Carbohydrates.ppt
- Количество слайдов: 40
Carbohydrates Novosibirsk State Agrarian University Professor Korotkevich O. S.

Contents: • The structure of carbohydrates • Functions of carbohydrates • Classification of carbohydrates

The structure of carbohydrates • Carbohydrates, sometimes referred to as sugars, consist of monosaccharides and their derivatives. • They are made up of carbon, hydrogen and oxygen, and were originally represented as hydrates of carbon with empirical formula Cn(H 2 O)n
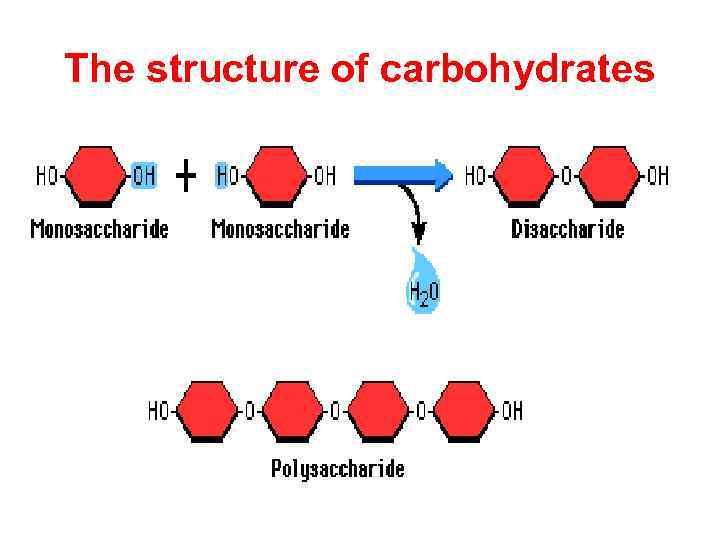
The structure of carbohydrates
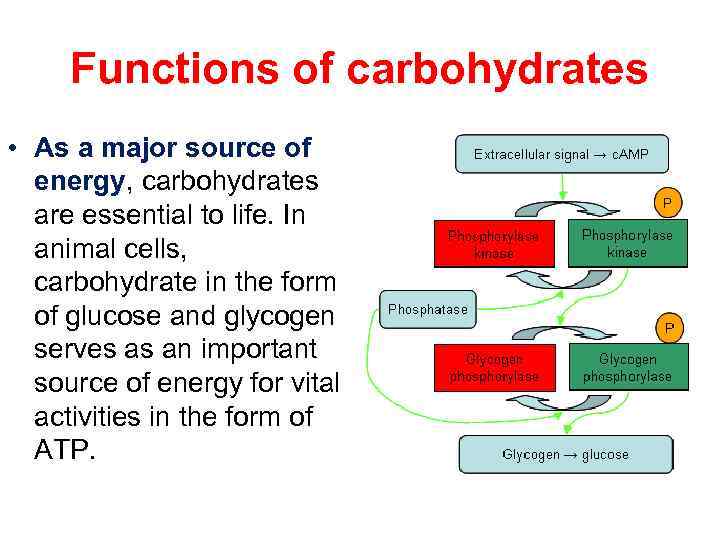
Functions of carbohydrates • As a major source of energy, carbohydrates are essential to life. In animal cells, carbohydrate in the form of glucose and glycogen serves as an important source of energy for vital activities in the form of ATP.

As structural components of cells • For example, cellulose, hemicellulose and pectic substances are common structural carbohydrates in plants chitin, hyaluronic acid, and chondroitin sulphates are important structural polysaccharides of animals.
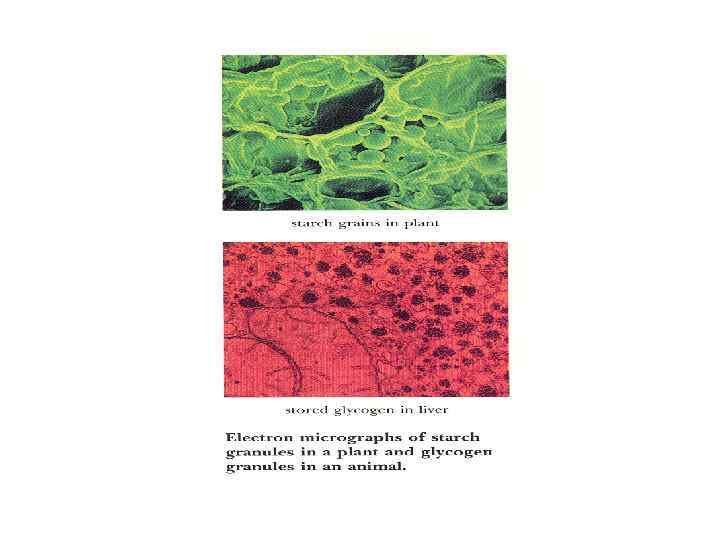
As reserve food • Carbohydrates are stored as reserve products in many tissues e. g. starch is stored in plants (seeds, tubes and rhizome), and glycogen is the storage polysaccharide in animal cells.
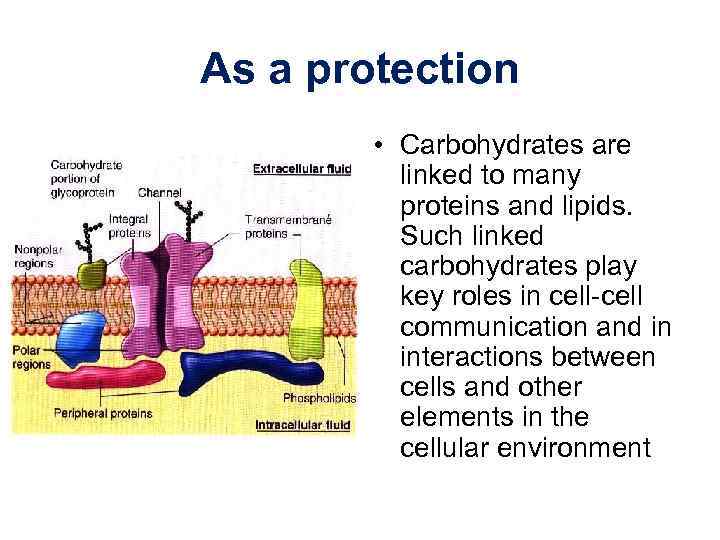
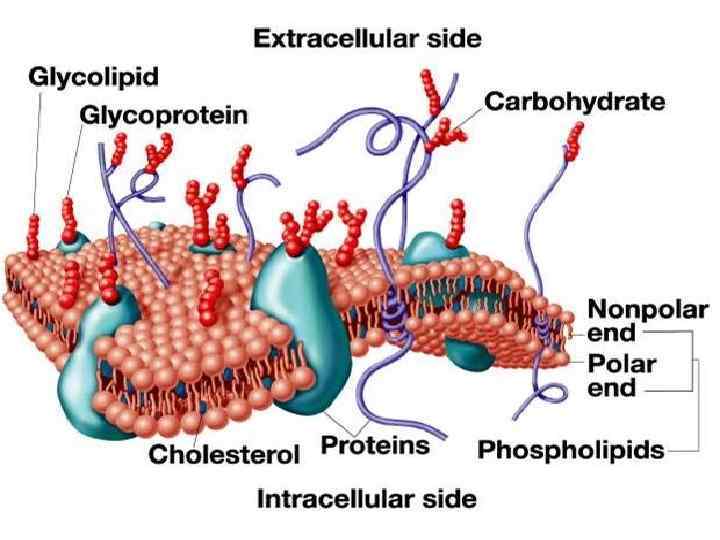
As a protection • Carbohydrates are linked to many proteins and lipids. Such linked carbohydrates play key roles in cell-cell communication and in interactions between cells and other elements in the cellular environment
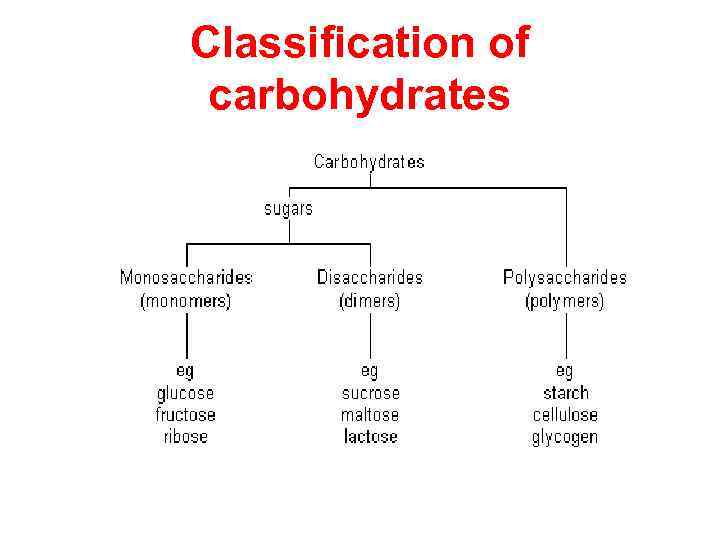
Classification of carbohydrates

Polysaccharides subdivided into: • Homopolysaccharides are polymers that consist of only one type of monosaccharide. • Heteropolysaccharides are polymers that consist of more than one type of monosaccharide

Monosaccharides • The simplest carbohydrates, are aldehydes or ketones that have two or more hydroxyl groups; the empirical formula of many is (C-H 2 O)n, literally a “carbon hydrate”. • Monosaccharides are important fuel molecules as well as building blocks for nucleic acids.
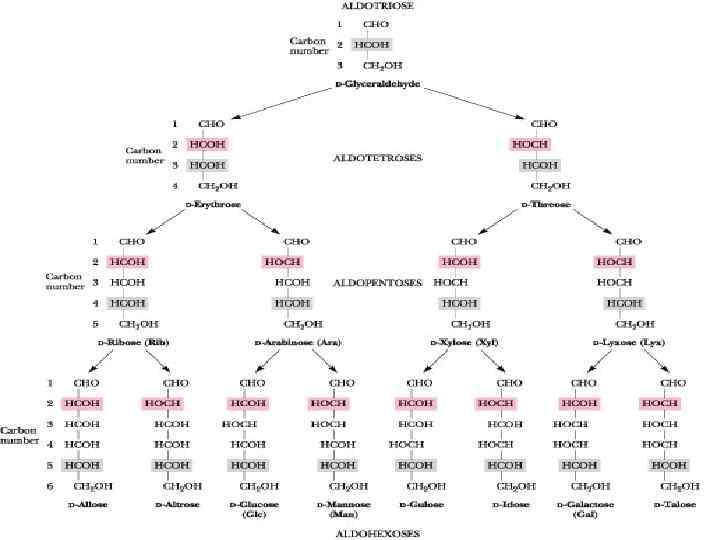
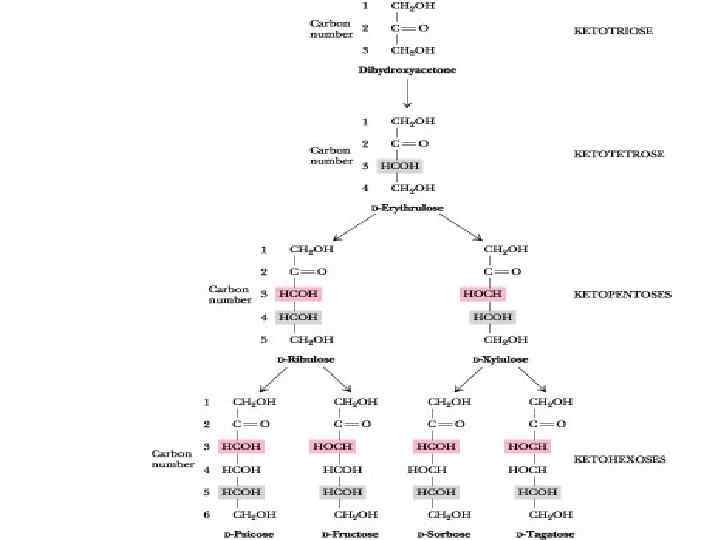

Monosaccharides may be subdivided into: • • • 3 carbons: trioses (glyceraldehydes) 4 carbons: tetroses (erythrose) 5 carbons: pentoses (ribose) 6 carbons: hexoses (glucose) 7 carbons: heptoses (sedoheptulose) 9 carbons: nonoses (neuraminic acid) depending on the number of carbon atoms they possess.

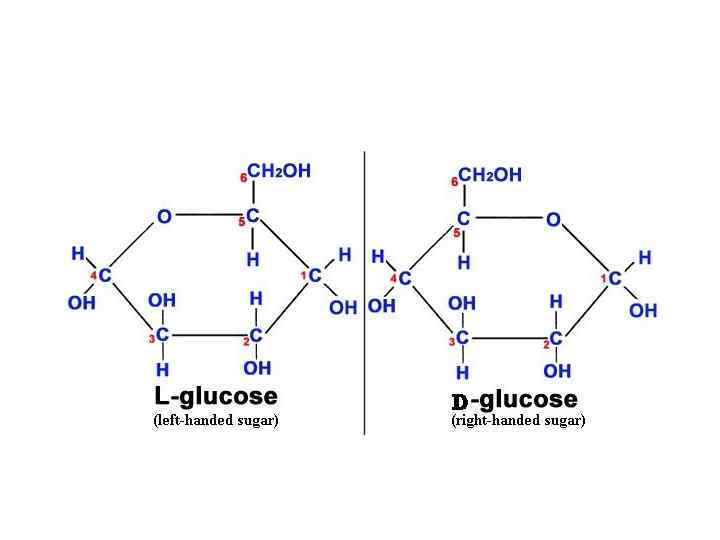
Isomers and epimers • Compounds that have the same chemical formula but have different structures are called isomers. For example, fructose, glucose, mannose and galactose are all isomers of each other, having the same chemical formula, C 6 H 12 O 6.
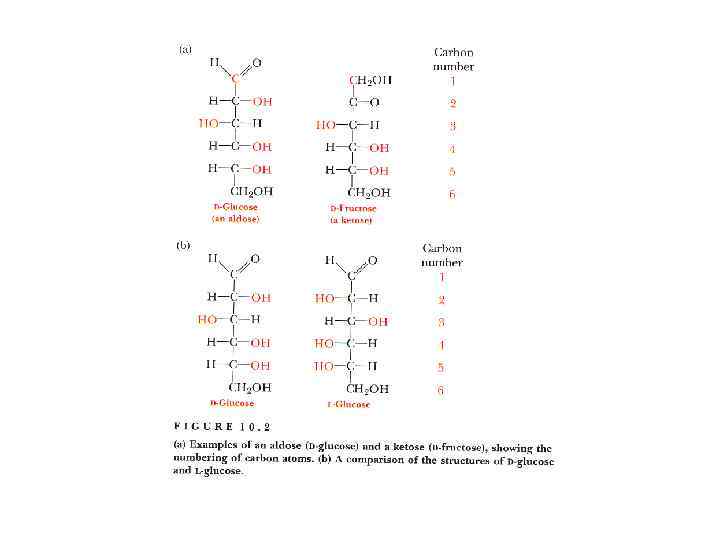

Epimers • If two monosaccharides differ in configuration around only one specific carbon atom (with the exception of carbonyl carbon, see “anomers” below), they are defined as epimers of each other. (Of course, they are also isomers!) For example, glucose and galactose are C-4 epimers – their structures differ only in the position of the – OH group at carbon 4.
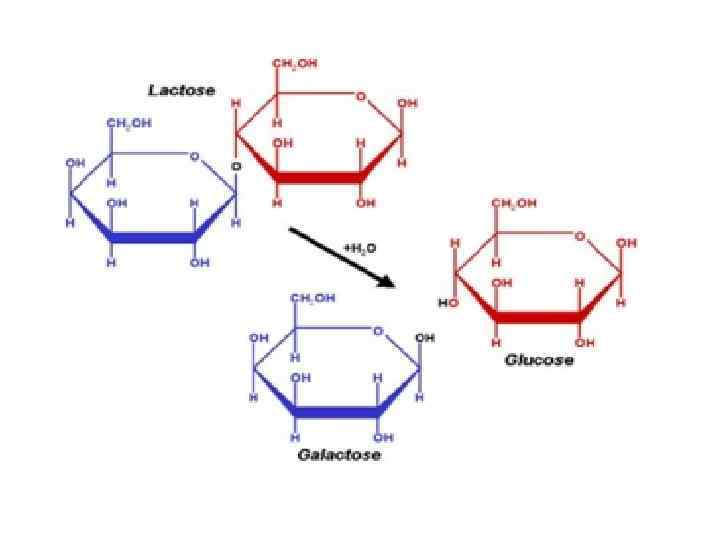
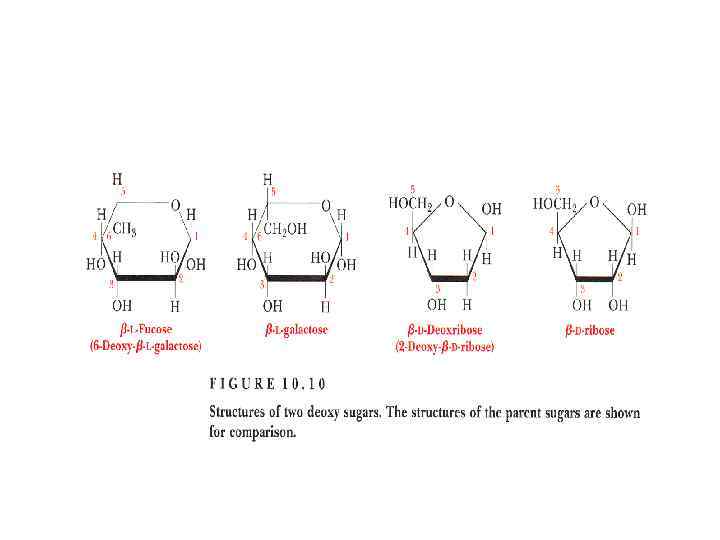

Disaccharides • They are carbohydrates that consist of two sugars joined by O-glycosidic bond. • The general formula is Cn(H 2 O)n-1 • Its examples are sucrose, lactose, maltose, cellobiose etc.
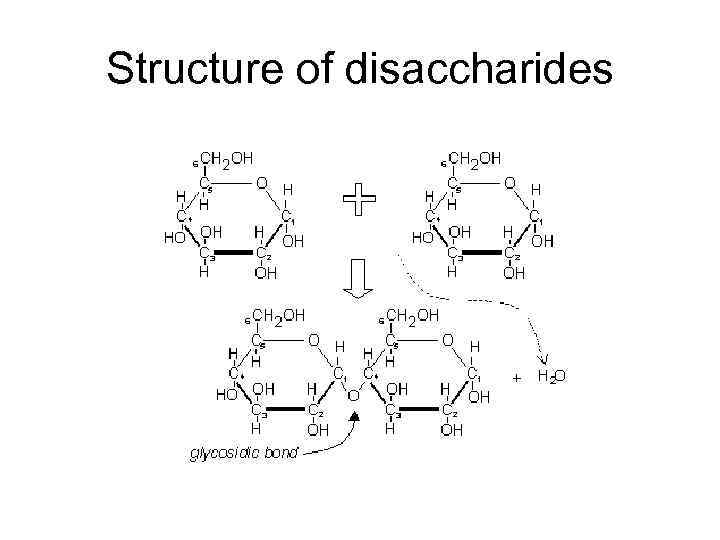
Structure of disaccharides
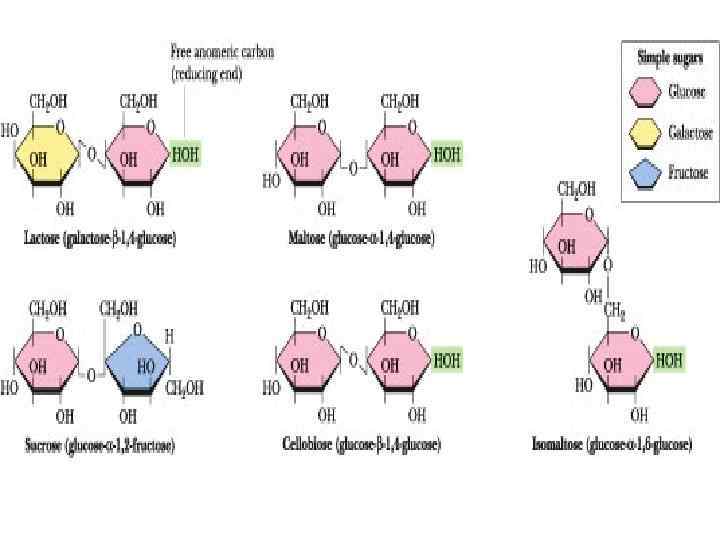
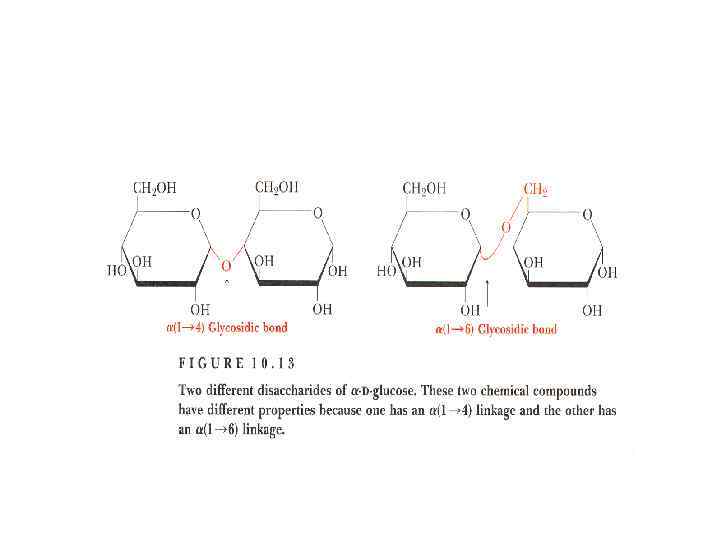
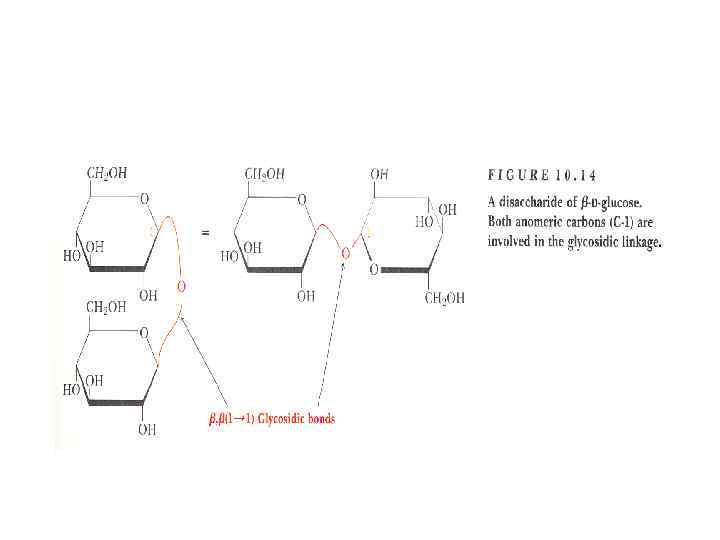
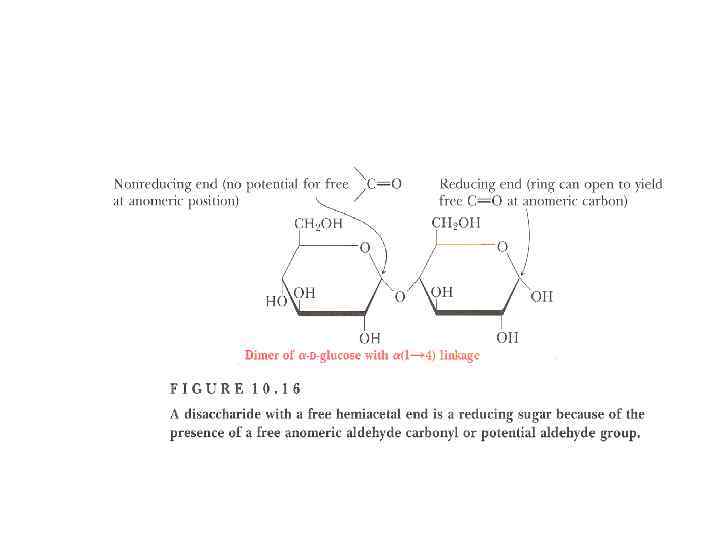
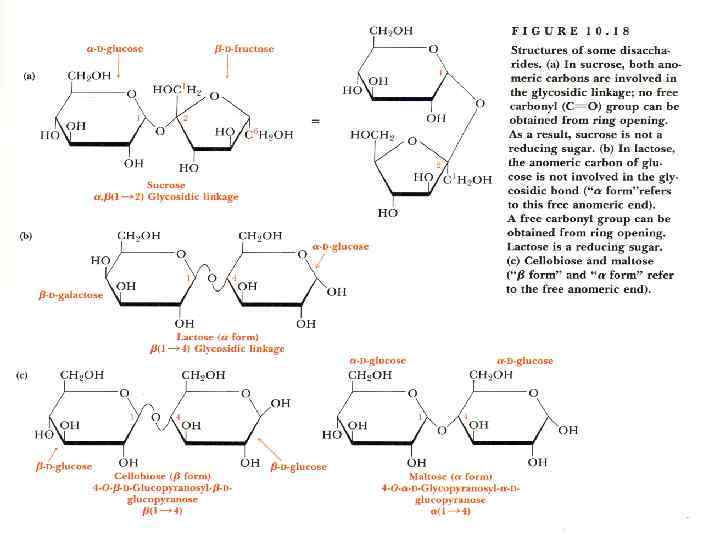

Disaccharides
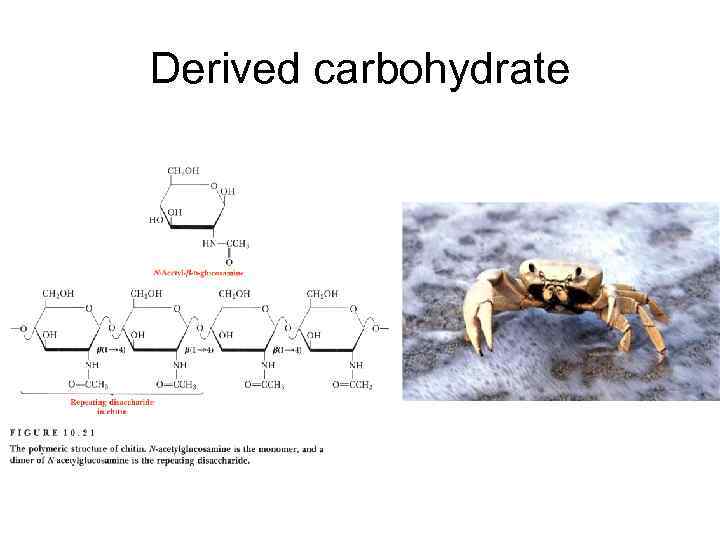
Derived carbohydrate

Oligosaccharides • They yield 3 -6 monosaccharide units on hydrolysis • Its examples are raffinose (trisaccharide), melezitose (trisaccharide), stachyose (tetrasaccharide) and so on

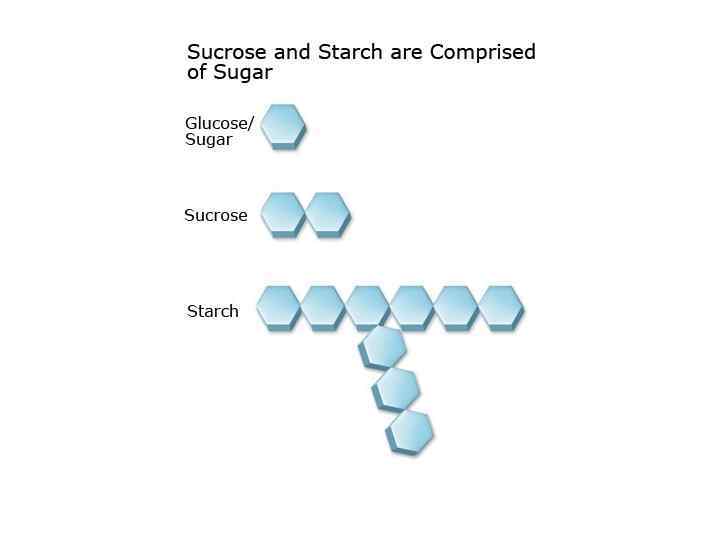
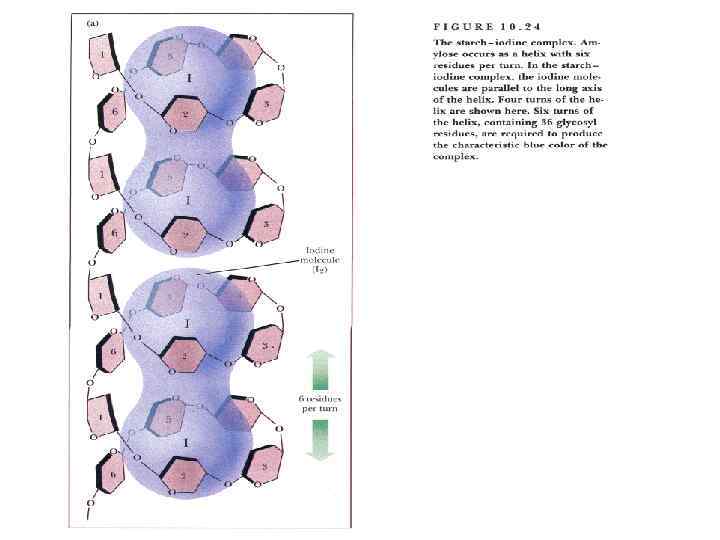
Polysaccharides • They yield more than 6 units of monosaccharides on hydrolysis. • Polysaccharides may be linear or branched. Its examples are starch and dextrin.

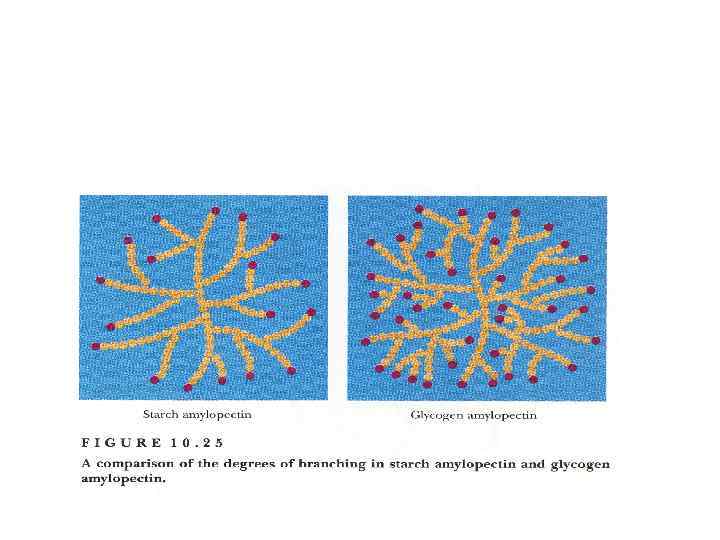
Polysaccharides Homopolysaccharides Heteropolysaccharides • Starch • Glycogen • cellulose • • • Hyaluronic acid Mucopolysaccharides Chondroitin sulfate Heparin Agar Pectin etc.
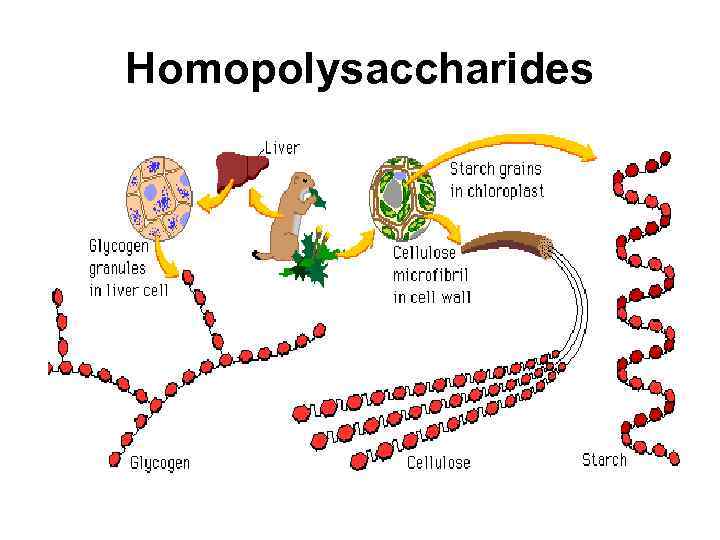
Homopolysaccharides
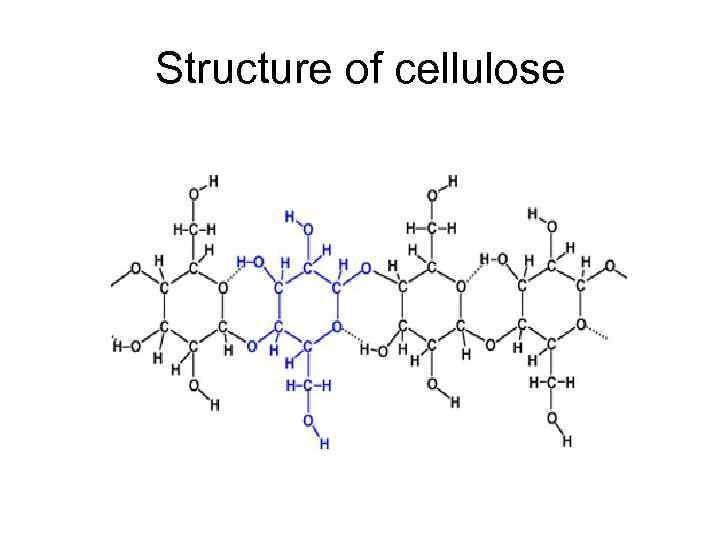
Structure of cellulose

Thank you for your attention
Carbohydrates.ppt