5cba0feceabe74b8fa01419878393f43.ppt
- Количество слайдов: 81
 CARDIO-RENAL Advisory Committee Meeting EXTRANEAL™ (7. 5% Icodextrin) Peritoneal Dialysis Solution NDA 21 -321 Orphan Drug Designation 97 -1056 August 9, 2001 Baxter Healthcare Corporation
CARDIO-RENAL Advisory Committee Meeting EXTRANEAL™ (7. 5% Icodextrin) Peritoneal Dialysis Solution NDA 21 -321 Orphan Drug Designation 97 -1056 August 9, 2001 Baxter Healthcare Corporation
 Baxter Participants n Marsha Wolfson, M. D. u n Salim Mujais, M. D. u n Director, Solutions R&D Richard Newman, Ph. D. u n Vice President, Solution Development James Moberly, Ph. D. u n Clinical Statistician Leo Martis, Ph. D. u n Vice President, Global Medical Affairs Frank Ogrinc, Ph. D. u n Vice President, Global Clinical Affairs Vice President, Global Product Development Mary Kay Rybicki u Associate Director, Regulatory Affairs 2
Baxter Participants n Marsha Wolfson, M. D. u n Salim Mujais, M. D. u n Director, Solutions R&D Richard Newman, Ph. D. u n Vice President, Solution Development James Moberly, Ph. D. u n Clinical Statistician Leo Martis, Ph. D. u n Vice President, Global Medical Affairs Frank Ogrinc, Ph. D. u n Vice President, Global Clinical Affairs Vice President, Global Product Development Mary Kay Rybicki u Associate Director, Regulatory Affairs 2
 Consultants n John Burkart, M. D. u n Allan Collins, M. D. u n Karl Nolph, M. D. u n Professor of Medicine, University of Minnesota, Director Nephrology Analytical Services, Minneapolis Medical n Research Foundation Dept. of Nephrology & Hypertension, University Hospital of Antwerp n u Consultant Nephrologist, University of Manchester u Professor of Biostatistics, Mayo Medical School, Department of Health Sciences Research Robert Stern, M. D. u n Chairman & Professor of Medicine, New York Medical College, Department of Medicine Peter O’Brien, Ph. D. Ram Gokal, M. D. u Curator Professor Emeritus, University of Missouri, Columbia William Frishman, M. D. Marc De. Broe, M. D. , Ph. D. u n Professor of Internal Medicine/Nephrology, Bowman Gray School of Medicine n Carl J. Herzog Professor of Dermatology, Beth Israel Deaconess Medical Center Jeff Trotter, M. M. u President, Ovation Research Group 3
Consultants n John Burkart, M. D. u n Allan Collins, M. D. u n Karl Nolph, M. D. u n Professor of Medicine, University of Minnesota, Director Nephrology Analytical Services, Minneapolis Medical n Research Foundation Dept. of Nephrology & Hypertension, University Hospital of Antwerp n u Consultant Nephrologist, University of Manchester u Professor of Biostatistics, Mayo Medical School, Department of Health Sciences Research Robert Stern, M. D. u n Chairman & Professor of Medicine, New York Medical College, Department of Medicine Peter O’Brien, Ph. D. Ram Gokal, M. D. u Curator Professor Emeritus, University of Missouri, Columbia William Frishman, M. D. Marc De. Broe, M. D. , Ph. D. u n Professor of Internal Medicine/Nephrology, Bowman Gray School of Medicine n Carl J. Herzog Professor of Dermatology, Beth Israel Deaconess Medical Center Jeff Trotter, M. M. u President, Ovation Research Group 3
 Extraneal Development Milestones n First market approval UK 1992 by ML Labs n Licensed by Baxter 1996 n Marketing approval in 31 countries u ~8200 patients currently treated worldwide u 30% of PD patients in Europe n US clinical trials began 1997 n Orphan Drug designation granted 1997 n NDA submitted December 2000 4
Extraneal Development Milestones n First market approval UK 1992 by ML Labs n Licensed by Baxter 1996 n Marketing approval in 31 countries u ~8200 patients currently treated worldwide u 30% of PD patients in Europe n US clinical trials began 1997 n Orphan Drug designation granted 1997 n NDA submitted December 2000 4
 Proposed Indication Extraneal is indicated for a single daily exchange for the long (8 -16 hour) dwell during continuous ambulatory peritoneal dialysis (CAPD) or automated peritoneal dialysis (APD) for the management of chronic renal failure. 5
Proposed Indication Extraneal is indicated for a single daily exchange for the long (8 -16 hour) dwell during continuous ambulatory peritoneal dialysis (CAPD) or automated peritoneal dialysis (APD) for the management of chronic renal failure. 5
 Topics Identified in FDA Briefing Document & Questions n Dialysis Efficacy n Quality of Life n Size of Database n Safety Profile u Mortality u Rash u Peritonitis u Membrane Transport Characteristics 6
Topics Identified in FDA Briefing Document & Questions n Dialysis Efficacy n Quality of Life n Size of Database n Safety Profile u Mortality u Rash u Peritonitis u Membrane Transport Characteristics 6
 AGENDA Cardio-Renal Advisory Committee NDA 21 -321 Extraneal (7. 5% icodextrin) Introduction and Rationale for Extraneal Salim Mujais, M. D. , Vice President Global Medical Affairs, Baxter Clinical Trial Experience with Extraneal Marsha Wolfson, M. D. , Vice President Global Clinical Affairs, Baxter Frank Ogrinc, Ph. D. , Clinical Statistician, Baxter Conclusions Salim Mujais, M. D. , Vice President Global Medical Affairs, Baxter 7
AGENDA Cardio-Renal Advisory Committee NDA 21 -321 Extraneal (7. 5% icodextrin) Introduction and Rationale for Extraneal Salim Mujais, M. D. , Vice President Global Medical Affairs, Baxter Clinical Trial Experience with Extraneal Marsha Wolfson, M. D. , Vice President Global Clinical Affairs, Baxter Frank Ogrinc, Ph. D. , Clinical Statistician, Baxter Conclusions Salim Mujais, M. D. , Vice President Global Medical Affairs, Baxter 7
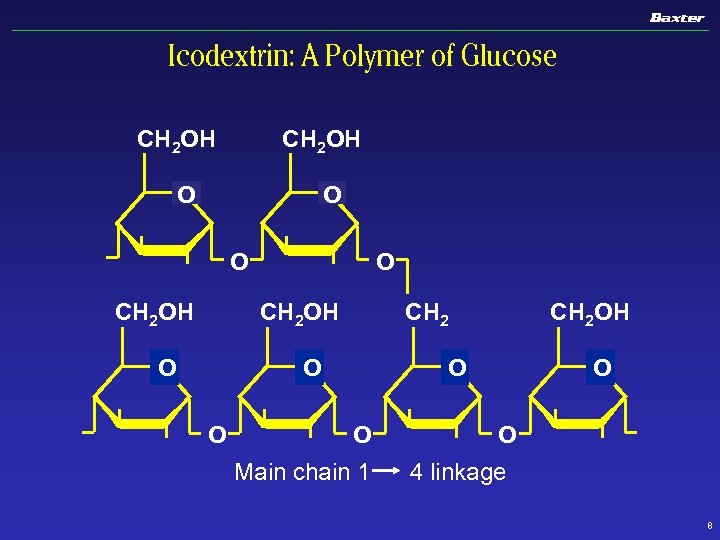 Icodextrin: A Polymer of Glucose CH 2 OH O O O CH 2 OH O CH 2 O O CH 2 OH O O Main chain 1 4 linkage 8
Icodextrin: A Polymer of Glucose CH 2 OH O O O CH 2 OH O CH 2 O O CH 2 OH O O Main chain 1 4 linkage 8
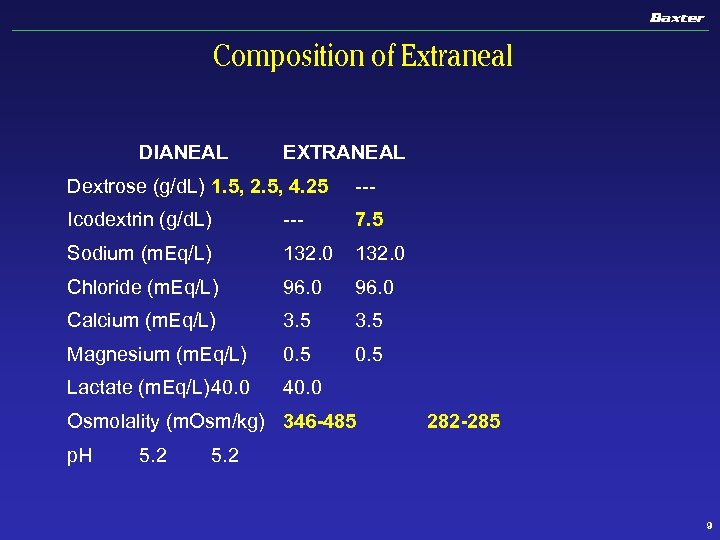 Composition of Extraneal DIANEAL EXTRANEAL Dextrose (g/d. L) 1. 5, 2. 5, 4. 25 --- Icodextrin (g/d. L) --- 7. 5 Sodium (m. Eq/L) 132. 0 Chloride (m. Eq/L) 96. 0 Calcium (m. Eq/L) 3. 5 Magnesium (m. Eq/L) 0. 5 Lactate (m. Eq/L)40. 0 Osmolality (m. Osm/kg) 346 -485 p. H 5. 2 282 -285 5. 2 9
Composition of Extraneal DIANEAL EXTRANEAL Dextrose (g/d. L) 1. 5, 2. 5, 4. 25 --- Icodextrin (g/d. L) --- 7. 5 Sodium (m. Eq/L) 132. 0 Chloride (m. Eq/L) 96. 0 Calcium (m. Eq/L) 3. 5 Magnesium (m. Eq/L) 0. 5 Lactate (m. Eq/L)40. 0 Osmolality (m. Osm/kg) 346 -485 p. H 5. 2 282 -285 5. 2 9
 Clinical Rationale for Extraneal n Unmet clinical need u Limitations of fluid management in PD n Limitations of current osmotic agents u Necessity of the long dwell u Kinetics of peritoneal ultrafiltration n Extraneal as a new osmotic agent u Kinetics matching clinical requirement 10
Clinical Rationale for Extraneal n Unmet clinical need u Limitations of fluid management in PD n Limitations of current osmotic agents u Necessity of the long dwell u Kinetics of peritoneal ultrafiltration n Extraneal as a new osmotic agent u Kinetics matching clinical requirement 10
 Unmet Clinical Need Limitations of Current Fluid Management in PD n Symptomatic fluid retention occurs in 25% of all PD patients 1. In these patients: u 98. 6% u Pleural effusions 76. 1% u n Lower extremity edema Pulmonary congestion 80. 3% Similar clinical observations have been reported from Japan 2, the Netherlands 3 and Sweden 4 1 Tzamaloukas et al. JASN 1995; 2 Kawaguchi et al. Kidney Int 1997; 3 Ho-dac-Pannekeet et al. Perit Dial Int 1997; 4 Heimbürger et al. Perit Dial Int 1999 11
Unmet Clinical Need Limitations of Current Fluid Management in PD n Symptomatic fluid retention occurs in 25% of all PD patients 1. In these patients: u 98. 6% u Pleural effusions 76. 1% u n Lower extremity edema Pulmonary congestion 80. 3% Similar clinical observations have been reported from Japan 2, the Netherlands 3 and Sweden 4 1 Tzamaloukas et al. JASN 1995; 2 Kawaguchi et al. Kidney Int 1997; 3 Ho-dac-Pannekeet et al. Perit Dial Int 1997; 4 Heimbürger et al. Perit Dial Int 1999 11
 Limitations of Fluid Management in PD Hampered by Inflexibility n Complexity of dietary counseling u u n Hampered by compliance issues May complicate management Constrained renal excretion 1 u u n Gradual decline to anuria Diuretic resistance Peritoneal Ultrafiltration u Challenge of the long dwell 1 Medcalf et al, Kidney International 59: 1128, 2001 12
Limitations of Fluid Management in PD Hampered by Inflexibility n Complexity of dietary counseling u u n Hampered by compliance issues May complicate management Constrained renal excretion 1 u u n Gradual decline to anuria Diuretic resistance Peritoneal Ultrafiltration u Challenge of the long dwell 1 Medcalf et al, Kidney International 59: 1128, 2001 12
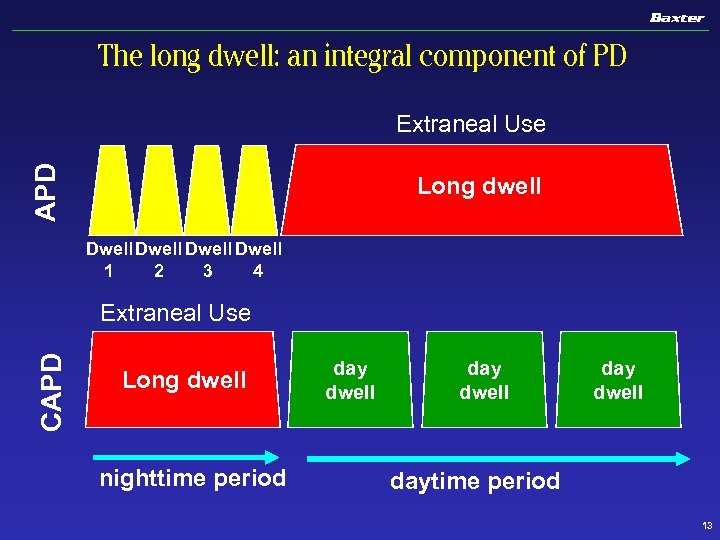 The long dwell: an integral component of PD APD Extraneal Use Long dwell Dwell 1 2 3 4 CAPD Extraneal Use Long dwell nighttime period day dwell daytime period 13
The long dwell: an integral component of PD APD Extraneal Use Long dwell Dwell 1 2 3 4 CAPD Extraneal Use Long dwell nighttime period day dwell daytime period 13
 Rationale for the long dwell in PD: Intersection of two imperatives n Toxin removal imperative: u u Middle and large molecular weight toxins are time dependent u n Small solutes removal fluid flow dependent Continuously wet abdomen required for therapy success Realistic therapy imperative u Logistic burden and compliance 14
Rationale for the long dwell in PD: Intersection of two imperatives n Toxin removal imperative: u u Middle and large molecular weight toxins are time dependent u n Small solutes removal fluid flow dependent Continuously wet abdomen required for therapy success Realistic therapy imperative u Logistic burden and compliance 14
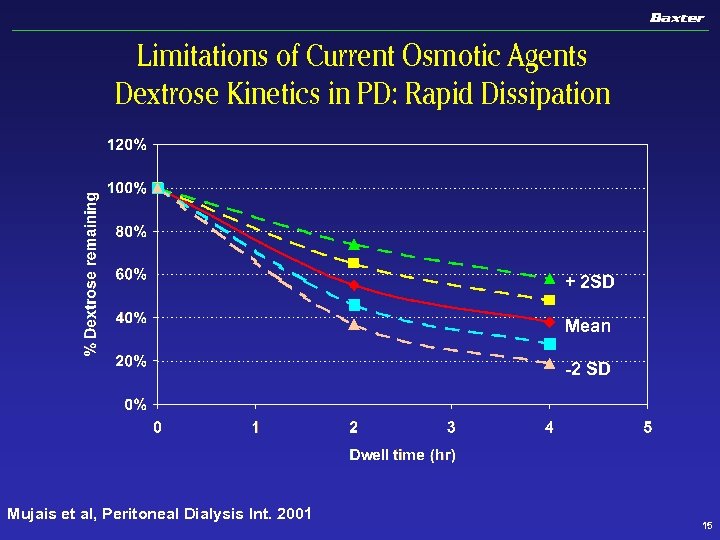 Limitations of Current Osmotic Agents Dextrose Kinetics in PD: Rapid Dissipation + 2 SD Mean -2 SD Mujais et al, Peritoneal Dialysis Int. 2001 15
Limitations of Current Osmotic Agents Dextrose Kinetics in PD: Rapid Dissipation + 2 SD Mean -2 SD Mujais et al, Peritoneal Dialysis Int. 2001 15
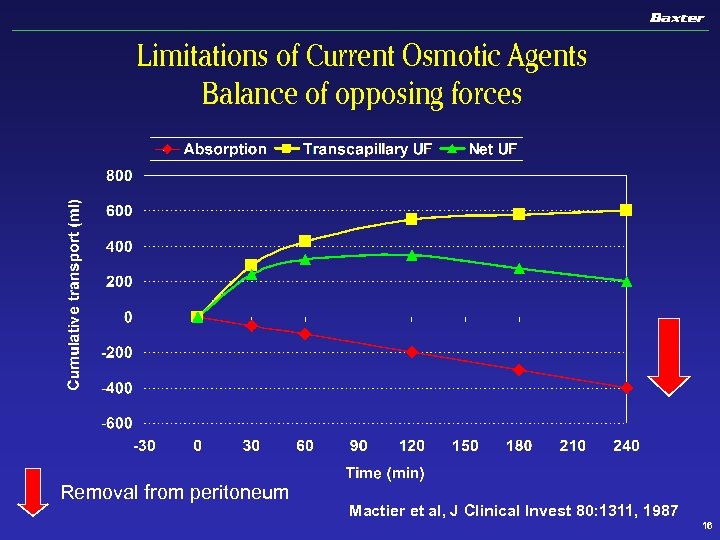 Limitations of Current Osmotic Agents Balance of opposing forces Removal from peritoneum Mactier et al, J Clinical Invest 80: 1311, 1987 16
Limitations of Current Osmotic Agents Balance of opposing forces Removal from peritoneum Mactier et al, J Clinical Invest 80: 1311, 1987 16
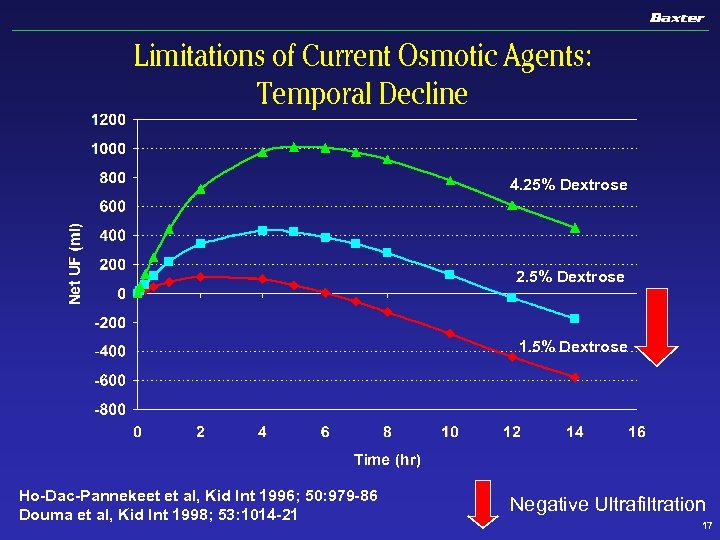 Limitations of Current Osmotic Agents: Temporal Decline 4. 25% Dextrose 2. 5% Dextrose 1. 5% Dextrose Ho-Dac-Pannekeet et al, Kid Int 1996; 50: 979 -86 Douma et al, Kid Int 1998; 53: 1014 -21 Negative Ultrafiltration 17
Limitations of Current Osmotic Agents: Temporal Decline 4. 25% Dextrose 2. 5% Dextrose 1. 5% Dextrose Ho-Dac-Pannekeet et al, Kid Int 1996; 50: 979 -86 Douma et al, Kid Int 1998; 53: 1014 -21 Negative Ultrafiltration 17
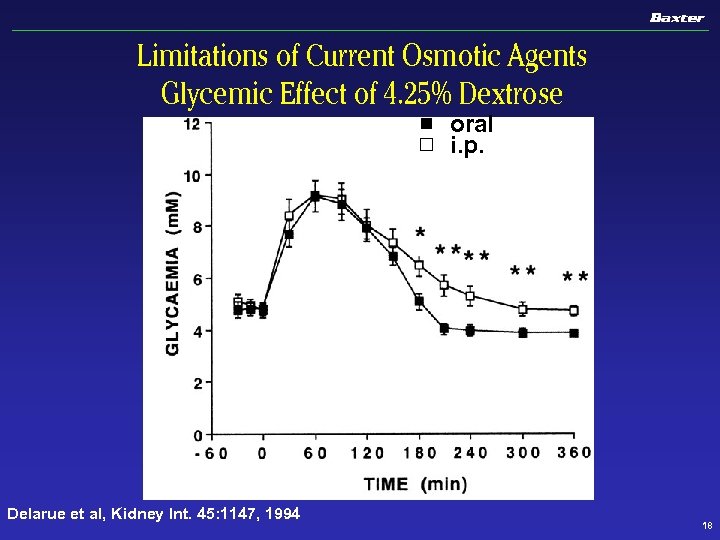 Limitations of Current Osmotic Agents Glycemic Effect of 4. 25% Dextrose oral i. p. Delarue et al, Kidney Int. 45: 1147, 1994 18
Limitations of Current Osmotic Agents Glycemic Effect of 4. 25% Dextrose oral i. p. Delarue et al, Kidney Int. 45: 1147, 1994 18
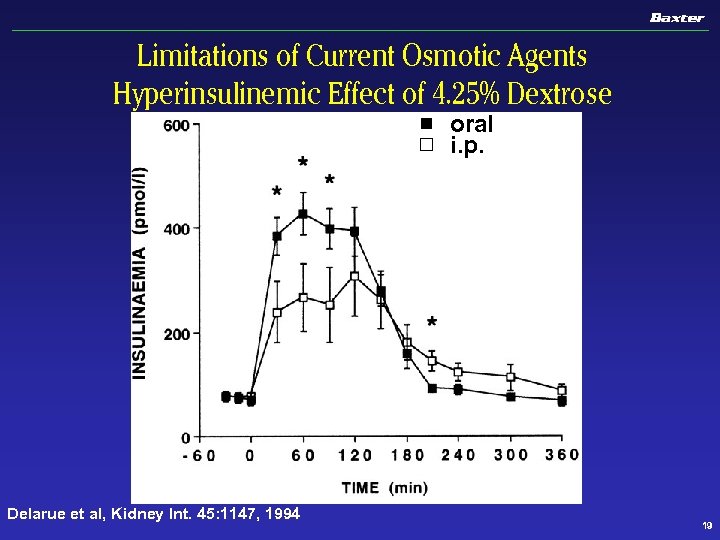 Limitations of Current Osmotic Agents Hyperinsulinemic Effect of 4. 25% Dextrose oral i. p. Delarue et al, Kidney Int. 45: 1147, 1994 19
Limitations of Current Osmotic Agents Hyperinsulinemic Effect of 4. 25% Dextrose oral i. p. Delarue et al, Kidney Int. 45: 1147, 1994 19
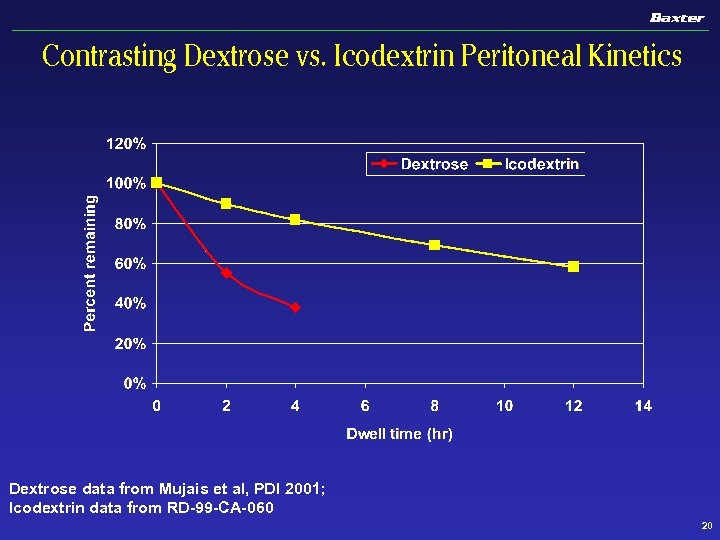 Contrasting Dextrose vs. Icodextrin Peritoneal Kinetics Dextrose data from Mujais et al, PDI 2001; Icodextrin data from RD-99 -CA-060 20
Contrasting Dextrose vs. Icodextrin Peritoneal Kinetics Dextrose data from Mujais et al, PDI 2001; Icodextrin data from RD-99 -CA-060 20
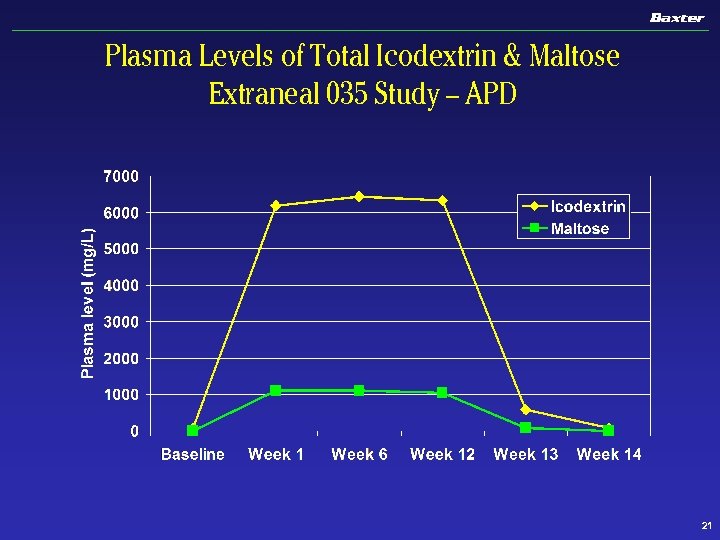 Plasma Levels of Total Icodextrin & Maltose Extraneal 035 Study – APD 21
Plasma Levels of Total Icodextrin & Maltose Extraneal 035 Study – APD 21
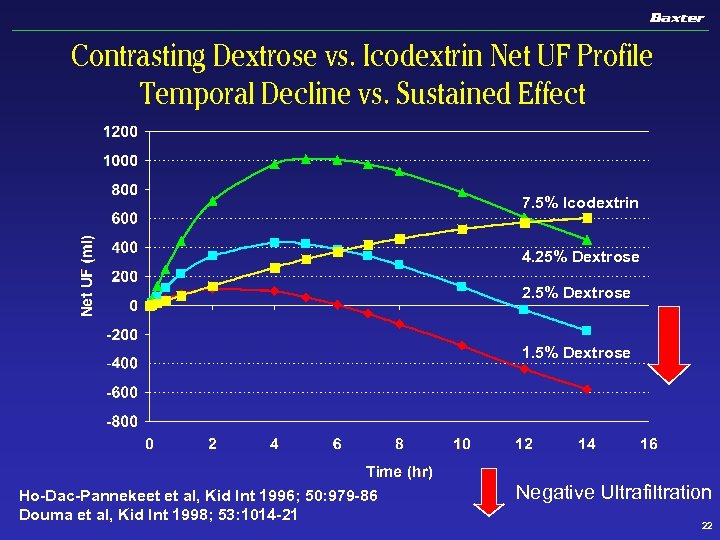 Contrasting Dextrose vs. Icodextrin Net UF Profile Temporal Decline vs. Sustained Effect 7. 5% Icodextrin 4. 25% Dextrose 2. 5% Dextrose 1. 5% Dextrose Ho-Dac-Pannekeet et al, Kid Int 1996; 50: 979 -86 Douma et al, Kid Int 1998; 53: 1014 -21 Negative Ultrafiltration 22
Contrasting Dextrose vs. Icodextrin Net UF Profile Temporal Decline vs. Sustained Effect 7. 5% Icodextrin 4. 25% Dextrose 2. 5% Dextrose 1. 5% Dextrose Ho-Dac-Pannekeet et al, Kid Int 1996; 50: 979 -86 Douma et al, Kid Int 1998; 53: 1014 -21 Negative Ultrafiltration 22
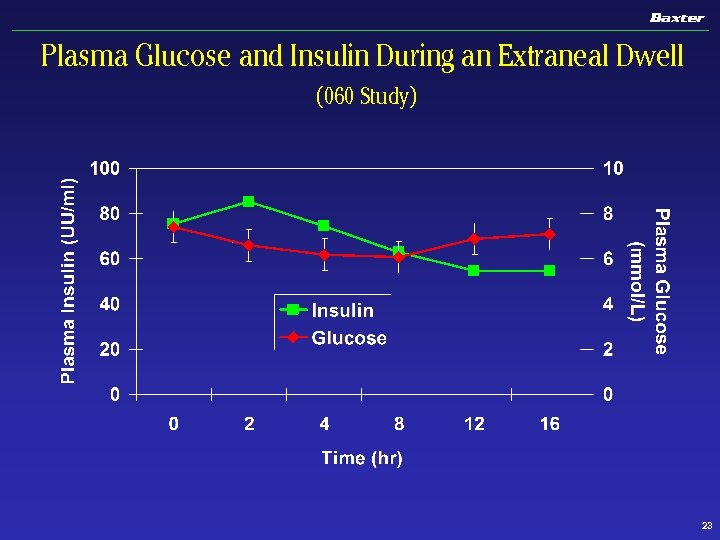 Plasma Glucose and Insulin During an Extraneal Dwell (060 Study) 23
Plasma Glucose and Insulin During an Extraneal Dwell (060 Study) 23
 Rationale for Extraneal n Fluid management in PD is constrained by the consequences of the underlying disease resulting in a necessary high reliance on peritoneal ultrafiltration. n With dextrose-based solutions a long dwell can compound the difficulties with fluid management n There is an unmet need in fluid management in PD n Extraneal is uniquely suited for successful ultrafiltration during the long dwell, and can contribute significantly to fluid management in these critically ill patients 24
Rationale for Extraneal n Fluid management in PD is constrained by the consequences of the underlying disease resulting in a necessary high reliance on peritoneal ultrafiltration. n With dextrose-based solutions a long dwell can compound the difficulties with fluid management n There is an unmet need in fluid management in PD n Extraneal is uniquely suited for successful ultrafiltration during the long dwell, and can contribute significantly to fluid management in these critically ill patients 24
 Clinical Trial Experience with Extraneal Efficacy and Safety Marsha Wolfson, MD, FACP VP, Global Clinical Affairs Francis G. Ogrinc, Ph. D. Clinical Statistician
Clinical Trial Experience with Extraneal Efficacy and Safety Marsha Wolfson, MD, FACP VP, Global Clinical Affairs Francis G. Ogrinc, Ph. D. Clinical Statistician
 Presentation Plan n Efficacy of Extraneal u u Peritoneal Clearance u n Net Ultrafiltration Special Assessments in Study 131 Safety Profile of Extraneal u Database u Observational Mortality Data u Adverse Events u Laboratory Values 26
Presentation Plan n Efficacy of Extraneal u u Peritoneal Clearance u n Net Ultrafiltration Special Assessments in Study 131 Safety Profile of Extraneal u Database u Observational Mortality Data u Adverse Events u Laboratory Values 26
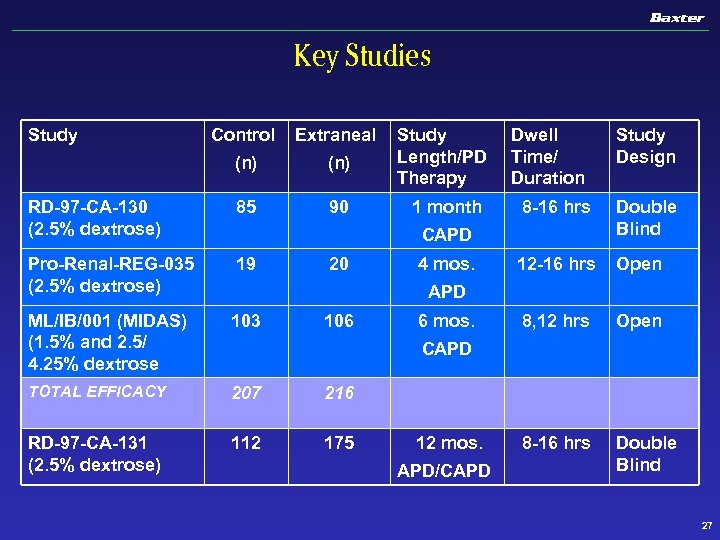 Key Studies Study Control Extraneal (n) Study Length/PD Therapy RD-97 -CA-130 (2. 5% dextrose) 85 90 1 month Pro-Renal-REG-035 (2. 5% dextrose) 19 ML/IB/001 (MIDAS) (1. 5% and 2. 5/ 4. 25% dextrose 103 TOTAL EFFICACY 207 216 RD-97 -CA-131 (2. 5% dextrose) 112 175 Dwell Time/ Duration 8 -16 hrs Double Blind 12 -16 hrs Open 8, 12 hrs Open 8 -16 hrs Double Blind CAPD 20 4 mos. Study Design APD 106 6 mos. CAPD 12 mos. APD/CAPD 27
Key Studies Study Control Extraneal (n) Study Length/PD Therapy RD-97 -CA-130 (2. 5% dextrose) 85 90 1 month Pro-Renal-REG-035 (2. 5% dextrose) 19 ML/IB/001 (MIDAS) (1. 5% and 2. 5/ 4. 25% dextrose 103 TOTAL EFFICACY 207 216 RD-97 -CA-131 (2. 5% dextrose) 112 175 Dwell Time/ Duration 8 -16 hrs Double Blind 12 -16 hrs Open 8, 12 hrs Open 8 -16 hrs Double Blind CAPD 20 4 mos. Study Design APD 106 6 mos. CAPD 12 mos. APD/CAPD 27
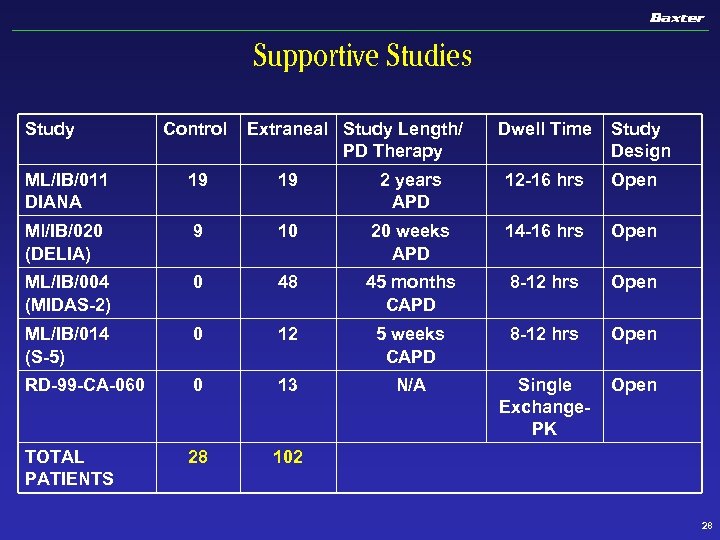 Supportive Studies Study Control Extraneal Study Length/ PD Therapy Dwell Time Study Design ML/IB/011 DIANA 19 19 2 years APD 12 -16 hrs Open Ml/IB/020 (DELIA) 9 10 20 weeks APD 14 -16 hrs Open ML/IB/004 (MIDAS-2) 0 48 45 months CAPD 8 -12 hrs Open ML/IB/014 (S-5) 0 12 5 weeks CAPD 8 -12 hrs Open RD-99 -CA-060 0 13 N/A Single Exchange. PK Open TOTAL PATIENTS 28 102 28
Supportive Studies Study Control Extraneal Study Length/ PD Therapy Dwell Time Study Design ML/IB/011 DIANA 19 19 2 years APD 12 -16 hrs Open Ml/IB/020 (DELIA) 9 10 20 weeks APD 14 -16 hrs Open ML/IB/004 (MIDAS-2) 0 48 45 months CAPD 8 -12 hrs Open ML/IB/014 (S-5) 0 12 5 weeks CAPD 8 -12 hrs Open RD-99 -CA-060 0 13 N/A Single Exchange. PK Open TOTAL PATIENTS 28 102 28
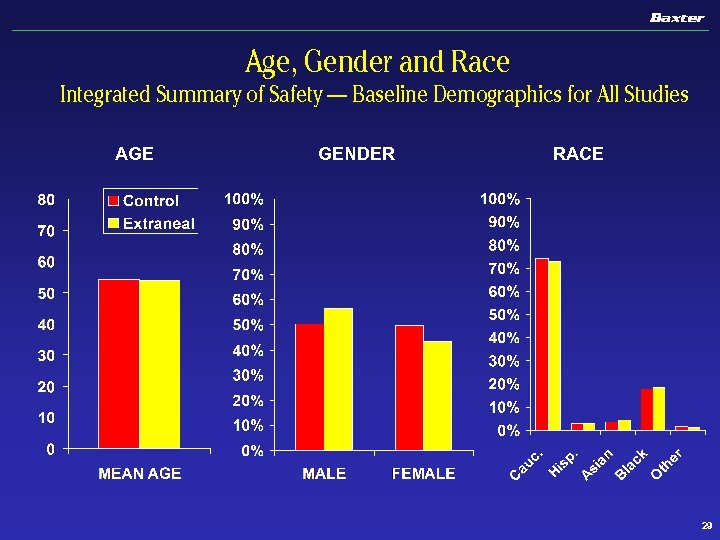 Age, Gender and Race Integrated Summary of Safety — Baseline Demographics for All Studies AGE GENDER RACE 29
Age, Gender and Race Integrated Summary of Safety — Baseline Demographics for All Studies AGE GENDER RACE 29
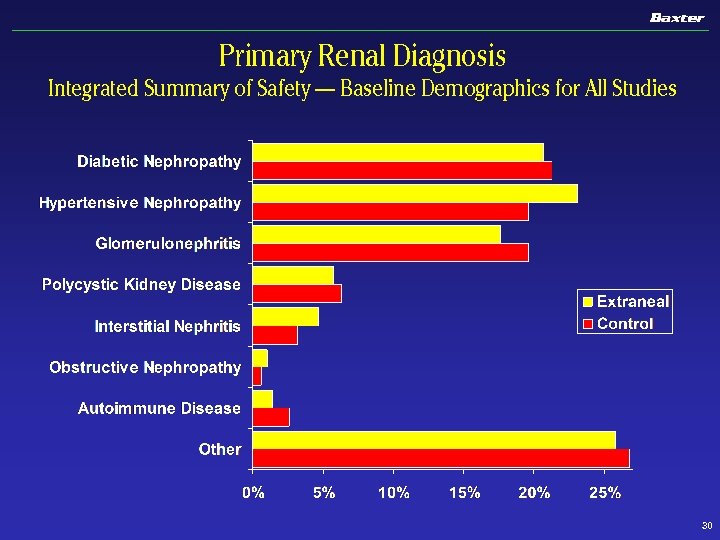 Primary Renal Diagnosis Integrated Summary of Safety — Baseline Demographics for All Studies 30
Primary Renal Diagnosis Integrated Summary of Safety — Baseline Demographics for All Studies 30
 Efficacy Endpoints n Primary Endpoint u n Net Ultrafiltration Secondary Endpoints u u n Peritoneal Creatinine Clearance Peritoneal Urea Clearance Special Assessments from Study 131 u Edema u Body weight u Qo. L 31
Efficacy Endpoints n Primary Endpoint u n Net Ultrafiltration Secondary Endpoints u u n Peritoneal Creatinine Clearance Peritoneal Urea Clearance Special Assessments from Study 131 u Edema u Body weight u Qo. L 31
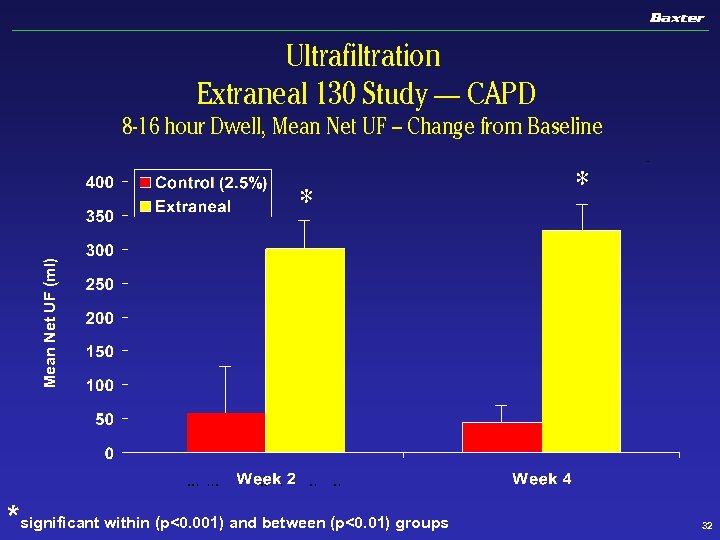 Ultrafiltration Extraneal 130 Study — CAPD 8 -16 hour Dwell, Mean Net UF – Change from Baseline Mean Net UF (ml) * * *significant within (p<0. 001) and between (p<0. 01) groups 32
Ultrafiltration Extraneal 130 Study — CAPD 8 -16 hour Dwell, Mean Net UF – Change from Baseline Mean Net UF (ml) * * *significant within (p<0. 001) and between (p<0. 01) groups 32
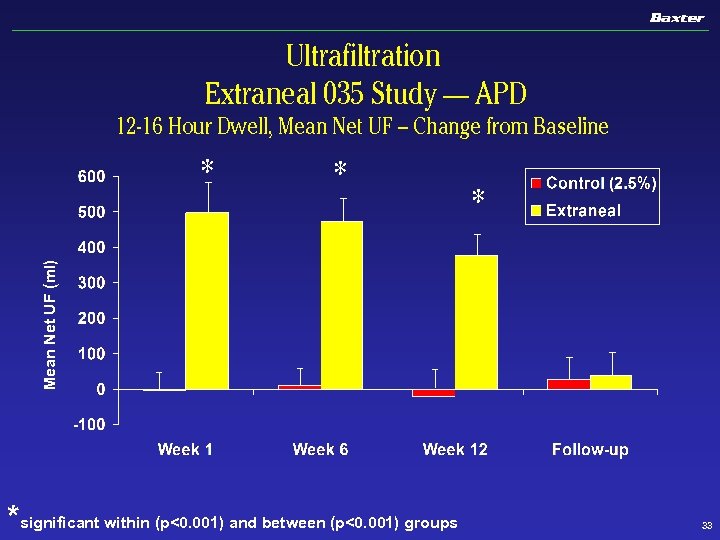 Ultrafiltration Extraneal 035 Study — APD 12 -16 Hour Dwell, Mean Net UF – Change from Baseline * * Mean Net UF (ml) * *significant within (p<0. 001) and between (p<0. 001) groups 33
Ultrafiltration Extraneal 035 Study — APD 12 -16 Hour Dwell, Mean Net UF – Change from Baseline * * Mean Net UF (ml) * *significant within (p<0. 001) and between (p<0. 001) groups 33
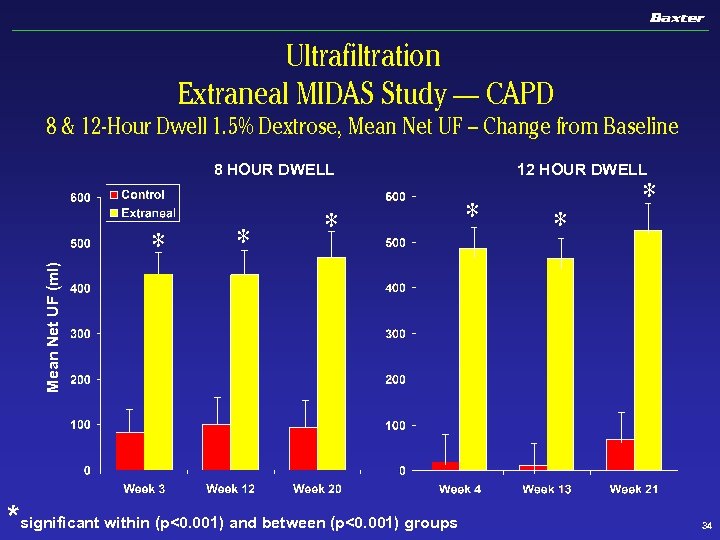 Ultrafiltration Extraneal MIDAS Study — CAPD 8 & 12 -Hour Dwell 1. 5% Dextrose, Mean Net UF – Change from Baseline 12 HOUR DWELL Mean Net UF (ml) 8 HOUR DWELL * *significant within (p<0. 001) and between (p<0. 001) groups * * * 34
Ultrafiltration Extraneal MIDAS Study — CAPD 8 & 12 -Hour Dwell 1. 5% Dextrose, Mean Net UF – Change from Baseline 12 HOUR DWELL Mean Net UF (ml) 8 HOUR DWELL * *significant within (p<0. 001) and between (p<0. 001) groups * * * 34
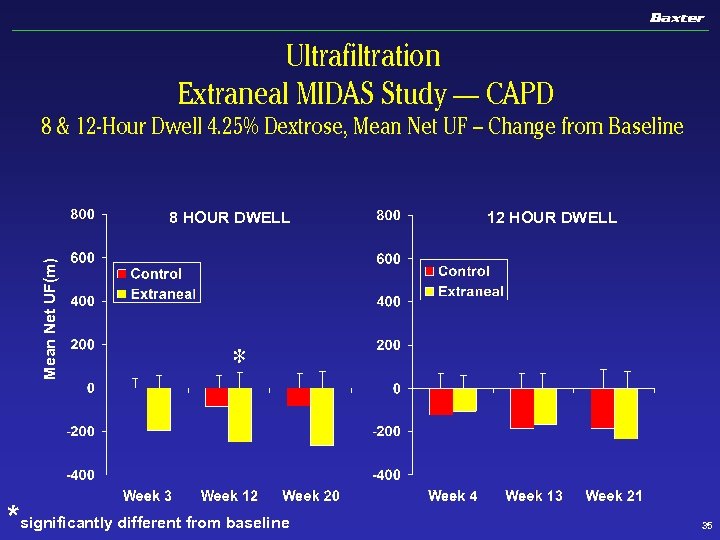 Ultrafiltration Extraneal MIDAS Study — CAPD 8 & 12 -Hour Dwell 4. 25% Dextrose, Mean Net UF – Change from Baseline Mean Net UF(m) 8 HOUR DWELL 12 HOUR DWELL * *significantly different from baseline 35
Ultrafiltration Extraneal MIDAS Study — CAPD 8 & 12 -Hour Dwell 4. 25% Dextrose, Mean Net UF – Change from Baseline Mean Net UF(m) 8 HOUR DWELL 12 HOUR DWELL * *significantly different from baseline 35
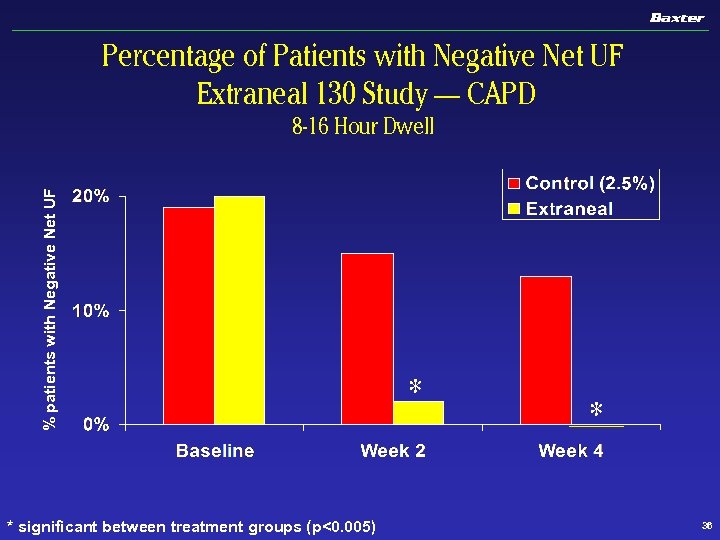 Percentage of Patients with Negative Net UF Extraneal 130 Study — CAPD % patients with Negative Net UF 8 -16 Hour Dwell * significant between treatment groups (p<0. 005) * * 36
Percentage of Patients with Negative Net UF Extraneal 130 Study — CAPD % patients with Negative Net UF 8 -16 Hour Dwell * significant between treatment groups (p<0. 005) * * 36
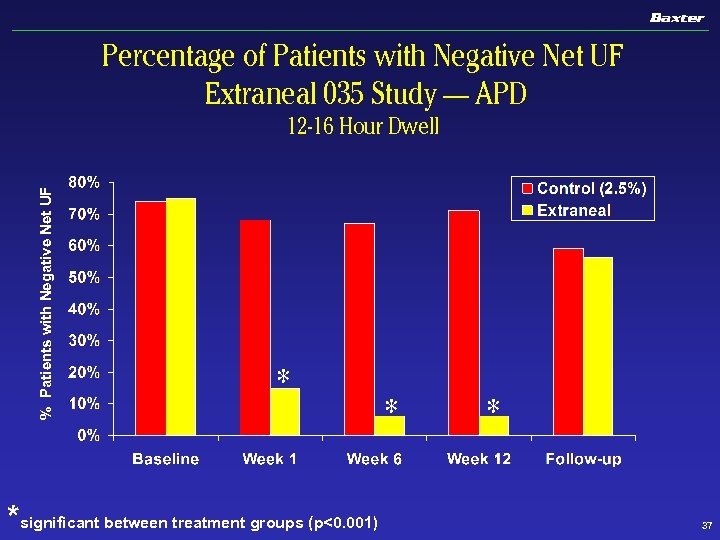 Percentage of Patients with Negative Net UF Extraneal 035 Study — APD % Patients with Negative Net UF 12 -16 Hour Dwell * *significant between treatment groups (p<0. 001) * * 37
Percentage of Patients with Negative Net UF Extraneal 035 Study — APD % Patients with Negative Net UF 12 -16 Hour Dwell * *significant between treatment groups (p<0. 001) * * 37
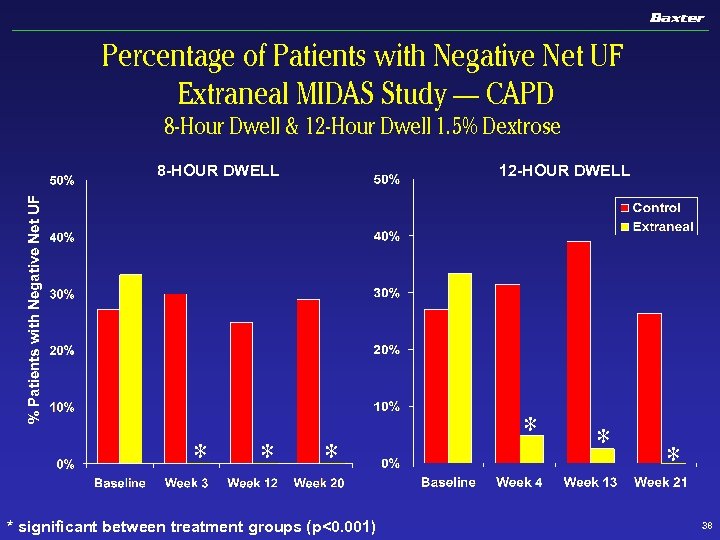 Percentage of Patients with Negative Net UF Extraneal MIDAS Study — CAPD 8 -Hour Dwell & 12 -Hour Dwell 1. 5% Dextrose 12 -HOUR DWELL % Patients with Negative Net UF 8 -HOUR DWELL * * significant between treatment groups (p<0. 001) * * * 38
Percentage of Patients with Negative Net UF Extraneal MIDAS Study — CAPD 8 -Hour Dwell & 12 -Hour Dwell 1. 5% Dextrose 12 -HOUR DWELL % Patients with Negative Net UF 8 -HOUR DWELL * * significant between treatment groups (p<0. 001) * * * 38
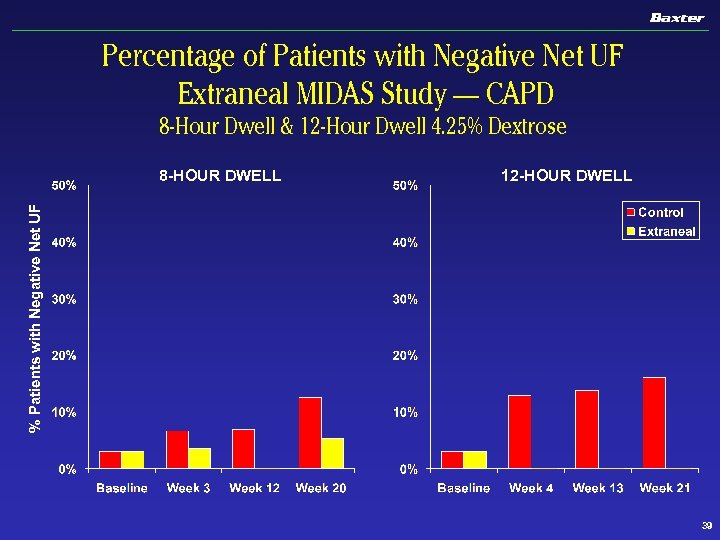 Percentage of Patients with Negative Net UF Extraneal MIDAS Study — CAPD 8 -Hour Dwell & 12 -Hour Dwell 4. 25% Dextrose 12 -HOUR DWELL % Patients with Negative Net UF 8 -HOUR DWELL 39
Percentage of Patients with Negative Net UF Extraneal MIDAS Study — CAPD 8 -Hour Dwell & 12 -Hour Dwell 4. 25% Dextrose 12 -HOUR DWELL % Patients with Negative Net UF 8 -HOUR DWELL 39
 Efficacy Endpoints n Primary Endpoint u n Net Ultrafiltration Secondary Endpoints u u n Peritoneal Creatinine Clearance Peritoneal Urea Clearance Special Assessments from Study 131 u Edema u Body weight u Qo. L 40
Efficacy Endpoints n Primary Endpoint u n Net Ultrafiltration Secondary Endpoints u u n Peritoneal Creatinine Clearance Peritoneal Urea Clearance Special Assessments from Study 131 u Edema u Body weight u Qo. L 40
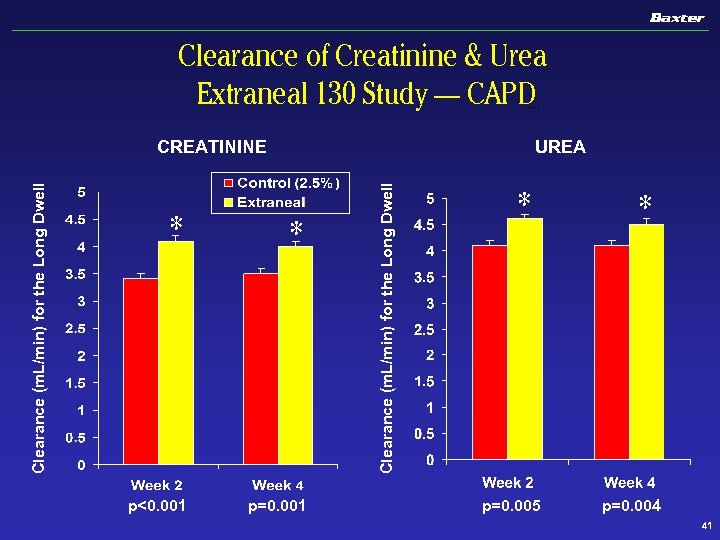 Clearance of Creatinine & Urea Extraneal 130 Study — CAPD UREA * * p<0. 001 p=0. 001 Clearance (m. L/min) for the Long Dwell CREATININE * p=0. 005 * p=0. 004 41
Clearance of Creatinine & Urea Extraneal 130 Study — CAPD UREA * * p<0. 001 p=0. 001 Clearance (m. L/min) for the Long Dwell CREATININE * p=0. 005 * p=0. 004 41
 Special Assessments from Long-term Study 131 n Edema n Body Weight n Quality of Life 42
Special Assessments from Long-term Study 131 n Edema n Body Weight n Quality of Life 42
 Peripheral Edema Assessment Extraneal 131 Study — CAPD & APD n Usually assessed by same individual n 0 - 3+ - Recorded on CRF n 4+ - Recorded as Adverse Event 43
Peripheral Edema Assessment Extraneal 131 Study — CAPD & APD n Usually assessed by same individual n 0 - 3+ - Recorded on CRF n 4+ - Recorded as Adverse Event 43
 Adverse Events for Edema Extraneal 131 Study — CAPD & APD COSTART Term Control N=112 Extraneal N=175 Peripheral Edema (p=0. 002) 17. 9% 6. 3% Other Edema 13. 4% 10. 3% 44
Adverse Events for Edema Extraneal 131 Study — CAPD & APD COSTART Term Control N=112 Extraneal N=175 Peripheral Edema (p=0. 002) 17. 9% 6. 3% Other Edema 13. 4% 10. 3% 44
 Special Assessments from Study 131 n Edema n Body Weight n Quality of Life 45
Special Assessments from Study 131 n Edema n Body Weight n Quality of Life 45
 Body Weight n Important parameter in ESRD n Assesses: u Fluid balance - short term u Body composition - long term 46
Body Weight n Important parameter in ESRD n Assesses: u Fluid balance - short term u Body composition - long term 46
 Body Weight – Before Drain Extraneal 131 Study — CAPD & APD (N=47 Control, N=88 Extraneal) n Extraneal patients - maintained body weight at 52 weeks (mean change -0. 03 kg) n Control patients - average increase of 2. 33 kg at 52 weeks u (p=0. 022 at 52 weeks) 47
Body Weight – Before Drain Extraneal 131 Study — CAPD & APD (N=47 Control, N=88 Extraneal) n Extraneal patients - maintained body weight at 52 weeks (mean change -0. 03 kg) n Control patients - average increase of 2. 33 kg at 52 weeks u (p=0. 022 at 52 weeks) 47
 Special Assessments from Long-term Study 131 n Edema n Body Weight n Quality of Life 48
Special Assessments from Long-term Study 131 n Edema n Body Weight n Quality of Life 48
 Quality of Life — KDQo. L Extraneal 131 Study — CAPD & APD n Patients completing both Baseline and Week 52 (N=25 Control, 41 Extraneal): u Queried on 35 kidney-specific symptoms/problems u Short Form 36 49
Quality of Life — KDQo. L Extraneal 131 Study — CAPD & APD n Patients completing both Baseline and Week 52 (N=25 Control, 41 Extraneal): u Queried on 35 kidney-specific symptoms/problems u Short Form 36 49
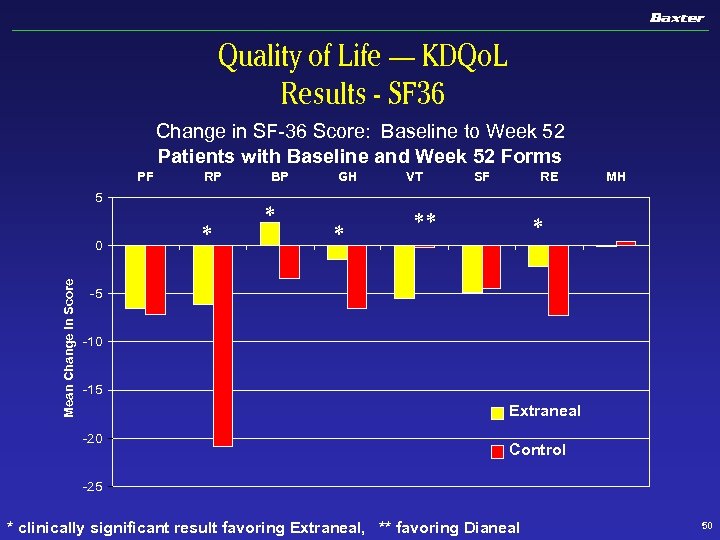 Quality of Life — KDQo. L Results - SF 36 Change in SF-36 Score: Baseline to Week 52 Patients with Baseline and Week 52 Forms RP 5 Mean Change in Score 0 * BP * GH * VT SF RE ** PF MH * -5 -10 -15 Extraneal -20 Control -25 * clinically significant result favoring Extraneal, ** favoring Dianeal 50
Quality of Life — KDQo. L Results - SF 36 Change in SF-36 Score: Baseline to Week 52 Patients with Baseline and Week 52 Forms RP 5 Mean Change in Score 0 * BP * GH * VT SF RE ** PF MH * -5 -10 -15 Extraneal -20 Control -25 * clinically significant result favoring Extraneal, ** favoring Dianeal 50
 Quality of Life — KDQOL Extraneal 131 Study — CAPD & APD For patients completing both Baseline and Week 52 (N=25 Control, N=41 Extraneal): n 35 Symptoms/Problems: 10 favored Extraneal, 5 favored Dextrose n Short Form 36 u Domains: 4 favored Extraneal, 1 favored Dextrose u Health Transition Summary: 30% of Extraneal patients vs 4% of Control patients reported their health was much better as compared to one year ago (p=0. 032) 51
Quality of Life — KDQOL Extraneal 131 Study — CAPD & APD For patients completing both Baseline and Week 52 (N=25 Control, N=41 Extraneal): n 35 Symptoms/Problems: 10 favored Extraneal, 5 favored Dextrose n Short Form 36 u Domains: 4 favored Extraneal, 1 favored Dextrose u Health Transition Summary: 30% of Extraneal patients vs 4% of Control patients reported their health was much better as compared to one year ago (p=0. 032) 51
 Extraneal Efficacy Conclusions n Superior UF compared to 1. 5% or 2. 5% Dextrose and comparable to 4. 25% Dextrose n Significantly reduced number of patients with negative net UF compared to both 1. 5% and 2. 5% Dextrose and comparable to 4. 25% Dextrose n Significantly increased peritoneal clearance of urea and creatinine compared to 2. 5% Dextrose n Potential benefit in preventing weight gain and edema, and improving quality of life 52
Extraneal Efficacy Conclusions n Superior UF compared to 1. 5% or 2. 5% Dextrose and comparable to 4. 25% Dextrose n Significantly reduced number of patients with negative net UF compared to both 1. 5% and 2. 5% Dextrose and comparable to 4. 25% Dextrose n Significantly increased peritoneal clearance of urea and creatinine compared to 2. 5% Dextrose n Potential benefit in preventing weight gain and edema, and improving quality of life 52
 Safety Profile of Extraneal n Database n Observational Mortality Data n Adverse Events n Laboratory Values n Membrane Transport Characteristics 53
Safety Profile of Extraneal n Database n Observational Mortality Data n Adverse Events n Laboratory Values n Membrane Transport Characteristics 53
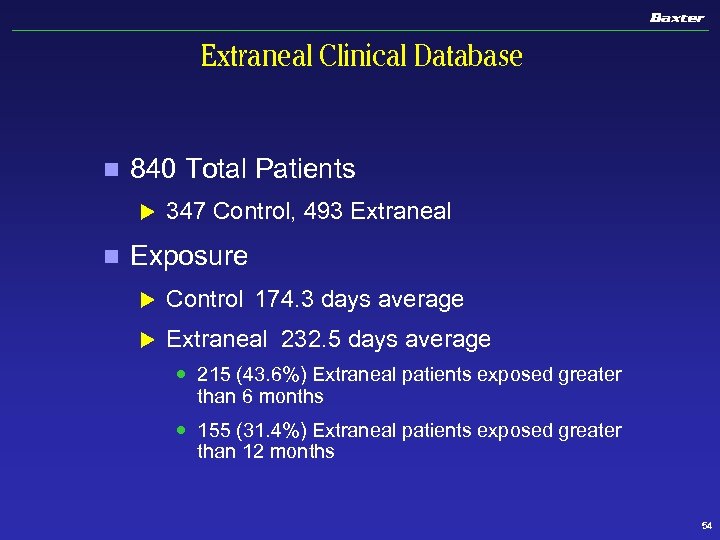 Extraneal Clinical Database n 840 Total Patients u n 347 Control, 493 Extraneal Exposure u Control 174. 3 days average u Extraneal 232. 5 days average 215 (43. 6%) Extraneal patients exposed greater than 6 months 155 (31. 4%) Extraneal patients exposed greater than 12 months 54
Extraneal Clinical Database n 840 Total Patients u n 347 Control, 493 Extraneal Exposure u Control 174. 3 days average u Extraneal 232. 5 days average 215 (43. 6%) Extraneal patients exposed greater than 6 months 155 (31. 4%) Extraneal patients exposed greater than 12 months 54
 Patient Disposition (Withdrawals/Completions, All Studies) CONTROL N=347 EXTRANEAL N=493 Completed 70. 9% 65. 5% Prematurely Discontinued 29. 1% 34. 5% Transplantation 6. 1% 8. 5% Adverse Event 11. 8% 13. 4% Deaths 3. 2% 3. 7% Protocol Violations 3. 2% 1. 4% Other 4. 9% 7. 5% 55
Patient Disposition (Withdrawals/Completions, All Studies) CONTROL N=347 EXTRANEAL N=493 Completed 70. 9% 65. 5% Prematurely Discontinued 29. 1% 34. 5% Transplantation 6. 1% 8. 5% Adverse Event 11. 8% 13. 4% Deaths 3. 2% 3. 7% Protocol Violations 3. 2% 1. 4% Other 4. 9% 7. 5% 55
 Safety Profile of Extraneal n Database n Observational Mortality Data n Adverse Events n Laboratory Values n Membrane Transport Characteristics 56
Safety Profile of Extraneal n Database n Observational Mortality Data n Adverse Events n Laboratory Values n Membrane Transport Characteristics 56
 Observational Mortality Extraneal 131 Study: Original Data Collection n Each patient was followed until drop out or completion of 12 months. n Once a patient discontinued (early drop out or completer), he was observed for an additional 30 days to collect adverse events, including death. n Based on these data collection methods: Treatment Group Number of Patients Control 112 Deaths N % 5 4. 5 EXTRANEAL 175 13 7. 4 57
Observational Mortality Extraneal 131 Study: Original Data Collection n Each patient was followed until drop out or completion of 12 months. n Once a patient discontinued (early drop out or completer), he was observed for an additional 30 days to collect adverse events, including death. n Based on these data collection methods: Treatment Group Number of Patients Control 112 Deaths N % 5 4. 5 EXTRANEAL 175 13 7. 4 57
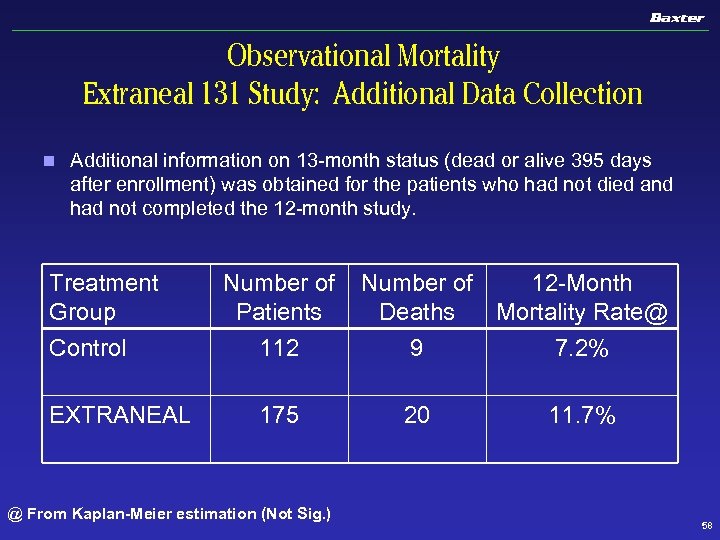 Observational Mortality Extraneal 131 Study: Additional Data Collection n Additional information on 13 -month status (dead or alive 395 days after enrollment) was obtained for the patients who had not died and had not completed the 12 -month study. Treatment Group Number of 12 -Month Patients Deaths Mortality Rate@ Control 112 9 7. 2% EXTRANEAL 175 20 11. 7% @ From Kaplan-Meier estimation (Not Sig. ) 58
Observational Mortality Extraneal 131 Study: Additional Data Collection n Additional information on 13 -month status (dead or alive 395 days after enrollment) was obtained for the patients who had not died and had not completed the 12 -month study. Treatment Group Number of 12 -Month Patients Deaths Mortality Rate@ Control 112 9 7. 2% EXTRANEAL 175 20 11. 7% @ From Kaplan-Meier estimation (Not Sig. ) 58
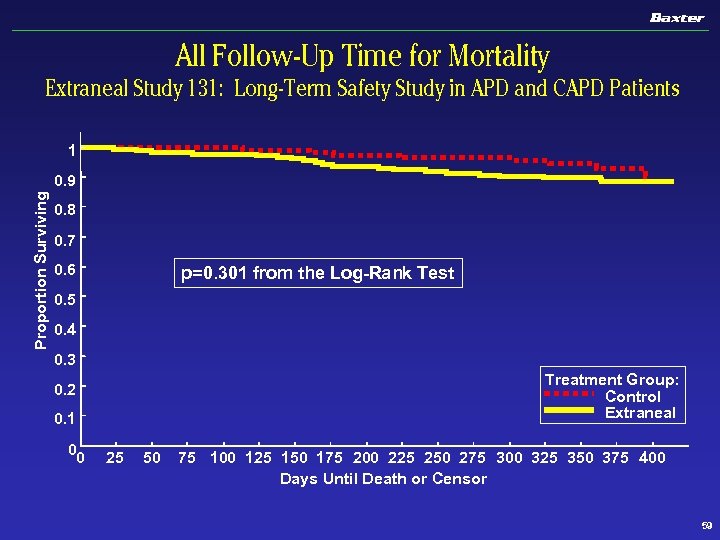 All Follow-Up Time for Mortality Extraneal Study 131: Long-Term Safety Study in APD and CAPD Patients 1 Proportion Surviving 0. 9 0. 8 0. 7 0. 6 p=0. 301 from the Log-Rank Test 0. 5 0. 4 0. 3 Treatment Group: Control Extraneal 0. 2 0. 1 0 0 25 50 75 100 125 150 175 200 225 250 275 300 325 350 375 400 Days Until Death or Censor 59
All Follow-Up Time for Mortality Extraneal Study 131: Long-Term Safety Study in APD and CAPD Patients 1 Proportion Surviving 0. 9 0. 8 0. 7 0. 6 p=0. 301 from the Log-Rank Test 0. 5 0. 4 0. 3 Treatment Group: Control Extraneal 0. 2 0. 1 0 0 25 50 75 100 125 150 175 200 225 250 275 300 325 350 375 400 Days Until Death or Censor 59
 Observational Mortality All Studies (Other Than MIDAS-2) n Because of the limited database in Study 131, mortality results from all studies, other than MIDAS-2, were combined to describe the overall mortality. n Intent-to-treat methods were applied and all deaths were included. Some of these were after study participation. Treatment Group Number of Number Total Follow-Up Deaths per Patients of Deaths Time (Patient-Yrs) 1000 Pt-Year Control 285 20 189 106 EXTRANEAL 366 26 244 107 60
Observational Mortality All Studies (Other Than MIDAS-2) n Because of the limited database in Study 131, mortality results from all studies, other than MIDAS-2, were combined to describe the overall mortality. n Intent-to-treat methods were applied and all deaths were included. Some of these were after study participation. Treatment Group Number of Number Total Follow-Up Deaths per Patients of Deaths Time (Patient-Yrs) 1000 Pt-Year Control 285 20 189 106 EXTRANEAL 366 26 244 107 60
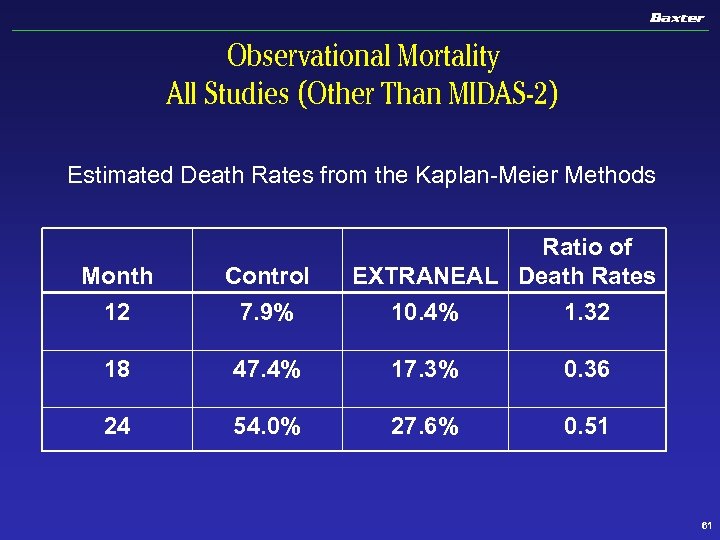 Observational Mortality All Studies (Other Than MIDAS-2) Estimated Death Rates from the Kaplan-Meier Methods Ratio of EXTRANEAL Death Rates Month Control 12 7. 9% 10. 4% 1. 32 18 47. 4% 17. 3% 0. 36 24 54. 0% 27. 6% 0. 51 61
Observational Mortality All Studies (Other Than MIDAS-2) Estimated Death Rates from the Kaplan-Meier Methods Ratio of EXTRANEAL Death Rates Month Control 12 7. 9% 10. 4% 1. 32 18 47. 4% 17. 3% 0. 36 24 54. 0% 27. 6% 0. 51 61
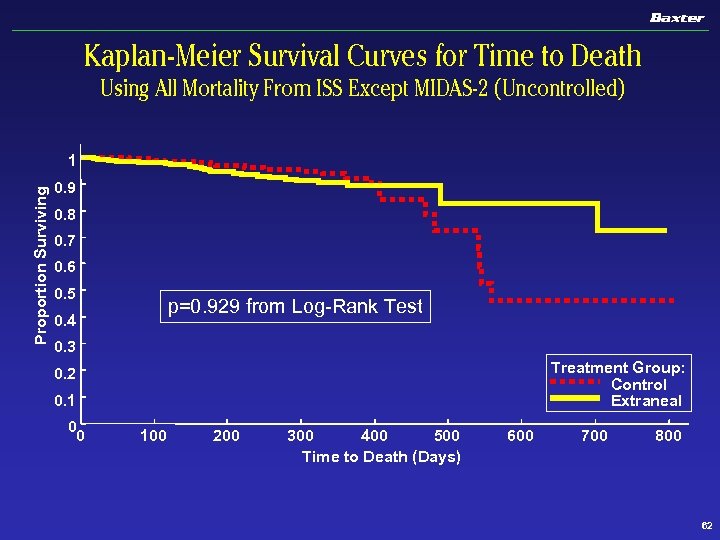 Kaplan-Meier Survival Curves for Time to Death Using All Mortality From ISS Except MIDAS-2 (Uncontrolled) Proportion Surviving 1 0. 9 0. 8 0. 7 0. 6 0. 5 p=0. 929 from Log-Rank Test 0. 4 0. 3 Treatment Group: Control Extraneal 0. 2 0. 1 0 0 100 200 300 400 500 Time to Death (Days) 600 700 800 62
Kaplan-Meier Survival Curves for Time to Death Using All Mortality From ISS Except MIDAS-2 (Uncontrolled) Proportion Surviving 1 0. 9 0. 8 0. 7 0. 6 0. 5 p=0. 929 from Log-Rank Test 0. 4 0. 3 Treatment Group: Control Extraneal 0. 2 0. 1 0 0 100 200 300 400 500 Time to Death (Days) 600 700 800 62
 Observational Mortality Summary n Mortality information from all clinical studies was combined to better describe the experience for Extraneal. n There were 366 Extraneal patients in this integrated safety database. n These provide 244 patient-years of experience with Extraneal. n Survival times were comparable for Control and Extraneal u Hazard Ratio: 1. 03 u 90% Confidence Interval: [0. 63, 1. 68] 63
Observational Mortality Summary n Mortality information from all clinical studies was combined to better describe the experience for Extraneal. n There were 366 Extraneal patients in this integrated safety database. n These provide 244 patient-years of experience with Extraneal. n Survival times were comparable for Control and Extraneal u Hazard Ratio: 1. 03 u 90% Confidence Interval: [0. 63, 1. 68] 63
 Safety Profile of Extraneal n Database n Observational Mortality Data n Adverse Events n Laboratory Values n Membrane Transport Characteristics 64
Safety Profile of Extraneal n Database n Observational Mortality Data n Adverse Events n Laboratory Values n Membrane Transport Characteristics 64
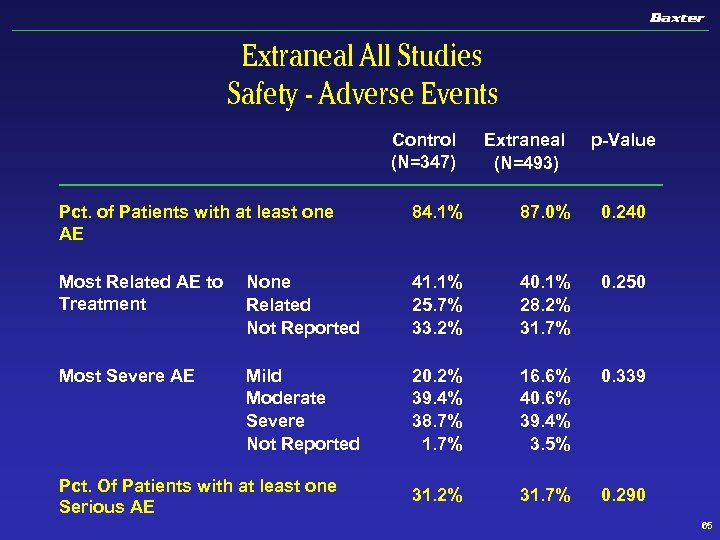 Extraneal All Studies Safety - Adverse Events Pct. of Patients with at least one AE Control (N=347) Extraneal (N=493) p-Value 84. 1% 87. 0% 0. 240 Most Related AE to Treatment None Related Not Reported 41. 1% 25. 7% 33. 2% 40. 1% 28. 2% 31. 7% 0. 250 Most Severe AE Mild Moderate Severe Not Reported 20. 2% 39. 4% 38. 7% 16. 6% 40. 6% 39. 4% 3. 5% 0. 339 31. 2% 31. 7% 0. 290 Pct. Of Patients with at least one Serious AE 65
Extraneal All Studies Safety - Adverse Events Pct. of Patients with at least one AE Control (N=347) Extraneal (N=493) p-Value 84. 1% 87. 0% 0. 240 Most Related AE to Treatment None Related Not Reported 41. 1% 25. 7% 33. 2% 40. 1% 28. 2% 31. 7% 0. 250 Most Severe AE Mild Moderate Severe Not Reported 20. 2% 39. 4% 38. 7% 16. 6% 40. 6% 39. 4% 3. 5% 0. 339 31. 2% 31. 7% 0. 290 Pct. Of Patients with at least one Serious AE 65
 Adverse Events n Peritonitis n Rash 66
Adverse Events n Peritonitis n Rash 66
 Adverse Events n Peritonitis most frequent adverse event u u n Control – 25. 4% Extraneal – 26. 4% Peritonitis most frequent serious adverse event - resulting in hospitalization u Control - 8. 6% u Extraneal - 5. 3% (p=0. 013) 67
Adverse Events n Peritonitis most frequent adverse event u u n Control – 25. 4% Extraneal – 26. 4% Peritonitis most frequent serious adverse event - resulting in hospitalization u Control - 8. 6% u Extraneal - 5. 3% (p=0. 013) 67
 Adverse Events n Of all adverse events, only Rash showed greater than 5 percentage points difference between groups 68
Adverse Events n Of all adverse events, only Rash showed greater than 5 percentage points difference between groups 68
 Total Skin Events Incidence Rates – All Studies, Unrelated and Related AEs COSTART Code Control (N=347) Extraneal (N=493) All SKIN EVENTS 22. 5% 27. 0% 4. 6% 10. 1% 0. 3% 1. 8% RASH DERM EXFOL 69
Total Skin Events Incidence Rates – All Studies, Unrelated and Related AEs COSTART Code Control (N=347) Extraneal (N=493) All SKIN EVENTS 22. 5% 27. 0% 4. 6% 10. 1% 0. 3% 1. 8% RASH DERM EXFOL 69
 Total Skin Events Incidence Rates – All Studies, Related AEs COSTART Code Control (N=347) Extraneal (N=493) All SKIN EVENTS 4. 0% 9. 9% RASH 1. 7% 5. 5% DERM EXFOL 0. 0% 1. 6% 70
Total Skin Events Incidence Rates – All Studies, Related AEs COSTART Code Control (N=347) Extraneal (N=493) All SKIN EVENTS 4. 0% 9. 9% RASH 1. 7% 5. 5% DERM EXFOL 0. 0% 1. 6% 70
 Outcomes for Rash in Clinical Trials n 6 patients withdrew/discontinued for rash, 1 for exfoliative dermatitis n All were in the Extraneal Group n No patient was hospitalized for rash n All rash events resolved n No anaphylaxis n No Stevens-Johnson Syndrome 71
Outcomes for Rash in Clinical Trials n 6 patients withdrew/discontinued for rash, 1 for exfoliative dermatitis n All were in the Extraneal Group n No patient was hospitalized for rash n All rash events resolved n No anaphylaxis n No Stevens-Johnson Syndrome 71
 Safety Profile of Extraneal n Database n Observational Mortality Data n Adverse Events n Laboratory Values n Membrane Transport Characteristics 72
Safety Profile of Extraneal n Database n Observational Mortality Data n Adverse Events n Laboratory Values n Membrane Transport Characteristics 72
 Laboratory Value Changes At Last Visit, Between Groups INCREASED: n Alkaline Phosphatase DECREASED: n Amylase-Assay Interference n Sodium n Chloride (130, 131 and 035 studies) 73
Laboratory Value Changes At Last Visit, Between Groups INCREASED: n Alkaline Phosphatase DECREASED: n Amylase-Assay Interference n Sodium n Chloride (130, 131 and 035 studies) 73
 Serum Alkaline Phosphatase (U/L) (130, 131 and 035 Studies) n Mean change u u n +4. 039 Control +19. 073 Extraneal Patients above normal range (31. 0 - 115. 0 U/L) u u n 23. 6% Control 33. 5% Extraneal Adverse Events for increased Alk. Phos. u 1. 7% Control u 2. 8% Extraneal 74
Serum Alkaline Phosphatase (U/L) (130, 131 and 035 Studies) n Mean change u u n +4. 039 Control +19. 073 Extraneal Patients above normal range (31. 0 - 115. 0 U/L) u u n 23. 6% Control 33. 5% Extraneal Adverse Events for increased Alk. Phos. u 1. 7% Control u 2. 8% Extraneal 74
 Serum Sodium (mmol/L) All Studies n Mean change u u n -0. 272 Control -2. 771 Extraneal Patients below normal range (135 -148 mmol/L) u u n 15. 8% Control 32. 5% Extraneal Adverse Events for Hyponatremia u 2. 0% Control u 2. 2% Extraneal 75
Serum Sodium (mmol/L) All Studies n Mean change u u n -0. 272 Control -2. 771 Extraneal Patients below normal range (135 -148 mmol/L) u u n 15. 8% Control 32. 5% Extraneal Adverse Events for Hyponatremia u 2. 0% Control u 2. 2% Extraneal 75
 Serum Chloride (mmol/L) All Studies n Mean change u u n +0. 610 Control -2. 003 Extraneal Patients below normal range (96 -112 mmol/L) u u n 32. 9% Control 51. 7% Extraneal Adverse Events for hypochloremia u 0. 9% Control u 1. 6% Extraneal 76
Serum Chloride (mmol/L) All Studies n Mean change u u n +0. 610 Control -2. 003 Extraneal Patients below normal range (96 -112 mmol/L) u u n 32. 9% Control 51. 7% Extraneal Adverse Events for hypochloremia u 0. 9% Control u 1. 6% Extraneal 76
 Safety Profile of Extraneal n Database n Observational Mortality Data n Adverse Events n Laboratory Values n Membrane Transport Characteristics 77
Safety Profile of Extraneal n Database n Observational Mortality Data n Adverse Events n Laboratory Values n Membrane Transport Characteristics 77
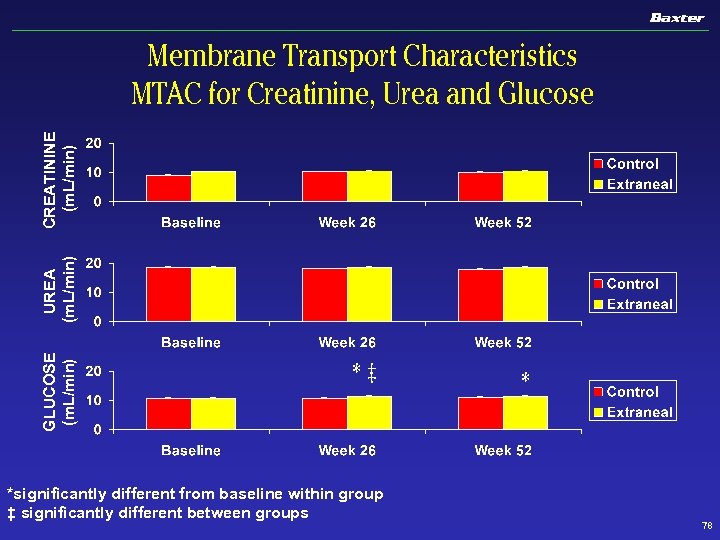 GLUCOSE (m. L/min) UREA (m. L/min) CREATININE (m. L/min) Membrane Transport Characteristics MTAC for Creatinine, Urea and Glucose *‡ *significantly different from baseline within group ‡ significantly different between groups * 78
GLUCOSE (m. L/min) UREA (m. L/min) CREATININE (m. L/min) Membrane Transport Characteristics MTAC for Creatinine, Urea and Glucose *‡ *significantly different from baseline within group ‡ significantly different between groups * 78
 Summary of Clinical Trial Results EFFICACY SAFETY n Increased Ultrafiltration n n Reduced percentage of patients with negative net UF Safety profile comparable to current therapy n Rash most frequent related AE n Increases in alkaline phosphatase, decreases in sodium and chloride, with no known clinical relevance n Increased peritoneal creatinine and urea clearance n Potential benefit in preventing weight gain and edema, and improving quality of life 79
Summary of Clinical Trial Results EFFICACY SAFETY n Increased Ultrafiltration n n Reduced percentage of patients with negative net UF Safety profile comparable to current therapy n Rash most frequent related AE n Increases in alkaline phosphatase, decreases in sodium and chloride, with no known clinical relevance n Increased peritoneal creatinine and urea clearance n Potential benefit in preventing weight gain and edema, and improving quality of life 79
 Extraneal Overall Summary n Patients on PD have limited therapy options for fluid management n Extraneal provides patients and their physicians a new dialysis solution that expands these options by sustaining effective ultrafiltration during the long dwell n The enhanced efficacy of the new solution is coupled with a safety profile comparable to existing solutions 80
Extraneal Overall Summary n Patients on PD have limited therapy options for fluid management n Extraneal provides patients and their physicians a new dialysis solution that expands these options by sustaining effective ultrafiltration during the long dwell n The enhanced efficacy of the new solution is coupled with a safety profile comparable to existing solutions 80
 Proposed Indication Extraneal is indicated for a single daily exchange for the long (8 -16 hour) dwell during continuous ambulatory peritoneal dialysis (CAPD) or automated peritoneal dialysis (APD) for the management of chronic renal failure. 81
Proposed Indication Extraneal is indicated for a single daily exchange for the long (8 -16 hour) dwell during continuous ambulatory peritoneal dialysis (CAPD) or automated peritoneal dialysis (APD) for the management of chronic renal failure. 81


