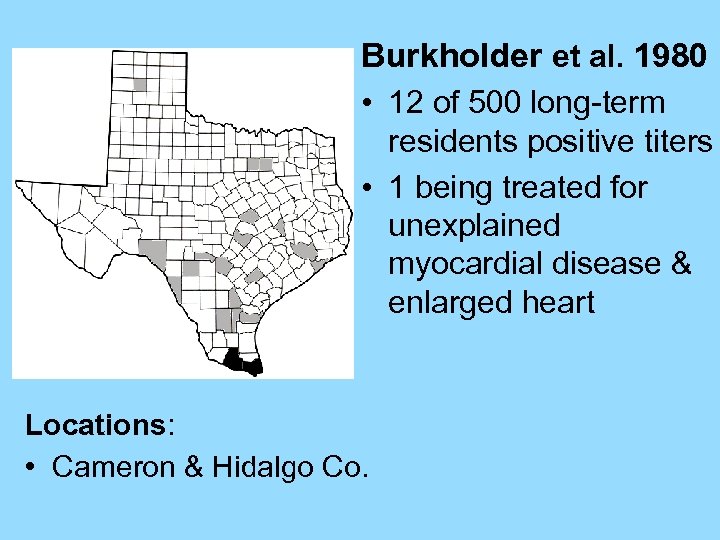 Burkholder et al. 1980 • 12 of 500 long-term residents positive titers • 1 being treated for unexplained myocardial disease & enlarged heart Locations: • Cameron & Hidalgo Co.
Burkholder et al. 1980 • 12 of 500 long-term residents positive titers • 1 being treated for unexplained myocardial disease & enlarged heart Locations: • Cameron & Hidalgo Co.
 rd 3 Indigenous Case Betz 1984 • 7 -mo old Hispanic male fatality July 30, 1983 • April 1984 pathology diagnosis: acute Chagas myocarditis • Likely infected Mathis (SP) or Alice (JW) • Family all seronegative in 1984 Locations: • San Patricio or Jim Wells Co.
rd 3 Indigenous Case Betz 1984 • 7 -mo old Hispanic male fatality July 30, 1983 • April 1984 pathology diagnosis: acute Chagas myocarditis • Likely infected Mathis (SP) or Alice (JW) • Family all seronegative in 1984 Locations: • San Patricio or Jim Wells Co.
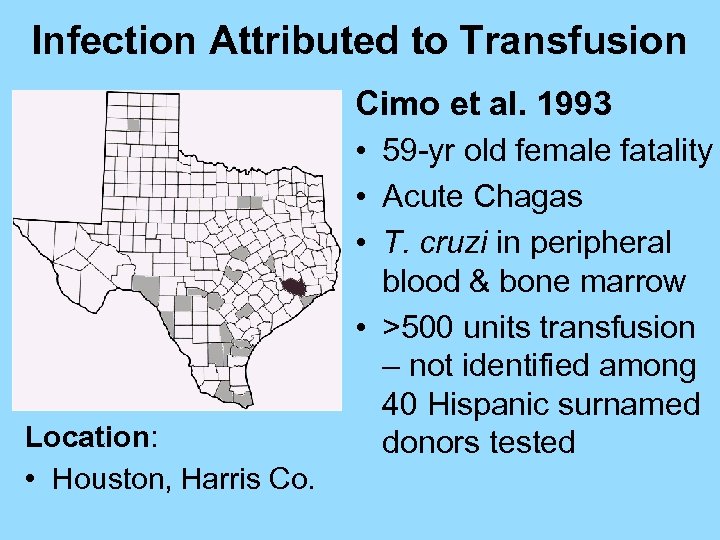 Infection Attributed to Transfusion Cimo et al. 1993 • 59 -yr old female fatality • Acute Chagas • T. cruzi in peripheral Location: • Houston, Harris Co. blood & bone marrow • >500 units transfusion – not identified among 40 Hispanic surnamed donors tested
Infection Attributed to Transfusion Cimo et al. 1993 • 59 -yr old female fatality • Acute Chagas • T. cruzi in peripheral Location: • Houston, Harris Co. blood & bone marrow • >500 units transfusion – not identified among 40 Hispanic surnamed donors tested
 Cross-Section Study in Houston Di. Pentima et al. 1999 Location: • Harris Co. • Pregnant women (’ 93’ 96) ages 13 - 44 • 2107 Hispanic, 1658 non • 22 positive (18 >age 20) 13 (0. 6%) Hispanic & 9 (0. 5%) White & Black • Risk factors & points of exposure unknown • Congenital not reported
Cross-Section Study in Houston Di. Pentima et al. 1999 Location: • Harris Co. • Pregnant women (’ 93’ 96) ages 13 - 44 • 2107 Hispanic, 1658 non • 22 positive (18 >age 20) 13 (0. 6%) Hispanic & 9 (0. 5%) White & Black • Risk factors & points of exposure unknown • Congenital not reported
 Serologic Tests & Look-back Leiby et al. 1999 Location: • Mc. Clennan Co. • 3 EIA repeatably reactive and RIPA seropositive – all from Waco • 1 from Durango MX • 2 TX natives (17 & 40) • All 3 families: history of heart ailments & complications (enlarged heart & arrhythmias)
Serologic Tests & Look-back Leiby et al. 1999 Location: • Mc. Clennan Co. • 3 EIA repeatably reactive and RIPA seropositive – all from Waco • 1 from Durango MX • 2 TX natives (17 & 40) • All 3 families: history of heart ailments & complications (enlarged heart & arrhythmias)
 Cardiac Surgery Patients Leiby et al. 2000 • 23 repeatably reactive Location: • Harris Co. 6 confirmed at 3 hospitals, including: Methodist Hospital & St. Luke’s Episcopal in Houston • Original source of infection unknown; all had received blood transfusions
Cardiac Surgery Patients Leiby et al. 2000 • 23 repeatably reactive Location: • Harris Co. 6 confirmed at 3 hospitals, including: Methodist Hospital & St. Luke’s Episcopal in Houston • Original source of infection unknown; all had received blood transfusions
 El. Munzer et al. 2004 • 70 -yr old Hispanic male • Immigrated to TX 20 yrs • Oct 2002 presented to Location: • Dallas Co. ER Parkland Memorial • History of acute MI, with right bundle branch block on ECG, ventricular arrythmia • Diagnosis confirmed by Complement Fixation
El. Munzer et al. 2004 • 70 -yr old Hispanic male • Immigrated to TX 20 yrs • Oct 2002 presented to Location: • Dallas Co. ER Parkland Memorial • History of acute MI, with right bundle branch block on ECG, ventricular arrythmia • Diagnosis confirmed by Complement Fixation
 T. Cruzi Reactivation by AIDS Rivera et al. 2004 Location: • Dallas Co. • 29 -yr old male Honduran fatality • 5 -yr Immigrant was diagnosed 5 -mo. with HIV • Developed acute congestive heart failure secondary to cardiac Chagas Disease • Necropsy showed T. cruzi amastigotes in myocardium – no atherosclerosis
T. Cruzi Reactivation by AIDS Rivera et al. 2004 Location: • Dallas Co. • 29 -yr old male Honduran fatality • 5 -yr Immigrant was diagnosed 5 -mo. with HIV • Developed acute congestive heart failure secondary to cardiac Chagas Disease • Necropsy showed T. cruzi amastigotes in myocardium – no atherosclerosis
 Hosts & Reservoirs in TX v Woodrats (Neotoma) v Opossums (Didelphis) v Armadillos (Dasypus) v Coyotes (Canis) v Others (+ humans)
Hosts & Reservoirs in TX v Woodrats (Neotoma) v Opossums (Didelphis) v Armadillos (Dasypus) v Coyotes (Canis) v Others (+ humans)
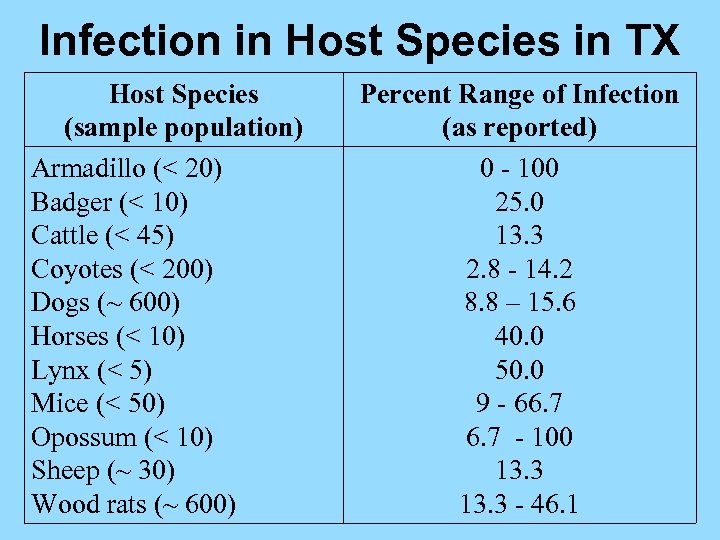 Infection in Host Species in TX Host Species (sample population) Armadillo (< 20) Badger (< 10) Cattle (< 45) Coyotes (< 200) Dogs (~ 600) Horses (< 10) Lynx (< 5) Mice (< 50) Opossum (< 10) Sheep (~ 30) Wood rats (~ 600) Percent Range of Infection (as reported) 0 - 100 25. 0 13. 3 2. 8 - 14. 2 8. 8 – 15. 6 40. 0 50. 0 9 - 66. 7 - 100 13. 3 - 46. 1
Infection in Host Species in TX Host Species (sample population) Armadillo (< 20) Badger (< 10) Cattle (< 45) Coyotes (< 200) Dogs (~ 600) Horses (< 10) Lynx (< 5) Mice (< 50) Opossum (< 10) Sheep (~ 30) Wood rats (~ 600) Percent Range of Infection (as reported) 0 - 100 25. 0 13. 3 2. 8 - 14. 2 8. 8 – 15. 6 40. 0 50. 0 9 - 66. 7 - 100 13. 3 - 46. 1
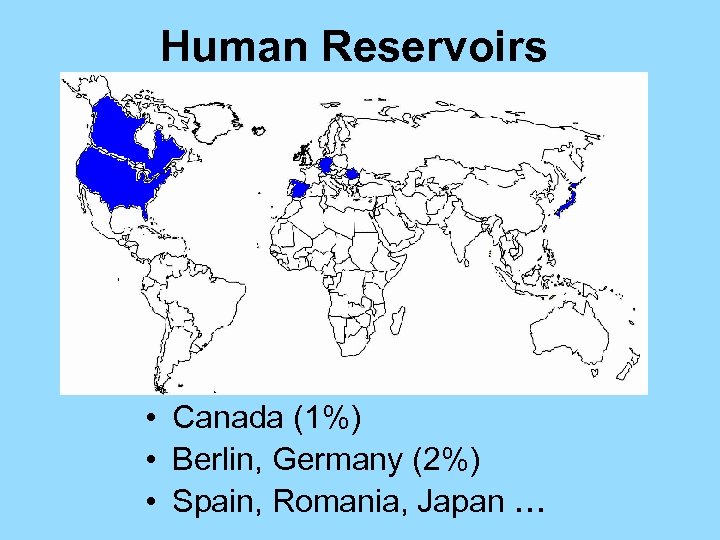 Human Reservoirs • Canada (1%) • Berlin, Germany (2%) • Spain, Romania, Japan …
Human Reservoirs • Canada (1%) • Berlin, Germany (2%) • Spain, Romania, Japan …
 Chagas Disease in the U. S. • Blood Transfusion Immuno-compromised individuals ? Immuno-competent • Organ Transplants 2002 = 3 from 1 donor 2006 = 2 cases, 6 monitor from 2 donors • 6 Autochthonous cases: TX, CA, TN, LA • Unrecognized ? ?
Chagas Disease in the U. S. • Blood Transfusion Immuno-compromised individuals ? Immuno-competent • Organ Transplants 2002 = 3 from 1 donor 2006 = 2 cases, 6 monitor from 2 donors • 6 Autochthonous cases: TX, CA, TN, LA • Unrecognized ? ?
 Triatomid Species in Texas T. gerstaeckeri T. rubida T. recurva T. sanguisuga T. protracta
Triatomid Species in Texas T. gerstaeckeri T. rubida T. recurva T. sanguisuga T. protracta
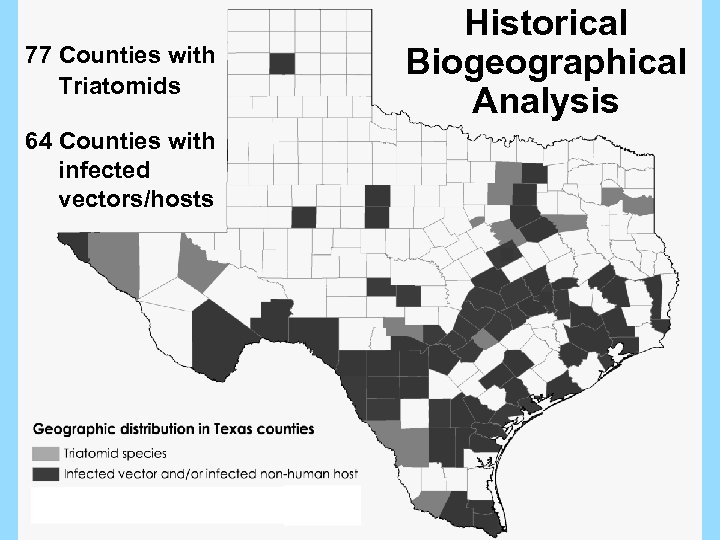 77 Counties with Triatomids 64 Counties with infected vectors/hosts Historical Biogeographical Analysis
77 Counties with Triatomids 64 Counties with infected vectors/hosts Historical Biogeographical Analysis
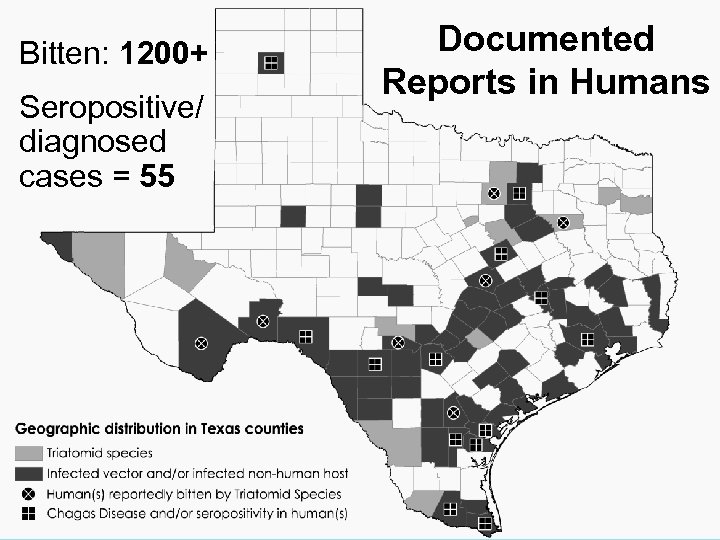 Bitten: 1200+ Seropositive/ diagnosed cases = 55 Documented Reports in Humans
Bitten: 1200+ Seropositive/ diagnosed cases = 55 Documented Reports in Humans
 Demographics & Estimates • Latin America: DALY 2. 7 m ~ U. S. $6. 5 b United States: 16 m (incl 6. 2 undoc) Latin Am immigrants ~ 7% infection rate 1 m + infected 150, 000+ chronic TEXAS: ~ 300, 000 - 600, 000 infected ~ 50, 000 - 100, 000 chronic
Demographics & Estimates • Latin America: DALY 2. 7 m ~ U. S. $6. 5 b United States: 16 m (incl 6. 2 undoc) Latin Am immigrants ~ 7% infection rate 1 m + infected 150, 000+ chronic TEXAS: ~ 300, 000 - 600, 000 infected ~ 50, 000 - 100, 000 chronic
 Under-Estimation ? ? Ø Actual infection rate Ø Number of undocumented immigrants Ø Number of congenital cases Ø Multi-generational transmission Ø Ø Ø Infected immuno-competent Sero-negative but still infected More aggressive genotype
Under-Estimation ? ? Ø Actual infection rate Ø Number of undocumented immigrants Ø Number of congenital cases Ø Multi-generational transmission Ø Ø Ø Infected immuno-competent Sero-negative but still infected More aggressive genotype
 Significance = Emerging Disease More is needed in Texas: • Education & Prevention • Research & Development • Awareness / Recognition And in other states • Endemic regions • Introduced
Significance = Emerging Disease More is needed in Texas: • Education & Prevention • Research & Development • Awareness / Recognition And in other states • Endemic regions • Introduced
 Research & Development • Improved understanding of environmental ecology of vectors & hosts (adaptability) • Field studies to determine infection rates and ranges of endemic vector and host species • Field studies to monitor introduced/migrating vectors and hosts & interactions with native species • FDA-approved tests for diagnosis & screening of blood supply & donor organs • Preventative vaccine • Pharmaceuticals to control disease progression or to produce a “cure”
Research & Development • Improved understanding of environmental ecology of vectors & hosts (adaptability) • Field studies to determine infection rates and ranges of endemic vector and host species • Field studies to monitor introduced/migrating vectors and hosts & interactions with native species • FDA-approved tests for diagnosis & screening of blood supply & donor organs • Preventative vaccine • Pharmaceuticals to control disease progression or to produce a “cure”
 What is needed… Recognition as – endemic zoonotic risk for humans – introduced disease associated with changing human demographics & genotypes Education Prevention & Awareness – public health, physicians & cardiologists – veterinary & animal care workers Recommendations – Inclusion as Communicable / Reportable to TDSHS – Serologic screening test
What is needed… Recognition as – endemic zoonotic risk for humans – introduced disease associated with changing human demographics & genotypes Education Prevention & Awareness – public health, physicians & cardiologists – veterinary & animal care workers Recommendations – Inclusion as Communicable / Reportable to TDSHS – Serologic screening test


