41b5fde394d5cfab039ed337978d3de8.ppt
- Количество слайдов: 46

Carbon Nanotubes

CNTs - OUTLINE • • • Formation Synthesis Chemically modified CNTs Properties Applications Carbon arc synthesis • Andrzej Huczko, Hubert Lange Laboratory of Plasma Chemistry Department of Chemistry, Warsaw University
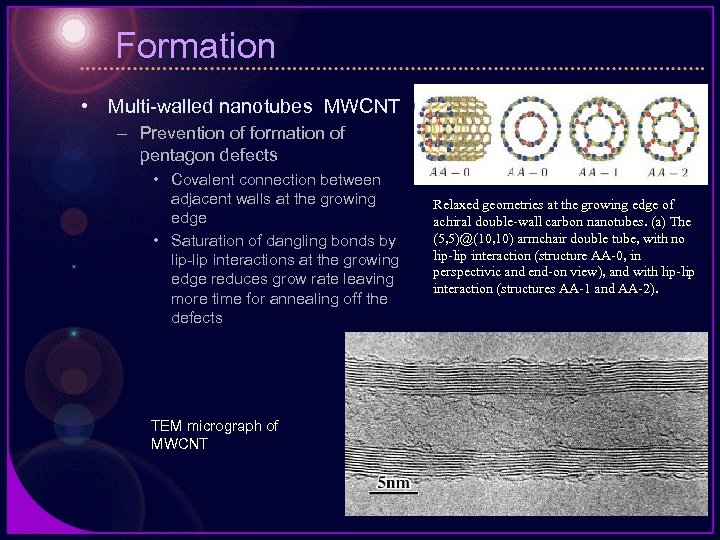
Formation • Multi-walled nanotubes MWCNT – Prevention of formation of pentagon defects • Covalent connection between adjacent walls at the growing edge • Saturation of dangling bonds by lip-lip interactions at the growing edge reduces grow rate leaving more time for annealing off the defects TEM micrograph of MWCNT Relaxed geometries at the growing edge of achiral double-wall carbon nanotubes. (a) The (5, 5)@(10, 10) armchair double tube, with no lip-lip interaction (structure AA-0, in perspectivic and end-on view), and with lip-lip interaction (structures AA-1 and AA-2).
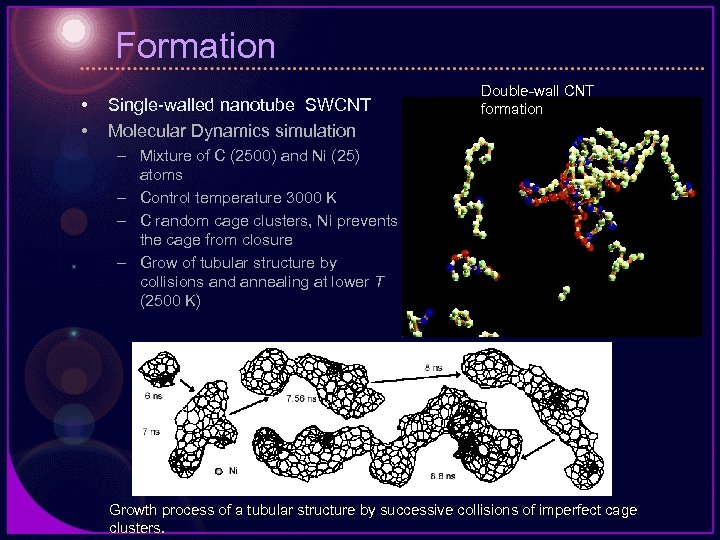
Formation • • Single-walled nanotube SWCNT Molecular Dynamics simulation Double-wall CNT formation – Mixture of C (2500) and Ni (25) atoms – Control temperature 3000 K – C random cage clusters, Ni prevents the cage from closure – Grow of tubular structure by collisions and annealing at lower T (2500 K) Growth process of a tubular structure by successive collisions of imperfect cage clusters.

Formation • • Single-walled nanotube SWCNT Gas-phase catalytic growth – Transition metal catalysts (Co, Ni) – C, metal and metal carbide clusters (aggregates) – Metal carbide clusters saturated with C – Nanotube grows out of the cluster – Computer simulation • Ni atoms block adjacent sites of pentagon • Ni atoms anneal existing defects
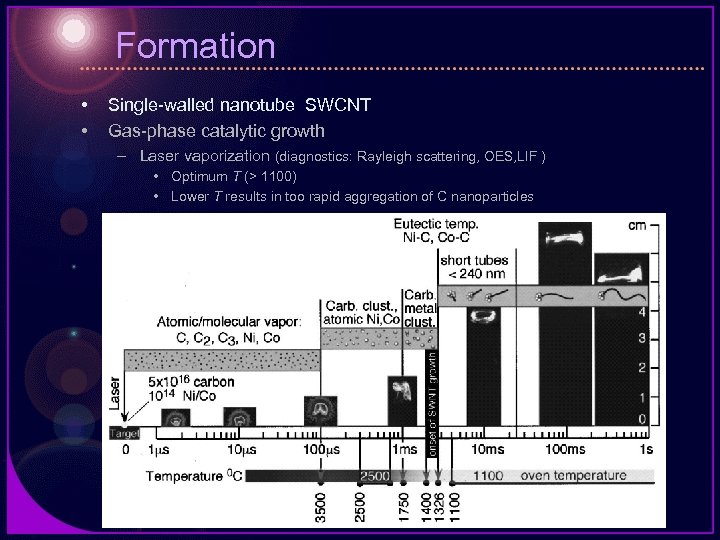
Formation • • Single-walled nanotube SWCNT Gas-phase catalytic growth – Laser vaporization (diagnostics: Rayleigh scattering, OES, LIF ) • Optimum T (> 1100) • Lower T results in too rapid aggregation of C nanoparticles

Formation • Single-walled nanotubes SWCNT – Electrode or metallic particle surface • Small flat graphene patches – How the graphene sheet can curl into nanotube without pentagons? • Spontaneous opening of double-layered graphitic patches – Bridging the opposite edges of parallel patches – Extreme curvature forms without pentagons

Synthesis • Carbon arc – 1991 Iijima in carbon soot – 1988 SEM images of MWCNTs from catalytic pyrolysis of hydrocarbons – 1889 US patent: ‘hair-like carbon filaments’ from CH 4 decomposition in iron crucible • DC arc sublimation of anode – MWCNT • He, 500 torr • Cathode deposit – Outer glossy gray hard-shell – Inner dark black soft-core with nanotubes – SWNT • Metal catalyst (Fe, Ni, Y, Co) – Vapor phase formation of SWCNT – Anode filled with a metal powder • Binary catalyst – Hydrogen arc with a mixture of Ni, Fe, Co and Fe. S: 1 g nanotubes/hour
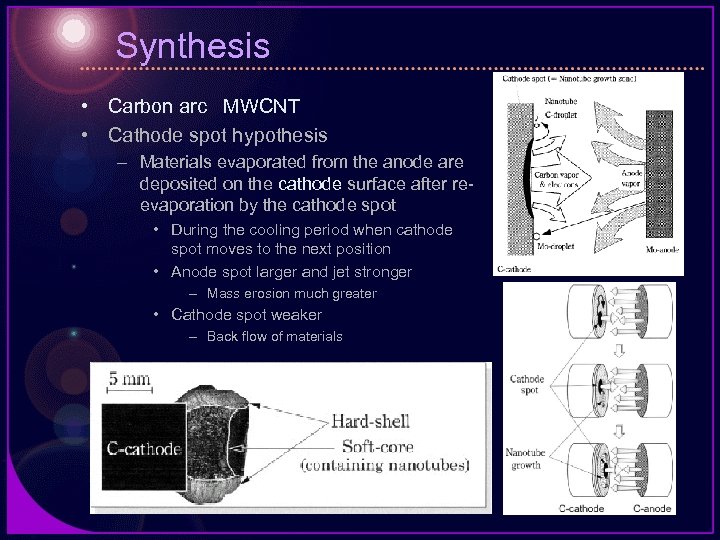
Synthesis • Carbon arc MWCNT • Cathode spot hypothesis – Materials evaporated from the anode are deposited on the cathode surface after reevaporation by the cathode spot • During the cooling period when cathode spot moves to the next position • Anode spot larger and jet stronger – Mass erosion much greater • Cathode spot weaker – Back flow of materials

Synthesis • Carbon arc SWCNT • Occurrence – Web-like deposits on the walls near the cathode – Collaret around the cathode’s edge – Soot • Temperature control of SWCNT – Variation in conductance of the gap – Variation in composition of Ar/He mixture • T~x. He/x. Ar • Thermal conductivity of Ar 8 times smaller – Optimal regime for maximum yield • The gap distance set to obtain strong visible vortices at the cathode edge – dnanotube from 1. 27 (Ar) to 1. 37 nm (He)
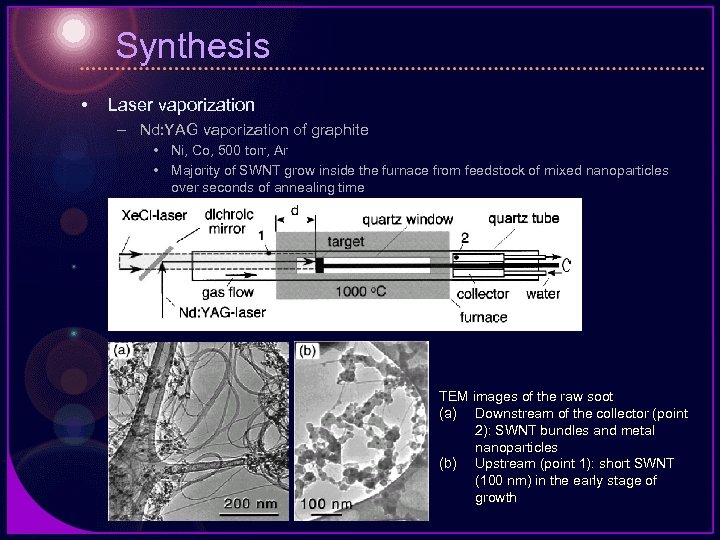
Synthesis • Laser vaporization – Nd: YAG vaporization of graphite • Ni, Co, 500 torr, Ar • Majority of SWNT grow inside the furnace from feedstock of mixed nanoparticles over seconds of annealing time TEM images of the raw soot (a) Downstream of the collector (point 2): SWNT bundles and metal nanoparticles (b) Upstream (point 1): short SWNT (100 nm) in the early stage of growth

Synthesis • Catalytic Chemical Vapor Decomposition CCVD (pyrolysis) – Carbon bearing precursors in the presence of catalysts (Fe, Co, Ni, Al) – Substrate e. g. porous Al 2 O 3 – Example • CH 4, 850 -1000 °C, Al – high quality SWNT – Large scale synthesis • Seeded catalyst – – M/SWCNT Benzene vapors over Fe catalyst at 1100 ºC Nanotube diameter varies with the size of active particles CNT irregular shapes and amorphous coating and catalyst particles embedded • Floating catalyst – SWCNT – Pyrolysis of acetylene in two-stage furnace, ferrocene precursor, sulphur-containing additive
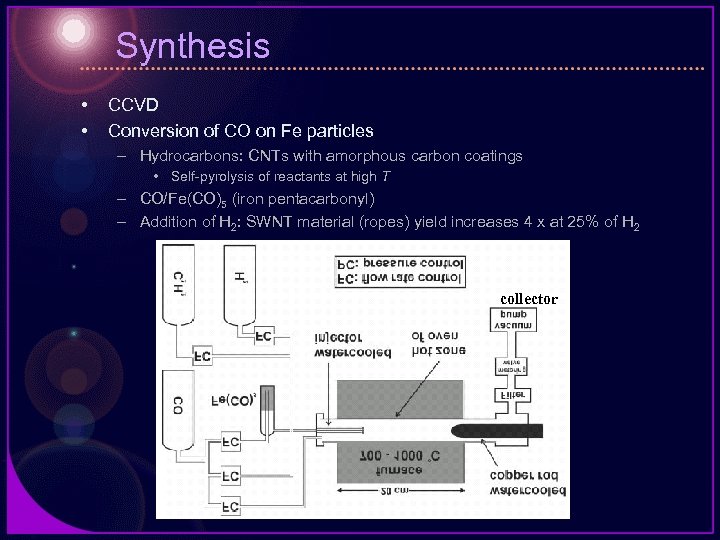
Synthesis • • CCVD Conversion of CO on Fe particles – Hydrocarbons: CNTs with amorphous carbon coatings • Self-pyrolysis of reactants at high T – CO/Fe(CO)5 (iron pentacarbonyl) – Addition of H 2: SWNT material (ropes) yield increases 4 x at 25% of H 2 collector

Synthesis • • CCVD Hi. Pco High-pressure conversion of CO – Thermal decomposition of Fe(CO)5 – Fe(CO)n (n=0 -4) Fe clusters in gas phase – Solid C on Fe clusters produced by CO+CO C(s)+CO 2 – Rapid heating of CO/Fe(CO)5 mixture enhances production of SWCNTs • Running conditions – – p. CO: 30 atm Tshowerhead: 1050 °C Run time: 24 -72 h Production rate: 450 mg/h (10. 8 g/day) SWNT of 97 mol % purity
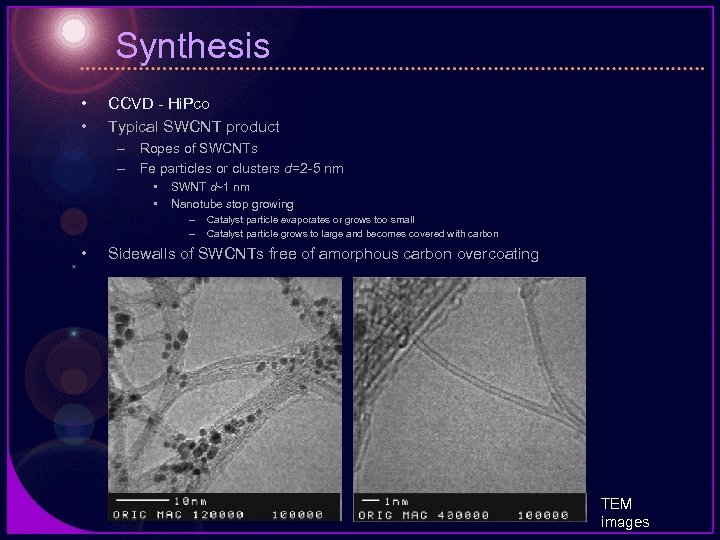
Synthesis • • CCVD - Hi. Pco Typical SWCNT product – Ropes of SWCNTs – Fe particles or clusters d=2 -5 nm • • SWNT d~1 nm Nanotube stop growing – – • Catalyst particle evaporates or grows too small Catalyst particle grows to large and becomes covered with carbon Sidewalls of SWCNTs free of amorphous carbon overcoating TEM images
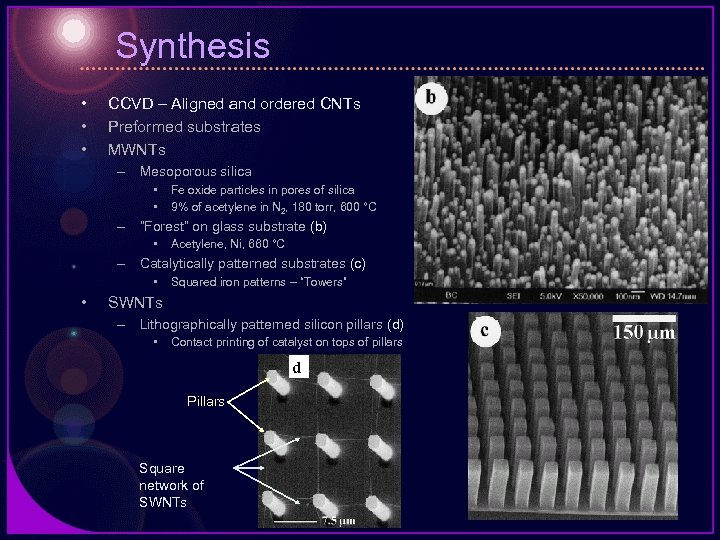
Synthesis • • • CCVD – Aligned and ordered CNTs Preformed substrates MWNTs – Mesoporous silica • • Fe oxide particles in pores of silica 9% of acetylene in N 2, 180 torr, 600 °C – “Forest” on glass substrate (b) • Acetylene, Ni, 660 °C – Catalytically patterned substrates (c) • • Squared iron patterns – “Towers” SWNTs – Lithographically patterned silicon pillars (d) • Contact printing of catalyst on tops of pillars d Pillars Square network of SWNTs

Synthesis • • Plasma-enhanced chemical vapor deposition PECVD Microwave PECVD of methane – – – Large-scale synthesis 600 W, 15 torr Mixture of CH 4 and H 2 Al 2 O 3 substrate coated with ferric nitrate solution, 850÷ 900 ºC Nucleation at the surface of Fe catalyst particles Nanotube grows from the catalyst particle staying on the substrate surface Tangled C nanotubes of uniform diameter (10÷ 150 nm), 20 m
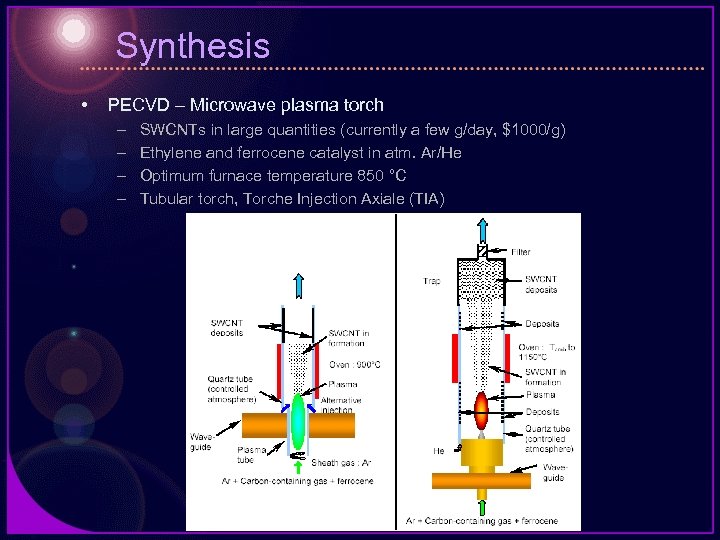
Synthesis • PECVD – Microwave plasma torch – – SWCNTs in large quantities (currently a few g/day, $1000/g) Ethylene and ferrocene catalyst in atm. Ar/He Optimum furnace temperature 850 °C Tubular torch, Torche Injection Axiale (TIA)
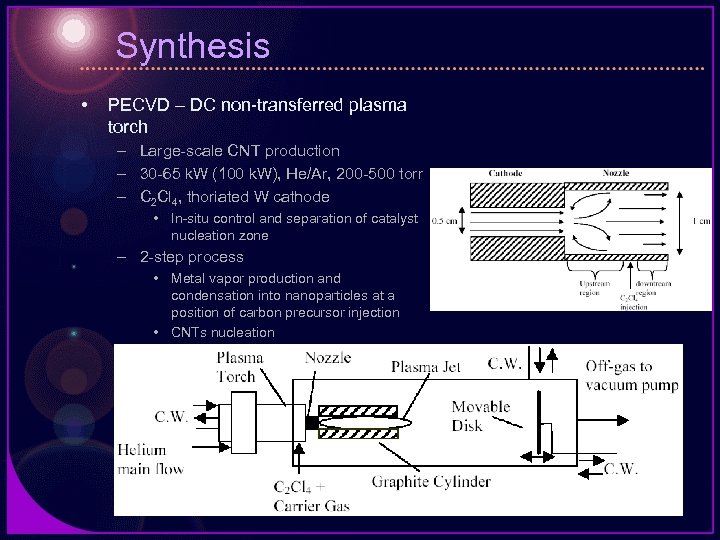
Synthesis • PECVD – DC non-transferred plasma torch – Large-scale CNT production – 30 -65 k. W (100 k. W), He/Ar, 200 -500 torr – C 2 Cl 4, thoriated W cathode • In-situ control and separation of catalyst nucleation zone – 2 -step process • Metal vapor production and condensation into nanoparticles at a position of carbon precursor injection • CNTs nucleation

Synthesis • Pulsed RF PECVD – Vertically aligned CNTs – CH 4 RF glow discharge • 100 W peak power, 53 Pa – Ni catalyst thin films on Si 3 N 4/Si substrates (650 °C) – Alignment mechanism turns on by switching the plasma source for 0. 1 s – Sharp transition • Pulsed plasma-grown straight NTs • Continuous plasma-grown curly NTs Continuous mode pulsed mode
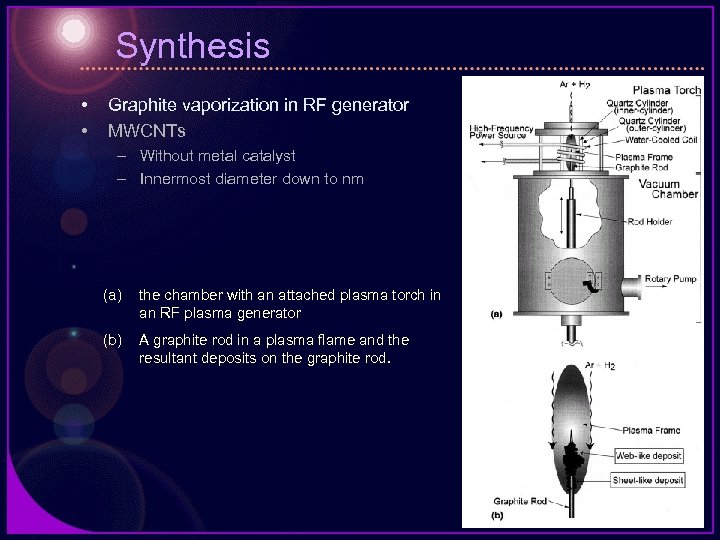
Synthesis • • Graphite vaporization in RF generator MWCNTs – Without metal catalyst – Innermost diameter down to nm (a) the chamber with an attached plasma torch in an RF plasma generator (b) A graphite rod in a plasma flame and the resultant deposits on the graphite rod.
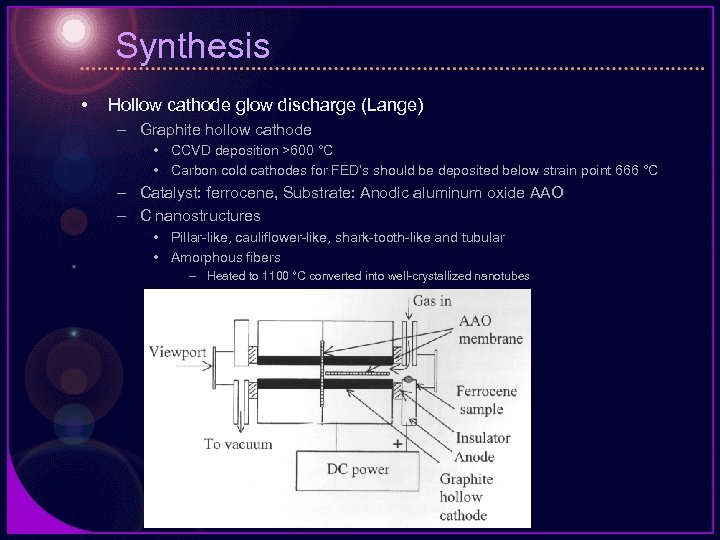
Synthesis • Hollow cathode glow discharge (Lange) – Graphite hollow cathode • CCVD deposition >600 °C • Carbon cold cathodes for FED’s should be deposited below strain point 666 °C – Catalyst: ferrocene, Substrate: Anodic aluminum oxide AAO – C nanostructures • Pillar-like, cauliflower-like, shark-tooth-like and tubular • Amorphous fibers – Heated to 1100 °C converted into well-crystallized nanotubes
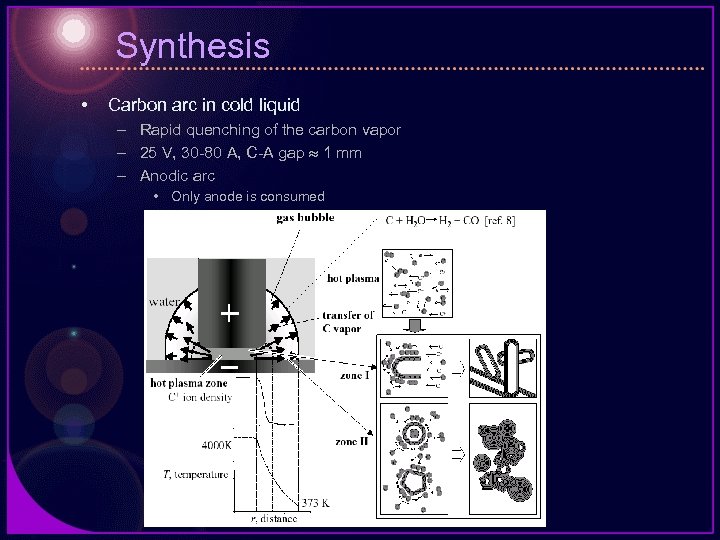
Synthesis • Carbon arc in cold liquid – Rapid quenching of the carbon vapor – 25 V, 30 -80 A, C-A gap 1 mm – Anodic arc • Only anode is consumed
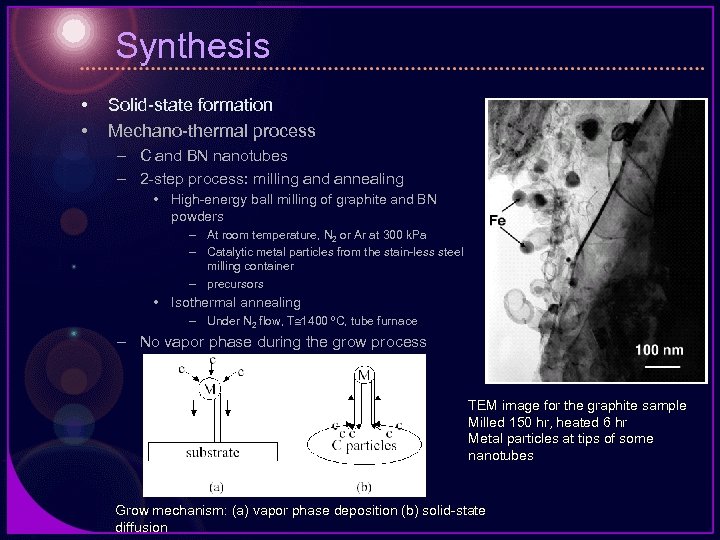
Synthesis • • Solid-state formation Mechano-thermal process – C and BN nanotubes – 2 -step process: milling and annealing • High-energy ball milling of graphite and BN powders – At room temperature, N 2 or Ar at 300 k. Pa – Catalytic metal particles from the stain-less steel milling container – precursors • Isothermal annealing – Under N 2 flow, T 1400 ºC, tube furnace – No vapor phase during the grow process TEM image for the graphite sample Milled 150 hr, heated 6 hr Metal particles at tips of some nanotubes Grow mechanism: (a) vapor phase deposition (b) solid-state diffusion

Synthesis • Electrolysis – Electrolytic conversion of graphite cathode in fused salts • MWCNT – Crystalline lithium carbide catalyst • Reaction of electrodeposited lithium with the carbon cathode • Cost: 10 times the price of gold
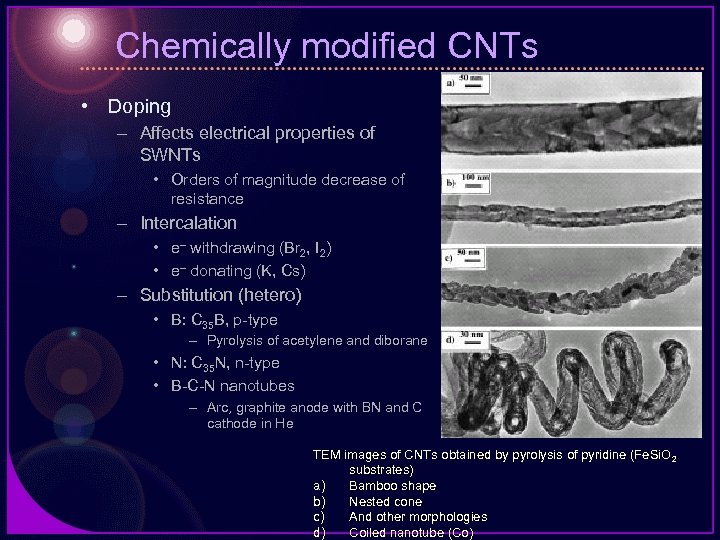
Chemically modified CNTs • Doping – Affects electrical properties of SWNTs • Orders of magnitude decrease of resistance – Intercalation • e– withdrawing (Br 2, I 2) • e– donating (K, Cs) – Substitution (hetero) • B: C 35 B, p-type – Pyrolysis of acetylene and diborane • N: C 35 N, n-type • B-C-N nanotubes – Arc, graphite anode with BN and C cathode in He TEM images of CNTs obtained by pyrolysis of pyridine (Fe. Si. O 2 substrates) a) Bamboo shape b) Nested cone c) And other morphologies d) Coiled nanotube (Co)
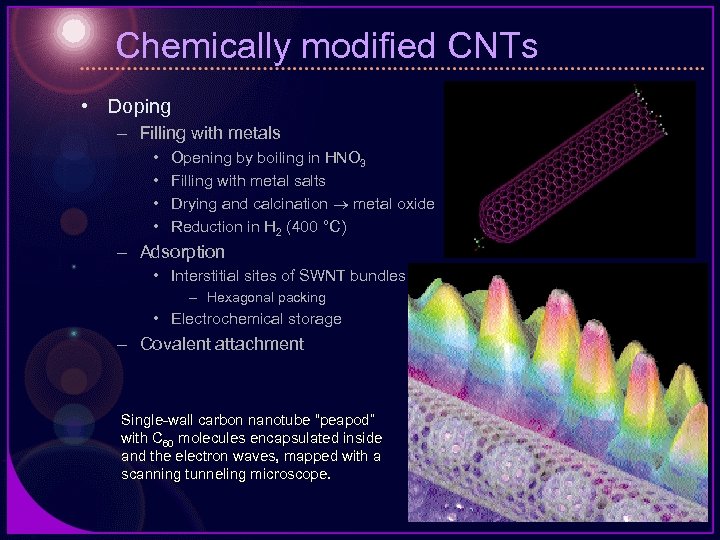
Chemically modified CNTs • Doping – Filling with metals • • Opening by boiling in HNO 3 Filling with metal salts Drying and calcination metal oxide Reduction in H 2 (400 °C) – Adsorption • Interstitial sites of SWNT bundles – Hexagonal packing • Electrochemical storage – Covalent attachment Single-wall carbon nanotube “peapod” with C 60 molecules encapsulated inside and the electron waves, mapped with a scanning tunneling microscope.
Carbon fibers • Organic polymers e. g. poly(acrylonitrile) – stretching – Oxidation in air (200 -300 °C) • Nonmeltable precursor fiber – Heating in nitrogen (1000 -2500 °C) • Until 92% C • D = 6 -10 m – 5 x thinner than human hair • Adding epoxy resin

Carbon fibers • Dispersion of SWCNTs in petroleum pitch – Tensile strength improved by 90% – Elastic modulus by 150% – Electric conductivity increased by 340% • CNTs dispersed in surfactant solution – A soluble compound that reduces the surface tension – recondensed in stream of polymer solution Knotted nanotube fibers, Dfiber 10
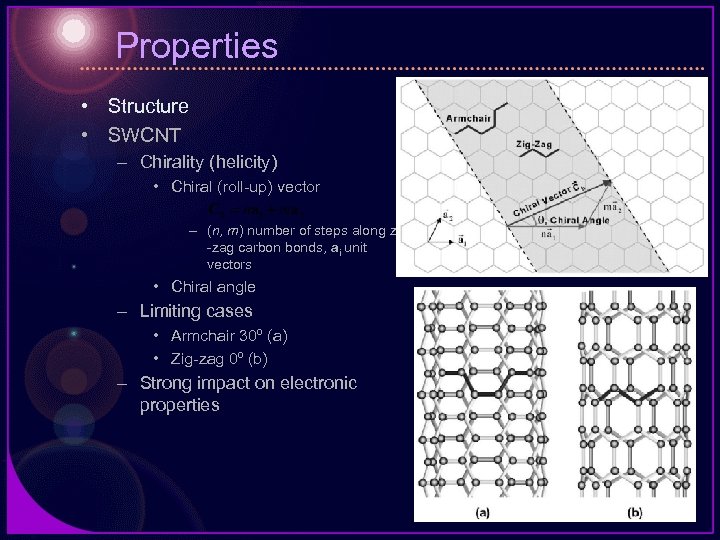
Properties • Structure • SWCNT – Chirality (helicity) • Chiral (roll-up) vector – (n, m) number of steps along zig -zag carbon bonds, ai unit vectors • Chiral angle – Limiting cases • Armchair 30º (a) • Zig-zag 0º (b) – Strong impact on electronic properties

Properties • SWCNT Ropes – Tens of SWNTs packed into hexagonal crystals (van der Waals) TEM image of cross-section of a bundle of SWNTs

Properties • MWCNT – – Concentric SWCNT Each tube can have different chirality Van der Waals bonding Easier and less expensive to produce but more defects – Inner tubes can spin with nearly zero friction • Nano machines • Mechanical properties – Elastic (Young) modulus • > 1 TPa (diamond 1. 2 TPa) – Tensile strength • 10 -100 times > than steel at a fraction of the weight • Thermal properties – Stable up to 2800 ºC – Thermal conductivity 2 x as diamond Axial compression of SWCNT

Properties • Electrical properties – Electric properties ~ diameter and chirality • Metallic (armchair, zigzag) • Semiconducting (zigzag) – Electrical conductivity similar to Cu – Electric-current-carrying capacity • 1000 times higher than copper wires • Optical properties – Nonlinear – Fluorescence • Wavelength depends on diameter – Biosensors, nanomedicine – Remotely triggered exposives – combustion SWNTs exposed to a photographic flash - photo-acoustic effect (expansion and contraction of surrounding gas) - ignition
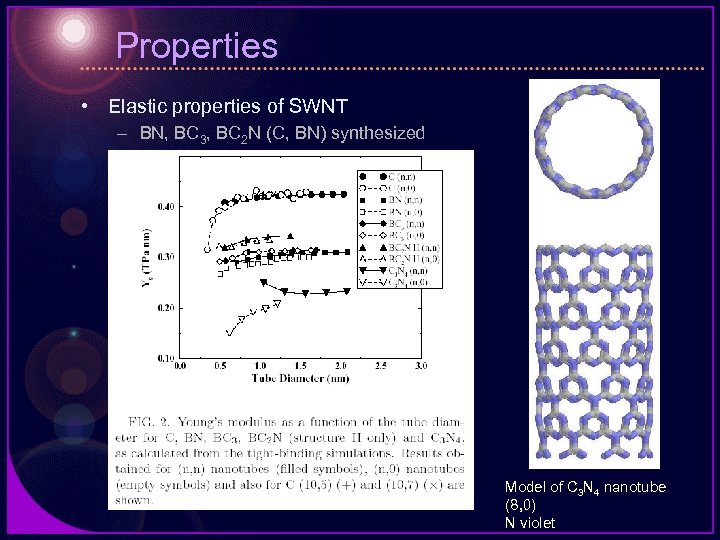
Properties • Elastic properties of SWNT – BN, BC 3, BC 2 N (C, BN) synthesized Model of C 3 N 4 nanotube (8, 0) N violet

Applications • Bulk CNTs – High-capacity hydrogen storage • Aligned CNTs Batteries used in about 60% of cell phones and notebook computers contain MWCNTs. – Field emission based flat-panel displays – Composite materials (polymer resin, metal, ceramic-matrix). – Electromechanical actuators • Individual SWCNTs – – – • Field emission sources Tips for scanning microscopy Nanotweezers Chemical sensors Central elements of miniaturized electronic devices Doped SWCNTs – Chemical sensors • Semiconducting SWCNT: conductance sensitive to doping and adsorption – Small conc. of NO 2 NH 3 (200 ppm): el. conductance increases 3 orders of mag. – SET: single electron transistor Field-effect transistor (FET) - much faster than Si transistors (MOSFET) - much better V-I characteristics - 4 K: single-electron transistor (SET)
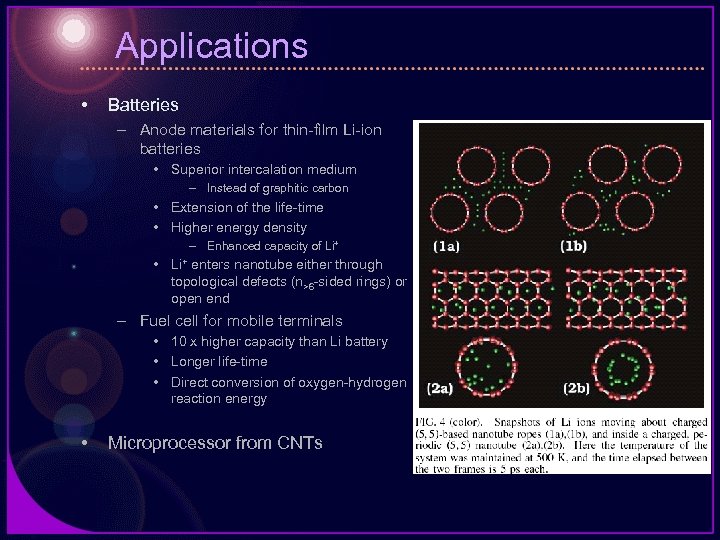
Applications • Batteries – Anode materials for thin-film Li-ion batteries • Superior intercalation medium – Instead of graphitic carbon • Extension of the life-time • Higher energy density – Enhanced capacity of Li+ • Li+ enters nanotube either through topological defects (n>6 -sided rings) or open end – Fuel cell for mobile terminals • 10 x higher capacity than Li battery • Longer life-time • Direct conversion of oxygen-hydrogen reaction energy • Microprocessor from CNTs
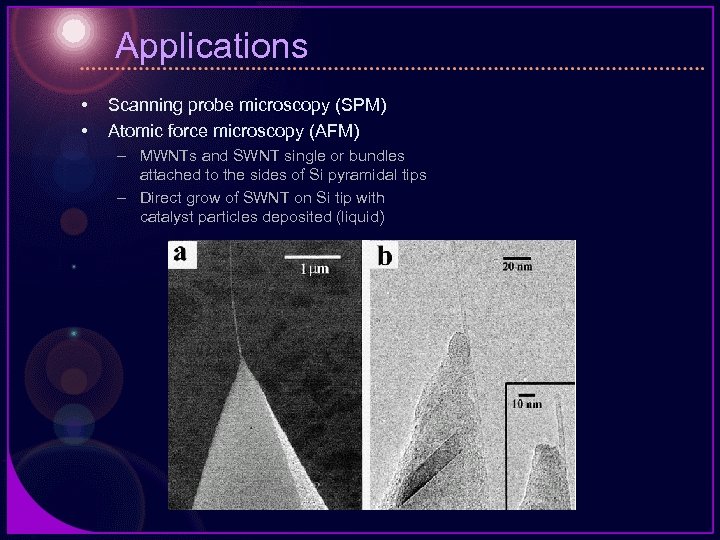
Applications • • Scanning probe microscopy (SPM) Atomic force microscopy (AFM) – MWNTs and SWNT single or bundles attached to the sides of Si pyramidal tips – Direct grow of SWNT on Si tip with catalyst particles deposited (liquid)

Applications • Hydrogen storage – – Interstitial and inside Low cost and high capacity (5. 5 wt%) at room temperature Portable devices Transition metals and hydrogen bonding clusters doping • Uptake and release of hydrogen – H adsorption increases below 77 K • Quantum mechanical nature of interaction
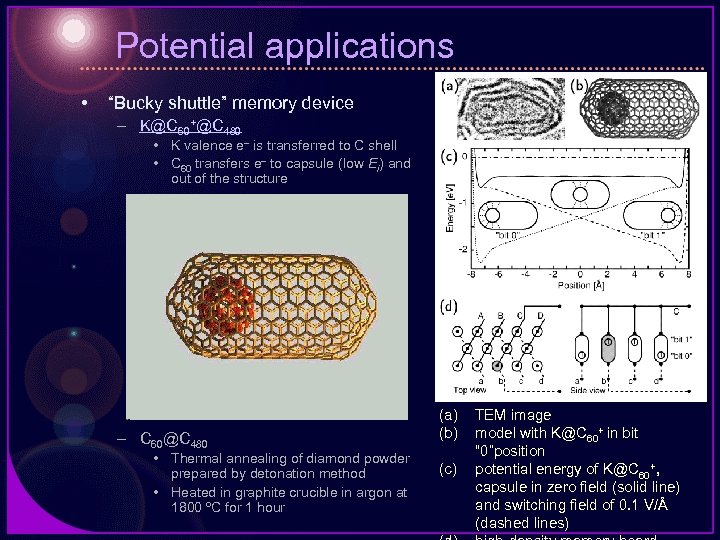
Potential applications • “Bucky shuttle” memory device – K@C 60+@C 480 • K valence e– is transferred to C shell • C 60 transfers e– to capsule (low Ei) and out of the structure – C 60@C 480 • Thermal annealing of diamond powder prepared by detonation method • Heated in graphite crucible in argon at 1800 ºC for 1 hour (a) (b) (c) TEM image model with K@C 60+ in bit “ 0”position potential energy of K@C 60+, capsule in zero field (solid line) and switching field of 0. 1 V/Å (dashed lines)

Potential applications • Electro-mechanical actuators – Actuator effect: the tube increases its length by charge transfer on the tube • Expansion of C-C bond – Artificial muscles • Sheets of SWCNTs – bucky paper • More efficient than natural or ferroelectric muscles The strip actuator - Strips of bucky paper on both sides of a scotch tape - One side is charged negatively and the other positively - Both sides expand but the positive side expands more than the negative
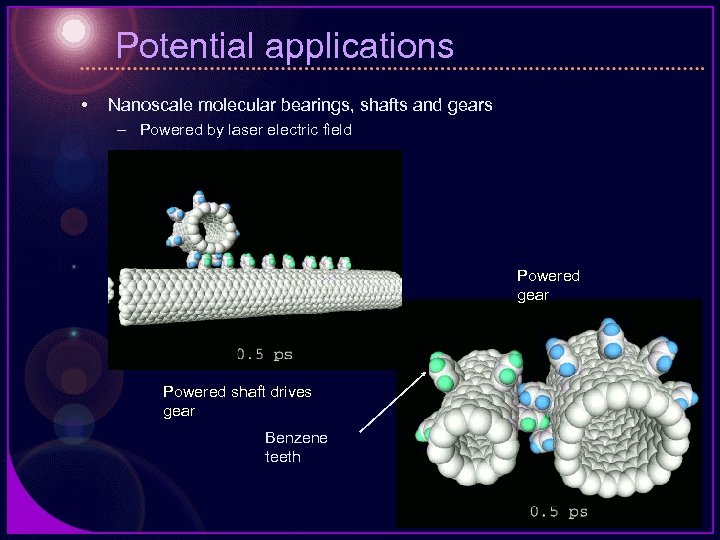
Potential applications • Nanoscale molecular bearings, shafts and gears – Powered by laser electric field Powered gear Powered shaft drives gear Benzene teeth

Potential applications • Nanoscale molecular bearings, shafts and gears Planetary gear
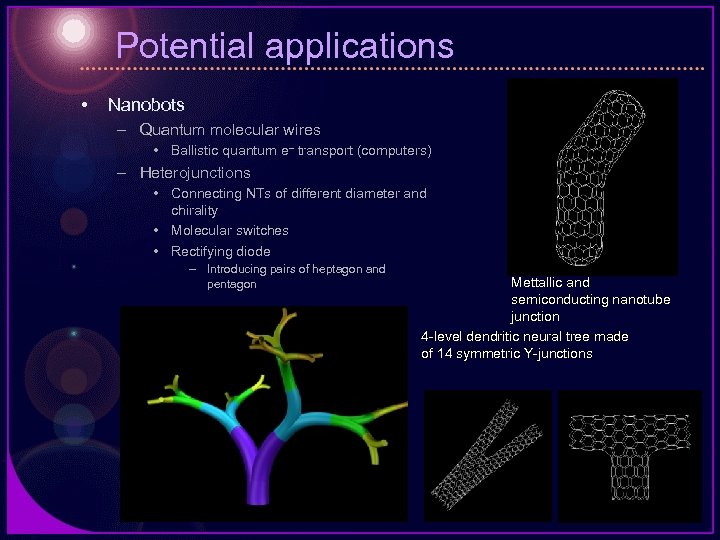
Potential applications • Nanobots – Quantum molecular wires • Ballistic quantum e– transport (computers) – Heterojunctions • Connecting NTs of different diameter and chirality • Molecular switches • Rectifying diode – Introducing pairs of heptagon and pentagon Mettallic and semiconducting nanotube junction 4 -level dendritic neural tree made of 14 symmetric Y-junctions

Potential applications • Nanobots – Chemical adsorption or mechanical deformation of NTs • Chemical reactivity and electronic properties The Steward platform Molecular actuator - CNT nested in an open CNT
Potential applications • Nanobots Nanobot in-body voyage: destroying cell
Potential applications • Nanobots Barber nanobots
41b5fde394d5cfab039ed337978d3de8.ppt